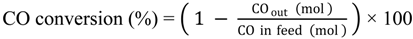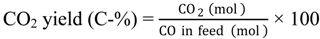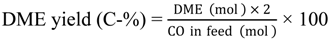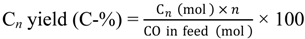Saturated hydrocarbons are produced from syngas over a hybrid catalyst consisting of Cu-ZnO and a metal-loaded ZSM-5 as follows: (1) the conversion of syngas to methanol over Cu-ZnO; (2) the subsequent conversion of the generated methanol to hydrocarbons through the formation of dimethyl ether (DME) over the acid sites of ZSM-5; and (3) the hydrogenation of primarily produced unsaturated hydrocarbons to saturated hydrocarbons over metal species on ZSM-5.
2.1. Hydrocarbon Synthesis from Syngas over Hybrid Catalyst Consisting of Cu-ZnO and Metal-Loaded ZSM-5
The catalytic properties of a hybrid catalyst consisting of Cu-ZnO and metal-loaded ZSM-5 were investigated in the conversion of syngas to hydrocarbons in a near-critical
n-hexane solvent. The metal-loaded ZSM-5 catalysts were prepared by loading 0.5 wt% Pd, 5 wt% Fe, 5 wt% Co or 5 wt% Cu on ZSM-5 with the SiO
2/Al
2O
3 ratio of 23.
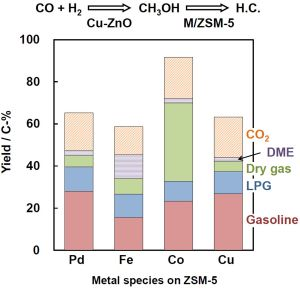
Figure 1 shows product yields after 6.5 h of the reaction at 543 K. Except for the hybrid catalyst with Co/ZSM-5, each catalyst exhibited almost the same yield of the sum of hydrocarbons and DME. Hydrocarbons and DME are produced through the formation of methanol, indicating that the conversion of methanol to DME followed by hydrocarbons is independent of metal species. Pd/ZSM-5 produced selectively hydrocarbons in the gasoline fractions (C
5–C
9 saturated hydrocarbons) among the hydrocarbons with a high CO conversion of 67%. When Fe/ZSM-5 was employed as a portion of the hybrid catalyst, the CO conversion as well as the hydrocarbons yield was smaller than those of the other metal-loaded ZSM-5 catalysts, while DME was selectively produced. Thus, the conversion of DME to hydrocarbons did not take place smoothly over the Fe/ZSM-5 catalyst, resulting in decreasing the hydrocarbons yield. In addition, the yield of CO
2 generated through the water-gas-shift (WGS) reaction was decreased due to a decrease in H
2O generated through the conversion of DME to hydrocarbons. By contrast, the hybrid catalyst with Co/ZSM-5 gave the highest CO conversion as well as the hydrocarbons yield among the four hybrid catalysts. Among the hydrocarbons, methane in hydrocarbons in dry gas fraction was dominantly produced with the yield of 38%. In the conversion of methanol to hydrocarbons, hydrocarbons with a carbon number of more than 1 are dominantly produced as a result of the cracking of intermediates [
9,
10]. These results indicated that the Co species on ZSM-5 caused selectively the methanation of CO, CO
2 and methanol under the present conditions. The hybrid catalyst with 5 wt% Cu/ZSM-5 showed the CO conversion of 64% and the 28% yield of hydrocarbons in the gasoline fraction, which were very similar to those over that with 0.5 wt% Pd/ZSM-5. The TOFs in terms of the carbon amount (C-mol) of hydrocarbon products per mole of the metal species were estimated to 478 h
−1 and 8521 h
−1 for 5 wt% Cu/ZSM-5 and 0.5 wt% Pd/ZSM-5, respectively. Although the TOFs were much different between the two catalysts, the hydrocarbon distributions were almost the same. Thus, the hydrocarbon formation from methanol or DME would be dependent on the acidity of ZSM-5, and 5 wt% Cu on ZSM-5 would have almost the same catalytic properties for the WGS reaction and the methanation as those of 0.5 wt% Pd on ZSM-5.
Figure 1.
Product yield resulting from conversion of syngas to hydrocarbons over hybrid catalysts consisting of Cu-ZnO and metal-loaded ZSM-5. Reaction conditions: catalyst, 1.0 g (0.5 g Cu-ZnO and 0.5 g metal-loaded ZSM-5); temperature, 543 K; Psyngas = 2.5 MPa; Pn-hexane = 1.5 MPa; W/Fsyngas = 9.7 g-catalyst h mol−1; H2/CO = 1.9.
Figure 1.
Product yield resulting from conversion of syngas to hydrocarbons over hybrid catalysts consisting of Cu-ZnO and metal-loaded ZSM-5. Reaction conditions: catalyst, 1.0 g (0.5 g Cu-ZnO and 0.5 g metal-loaded ZSM-5); temperature, 543 K; Psyngas = 2.5 MPa; Pn-hexane = 1.5 MPa; W/Fsyngas = 9.7 g-catalyst h mol−1; H2/CO = 1.9.
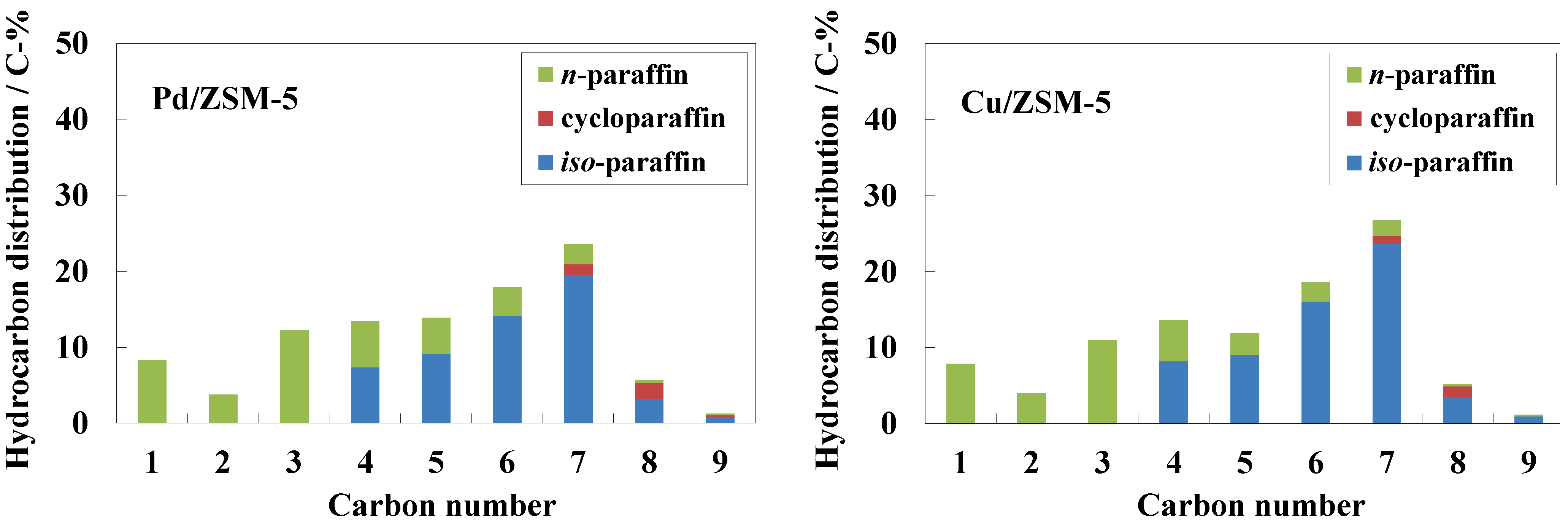
Figure 2 shows hydrocarbon distributions over the hybrid catalysts with 0.5 wt% Pd/ZSM-5 or 5 wt% Cu/ZSM-5. Both catalysts exhibited almost the same hydrocarbon distributions, and C
7 hydrocarbons were selectively produced. Only saturated hydrocarbons with a carbon number of less than 10 were obtained as products during the reaction. Among the gasoline-ranged hydrocarbons, the formation of
iso-paraffins was more favorable than
n-paraffins probably due to the isomerization of produced hydrocarbons over acid sites of ZSM-5. Furthermore, mono-branched paraffins such as 2-methylhexane and 3-methylpentane were mainly obtained as
iso-paraffins. The carbon ratios of (
iso-paraffins + cycloparaffins)/
n-paraffins were 4.6 and 7.2 for 0.5 wt% Pd/ZSM-5 and 5 wt% Cu/ZSM-5, respectively. Metal species such as Pt and Pd, with a high hydrogenation ability, coexisting with a solid acid catalyst supplies dissociated hydrogen species to the surface of the solid acid catalyst to generate active acid sites, which play an important role in the isomerization of alkane [
18,
19,
21,
22,
23,
24]. The amount of the Cu species on ZSM-5 was larger than that of the Pd species on ZSM-5. Thus, it is suggested that in the case of the hybrid catalyst with Cu/ZSM-5 the larger number of acid sites were generated from hydrogen dissociated on the metal species to cause the isomerization of hydrocarbon products without the cracking of hydrocarbons.
Figure 2.
Hydrocarbon distribution resulting from conversion of syngas to hydrocarbons over hybrid catalysts with 0.5 wt% Pd/ZSM-5 or 5 wt% Cu/ZSM-5. Reaction conditions: catalyst, 1.0 g (0.5 g Cu-ZnO and 0.5 g metal-loaded ZSM-5); temperature, 543 K; Psyngas = 2.5 MPa; Pn-hexane = 1.5 MPa; W/Fsyngas = 9.7 g-catalyst h mol−1; H2/CO = 1.9.
Figure 2.
Hydrocarbon distribution resulting from conversion of syngas to hydrocarbons over hybrid catalysts with 0.5 wt% Pd/ZSM-5 or 5 wt% Cu/ZSM-5. Reaction conditions: catalyst, 1.0 g (0.5 g Cu-ZnO and 0.5 g metal-loaded ZSM-5); temperature, 543 K; Psyngas = 2.5 MPa; Pn-hexane = 1.5 MPa; W/Fsyngas = 9.7 g-catalyst h mol−1; H2/CO = 1.9.
2.2. Effect of Copper Loaded on ZSM-5 on Catalytic Properties
The hydrogenation ability of the Cu species on ZSM-5 would play an important role in supplying acidic species to the surface of ZSM-5 as well as in hydrogenating olefins, which were primarily produced from DME, to paraffins. In addition, the hydrogenation ability is important to decompose carbonaceous species deposited in/on ZSM-5 because a rapid deactivation occurs due to the deposition of coke resulting from the further oligomerization of the carbonaceous species to cover the acid sites and/or to block the pores of the zeolite [
25]. In order to investigate the effects of the Cu species on ZSM-5 on the durability of the catalyst, the conversion of syngas to hydrocarbons was conducted over the hybrid catalysts with the different Cu loadings on ZSM-5.
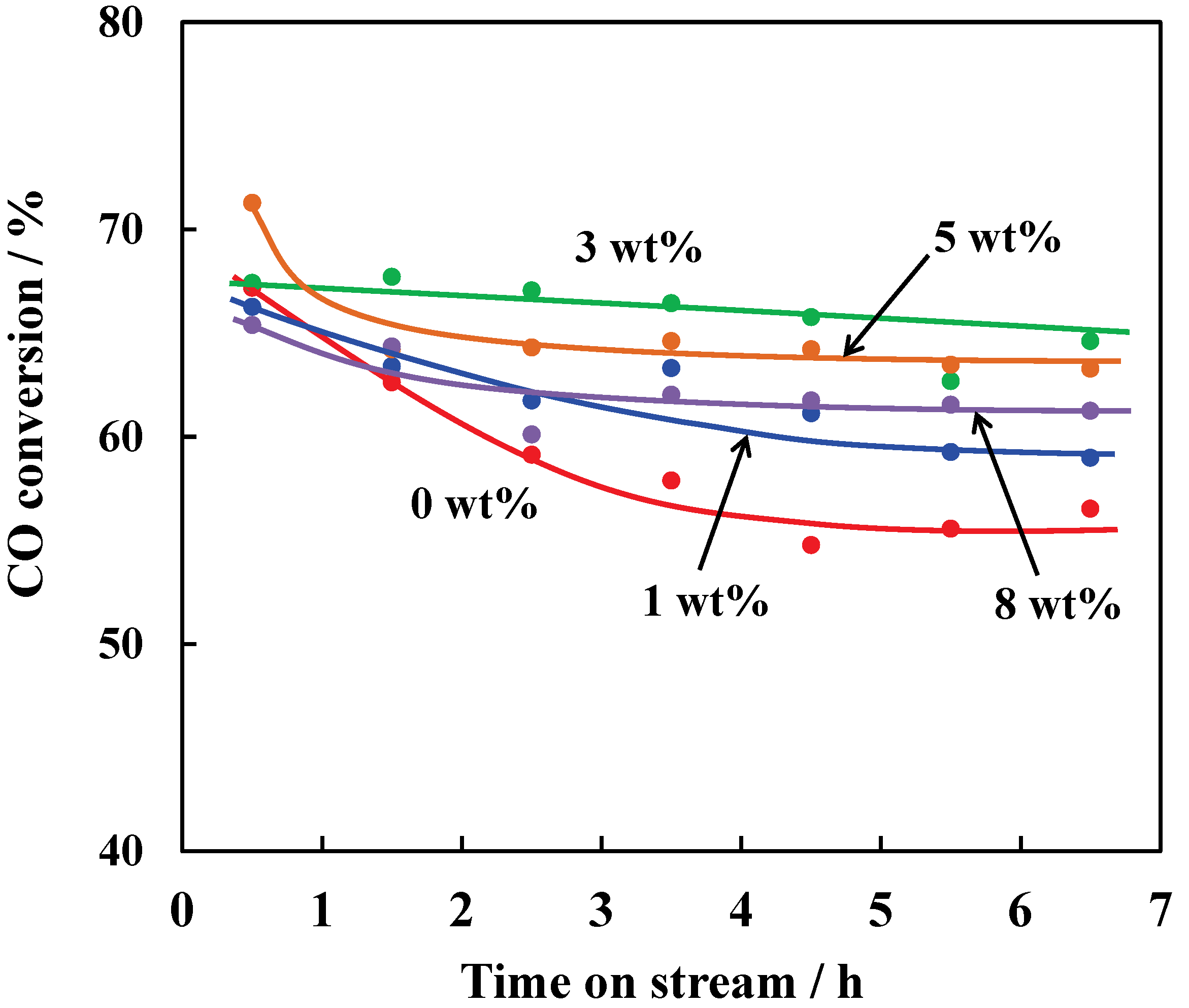
Figure 3 shows time course plots of the CO conversions over the hybrid catalysts containing Cu/ZSM-5 with the different Cu loadings. The initial CO conversion was reached to ca. 67%, independent of the Cu loadings, indicating that the Cu species on ZSM-5 did not influence the conversion of syngas to methanol followed by DME. In the case of employing ZSM-5 without the Cu species, the CO conversion rapidly decreased after the reaction started until the conversion reached to 57% after 6.5 h. By loading even small amount of the Cu species on ZSM-5, the deactivation became slow. When 3 wt% Cu species was loaded on ZSM-5, the rapid deactivation was obviously suppressed compared with 0 wt% and 1 wt% Cu/ZSM-5, resulting in the CO conversion of 65% after 6.5 h of the reaction time. The inhibition of the deactivation by loading the Cu species on ZSM-5 is consistent with the previous report employing hybrid catalysts with Pd/ZSM-5 [
15,
20]. By contrast, a further increase in the Cu loading caused the deactivation at the initial periods of the reaction again; in addition, decreased the CO conversion. However, the CO conversions of the hybrid catalysts with 5 wt% or 8 wt% Cu/ZSM-5 became constant after 2.5 h of the reaction time, while the slight deactivation was observed over the hybrid catalyst with 3 wt% Cu/ZSM-5 during the reaction.
Figure 3.
CO conversion over hybrid catalysts composed of Cu-ZnO and Cu/ZSM-5 with different Cu loading amounts. Reaction conditions: catalyst, 1.0 g (0.5 g Cu-ZnO and 0.5 g Cu/ZSM-5); temperature, 543 K; Psyngas = 2.5 MPa; Pn-hexane = 1.5 MPa; W/Fsyngas = 9.7 g-catalyst h mol−1; H2/CO = 1.9.
Figure 3.
CO conversion over hybrid catalysts composed of Cu-ZnO and Cu/ZSM-5 with different Cu loading amounts. Reaction conditions: catalyst, 1.0 g (0.5 g Cu-ZnO and 0.5 g Cu/ZSM-5); temperature, 543 K; Psyngas = 2.5 MPa; Pn-hexane = 1.5 MPa; W/Fsyngas = 9.7 g-catalyst h mol−1; H2/CO = 1.9.
Hydrogen uptakes determined from the hydrogen chemisorption measurement were 2.4 μmol g−1, 3.6 μmol g−1, and 3.6 μmol g−1 for 1 wt%, 5 wt%, and 8 wt% Cu/ZSM-5, respectively. The hydrogen uptake was increased by increasing the Cu loading. Thus, it is assumed that the Cu species on ZSM-5 inhibited the formation of heavy coke through the hydrogenation of carbonaceous species, leading to the suppression of the deactivation.
Figure 4 shows product yields at the initial period and after 6.5 h of the reaction at 543 K. In order to investigate the product distribution without the influence of the deactivation, the product yields at the initial period of the reaction were compared on the basis of a similar CO conversion. At the initial period, although the hydrocarbon yield of the hybrid catalyst with 1 wt% Cu/ZSM-5 was smaller than that of the hybrid catalyst with H-ZSM-5, the gasoline-ranged hydrocarbon yields of both the catalysts were almost the same values. Increasing the Cu loading resulted in an obvious increase in the gasoline-ranged hydrocarbons yield with the yields of hydrocarbons in LPG and dry gas fractions unchanged. Since CO
2 is generated from CO through the WGS reaction, a high hydrocarbon yield leads to the generation of a large amount of H
2O to encourage the CO
2 production through the WGS reaction. However, the CO
2 yield was decreased by increasing the Cu loading. It is indicated that the Cu species on ZSM-5 had less influence on the WGS reaction under the present conditions.
Figure 4.
Product yield resulting from conversion of syngas to hydrocarbons over hybrid catalysts with Cu/ZSM-5 with different Cu loadings. Reaction conditions: catalyst, 1.0 g (0.5 g Cu-ZnO and 0.5 g Cu/ZSM-5); temperature, 543 K; Psyngas = 2.5 MPa; Pn-hexane = 1.5 MPa; W/Fsyngas = 9.7 g-catalyst h mol−1; H2/CO = 1.9.
Figure 4.
Product yield resulting from conversion of syngas to hydrocarbons over hybrid catalysts with Cu/ZSM-5 with different Cu loadings. Reaction conditions: catalyst, 1.0 g (0.5 g Cu-ZnO and 0.5 g Cu/ZSM-5); temperature, 543 K; Psyngas = 2.5 MPa; Pn-hexane = 1.5 MPa; W/Fsyngas = 9.7 g-catalyst h mol−1; H2/CO = 1.9.
At 6.5 h after the reaction started, the product distribution was dependent on the Cu loading as well. An increase in the Cu loading resulted in increasing the gasoline-ranged hydrocarbons yield and in decreasing the DME yield. When increasing the Cu loading amount up to 5 wt%, the hydrocarbons yield slightly decreased with the slight increase in the gasoline-ranged hydrocarbons yield. It is implied that the Cu species on ZSM-5 would cover the acid sites causing the cracking of hydrocarbons, leading to the improvement in the yield of the gasoline-ranged hydrocarbons.
Acid sites of a catalyst should play an important role in the conversion of methanol to DME followed by hydrocarbons.
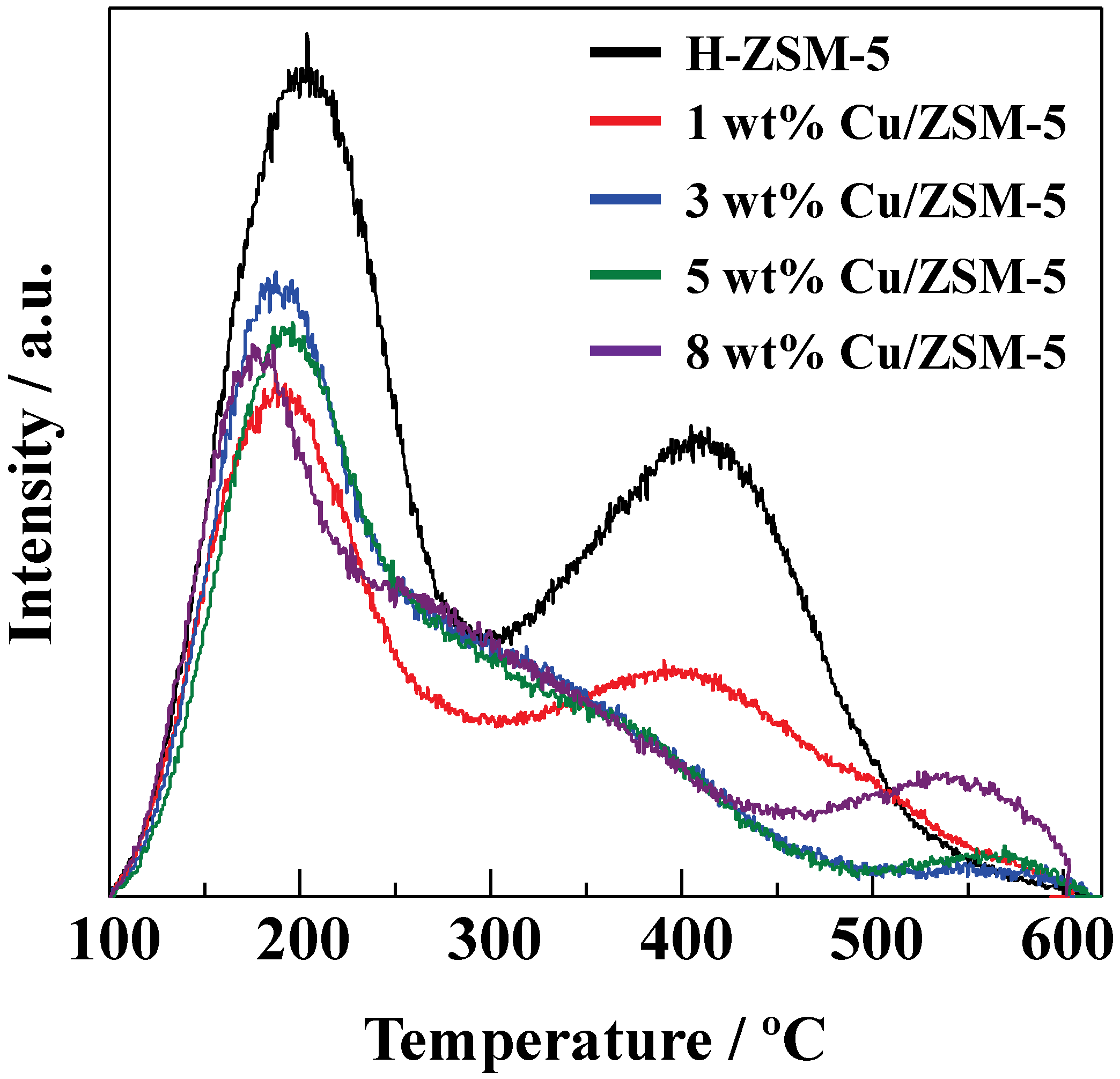
Figure 5 shows NH
3-TPD profiles of Cu/ZSM-5 with the different Cu loadings. The acid amounts estimated from the peak at higher temperature were 0.99 mmol g
−1, 0.63 mmol g
−1, 0.52 mmol g
−1, 0.48 mmol g
−1, and 0.59 mmol g
−1 for H-ZSM-5, 1 wt%, 3 wt%, 5 wt%, and 8 wt% Cu/ZSM-5, respectively. The peak at higher temperature was decreased by increasing the Cu loading. When the Cu loading was increased up to 3 wt%, a shoulder peak appeared at around 300 K. The peak at higher temperature (573–873 K) corresponds to NH
3 desorption from catalytically active acid sites [
26,
27]. It is suggested that the Cu species interacted with the acid sites of ZSM-5 to make their acid strength weaker. The high Cu loading (3–8 wt%) catalysts exhibited a new peak at around 550 K, and the peak was increased by increasing the Cu loading. Since the large peak was observed on Cu/ZSM-5 with the high Cu loading of 8 wt%, the peak can be derived from NH
3 adsorbed on the Cu species, which is consistent with the previous reports [
28,
29,
30]. As shown in
Figure 4, the gasoline-ranged hydrocarbons yield was drastically improved when the Cu loading was increased up to 3 wt%, while the slight increase in the gasoline-ranged hydrocarbons yield was observed by the further increase in the Cu loading, which was in line with the changes in the peak at the higher temperature in the NH
3-TPD profiles. Therefore, it is suggested that the weak acid sites generated by loading the Cu species on ZSM-5 played an important role in the selective synthesis of the gasoline-ranged hydrocarbons without the polymerization and cracking of products because of mild acid strength.
Figure 5.
Temperature programmed ammonia desorption (NH3-TPD) profiles of Cu/ZSM-5 with different Cu loading amounts.
Figure 5.
Temperature programmed ammonia desorption (NH3-TPD) profiles of Cu/ZSM-5 with different Cu loading amounts.
2.3. Durability of Hybrid Catalyst with Cu/ZSM-5
In order to investigate the durability of the hybrid catalyst with Cu/ZSM-5 in the syngas conversion, the changes in the CO conversion and product selectivities along with time-on-stream were examined in a long-run reaction at 553 K. It is reported that by loading Pd on ZSM-5, a high CO conversion was retained without the rapid deactivation and changes in product distributions in the conversion of syngas to the gasoline-ranged hydrocarbons [
15]. As shown in
Figure 6, the initial CO conversion of the hybrid catalyst with 0.5 wt% Pd/ZSM-5 reached to 80%, while the hybrid catalyst with 5 wt% Cu/ZSM-5 exhibited 77% initial CO conversion. In the case of the hybrid catalyst with Pd/ZSM-5, the CO conversion gradually decreased until around 7 h after the reaction started, and then, the conversion of
ca. 70% was kept until 30 h of the reaction time. By contrast, the CO conversion of the hybrid catalyst with Cu/ZSM-5 was kept constant with the value of
ca. 74% during the reaction. Although CO
2 selectivity of the hybrid catalyst with Cu/ZSM-5 was slightly smaller than that of the hybrid catalyst with Pd/ZSM-5, the hybrid catalyst with Cu/ZSM-5 exhibited the higher DME selectivity than the hybrid catalyst with Pd/ZSM-5 during the reaction. These findings indicate that Cu/ZSM-5 constantly transformed methanol generated from syngas into DME followed by hydrocarbons without the rapid deposition of carbonaceous species on the catalyst during the reaction. Furthermore, the Cu species on ZSM-5 did not encourage the CO consumption through the WGS reaction compared with Pd/ZSM-5. Owing to the advantages mentioned above, Cu species is promising for the development of bifunctional catalysts in the selective synthesis of hydrocarbons from syngas, instead of precious metals such as Pd and Pt.
Figure 6.
Conversion of syngas to hydrocarbons over hybrid catalysts with 0.5 wt% Pd/ZSM-5 and 5 wt% Cu/ZSM-5. Reaction conditions: catalyst, 1.0 g (0.5g Cu-ZnO and 0.5 g metal-loaded ZSM-5); temperature, 553 K; Psyngas = 2.5 MPa; Pn-hexane = 1.5 MPa; W/Fsyngas = 9.7 g-catalyst h mol−1; H2/CO = 1.9.
Figure 6.
Conversion of syngas to hydrocarbons over hybrid catalysts with 0.5 wt% Pd/ZSM-5 and 5 wt% Cu/ZSM-5. Reaction conditions: catalyst, 1.0 g (0.5g Cu-ZnO and 0.5 g metal-loaded ZSM-5); temperature, 553 K; Psyngas = 2.5 MPa; Pn-hexane = 1.5 MPa; W/Fsyngas = 9.7 g-catalyst h mol−1; H2/CO = 1.9.