Carbon Xerogel Catalyst for NO Oxidation
Abstract
:1. Introduction
2. Experimental Section
2.1. Preparation of Carbon Xerogels
2.2. Chemical Modification of Carbon Xerogels
2.2.1. Oxidation in Liquid Phase with HNO3 5 M
2.2.2. Oxidation in Gas Phase with 5% O2
2.2.3. Treatment with Urea
2.3. Catalyst Characterization
2.3.1. Textural Characterization
2.3.2. Chemical Characterization
2.4. Oxidation of NO
3. Results and Discussion
3.1. Catalyst Characterization
3.1.1. Textural Characterization

| Sample | SBET [m2 g-1] | Vmicro [cm3 g-1] | Smeso [m2 g-1] |
|---|---|---|---|
| CX-5.3-500 °C | 583 | 0.14 | 230 |
| CX-5.3-700 °C | 707 | 0.20 | 226 |
| CX-5.3-700 °C-HNO3 | 634 | 0.18 | 184 |
| CX-5.3-700 °C-O2 | 628 | 0.23 | 136 |
| CX-5.3-700 °C-U | 714 | 0.19 | 266 |
| CX-5.3-900 °C | 661 | 0.19 | 189 |
| CX-6-500 °C | 367 | 0.07 | 156 |
| CX-6-700 °C | 625 | 0.19 | 167 |
| CX-6-700 °C-HNO3 | 677 | 0.17 | 277 |
| CX-6-700 °C-O2 | 1049 | 0.29 | 342 |
| CX-6-700 °C-U | 708 | 0.19 | 246 |
| CX-6-900 °C | 567 | 0.07 | 380 |
| CX-6.9-500 °C | 486 | 0.10 | 174 |
| CX-6.9-500°C-HNO3 | 641 | 0.05 | 514 |
| CX-6.9-500 °C-O2 | 618 | 0.08 | 236 |
| CX-6.9-500 °C-U | 526 | 0.10 | 200 |
| CX-6.9-700 °C | 149 | 0.01 | 110 |
| CX-6.9-900 °C | 87 | 0 | 87 |

3.1.2. Thermal Analysis (TG)

3.1.3. Elemental Analysis
| Sample | N [wt.%] | C [wt.%] | H [wt.%] | O [wt.%] |
|---|---|---|---|---|
| CX-5.3-500 °C | - | 79.9 | 2.5 | 17.6 |
| CX-5.3-700 °C | - | 92.4 | 1.1 | 6.5 |
| CX-5.3-700 °C-HNO3 | 0.3 | 89.5 | 1.2 | 9.0 |
| CX-5.3-700 °C-O2 | - | 90.8 | 1.3 | 7.9 |
| CX-5.3-700 °C-U | 0.4 | 94.5 | 1.6 | 3.5 |
| CX-5.3-900 °C | - | 95.0 | 0.4 | 4.6 |
| CX-6-500 °C | - | 83.9 | 3.1 | 13.0 |
| CX-6-700 °C | - | 90.2 | 2.8 | 7.0 |
| CX-6-700 °C-HNO3 | 0.4 | 87.2 | 0.4 | 12.0 |
| CX-6-700 °C-O2 | - | 82.7 | 1.3 | 16.0 |
| CX-6-700 °C-U | 0.7 | 91.2 | 0.3 | 7.8 |
| CX-6-900 °C | - | 94.9 | 0.5 | 4.6 |
| CX-6.9-500 °C | - | 82.6 | 2.4 | 15.0 |
| CX-6.9-500 °C-HNO3 | 0.2 | 80.9 | 0.9 | 18.0 |
| CX-6.9-500 °C-O2 | - | 77.7 | 3.3 | 19.0 |
| CX-6.9-500 °C-U | 0.3 | 89.6 | 1.5 | 8.6 |
| CX-6.9-700 °C | - | 98.5 | 1.4 | 0.10 |
| CX-6.9-900 °C | - | 98.9 | 1.0 | 0.10 |
3.1.4. TPD and pHpzc
| Sample | pHpzc | CO [μmol g-1] | CO2 [μmol g-1] | CO/CO2 |
|---|---|---|---|---|
| CX-5.3-500 °C | 7.0 | 2989.2 | 548.4 | 5.5 |
| CX-5.3-700 °C | 7.6 | 865.3 | 550.8 | 1.6 |
| CX-5.3-700 °C-HNO3 | 6.2 | 907.6 | 1714.4 | 0.5 |
| CX-5.3-700 °C-O2 | 7.1 | 3178.7 | 317.5 | 10.0 |
| CX-5.3-700 °C-U | 7.6 | 1003.6 | 122.6 | 8.2 |
| CX-5.3-900°C | 8.1 | 231.6 | 464.4 | 0.5 |
| CX-6-500 °C | 6.9 | 1137.6 | 912.0 | 1.2 |
| CX-6-700 °C | 6.9 | 614.4 | 446.4 | 1.4 |
| CX-6-700 °C-HNO3 | 6.2 | 1640.4 | 3465.6 | 0.5 |
| CX-6-700 °C-O2 | 7.2 | 2944.6 | 805.0 | 3.7 |
| CX-6-700 °C-U | 7.5 | 1843.2 | 653.5 | 2.8 |
| CX-6-900 °C | 6.9 | 583.2 | 184.3 | 3.2 |
| CX-6.9-500 °C | 7.0 | 1226.0 | 444.0 | 2.8 |
| CX-6.9-500 °C-HNO3 | 6.5 | 1353.6 | 5373.9 | 0.3 |
| CX-6.9-500 °C-O2 | 7.5 | 1482.0 | 376.2 | 3.9 |
| CX-6.9-500 °C-U | 7.5 | 1791.3 | 482.3 | 3.7 |
| CX-6.9-700 °C | 7.3 | 855.8 | 421.3 | 2.0 |
| CX-6.9-900 °C | 7.3 | 720.6 | 311.5 | 2.3 |
3.2. Catalytic Tests
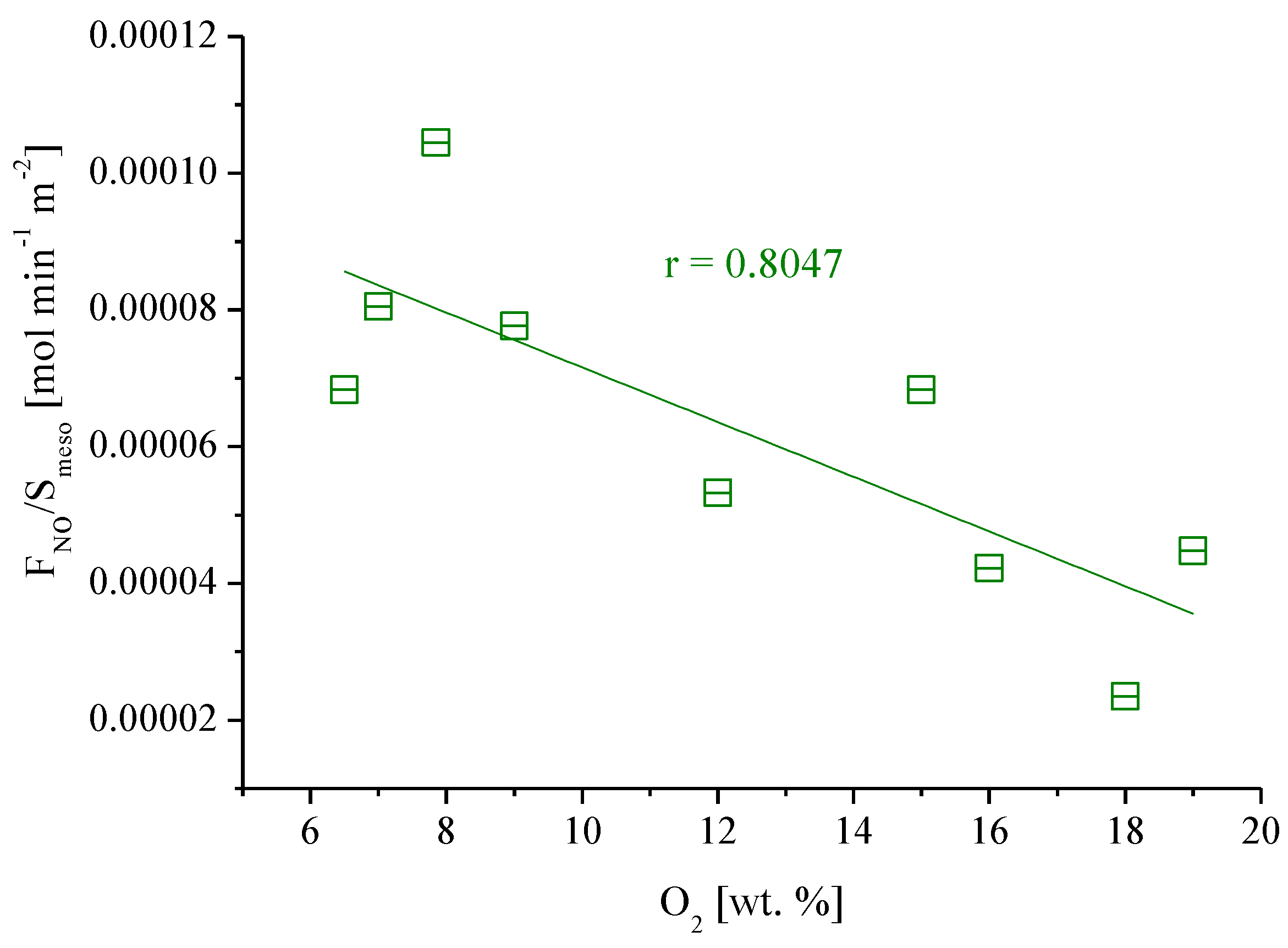
3.2.1. Influence of O2 Concentration on NO Oxidation


3.2.2. Influence of Surface Chemistry of the Carbon Xerogels on NO Oxidation
| Treatments | ||||
|---|---|---|---|---|
| Original | -HNO3 | -U | -O2 | |
| Sample | XNO [%] | |||
| CX-5.3-700 °C | 92 | 85 | 92 | 86 |
| CX-6-700 °C | 80 | 88 | 93 | 87 |
| CX-6.9-500 °C | 72 | 72 | 66 | 64 |

3.2.3. Influence of CO and CO2 on NO Oxidation
3.2.4. Stability of the Sample CX-5.3-900 °C for NO Oxidation

3.2.5. Influence of Reaction Temperature on NO Oxidation

4. Conclusions
Acknowledgements
Conflict of Interest
References
- Forzatti, P. Present status and perpectives in de-NOx SCR catalysis. Appl. Catal. A 2001, 222, 221–236. [Google Scholar] [CrossRef]
- Pârvulescu, V.I.; Grange, P.; Delmon, B. Catalytic removal of NO. Catal. Today 1998, 46, 233–316. [Google Scholar]
- Gómez-García, M.A.; Pitchon, V.; Kiennemann, A. Pollution by nitrogen oxides: an approach to NOx abatement by using sorbing catalytic materials. Environ. Int. 2005, 31, 445–467. [Google Scholar] [CrossRef]
- Skalska, K.; Miller, J.S.; Ledakowicz, S. Trends in NOx abatement: A review. Sci. Total Environ. 2010, 408, 3976–3989. [Google Scholar] [CrossRef]
- Barman, S.; Philip, L. Integrated System for the Treatment of Oxides of Nitrogen from Flue Gases. Environ. Sci. Tech. 2006, 40, 1035–1041. [Google Scholar] [CrossRef]
- Zawadzki, J.; Wiśniewski, M. Adsorption and decomposition of NO on carbon and carbon-supported catalysts. Carbon 2002, 40, 119–124. [Google Scholar] [CrossRef]
- Mochida, I.; Kisamori, S.; Hironaka, M.; Kawano, S.; Matsumura, Y.; Yoshikawa, M. Oxidation of NO into NO2 over Active Carbon Fibers. Energy Fuels 1994, 8, 1341–1344. [Google Scholar] [CrossRef]
- Sousa, J.P.S.; Pereira, M.F.R.; Figueiredo, J.L. Catalytic oxidation of NO to NO2 on N-doped activated carbons. Catal. Today 2011, 176, 383–387. [Google Scholar]
- Zhang, W.J.; Rabiei, S.; Bagreev, A.; Zhuang, M.S.; Rasouli, F. Study of NO adsorption on activated carbons. Appl. Catal. B 2008, 83, 63–71. [Google Scholar] [CrossRef]
- Wang, M.-X.; Huang, Z.-H.; Shimohara, T.; Kang, F.; Liang, K. NO removal by electrospun porous carbon nanofibers at room temperature. Chem. Eng. J. 2011, 170, 505–511. [Google Scholar] [CrossRef]
- Stanmore, B.R.; Tschamber, V.; Brilhac, J.F. Oxidation of carbon by NOx, with particular reference to NO2 and N2O. Fuel 2008, 87, 131–146. [Google Scholar] [CrossRef]
- Klose, W.; Rincón, S. Adsorption and reaction of NO on activated carbon in the presence of oxygen and water vapour. Fuel 2007, 86, 203–209. [Google Scholar] [CrossRef]
- Teng, H.; Hsu, Y.-F.; Tu, Y.-T. Reduction of NO with NH3 over carbon catalysts—the influence of carbon surface structures and the global kinetics. Appl. Catal. B 1999, 20, 145–154. [Google Scholar] [CrossRef]
- Mahata, N.; Silva, A.R.; Pereira, M.F.R.; Freire, C.; Castro, B.; Figueiredo, J.L. Anchoring of a [Mn(salen)Cl] complex onto mesoporous carbon xerogels. J. Colloid Interf. Sci. 2007, 311, 152–158. [Google Scholar] [CrossRef]
- Samant, P.V.; Gonçalves, F.; Freitas, M.M.A.; Pereira, M.F.R.; Figueiredo, J.L. Surface activation of a polymer based carbon. Carbon 2004, 42, 1321–1325. [Google Scholar] [CrossRef]
- Job, N.; Sabatier, F.; Pirard, J.-P.; Crine, M.; Léonard, A. Towards the production of carbon xerogel monoliths by optimizing convective drying conditions. Carbon 2006, 44, 2534–2542. [Google Scholar] [CrossRef]
- Maldonado-Hódar, F.J.; Moreno-Castilla, C.; Rivera-Utrilla, J. Synthesis, pore texture and surface acid–base character of TiO2/carbon composite xerogels and aerogels and their carbonized derivatives. Appl. Catal. A 2000, 203, 151–159. [Google Scholar] [CrossRef]
- ElKhatat, A.M.; Al-Muhtaseb, S.A. Advances in Tailoring Resorcinol-Formaldehyde Organic and Carbon Gels. Adv. Mater. 2011, 23, 2887–2903. [Google Scholar] [CrossRef]
- Al-Muhtaseb, S.A.; Ritter, J.A. Preparation and Properties of Resorcinol–Formaldehyde Organic and Carbon Gels. Adv. Mater. 2003, 15, 101–114. [Google Scholar] [CrossRef]
- Job, N.; Théry, A.; René, P.; Marien, J.; Kocon, J.L.; Rouzaud, J.-N.; Béguin, F.; Pirard, J.-P. Carbon aerogels, cryogels and xerogels: Influence of the drying method on the textural properties of porous carbon materials. Carbon 2005, 43, 2481–2494. [Google Scholar] [CrossRef]
- Job, N.; Pirard, R.; Marien, J.; Pirard, J.-P. Porous carbon xerogels with texture tailored by pH control during sol–gel process. Carbon 2004, 42, 619–628. [Google Scholar] [CrossRef]
- Léonard, A.; Job, N.; Blacher, S.; Pirard, J.-P.; Crine, M.; Jomaa, W. Suitability of convective air drying for the production of porous resorcinol–formaldehyde and carbon xerogels. Carbon 2005, 43, 1808–1811. [Google Scholar] [CrossRef]
- Zubizarreta, L.; Arenillas, A.; Pirard, J.-P.; Pis, J.J.; Job, N. Tailoring the textural properties of activated carbon xerogels by chemical activation with KOH. Micropor. Mesopor. Mat. 2008, 115, 480–490. [Google Scholar] [CrossRef]
- Job, N.; Heinrichs, B.; Ferauche, F.; Noville, F.; Marien, J.; Pirard, J.-P. Hydrodechlorination of 1,2-dichloroethane on Pd–Ag catalysts supported on tailored texture carbon xerogels. Catal. Today 2005, 102–103, 234–241. [Google Scholar]
- Job, N.; Heinrichs, B.; Lambert, S.; Pirard, J.-P.; Colomer, J.-F.; Vertruyen, B.; Marien, J. Carbon xerogels as catalyst supports: Study of mass transfer. AIChE J. 2006, 52, 2663–2676. [Google Scholar] [CrossRef]
- Jayne, D.; Zhang, Y.; Haji, S.; Erkey, C. Dynamics of removal of organosulfur compounds from diesel by adsorption on carbon aerogels for fuel cell applications. Int. J. Hydrog. Energy 2005, 30, 1287–1293. [Google Scholar] [CrossRef]
- Kang, K.Y.; Lee, B.I.; Lee, J.S. Hydrogen adsorption on nitrogen-doped carbon xerogels. Carbon 2009, 47, 1171–1180. [Google Scholar] [CrossRef]
- Frackowiak, E.; Béguin, F. Carbon materials for the electrochemical storage of energy in capacitors. Carbon 2001, 39, 937–950. [Google Scholar] [CrossRef]
- Fang, B.; Binder, L. A modified activated carbon aerogel for high-energy storage in electric double layer capacitors. J. Power Sources 2006, 163, 616–622. [Google Scholar] [CrossRef]
- Job, N.; Marie, J.; Lambert, S.; Berthon-Fabry, S.; Achard, P. Carbon xerogels as catalyst supports for PEM fuel cell cathode. Energy Convers. Manag. 2008, 49, 2461–2470. [Google Scholar] [CrossRef]
- Figueiredo, J.L.; Pereira, M.F.R.; Freitas, M.M.A.; Órfão, J.J.M. Modification of the surface chemistry of activated carbons. Carbon 1999, 37, 1379–1389. [Google Scholar] [CrossRef]
- Faria, P.C.C.; Órfão, J.J.M.; Pereira, M.F.R. Adsorption of anionic and cationic dyes on activated carbons with different surface chemistries. Water Res. 2004, 38, 2043–2052. [Google Scholar] [CrossRef]
- Alegre, C.; Gálvez, M.E.; Sebastián, D.; Moliner, R.; Lázaro, M.J. Influence of Synthesis pH on Textural Properties of Carbon Xerogels as Supports for Pt/CXs Catalysts for Direct Methanol Fuel Cells. Int. J. Electrochem. 2012. [Google Scholar] [CrossRef]
- Lin, C.; Ritter, J.A. Effect of synthesis pH on the structure of carbon xerogels. Carbon 1997, 35, 1271–1278. [Google Scholar] [CrossRef]
- Shimizu, T.; Sazawa, Y.; Adschiri, T.; Furusawa, T. Conversion of char-bound nitrogen to nitric oxide during combustion. Fuel 1992, 71, 361–365. [Google Scholar] [CrossRef]
- Illan-Goméz, M.J.; Linares-Solano, A.; de Lecea, C.S.M.; Calo, J.M. Nitrogen oxide (NO) reduction by activated carbons. 1. The role of carbon porosity and surface area. Energy Fuels 1993, 7, 146–154. [Google Scholar] [CrossRef]
- Jan, E.J. Formation and reduction of nitrogen oxides in fluidized-bed combustion. Fuel 1994, 73, 1398–1415. [Google Scholar] [CrossRef]
- Mochida, I.; Ogaki, M.; Fujitsu, H.; Komatsubara, Y.; Ida, S. Reduction of nitric oxide with activated PAN fibres. Fuel 1985, 64, 1054–1057. [Google Scholar] [CrossRef]
- Sousa, J.P.S.; Pereira, M.F.R.; Figueiredo, J.L. NO oxidation over nitrogen doped carbon xerogels. Appl. Catal. B 2012, 125, 398–408. [Google Scholar] [CrossRef]
- Boehm, H.P. Catalytic Properties of Nitrogen-Containing Carbons. In Carbon Materials for Catalysis; Serp, P., Figueiredo, J.L., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2009; Chapter 7. [Google Scholar]
- Stöhr, B.; Boehm, H.P.; Schlögl, R. Enhancement of the catalytic activity of activated carbons in oxidation reactions by thermal treatment with ammonia or hydrogen cyanide and observation of a superoxide species as a possible intermediate. Carbon 1991, 29, 707–720. [Google Scholar] [CrossRef]
- Seredych, M.; Hulicova-Jurcakova, D.; Lu, G.Q.; Bandosz, T.J. Surface functional groups of carbons and the effects of their chemical character, density and accessibility to ions on electrochemical performance. Carbon 2008, 46, 1475–1488. [Google Scholar] [CrossRef]
- Mangun, C.L.; Benak, K.R.; Economy, J.; Foster, K.L. Surface chemistry, pore sizes and adsorption properties of activated carbon fibers and precursors treated with ammonia. Carbon 2001, 39, 1809–1820. [Google Scholar] [CrossRef]
- Pels, J.R.; Kapteijn, F.; Moulijn, J.A.; Zhu, Q.; Thomas, K.M. Evolution of nitrogen functionalities in carbonaceous materials during pyrolysis. Carbon 1995, 33, 1641–1653. [Google Scholar] [CrossRef]
- Arrigo, R.; Hävecker, M.; Wrabetz, S.; Blume, R.; Lerch, M.; McGregor, J.; Parrot, E.P.J.; Zeitler, J.A.; Gladden, L.F.; Knop-Gericke, A.; et al. Tuning the Acid/Base Properties of Nanocarbons by Functionalization via Amination. J. Am. Chem. Soc. 2010, 33, 9616–9630. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sousa, J.P.S.; Pereira, M.F.R.; Figueiredo, J.L. Carbon Xerogel Catalyst for NO Oxidation. Catalysts 2012, 2, 447-465. https://doi.org/10.3390/catal2040447
Sousa JPS, Pereira MFR, Figueiredo JL. Carbon Xerogel Catalyst for NO Oxidation. Catalysts. 2012; 2(4):447-465. https://doi.org/10.3390/catal2040447
Chicago/Turabian StyleSousa, Juliana P. S., Manuel F. R. Pereira, and José L. Figueiredo. 2012. "Carbon Xerogel Catalyst for NO Oxidation" Catalysts 2, no. 4: 447-465. https://doi.org/10.3390/catal2040447







