Minimal Residual Disease as a Predictive Factor for Relapse after Allogeneic Hematopoietic Stem Cell Transplant in Adult Patients with Acute Myeloid Leukemia in First and Second Complete Remission
Abstract
:1. Introduction
2. Experimental Section
2.1. MRD Assessment
2.2. Statistical Analyses
3. Results
| Characteristics | Value | |||||
|---|---|---|---|---|---|---|
| CR1 | CR2 | Total (n/%) | ||||
| MRD+ (n/%) | MRD− (n/%) | MRD+ (n/%) | MRD− (n/%) | |||
| Patients (number, %) | 10 (100%) | 19 (100%) | 9 (100%) | 21 (100%) | 59 (100%) | |
| Sex | ||||||
| Male | 4 (40%) | 6 (31.6%) | 5 (55.5%) | 13 (90.5%) | 28 (47.5%) | |
| Female | 6 (60%) | 13 (68.4%) | 4 (44.4%) | 8 (38.1%) | 31 (52.5%) | |
| Age | ||||||
| Range | 28–62 | 20–61 | 29–65 | 30–65 | 20–65 | |
| Median | 51 | 44 | 48 | 43 | 48 | |
| Mean | 51.0 | 42.4 | 49.0 | 46.3 | 46.3 | |
| Initial cytogenetics | ||||||
| Favorable | / | / | / | 1 (4.8%) | 1 (1.7%) | |
| Intermediate | 4 (40%) | 7 (36.8%) | 5 (55.5%) | 17 (81%) | 33 (55.9%) | |
| Unfavorable | 6 (60%) | 10 (52.6%) | 2 (22.2%) | 1 (4.8%) | 19 (32.2%) | |
| Unknown | / | 2 (10.5%) | 2 (22.2%) | 2 (9.5%) | 6 (10.2%) | |
| Diagnosis | ||||||
| AML de novo | 4 (40%) | 13 (68.4%) | 7 (77.7%) | 17 (81%) | 41 (69.5%) | |
| Secondary AML | 6 (60%) | 6 (31.6%) | 2 (22.2%) | 4 (19%) | 18 (30.5%) | |
| Previous Stem Cell Transplant | 8 (13.6%) | |||||
| Autologous | / | / | 3 (33.3%) | 4 (19%) | 7 (11.9%) | |
| Allogeneic | / | / | / | 1 (4.8%) | 1 (1.7%) | |
| Disease status at transplantation | ||||||
| CR1 | 10 (100%) | 19 (100%) | / | / | 29 (49.1%) | |
| CR2 | / | / | 9 (100%) | 21 (100%) | 30 (50.8%) | |
| ECOG status | ||||||
| 0 | 3 (30%) | 10 (52.6%) | 1 (11.1%) | 4 (19%) | 18 (30.5%) | |
| 1 | 1 (10%) | 2 (10.5%) | 3 (33.3%) | 3 (14.3%) | 9 (15.2%) | |
| 2 | 1 (10%) | / | 1 (11.1%) | 3 (14.3%) | 5 (%) | |
| 3–4 | / | 1 (5.3%) | 1 (11.1%) | / | 2 (%) | |
| unknown | 5 (50%) | 6 (31.6%) | 3 (33.3%) | 11 (52.4%) | 25 (%) | |
| MRD defined by | ||||||
| Flow cytometry | 3 (30%) | / | 6 (66.6%) | / | 9 (15.2%) | |
| Cytogenetics | 4 (40%) | / | 4 (44.4%) | / | 8 (13.6%) | |
| FISH | 5 (50%) | / | 2 (22.2%) | / | 7 (11.9%) | |
| Leukemia cutis | / | / | 1 (11.1%) | / | 1 (1.7%) | |
| Type of transplant | ||||||
| Related | 6 (60%) | 10 (52.6%) | 6 (66.6%) | 12 (57.1%) | 34 (57.6%) | |
| Unrelated | 4 (40%) | 9 (47.4%) | 3 (33.3%) | 9 (42.9%) | 25 (42.4%) | |
| Graft type | ||||||
| PBSC | 6 (60%) | 11 (57.9%) | 4 (44.4%) | 16 (76.2%) | 37 (62.7%) | |
| Bone marrow | 1 (20%) | 3 (15.8%) | 3 (33.3%) | 3 (14.3%) | 10 (16.9%) | |
| Cord blood | 3 (30%) | 5 (26.3%) | 2 (22.2%) | 2 (9.5%) | 12 (20.3%) | |
| Donor-recipient HLA match | ||||||
| Matched (8/8, 6/6) | 7 (70%) | 14 (73.7%) | 7 (77.7%) | 18 (85.7%) | 46 (78%) | |
| Mismatched (7/8, 5/6) | 2 (20%) | 1 (5.3%) | 2 (22.2%) | 2 (9.5%) | 7 (11.9%) | |
| Mismatched (4/6) | 1 (10%) | 4 (21%) | / | 1 (4.8%) | 6 (10.2%) | |
| Condition intensity | ||||||
| Myeloablative | 9 (90%) | 19 (100%) | 7 (77.7%) | 17 (81%) | 52 (88.2%) | |
| Reduced intensity | 1 (10%) | / | 2 (22.2%) | 4 (19%) | 7 (11.9%) | |
| Conditioning protocol | ||||||
| Non TBI | 5 (50%) | 11 (57.9%) | 4 (44.4%) | 12 (57.2%) | 32 (54.3%) | |
| TBI | 5 (50%) | 8 (42.1%) | 5 (55.5%) | 9 (42.9%) | 27 (45.7%) | |
| Relapse | ||||||
| Yes | 5 (50%) | 2 (10.5%) | 6 (66.6%) | 4 (19%) | 17 (28.8%) | |
| No | 5 (50%) | 17 (89.5%) | 3 (33.3%) | 17 (81%) | 42 (71.2%) | |
| Present status | ||||||
| Alive | 2 (20%) | 12 (63.2%) | 2 (22.2%) | 11 (52.4%) | 27 (45.7%) | |
| Diseased | 8 (80%) | 7 (36.8%) | 7 (77.7%) | 10 (47.6%) | 32 (54.3%) | |
| Pt.# | Age | Cytogenetics (initial) | Dg. | Prior SCT | Rem. status | Flow cytometry | Cytogenetics | FISH | Type of transplant | Conditioning protocol/intensity | Relapse | aGVHD | cGVHD | TRM | Cause of death | OS |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 10 | 61 | Normal | AML | No | CR1 | 2–3% myeloblasts | ck | / | R 8/8 PBSC | NonTBI/Reduced intensity | +85 | 0 | Yes | No | L, G, OF | +138 |
| 11 | 62 | 46, xy, del(7)(p15)[23]/47,xy,+8[7] | sAML | No | CR1 | 1% coexpressing CD34+/CD117+mb | 47, xy,+8 | Abn. signal pattern, +8 | R 8/8 PBSC | TBI/Myeloablative | +102 | 4 | No | No | L | +196 |
| 12 | 45 | normal | AML | No | CR2 | 3.5% abn mb | 46, xx | Normal signal pattern | R 8/8 PBSC | TBI/Myeloablative | +210 | 1 | Yes | No | L, G | +299 |
| 14 | 48 | normal; FISHdel16p13 | AML | Auto SCT | CR2 | 4% myeloblasts | 46, xy, del16 (q23,q23) | Abn. signal pattern, del 16p | R 8/8 PBSC | NonTBI/Myeloablative | +88 | 4 | No | No | L, G | +96 |
| 21 | 43 | normal | AML | No | CR2 | 2% mb, 25–30% monocytes | 46, xy | / | R 8/8 PBSC | NonTBI/Reduced intensity | +245 | 1 | No | No | L | +370 |
| 22 | 50 | normal | AML | No | CR1 | 15–20% monocytes | 46, xy | / | R 8/8 PBSC | TBI/Myeloablative | No | 1 | No | +82 | I | +82 |
| 25 | 49 | normal | AML | Auto SCT | CR2 | <2% myeloblasts | ck | / | U 6/6 CB | NonTBI/Myeloablative | No | 0 | No | Alive | Alive | >+1512 |
| 28 | 46 | t(11q23, 17q25)/t(11,17) | sAML | No | CR1 | 1% myeloblasts | 46, xx | MLL gene rearrangement 11q23 | R 8/8 BM | TBI/Myeloablative | +260 | 3 | No | Alive | Alive | >+370 |
| 31 | 29 | unknown | AML | No | CR2 | 1% abn mb | 46, xy | / | R 8/8 PBSC | NonTBI/Myeloablative | +42 | 0 | No | No | L | +98 |
| 32 | 56 | ck | sAML | No | CR2 | / | ck | Abn. signal pattern, +9/del7q | R 8/8 BM | NonTBI/Reduced intensity | +110 | 1 | No | No | L | +126 |
| 35 | 61 | unknown | AML | Auto SCT | CR2 | 3% abn monocytic cells | 46, xx | / | U 7/8 BM | NonTBI/Reduced intensity | +179 | 2 | No | No | L | +184 |
| 39 | 45 | normal | AML | No | CR2 | 5% abn mb 10% CD34+/CD117+mb | 46, xx | / | U 5/6 CB | TBI/Myeloablative | No | 3 | No | Alive | Alive | >+1051 |
| 43 | 48 | ck | sAML | No | CR1 | 1% mb | ck | Abn. signal pattern 5q−, 7q− | U 4/6 CB | NonTBI/Myeloablative | +601 | 1 | No | No | L | +662 |
| 46 | 28 | del7q− | AML | No | CR1 | 10% CD34+/CD117+mb | 46, xx | Normal signal pattern | U 5/6CB | NonTBI/Reduced intensity | No | 0 | No | No | L | +269 |
| 50 | 41 | del5q,7q | sAML | No | CR1 | 4% abn.mb | / | / | U 8/8 PBSC | TBI/Myeloablative | +114 | 2 | No | No | L | +156 |
| 52 | 61 | normal | sAML | No | CR1 | Leukemia cutis | 46, xy | / | R 8/8 PBSC | TBI/Myeloablative | No | 4 | No | +86 | H, G | +86 |
| 53 | 52 | 47, xx,+8 | AML | No | CR1 | <2% mb | 46, xx | Abn. signal pattern, +8q | U 5/6 CB | TBI/Myeloablative | No | 2 | No | Alive | Alive | >+638 |
| 54 | 61 | FISH del7q | sAML | No | CR1 | 4% mb | 46, xy,+1, der (1,7) (q10;p10) | Abn. signal pattern 7q, +21q | R 8/8 PBSC | TBI/Myeloablative | No | 4 | No | +78 | I, G, OF | +78 |
| 58 | 65 | del20q | sAML | No | CR2 | 4% abn. mb | 46, xy, del (20) (q11.2q13.3) | / | U 8/8 BM | NonTBI/Myeloablative | No | 0 | Yes | +385 | G, OF | +385 |
3.1. Overall Survival
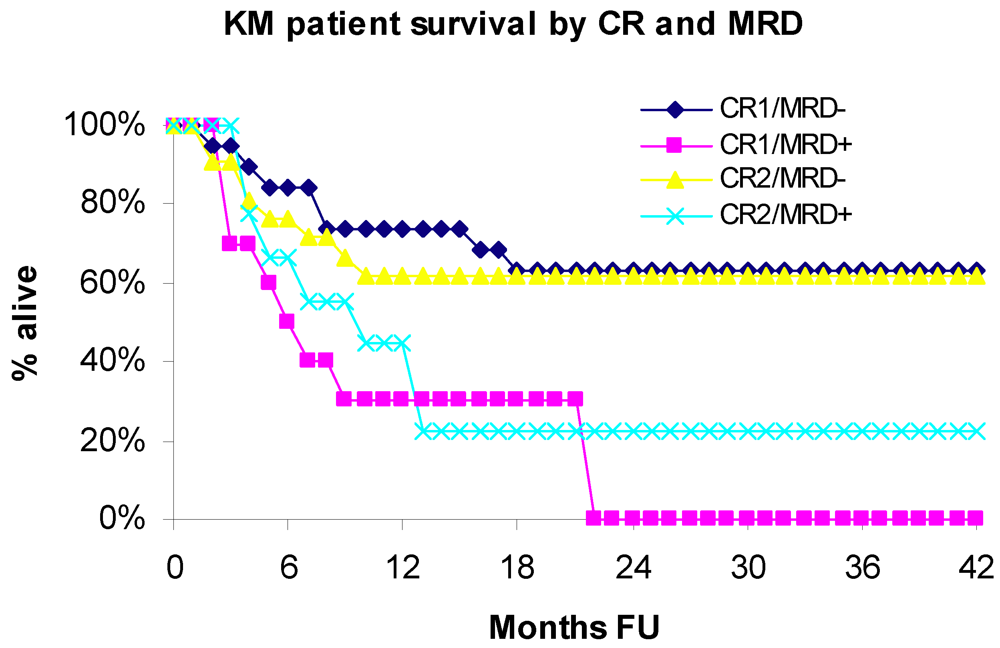
3.2. Leukemia Free Survival
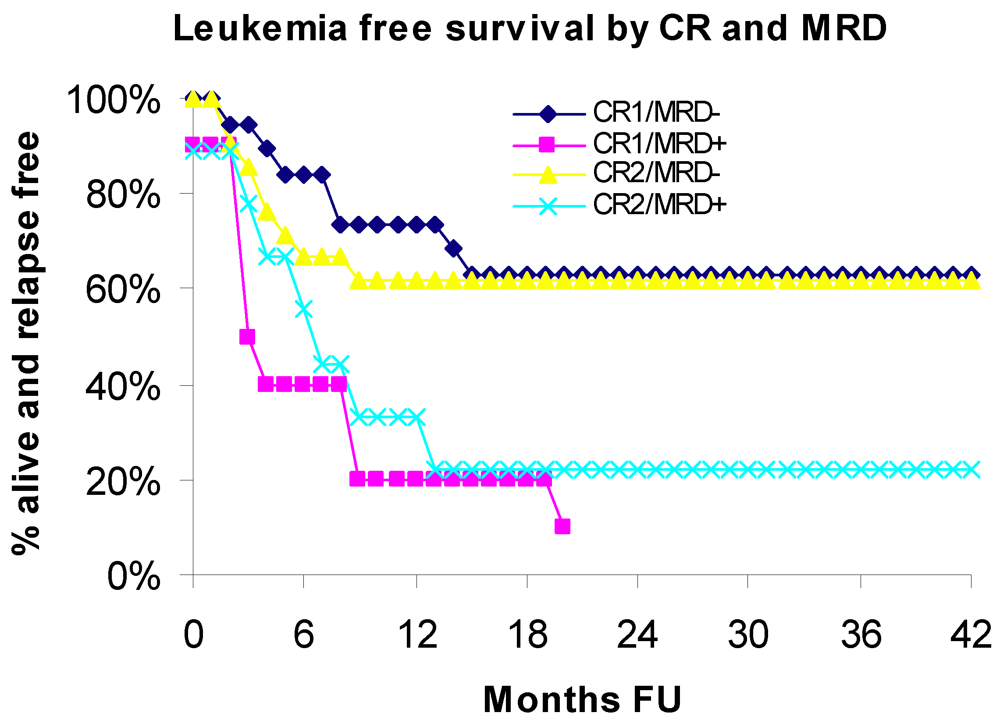
3.3. Relapse
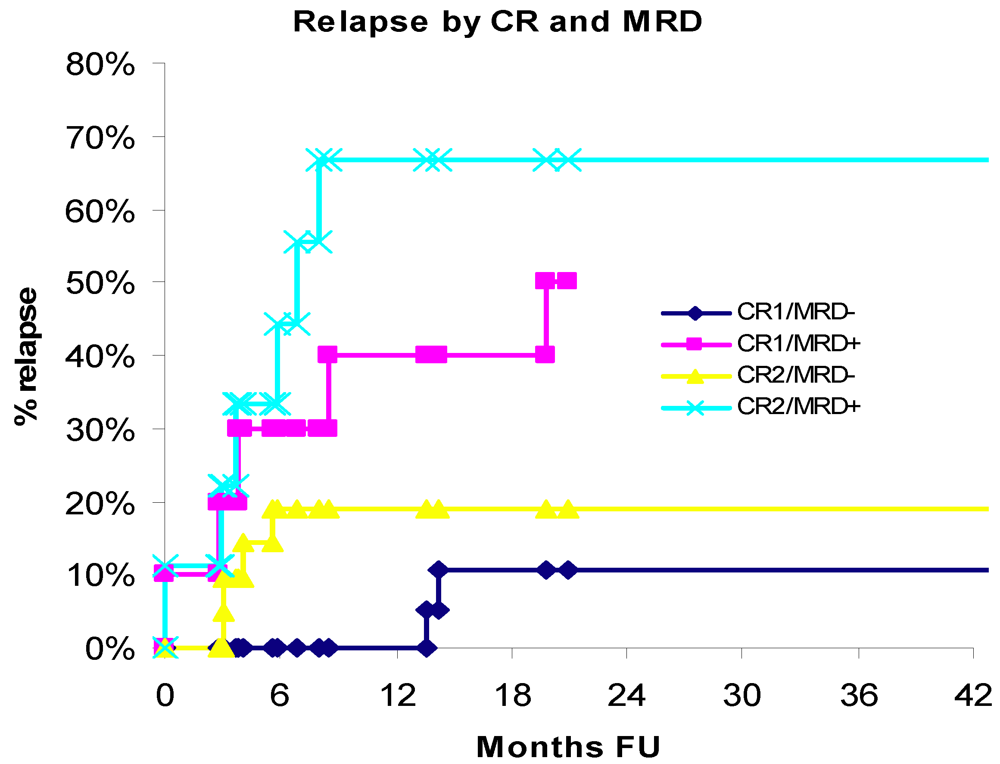
3.4. Treatment-Related Mortality

| MRD-negative | MRD-positive | ||||
| (n = 40) | (67.8%) | (n = 19) | (32.2%) | ||
| number | % | number | % | p-value | |
| CR | |||||
| 1 | 19 | 47.5 | 10 | 52.6 | 0.7126 |
| 2 | 21 | 52.5 | 9 | 47.4 | |
| Type/source of transplant | |||||
| Related, PBSC | 22 | 55.0 | 9 | 47.4 | 0.0707 |
| Related, BM | 0 | 0.0 | 3 | 15.8 | |
| Unrelated, PBSC | 5 | 12.5 | 1 | 5.3 | |
| Unrelated, BM | 6 | 15.0 | 1 | 5.3 | |
| Unrelated, CB | 7 | 17.5 | 5 | 26.3 | |
| HLA | |||||
| Matched (8/8, 6/6) | 32 | 80.0 | 14 | 73.7 | 0.7282 |
| Mismatched (7/8, 5/6) | 3 | 7.5 | 4 | 21.1 | |
| Mismatched (4/6) | 5 | 12.5 | 1 | 5.3 | |
| Initial cytogenetics | |||||
| Favorable | 1 | 2.5 | 0 | 0.0 | 0.2145 |
| Intermediate | 24 | 60.0 | 9 | 47.4 | |
| Unfavorable | 11 | 27.5 | 8 | 42.1 | |
| Unknown | 4 | 10.0 | 2 | 10.5 | |
| Previous disease/HSCT | |||||
| AML de novo | 27 | 67.5 | 8 | 42.1 | 0.1016 |
| Secondary AML | 8 | 20.0 | 8 | 42.1 | |
| Prior HSCT | 5 | 12.5 | 3 | 15.8 | |
| Conditioning protocol | |||||
| Non TBI | 23 | 57.5 | 9 | 47.4 | 0.4655 |
| TBI | 17 | 42.5 | 10 | 52.6 | |
| Conditioning intensity | |||||
| Myeleoblative | 36 | 90.0 | 16 | 84.2 | 0.6702 |
| Reduced intensity | 4 | 10.0 | 3 | 15.8 | |
| Mean | SD | Mean | SD | ||
| Age | 44.45 | 11.8 | 50.05 | 10.6 | 0.0836 |
| Factor | Unadjusted | |||
|---|---|---|---|---|
| Hazard Ratio | HRLowerCL | HRUpperCL | p-value | |
| MRD | 2.97 | 1.44 | 6.12 | 0.0032 |
| CR: 2 vs. 1 | 1.08 | 0.54 | 2.17 | 0.8277 |
| Initial cytogenetics | ||||
| Unknown vs. favorable/intermediate | 0.97 | 0.28 | 3.35 | 0.9655 |
| Unfavorable vs. favorable/intermediate | 1.78 | 0.85 | 3.72 | 0.1238 |
| Previous disease/prior HSCT | ||||
| Secondary vs. de novo AML | 1.52 | 0.71 | 3.22 | 0.2782 |
| Previous SCT vs. de novo AML | 0.71 | 0.21 | 2.42 | 0.5813 |
| Age (per year) | 1.03 | 1.00 | 1.07 | 0.0502 |
| Transplant type, source and HLA | ||||
| Type: unrelated vs. related | 0.49 | 0.23 | 1.04 | 0.0638 |
| Source: CB vs. PBSC and BM | 0.19 | 0.04 | 0.79 | 0.0225 |
| HLA: mismatched vs. matched | 0.27 | 0.08 | 0.89 | 0.0310 |
| Conditioning protocol | ||||
| TBI vs. non TBI | 0.83 | 0.41 | 1.67 | 0.5981 |
| Conditioning intensity | ||||
| Reduced intensity vs. myeloablative | 1.07 | 0.37 | 3.08 | 0.9018 |
4. Discussion
5. Conclusions
Conflict of Interest
References
- Verneris, M.R.; Burke, M.J. Novel approaches to prevent leukemia relapse following allogeneic hematopoietic cell transplantation. Curr. Hematol. Malig. Rep. 2010, 5, 157–162. [Google Scholar] [CrossRef]
- Gupta, V.; Tallman, S.; Weisdorf, D. Allogeneic hematopoietic cell transplantation for adults with acute myeloid leukemia: Myths, controversies, and unknowns. Blood 2001, 117, 2307–2318. [Google Scholar]
- Döhner, H.; Estey, E.H.; Amadori, S.; Appelbaum, F.R.; Büchner, T.; Burnett, A.K.; Dombret, H.; Fenaux, P.; Grimwade, D.; Larson, R.A.; et al. Diagnosis and management of acute myeloid leukemia in adults: Recommendations from an international expert panel, on behalf of the European LeukemiaNet. Blood 2010, 115, 453–474. [Google Scholar]
- Kern, W.; Bacher, U.; Haferlach, C.; Schnittger, S.; Haferlach, T. The role of multiparameter flow cytometry for disease monitoring in AML. Best Pract. Res. Clin. Hematol. 2010, 23, 379–390. [Google Scholar] [CrossRef]
- Freeman, S.D.; Jovanovic, J.V.; Grimwade, D. Development of minimal residual disease-directed therapy in acute myeloid leukemia. Semin. Oncol. 2008, 35, 388–400. [Google Scholar] [CrossRef]
- Shook, D.; Coustan-Smith, E.; Ribeiro, R.C.; Rubnitz, J.E.; Campana, D. Minimal residual disease quantitation in acute myeloid leukemia. Clin. Lymphoma Myeloma 2009, 9, S281–S285. [Google Scholar] [CrossRef]
- Wood, B.L. Myeloid malignancies: Myelodysplastic syndromes, myeloproliferative disorders, and acute myeloid leukemia. Clin. Lab. Med. 2007, 27, 551–575. [Google Scholar] [CrossRef]
- Liu Yin, J.A.; Grimwade, D. Minimal residual disease evaluation in acute myeloid leukemia. Lancet 2002, 360, 160–162. [Google Scholar]
- Liu Yin, J.A. Minimal residual disease in acute myeloid leukaemia. Best Prac. Res. Clin. Haematol. 2002, 15, 119–135. [Google Scholar] [CrossRef]
- Roboz, G.J.; Guzman, M. Acute myeloid leukemia stem cells: Seek and destroy. Expert Rev. Hematol. 2009, 2, 663–672. [Google Scholar] [CrossRef]
- Craig, F.E.; Foon, K.A. Flow cytometric immunophenotyping for hematologic neoplasms. Blood 2008, 111, 3941–3967. [Google Scholar] [CrossRef]
- Dohner, H.; Stilgenbauer, S.; Konstanze, F.; Schroder, M.; Bentz, M.; Lichter, P. Diagnosis and monitoring of chromosome aberrations in hematological malignancies by fluorescence in situ hybridization. Stem Cells 1995, 13, 76–82. [Google Scholar] [CrossRef]
- Smith, M.L.; Hills, R.K.; Grimwade, D. Independent prognostic variables in acute myeloid leukemia. Blood Rev. 2011, 25, 39–51. [Google Scholar] [CrossRef]
- Schiller, G.; Lee, M.; Paquete, R.; Sawyers, C.; Khoubian, F.; Territo, M. Transplantation of autologous peripheral blood progenitor cells procured after high-dose cytarabine-based consolidation chemotherapy for adults with secondary acute myelogenous leukemia in first remission. Leuk. Lymphoma 1999, 33, 475–484. [Google Scholar]
- Hurley, C.K.; Baxter Lowe, L.A.; Logan, B.; Karanes, C.; Anasetti, C.; Weisdorf, D.; Confer, D.L. National Marrow Donor Program HLA-matching guidelines for unrelated marrow transplants. Biol. Blood Marrow Transplant. 2003, 9, 610–615. [Google Scholar] [CrossRef]
- Walter, R.B.; Gooley, T.A.; Wood, B.L.; Milano, F.; Fang, M.; Sorror, M.L.; Estey, E.H.; Salter, A.I.; Lansverk, E.; Chien, J.W.; et al. Impact of pretransplantation minimal residual disease, as detected by multiparametric flow cytometry, on outcome of myeloablative hematopoietic cell transplantation for acute myeloid leukemia. J. Clin. Oncol. 2011, 29, 1190–1197. [Google Scholar]
- Marcucci, G.; Mrózek, K.; Ruppert, A.S.; Archer, K.J.; Pettenati, M.J.; Heerema, N.A.; Carroll, A.J.; Koduru, P.R.; Kolitz, J.E.; Sterling, L.J.; et al. Abnormal cytogenetics at date of morphologic complete remission predicts short overall and disease-free survival, and higher relapse rate in adult acute myeloid leukemia: Results from cancer and leukemia group B study 8461. J. Clin. Oncol. 2004, 22, 2410–2418. [Google Scholar]
- Fröhling, S.; Skelin, S.; Liebisch, C.; Scholl, C.; Schlenk, R.F.; Döhner, H.; Döhner, K. Acute Myeloid Leukemia Study Group, Ulm. Comparison of cytogenetic and molecular cytogenetic detection of chromosome abnormalities in 240 consecutive adult patients with acute myeloid leukemia. J. Clin. Oncol. 2002, 20, 2480–2485. [Google Scholar]
- Gallo, J.H.; Robson, L.G.; Watson, N.W.; Sharma, P.; Smith, A. Comparison of metaphase and interphase FISH monitoring of minimal residual disease with MLL gene probe: Case study of AML with t(9;11). Ann. Genet. 1999, 42, 109–112. [Google Scholar]
- Baer, M.R.; Stewart, C.C.; Dodge, R.K.; Leget, G.; Sulé, N.; Mrózek, K.; Schiffer, C.A.; Powell, B.L.; Kolitz, J.E.; Moore, J.O.; et al. High frequency of immunophenotype changes in acute myeloid leukemia at relapse: Implications for residual disease detection (Cancer and Leukemia Group B Study 8361). Blood 2001, 97, 3574–3580. [Google Scholar] [CrossRef]
- Stone, R.M. Should the presence of minimal residual disease (MRD) in morphologic complete remission alters post-remission strategy in AML? Best Pract. Res. Hematol. 2011, 24, 509–514. [Google Scholar]
- Kern, W.; Voskova, D.; Schoch, C.; Schnittger, S.; Hiddemann, W.; Haferlach, T. Prognostic impact of early response to induction therapy as assessed by multiparameter flow cytometry in acute myeloid leukemia. Haematologica 2004, 89, 528–540. [Google Scholar]
- Buccisano, F.; Maurillo, L.; Spagnoli, A.; del Principe, M.I.; Ceresoli, E.; Lo Coco, F.; Arcese, W.; Amadori, S.; Venditti, A. Monitoring of minimal residual disease in acute myeloid leukemia. Curr. Opin. Oncol. 2009, 21, 582–588. [Google Scholar] [CrossRef]
- Perea, G.; Lasa, A.; Aventín, A.; Domingo, A.; Villamor, N.; Queipo de Llano, M.P.; Llorente, A.; Juncà, J.; Palacios, C.; Fernández, C.; et al. Grupo Cooperativo para el Estudio y Tratamiento de las Leucemias Agudas y Miel. Prognostic value of minimal residual disease (MRD) in acute myeloid leukemia (AML) with favorable cytogenetics [t(8;21) and inv(16)]. Leukemia 2006, 20, 87–94. [Google Scholar]
- Díez-Campelo, M.; Pérez-Simón, J.A.; Pérez, J.; Alcoceba, M.; Richtmon, J.; Vidriales, B.; San Miguel, J. Minimal residual disease monitoring after allogeneic transplantation may help to individualize post-transplant therapeutic strategies in acute myeloid malignancies. Am. J. Hematol. 2009, 84, 149–152. [Google Scholar] [CrossRef]
- Venditti, A.; Maurillo, L.; Buccisano, F.; del Poeta, G.; Mazzone, C.; Tamburini, A.; del Principe, M.I.; Consalvo, M.I.; de Fabritiis, P.; Cudillo, L.; et al. Pretransplant minimal residual disease level predicts clinical outcome in patients with acute myeloid leukemia receiving high-dose chemotherapy and autologous stem cell transplantation. Leukemia 2003, 17, 2178–2182. [Google Scholar]
- Maurillo, L.; Buccisano, F.; del Principe, M.I.; del Poeta, G.; Spagnoli, A.; Panetta, P.; Ammatuna, E.; Neri, B.; Ottaviani, L.; et al. Toward optimization of postremission therapy for residual disease-positive patients with acute myeloid leukemia. J. Clin. Oncol. 2008, 26, 4944–4951. [Google Scholar]
- Hokland, P.; Ommen, H.B. Towards individualized follow-up in adult acute myeloid leukemia in remission. Blood 2011, 117, 2577–2584. [Google Scholar]
- Shaw, B.E.; Russell, N.H. Treatment options for the management of acute leukemia relapsing following an allogeneic transplant. Bone Marrow Transplant. 2008, 41, 495–503. [Google Scholar] [CrossRef]
- Oran, B.; de Lima, M. Prevention and treatment of acute myeloid leukemia relapse after allogeneic stem cell transplantation. Curr. Opin. Hematol. 2011, 18, 388–394. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Grubovikj, R.M.; Alavi, A.; Koppel, A.; Territo, M.; Schiller, G.J. Minimal Residual Disease as a Predictive Factor for Relapse after Allogeneic Hematopoietic Stem Cell Transplant in Adult Patients with Acute Myeloid Leukemia in First and Second Complete Remission. Cancers 2012, 4, 601-617. https://doi.org/10.3390/cancers4020601
Grubovikj RM, Alavi A, Koppel A, Territo M, Schiller GJ. Minimal Residual Disease as a Predictive Factor for Relapse after Allogeneic Hematopoietic Stem Cell Transplant in Adult Patients with Acute Myeloid Leukemia in First and Second Complete Remission. Cancers. 2012; 4(2):601-617. https://doi.org/10.3390/cancers4020601
Chicago/Turabian StyleGrubovikj, Rada M., Asif Alavi, Ahrin Koppel, Mary Territo, and Gary J. Schiller. 2012. "Minimal Residual Disease as a Predictive Factor for Relapse after Allogeneic Hematopoietic Stem Cell Transplant in Adult Patients with Acute Myeloid Leukemia in First and Second Complete Remission" Cancers 4, no. 2: 601-617. https://doi.org/10.3390/cancers4020601





