Visible Light-Photocatalytic Activity of Sulfate-Doped Titanium Dioxide Prepared by the Sol−Gel Method
Abstract
:1. Introduction
2. Results and Discussion
2.1. Characterization of the Photocatalysts
| Sample | T | S0 | S300 | S400 | S600 |
|---|---|---|---|---|---|
| crystallite size/nm | 17 ± 2 | - | 5.4 ± 0.3 | 6.7 ± 0.3 | 11 ± 1 |
| specific surface area/m2g−1 | 86 | - | 155 | 120 | 98 |
| TCE adsorption/mol g−1 | 1.0 × 10−5 | 9.5 × 10−5 | 2.1 × 10−5 | 1.6 × 10−5 | 1.0 × 10−5 |
| S/Ti ratio | 0 | 0.078 | 0.046 | 0.017 | 0.004 |





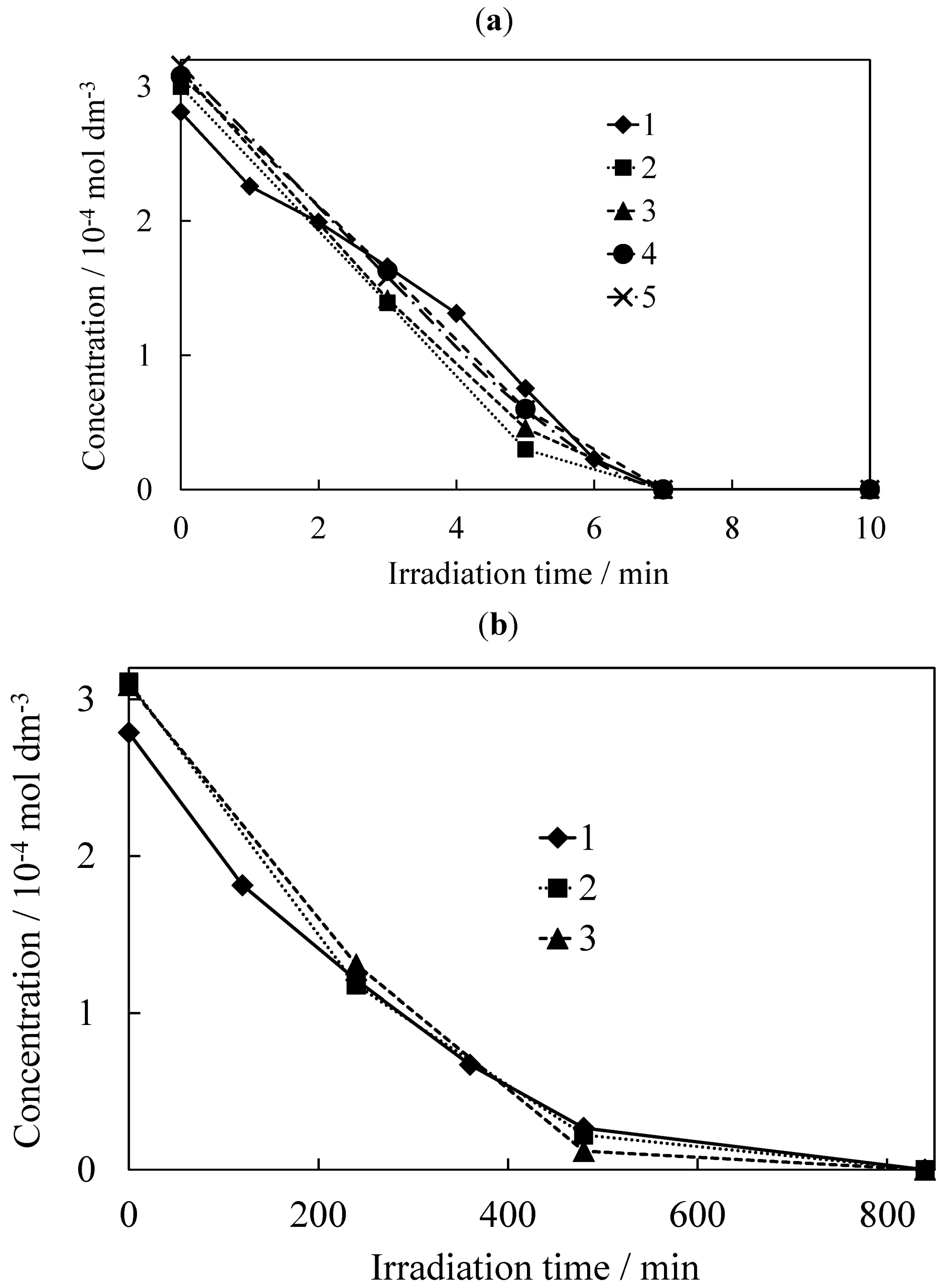
2.2. Photocatalytic Degradation of TCE
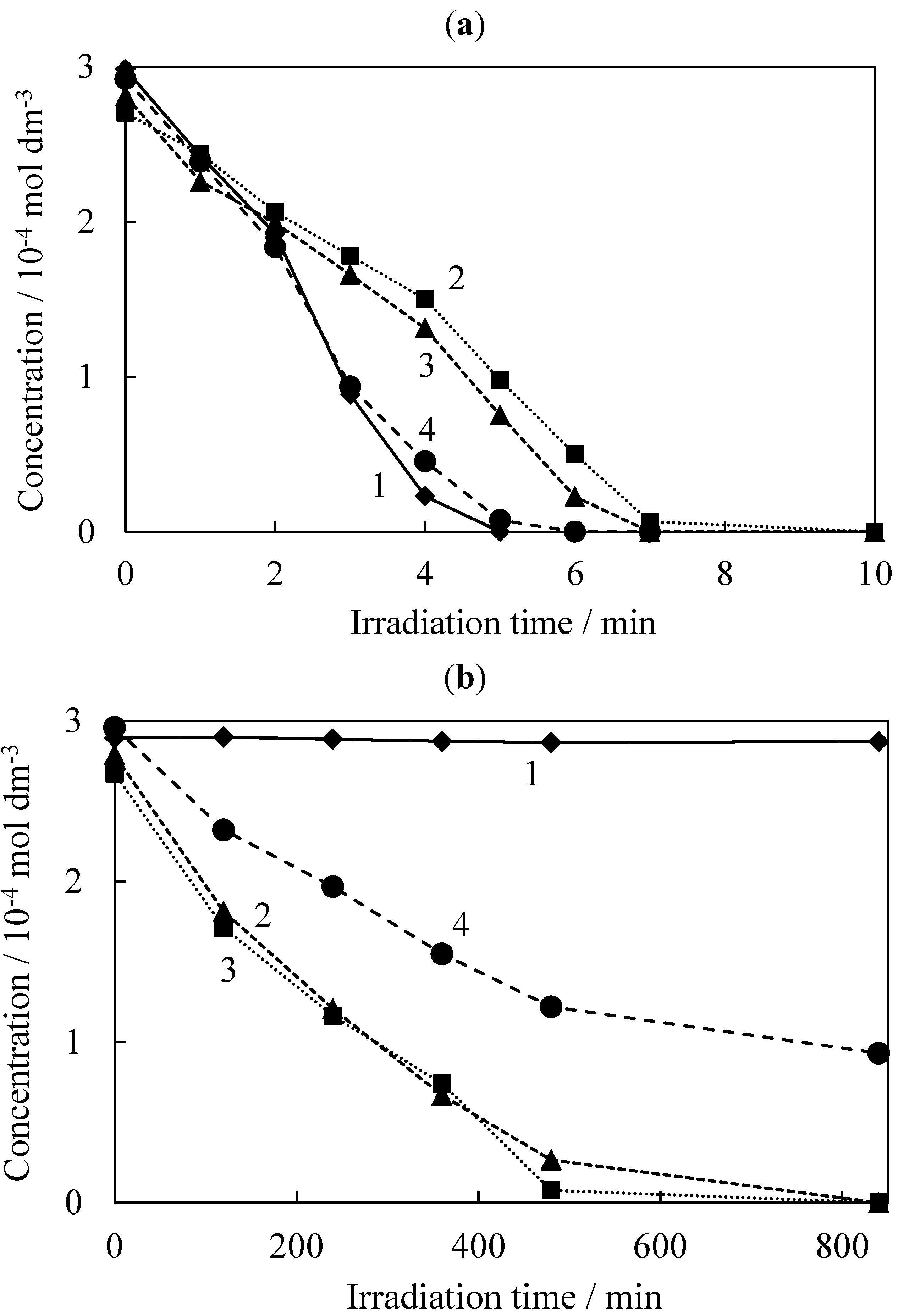
| Sample | T | S300 | S400 | S600 |
|---|---|---|---|---|
| degradation rate/mol dm−3 min−1 | ||||
| UV irradiation | 0.64 | 0.31 | 0.31 | 0.45 |
| visible irradiation | 3.6 × 10−5 | 5.1 × 10−3 | 5.2 × 10−3 | 2.4 × 10−3 |
| degradation rate per unit surface area/mol dm−3 min−1 m−2 | - | - | - | - |
| UV irradiation | 3.0 × 10−2 | 8.0 × 10−3 | 1.0 × 10−2 | 1.9 × 10−2 |
| visible irradiation | 1.7 × 10−6 | 1.3 × 10−4 | 1.7 × 10−4 | 9.6 × 10−5 |

3. Experimental Section
3.1. Materials
3.2. Preparation of Photocatalysts
3.3. Characterization of Photocatalysts
3.4. Photocatalytic Degradation of TCE
4. Conclusions
Acknowledgments
References
- Chatterjee, D.; Dasgupta, S. Visible light induced photocatalytic degradation of organic pollutants. J. Photochem. Photobiol. C 2005, 6, 186–205. [Google Scholar] [CrossRef]
- Fujishima, A.; Rao, T.N.; Tryk, D.A. Titanium dioxide photocatalysis. J. Photochem. Photobiol. C 2000, 1, 1–21. [Google Scholar] [CrossRef]
- Sato, S. Photocatalytic activity of NOx-doped TiO2 in the visible light region. Chem. Phys. Lett. 1986, 123, 126–128. [Google Scholar] [CrossRef]
- Ihara, T.; Miyoshi, M.; Iriyama, Y.; Matsumoto, O.; Sugihara, S. Visible-light-active titanium oxide photocatalyst realized by an oxygen-deficient structure and by nitrogen doping. Appl. Catal. B 2003, 42, 403–409. [Google Scholar] [CrossRef]
- Subagio, D.P.; Srinivasan, M.; Lim, M.; Lim, T. Photocatalytic degradation of bisphenol-A by nitrogen-doped TiO2 hollow sphere in a vis-LED photoreactor. Appl. Catal. B 2010, 95, 414–422. [Google Scholar] [CrossRef]
- Gandhe, A.R.; Naik, S.P.; Fernandes, J.B. Selective synthesis of N-doped mesoporous TiO2 phases having enhanced photocatalytic activity. Microporous Mesoporous Mater. 2005, 87, 103–109. [Google Scholar] [CrossRef]
- Choi, H.; Antoniou, M.G.; Pelaez, M.; De la Cruz, A.A.; Shoemaker, J.A.; Dionysiou, D.D. Mesoporous nitrogen-doped TiO2 for the photocatalytic destruction of the cyanobacterial toxin microcystin-LR under visible light irradiation. Environ. Sci. Technol. 2007, 41, 7530–7535. [Google Scholar] [CrossRef]
- Jagadale, T.C.; Takale, S.P.; Sonawane, R.S.; Joshi, H.M.; Patil, S.I.; Kale, B.B.; Ogale, S.B. N-Doped TiO2 nanoparticle based visible light photocatalyst by modified peroxide sol-gel method. J. Phys. Chem. C 2008, 112, 14595–14602. [Google Scholar]
- Ananpattarachai, J.; Kajitvichyanukul, P.; Seraphin, S. Visible light absorption ability and photocatalytic oxidation activity of various interstitial N-doped TiO2 prepared from different nitrogen dopants. J. Hazardous Mater. 2009, 168, 253–261. [Google Scholar] [CrossRef]
- Yokosuka, Y.; Oki, K.; Nishikiori, H.; Tatsumi, Y.; Tanaka, N.; Fujii, T. Photocatalytic degradation of trichloroethylene using N-doped TiO2 prepared by a simple sol-gel process. Res. Chem. Intermed. 2009, 35, 43–53. [Google Scholar] [CrossRef]
- Nishikiori, H.; Fukasawa, Y.; Yokosuka, Y.; Fujii, T. Nitrogen doping into titanium dioxide by the sol-gel method using nitric acid. Res. Chem. Intermed. 2011, 37, 869–881. [Google Scholar] [CrossRef]
- Dong, C.X.; Xian, A.P.; Ham, E.H.; Shang, J.K. Acid-mediated sol-gel synthesis of visible-light active photocatalysts. J. Mater. Sci. 2006, 41, 6168–6170. [Google Scholar] [CrossRef]
- Oki, K.; Tsuchida, S.; Nishikiori, H.; Tanaka, N.; Fujii, T. Photocatalytic degradation of chlorinated ethenes. Int. J. Photoenergy 2003, 5, 11–15. [Google Scholar] [CrossRef]
- Oki, K.; Yamada, S.; Tsuchida, S.; Nishikiori, H.; Tanaka, N.; Fujii, T. Degradation and isomerization of 1,2-dichloroethenes by photocatalytic reactions. Res. Chem. Intermed. 2003, 29, 827–837. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, J.; Lin, Y.; Zhang, Y.; Wei, Y. Simple fabrication and photocatalytic activity of S-doped TiO2 under low power LED visible light irradiation. Ceramics Int. 2009, 35, 3061–3065. [Google Scholar] [CrossRef]
- Han, C.; Pelaez, M.; Likodimos, V.; Kontos, A.G.; Falaras, P.; O’Shea, K.; Dionysiou, D.D. Innovative visible light-activated sulfur doped TiO2 films for water treatment. Appl. Catal. B 2011, 107, 77–87. [Google Scholar] [CrossRef]
- Muggli, D.S.; Ding, L. Photocatalytic performance of sulfated TiO2 and Degussa P-25 TiO2 during oxidation of organics. Appl. Catal. B 2001, 32, 181–194. [Google Scholar] [CrossRef]
- Noda, L.K.; de Almeida, R.M.; Gonçalves, N.S.; Probst, L.F.D.; Sala, O. TiO2 with a high sulfate content—thermogravimetric analysis, determination of acid sites by infrared spectroscopy and catalytic activity. Catal. Today 2003, 85, 69–74. [Google Scholar]
- Wang, X.; Yu, J.C.; Liu, P.; Wang, X.; Su, W.; Fu, X. Probing of photocatalytic surface sites on SO42−/TiO2 solid acids by in situ FT-IR spectroscopy and pyridine adsorption. J. Photochem. Photobiol. A 2006, 179, 339–347. [Google Scholar] [CrossRef]
- Maira, A.J.; Yeung, K.L.; Lee, C.Y.; Yue, P.L.; Chan, C.K. Size effects in gas-phase photo-oxidation of trichloroethylene using nanometer-sized TiO2 catalysts. J. Catal. 2000, 192, 185–196. [Google Scholar]
- Nishikiori, H.; Tanaka, N.; Kitsui, T.; Fujii, T. Photocurrent observed in dye-doped titania gel. J. Photochem. Photobiol. A 2006, 179, 125–129. [Google Scholar] [CrossRef]
- Nishikiori, H.; Uesugi, Y.; Takami, S.; Setiawan, R.A.; Fujii, T.; Qian, W.; El-Sayed, M.A. Influence of steam treatment on dye-titania complex formation and photoelectric conversion property of dye-doped titania gel. J. Phys. Chem. C 2011, 115, 2880–2887. [Google Scholar]
- Nishikiori, H.; Setiawan, R.A.; Miyamoto, K.; Sukmono, G.; Uesugi, Y.; Teshima, K.; Fujii, T. Photoinduced electron transport in dye-containing titania gel films. RSC Adv. 2012, 2, 4258–4267. [Google Scholar]
- Tarte, P. The Determination of Cation Co-cordination in Glasses by Infra-red Spectroscopy. In Physics of Non-Crystalline Solids; Prins, J.A., Ed.; North Holland: Amsterdam, The Netherlands, 1965; pp. 549–565. [Google Scholar]
- Galzada, M.L.; Delolmo, L. Sol-gel processing by inroganic route to obtain a TiO2-PbO xerogel as ceramic precursor. J. Non-Cryst. Solids 1990, 121, 413–416. [Google Scholar]
- Ben Amor, S.; Baud, G.; Besse, J.P.; Jacquet, M. Structural and optical properties of sputtered Titania films. Mater. Sci. Eng. 1997, 47, 110–118. [Google Scholar] [CrossRef]
- Castañeda, L.; Alonso, J.C.; Ortiz, A.; Andrade, E.; Saniger, J.M.; Bañuelos, J.G. Spray pyrolysis deposition and characterization of titanium oxide thin films. Mater. Chem. Phys. 2003, 77, 938–944. [Google Scholar] [CrossRef]
- Nam, S.; Kim, T.K.; Boo, J. Physical property and photo-catalytic activity of sulfur doped TiO2 catalysts responding to visible light. Catal. Today 2012, 185, 259–262. [Google Scholar]
- Yang, G.; Yan, Z.; Xiao, T. Low-temperature solvothermal synthesis of visible-light-responsive S-doped TiO2 nanocrystal. Appl. Surface Sci. 2012, 258, 4016–4022. [Google Scholar] [CrossRef]
- Ohno, T.; Mitsui, T.; Matsumura, M. Photocatalytic activity of S-doped TiO2 photocatalyst under visible light. Chem. Lett. 2003, 32, 364–365. [Google Scholar] [CrossRef]
- Nakahira, A.; Yokota, K.; Kubo, T.; Takahashi, M. Synthesis and characterization of S-doped TiO2 made by anodic oxidation of titanium in sulfuric acid. Chem. Lett. 2007, 36, 1318–1319. [Google Scholar] [CrossRef]
- Kim, J.S.; Itoh, K.; Murabayashi, M.; Kim, B.A. Pretreatment of the photocatalyst and the photocatalytic degradation of trichloroethylene in the gas-phase. Chemosphere 1999, 38, 2969–2978. [Google Scholar] [CrossRef]
- Amama, P.B.; Itoh, K.; Murabayashi, M. Photocatalytic oxidation of trichloroethylene in humidified atmosphere. J. Mol. Catal. A 2001, 176, 165–172. [Google Scholar] [CrossRef]
- Kang, M.; Lee, J.H.; Lee, S.H.; Chung, C.H.; Yoon, K.J.; Ogino, K.; Miyata, S.; Choung, S.J. Preparation of TiO2 film by the MOCVD method and analysis for decomposition of trichloroethylene using in situ FT-IR spectroscopy. J. Mol. Catal. A 2003, 193, 273–283. [Google Scholar] [CrossRef]
- Nakamura, R.; Tanaka, T.; Nakato, Y. Mechanism for visible light responses in anodic photocurrents at N-doped TiO2 film electrodes. J. Phys. Chem. B 2004, 108, 10617–10620. [Google Scholar] [CrossRef]
- Joung, S.K.; Amemiya, T.; Murabayashi, M.; Itoh, K. Mechanistic studies of the photocatalytic oxidation of trichloroethylene with visible-light-driven N-doped TiO2 photocatalysts. Chemistry 2006, 12, 5526–5534. [Google Scholar] [CrossRef]
- Jacoby, W.A.; Nimlos, M.R.; Blake, D.M.; Noble, R.D.; Koval, C.A. Products, intermediates, mass balances, and reaction pathways for the oxidation of trichloroethylene in air via heterogeneous photocatalysis. Environ. Sci. Technol. 1994, 28, 1661–1668. [Google Scholar] [CrossRef]
- Fan, J.; Yates, J.T., Jr. Mechanism of photooxidation of trichloroethylene on TiO2: Detection of intermediates by infrared spectroscopy. J. Am. Chem. Soc. 1996, 118, 4686–4692. [Google Scholar]
- Driessen, M.D.; Goodman, A.L.; Miller, T.M.; Zaharias, G.A.; Grassian, V.V. Gas-phase photooxidation of trichloroethylene on TiO2 and ZnO: Influence of trichloroethylene pressure, oxygen pressure, and the photocatalyst surface on the product distribution. J. Phys. Chem. B 1998, 102, 549–556. [Google Scholar]
- Kim, J.S.; Itoh, K.; Murabayashi, M. Photocatalytic degradation of trichloroethylene in the gas phase over TiO2 sol-gel films: Analysis of products. Chemosphere 1998, 36, 483–495. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Nishikiori, H.; Hayashibe, M.; Fujii, T. Visible Light-Photocatalytic Activity of Sulfate-Doped Titanium Dioxide Prepared by the Sol−Gel Method. Catalysts 2013, 3, 363-377. https://doi.org/10.3390/catal3020363
Nishikiori H, Hayashibe M, Fujii T. Visible Light-Photocatalytic Activity of Sulfate-Doped Titanium Dioxide Prepared by the Sol−Gel Method. Catalysts. 2013; 3(2):363-377. https://doi.org/10.3390/catal3020363
Chicago/Turabian StyleNishikiori, Hiromasa, Maki Hayashibe, and Tsuneo Fujii. 2013. "Visible Light-Photocatalytic Activity of Sulfate-Doped Titanium Dioxide Prepared by the Sol−Gel Method" Catalysts 3, no. 2: 363-377. https://doi.org/10.3390/catal3020363




