Low PD-L1 Expression Strongly Correlates with Local Recurrence in Epstein-Barr Virus-Positive Nasopharyngeal Carcinoma after Radiation-Based Therapy
Abstract
:1. Introduction
2. Results
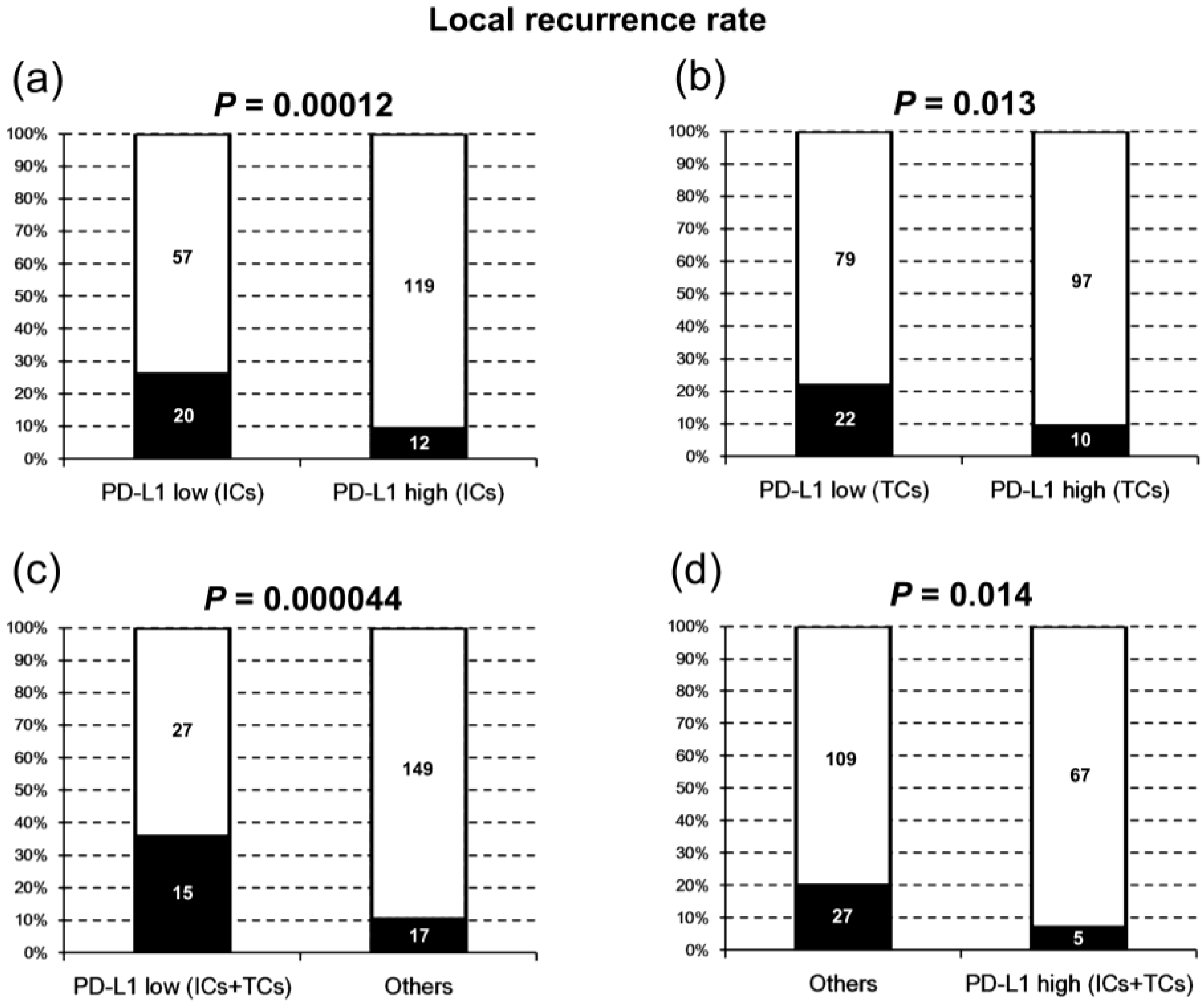
2.1. PD-L1 Expression and Clinicopathologic Characteristics
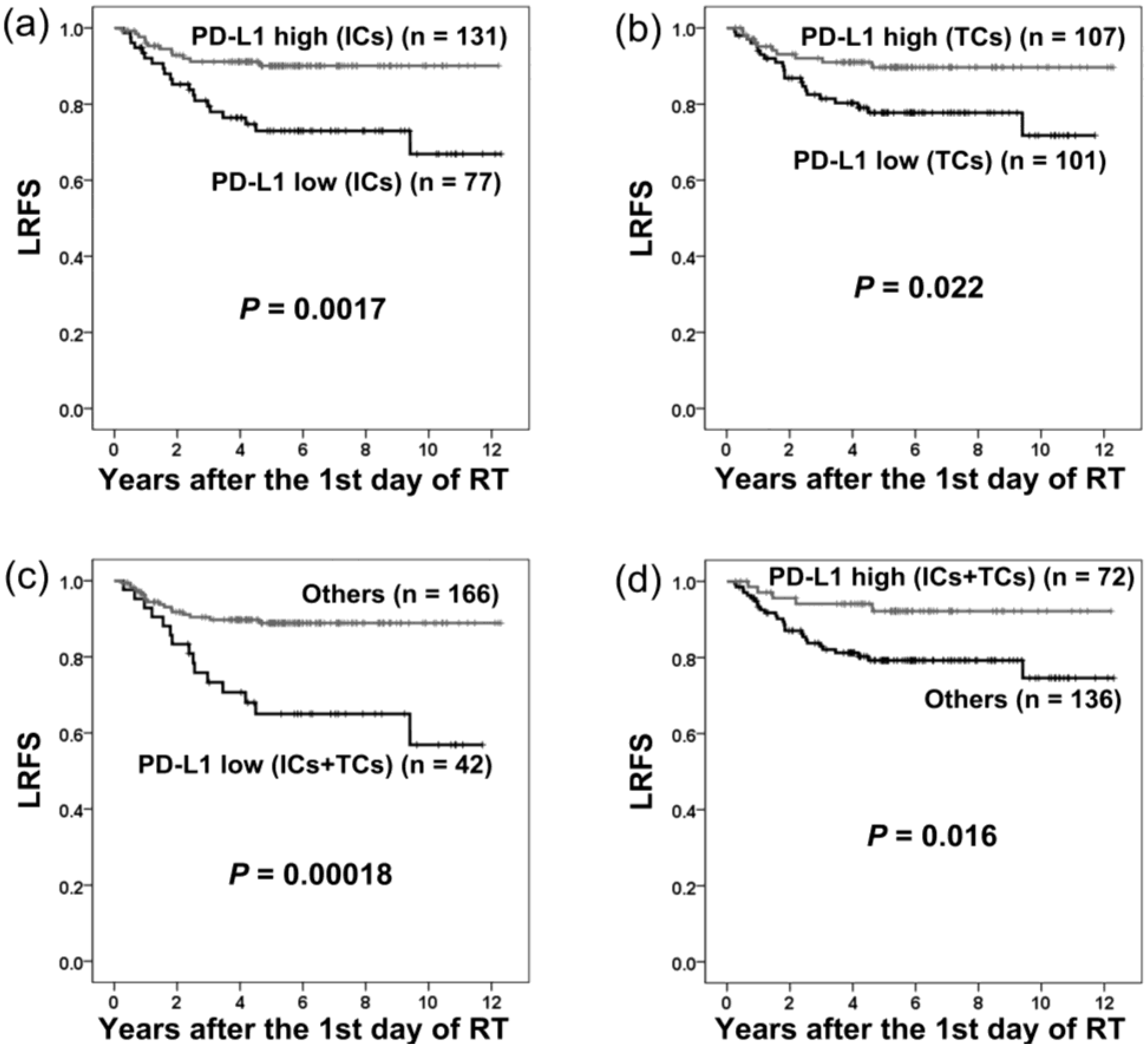
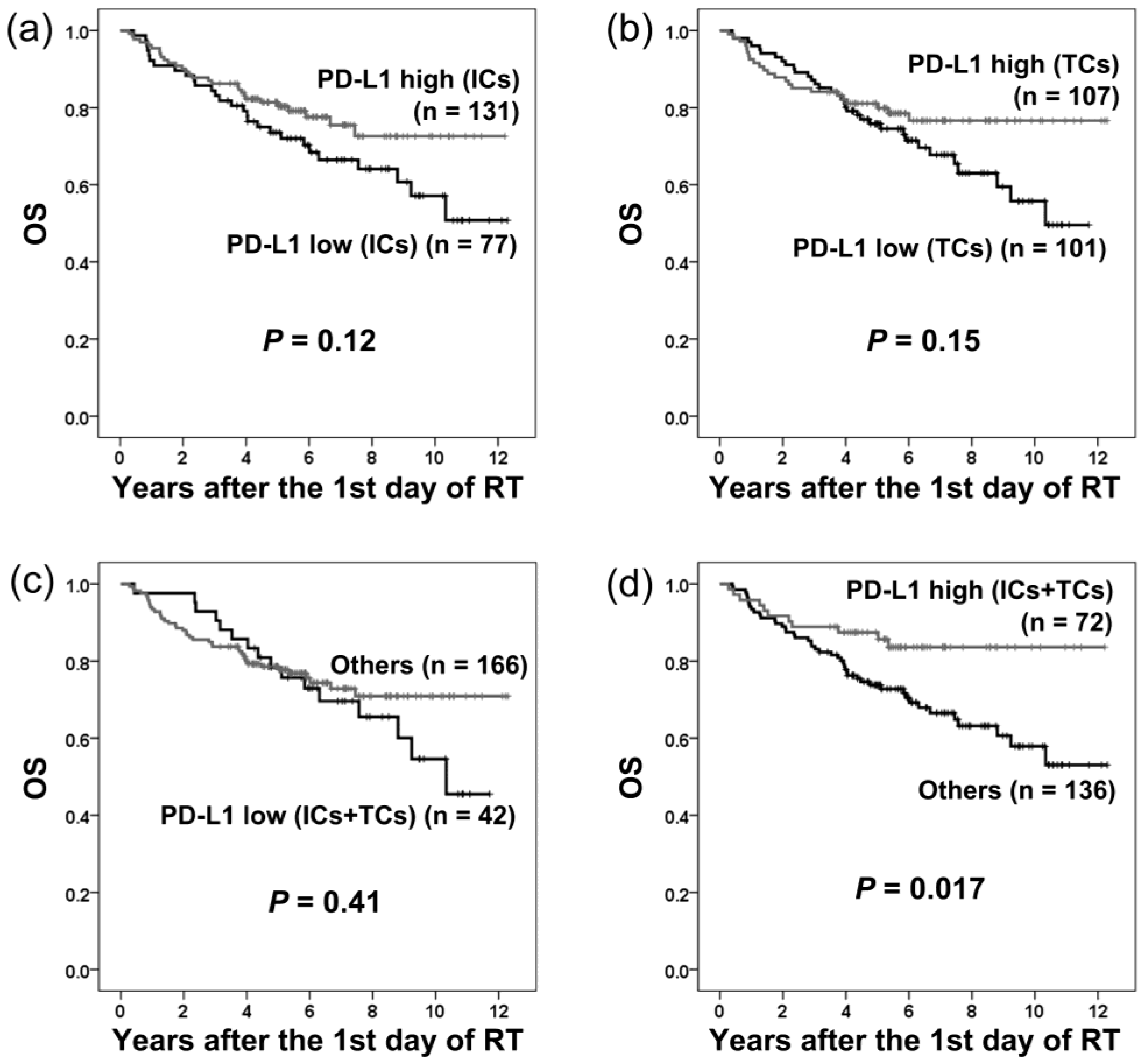
2.2. PD-L1 Expression and Local Recurrence-Free Survival
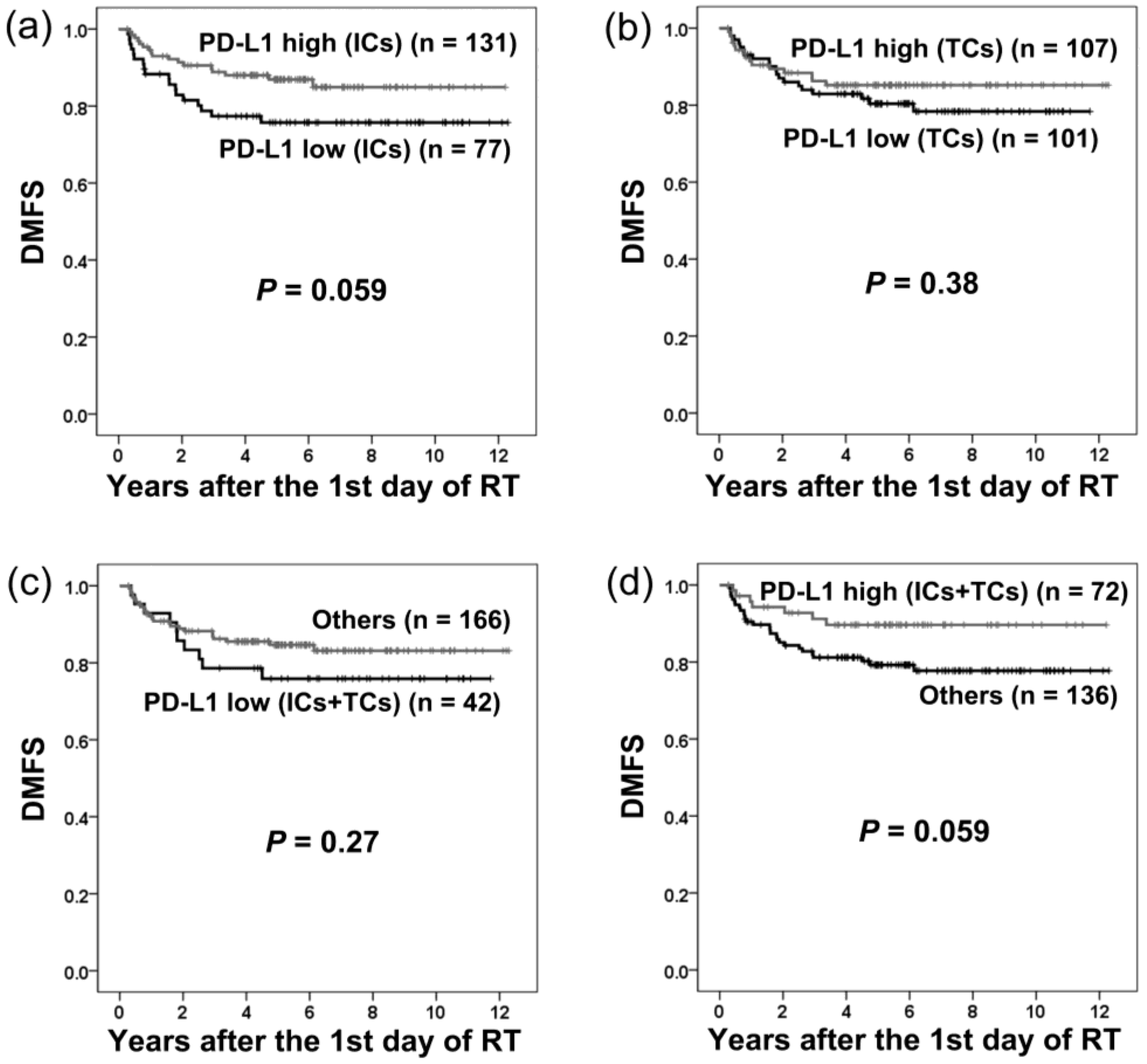
2.3. PD-L1 Expression and Distant Metastasis-Free Survival
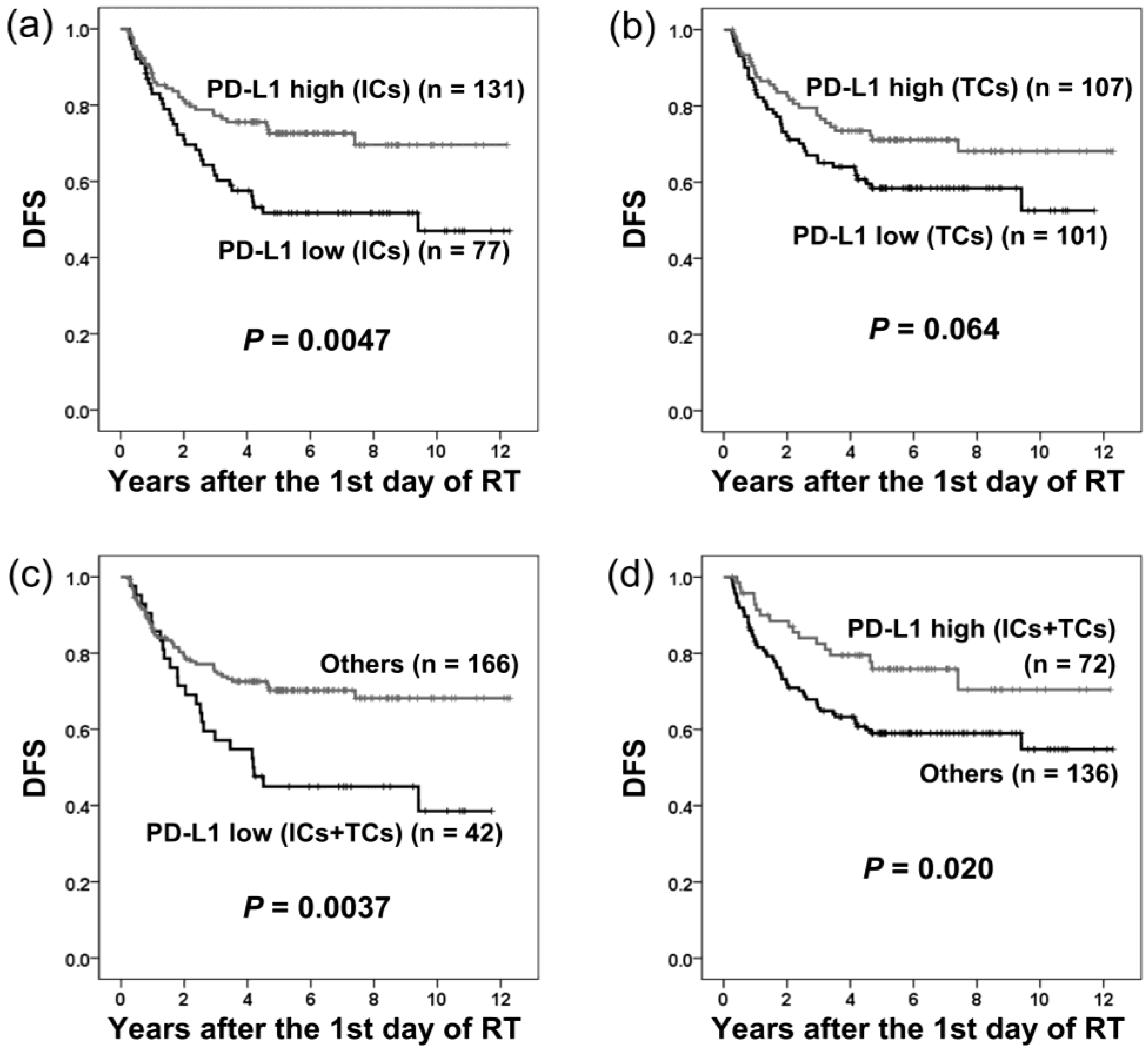
2.4. PD-L1 Expression and Disease-free Survival
2.5. PD-L1 Expression and Overall Survival
3. Discussion
4. Materials and Methods
4.1. Patients and Samples
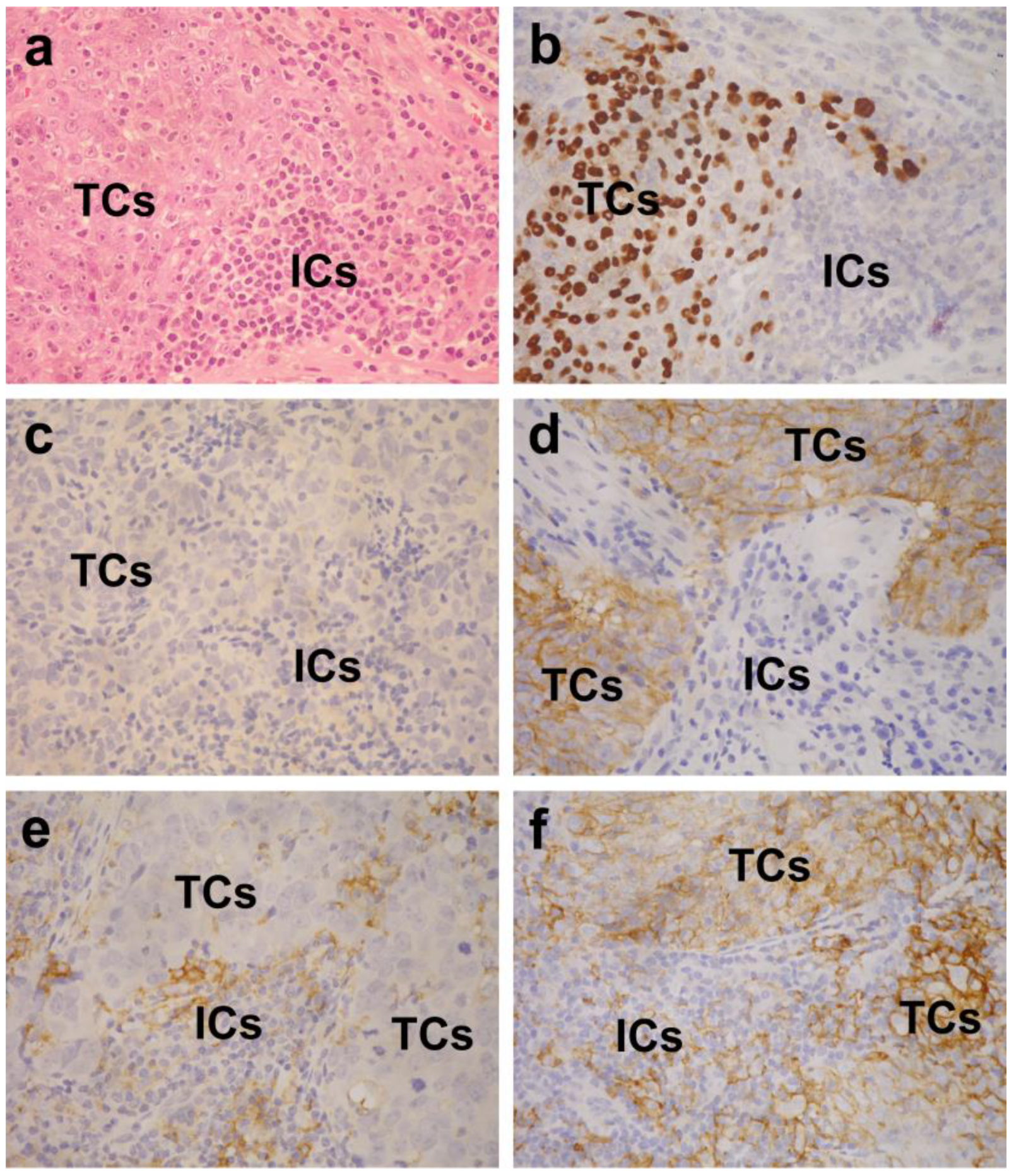
4.2. Evaluation of PD-L1 Expression
4.3. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef] [PubMed]
- Petersson, B.F.; Bell, D.; El-Mofty, S.K.; Gillison, M.; Lewis, J.S.; Nadal, A.; Nicolai, P.; Wenig, B.M. Nasopharyngeal carcinoma. In WHO Classification of Head and Neck Tumours, 4th ed.; El-Naggar, A.K., Chan, J.K.C., Grandis, J.R., Takata, T., Slootweg, P.J., Eds.; IARC: Lyon, France, 2017; pp. 65–70. [Google Scholar]
- Chiang, C.J.; Lo, W.C.; Yang, Y.W.; You, S.L.; Chen, C.J.; Lai, M.S. Incidence and survival of adult cancer patients in Taiwan, 2002-2012. J. Formos. Med. Assoc. 2016, 115, 1076–1088. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.W.; Ma, B.B.; Ng, W.T.; Chan, A.T. Management of Nasopharyngeal Carcinoma: Current Practice and Future Perspective. J. Clin. Oncol. 2015, 33, 3356–3364. [Google Scholar] [CrossRef] [PubMed]
- Andersson-Anvret, M.; Forsby, N.; Klein, G.; Henle, W. Relationship between the Epstein-Barr virus and undifferentiated nasopharyngeal carcinoma: Correlated nucleic acid hybridization and histopathological examination. Int. J. Cancer 1977, 20, 486–494. [Google Scholar] [CrossRef] [PubMed]
- Klein, G.; Giovanella, B.C.; Lindahl, T.; Fialkow, P.J.; Singh, S.; Stehlin, J.S. Direct evidence for the presence of Epstein-Barr virus DNA and nuclear antigen in malignant epithelial cells from patients with poorly differentiated carcinoma of the nasopharynx. Proc. Natl. Acad. Sci. USA 1974, 71, 4737–4741. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zeng, X.H.; Mo, H.Y.; Rolen, U.; Gao, Y.F.; Zhang, X.S.; Chen, Q.Y.; Zhang, L.; Zeng, M.S.; Li, M.Z.; et al. Functional inactivation of EBV-specific T-lymphocytes in nasopharyngeal carcinoma: Implications for tumor immunotherapy. PLoS ONE 2007, 2, e1122. [Google Scholar] [CrossRef] [PubMed]
- Ono, T.; Azuma, K.; Kawahara, A.; Sasada, T.; Matsuo, N.; Kakuma, T.; Kamimura, H.; Maeda, R.; Hattori, C.; On, K.; et al. Prognostic stratification of patients with nasopharyngeal carcinoma based on tumor immune microenvironment. Head Neck 2018. [Google Scholar] [CrossRef] [PubMed]
- Alsaab, H.O.; Sau, S.; Alzhrani, R.; Tatiparti, K.; Bhise, K.; Kashaw, S.K.; Iyer, A.K. PD-1 and PD-L1 Checkpoint Signaling Inhibition for Cancer Immunotherapy: Mechanism, Combinations, and Clinical Outcome. Front. Pharmacol. 2017, 8, 561. [Google Scholar] [CrossRef] [PubMed]
- Jain, A.; Chia, W.K.; Toh, H.C. Immunotherapy for nasopharyngeal cancer-a review. Chin. Clin. Oncol. 2016, 5, 22. [Google Scholar] [CrossRef] [PubMed]
- Ran, X.; Yang, K. Inhibitors of the PD-1/PD-L1 axis for the treatment of head and neck cancer: Current status and future perspectives. Drug Des. Dev. Ther. 2017, 11, 2007–2014. [Google Scholar] [CrossRef] [PubMed]
- Ritprajak, P.; Azuma, M. Intrinsic and extrinsic control of expression of the immunoregulatory molecule PD-L1 in epithelial cells and squamous cell carcinoma. Oral Oncol. 2015, 51, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Dave, V.; Harris, J.; Huang, Y. Response of advanced stage recurrent lymphoepithelioma-like carcinoma to nivolumab. Immunotherapy 2017, 9, 955–961. [Google Scholar] [CrossRef] [PubMed]
- Ansell, S.M.; Lesokhin, A.M.; Borrello, I.; Halwani, A.; Scott, E.C.; Gutierrez, M.; Schuster, S.J.; Millenson, M.M.; Cattry, D.; Freeman, G.J.; et al. PD-1 blockade with nivolumab in relapsed or refractory Hodgkin’s lymphoma. N. Engl. J. Med. 2015, 372, 311–319. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.S.; Irving, B.A.; Hodi, F.S. Molecular pathways: Next-generation immunotherapy—inhibiting programmed death-ligand 1 and programmed death-1. Clin. Cancer Res. 2012, 18, 6580–6587. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, J.E.; Hoffman-Censits, J.; Powles, T.; van der Heijden, M.S.; Balar, A.V.; Necchi, A.; Dawson, N.; O’Donnell, P.H.; Balmanoukian, A.; Loriot, Y.; et al. Atezolizumab in patients with locally advanced and metastatic urothelial carcinoma who have progressed following treatment with platinum-based chemotherapy: A single-arm, multicentre, phase 2 trial. Lancet 2016, 387, 1909–1920. [Google Scholar] [CrossRef]
- Taube, J.M.; Klein, A.; Brahmer, J.R.; Xu, H.; Pan, X.; Kim, J.H.; Chen, L.; Pardoll, D.M.; Topalian, S.L.; Anders, R.A. Association of PD-1, PD-1 ligands, and other features of the tumor immune microenvironment with response to anti-PD-1 therapy. Clin. Cancer Res. 2014, 20, 5064–5074. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.; Lee, S.H.; Ejadi, S.; Even, C.; Cohen, R.B.; Le Tourneau, C.; Mehnert, J.M.; Algazi, A.; van Brummelen, E.M.J.; Saraf, S.; et al. Safety and Antitumor Activity of Pembrolizumab in Patients With Programmed Death-Ligand 1-Positive Nasopharyngeal Carcinoma: Results of the KEYNOTE-028 Study. J. Clin. Oncol. 2017, 35, 4050–4056. [Google Scholar] [CrossRef] [PubMed]
- Chan, O.S.; Kowanetz, M.; Ng, W.T.; Koeppen, H.; Chan, L.K.; Yeung, R.M.; Wu, H.; Amler, L.; Mancao, C. Characterization of PD-L1 expression and immune cell infiltration in nasopharyngeal cancer. Oral Oncol. 2017, 67, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Chang, A.M.V.; Chiosea, S.I.; Altman, A.; Pagdanganan, H.A.; Ma, C. Programmed Death-Ligand 1 Expression, Microsatellite Instability, Epstein-Barr Virus, and Human Papillomavirus in Nasopharyngeal Carcinomas of Patients from the Philippines. Head Neck Pathol. 2017, 11, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Fang, W.; Zhang, J.; Hong, S.; Zhan, J.; Chen, N.; Qin, T.; Tang, Y.; Zhang, Y.; Kang, S.; Zhou, T.; et al. EBV-driven LMP1 and IFN-gamma up-regulate PD-L1 in nasopharyngeal carcinoma: Implications for oncotargeted therapy. Oncotarget 2014, 5, 12189–12202. [Google Scholar] [CrossRef] [PubMed]
- Lee, V.H.; Lo, A.W.; Leung, C.Y.; Shek, W.H.; Kwong, D.L.; Lam, K.O.; Tong, C.C.; Sze, C.K.; Leung, T.W. Correlation of PD-L1 Expression of Tumor Cells with Survival Outcomes after Radical Intensity-Modulated Radiation Therapy for Non-Metastatic Nasopharyngeal Carcinoma. PLoS ONE 2016, 11, e0157969. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.F.; Ding, J.W.; Liao, L.M.; Zhang, Z.L.; Liao, S.S.; Wu, Y.; Zhou, D.Y.; Liu, A.W.; Huang, L. Expression of programmed death ligand-1 predicts poor outcome in nasopharyngeal carcinoma. Mol. Clin. Oncol. 2017, 7, 378–382. [Google Scholar] [CrossRef] [PubMed]
- Ooft, M.L.; van Ipenburg, J.A.; Braunius, W.W.; Zuur, C.I.; Koljenovic, S.; Willems, S.M. Prognostic role of tumor infiltrating lymphocytes in EBV positive and EBV negative nasopharyngeal carcinoma. Oral Oncol. 2017, 71, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Fang, W.; Qin, T.; Yang, Y.; Hong, S.; Liang, W.; Ma, Y.; Zhao, H.; Huang, Y.; Xue, C.; et al. Co-expression of PD-1 and PD-L1 predicts poor outcome in nasopharyngeal carcinoma. Med. Oncol. 2015, 32, 86. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Shi, D.; Miao, J.; Wu, H.; Chen, J.; Zhou, X.; Hu, D.; Zhao, C.; Deng, W.; Xie, C. PD-L1 predicts poor prognosis for nasopharyngeal carcinoma irrespective of PD-1 and EBV-DNA load. Sci. Rep. 2017, 7, 43627. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zheng, L.; Cao, C.; Cheng, G.; Hu, Q.; Chen, X. Cytomembranic PD-L1 expression in locoregionally advanced nasopharyngeal carcinoma. Onco Targets Ther. 2017, 10, 5483–5487. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, Q.; Cai, M.Y.; Chen, C.L.; Hu, H.; Lin, H.X.; Li, M.; Weng, D.S.; Zhao, J.J.; Guo, L.; Xia, J.C. Tumor cells PD-L1 expression as a favorable prognosis factor in nasopharyngeal carcinoma patients with pre-existing intratumor-infiltrating lymphocytes. Oncoimmunology 2017, 6, e1312240. [Google Scholar] [CrossRef] [PubMed]
- Parra, E.R.; Villalobos, P.; Mino, B.; Rodriguez-Canales, J. Comparison of Different Antibody Clones for Immunohistochemistry Detection of Programmed Cell Death Ligand 1 (PD-L1) on Non-Small Cell Lung Carcinoma. Appl. Immunohistochem. Mol. Morphol. 2018, 26, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Schats, K.A.; Van Vre, E.A.; De Schepper, S.; Boeckx, C.; Schrijvers, D.M.; Waelput, W.; Fransen, E.; Vanden Bempt, I.; Neyns, B.; De Meester, I.; et al. Validated programmed cell death ligand 1 immunohistochemistry assays (E1L3N and SP142) reveal similar immune cell staining patterns in melanoma when using the same sensitive detection system. Histopathology 2017, 70, 253–263. [Google Scholar] [CrossRef] [PubMed]
- Birtalan, E.; Danos, K.; Gurbi, B.; Brauswetter, D.; Halasz, J.; Kalocsane Piurko, V.; Acs, B.; Antal, B.; Mihalyi, R.; Pato, A.; et al. Expression of PD-L1 on Immune Cells Shows Better Prognosis in Laryngeal, Oropharygeal, and Hypopharyngeal Cancer. Appl. Immunohistochem. Mol. Morphol. 2018, 26, e79–e85. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.R.; Ha, S.J.; Hong, M.H.; Heo, S.J.; Koh, Y.W.; Choi, E.C.; Kim, E.K.; Pyo, K.H.; Jung, I.; Seo, D.; et al. PD-L1 expression on immune cells, but not on tumor cells, is a favorable prognostic factor for head and neck cancer patients. Sci. Rep. 2016, 6, 36956. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, Y.M.; Sung, W.W.; Hsieh, M.J.; Tsai, S.C.; Lai, H.W.; Yang, S.M.; Shen, K.H.; Chen, M.K.; Lee, H.; Yeh, K.T.; et al. High PD-L1 Expression Correlates with Metastasis and Poor Prognosis in Oral Squamous Cell Carcinoma. PLoS ONE 2015, 10, e0142656. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.C.; Wu, C.T.; Wang, C.P.; Hsu, W.L.; Yang, T.L.; Lou, P.J.; Ko, J.Y.; Chang, Y.L. Associations among pretreatment tumor necrosis and the expression of HIF-1alpha and PD-L1 in advanced oral squamous cell carcinoma and the prognostic impact thereof. Oral Oncol. 2015, 51, 1004–1010. [Google Scholar] [CrossRef] [PubMed]
- Kogashiwa, Y.; Yasuda, M.; Sakurai, H.; Nakahira, M.; Sano, Y.; Gonda, K.; Ikeda, T.; Inoue, H.; Kuba, K.; Oba, S.; et al. PD-L1 Expression Confers Better Prognosis in Locally Advanced Oral Squamous Cell Carcinoma. Anticancer Res. 2017, 37, 1417–1424. [Google Scholar] [CrossRef] [PubMed]
- Barker, H.E.; Paget, J.T.; Khan, A.A.; Harrington, K.J. The tumour microenvironment after radiotherapy: Mechanisms of resistance and recurrence. Nat. Rev. Cancer 2015, 15, 409–425. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.C.M.; Tsao, S.W.; Tsang, C.M. Interplay of Viral Infection, Host Cell Factors and Tumor Microenvironment in the Pathogenesis of Nasopharyngeal Carcinoma. Cancers 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- Velapasamy, S.; Dawson, C.W.; Young, L.S.; Paterson, I.C.; Yap, L.F. The Dynamic Roles of TGF-beta Signalling in EBV-Associated Cancers. Cancers 2018, 10. [Google Scholar] [CrossRef] [PubMed]
- Vassilakopoulou, M.; Avgeris, M.; Velcheti, V.; Kotoula, V.; Rampias, T.; Chatzopoulos, K.; Perisanidis, C.; Kontos, C.K.; Giotakis, A.I.; Scorilas, A.; et al. Evaluation of PD-L1 Expression and Associated Tumor-Infiltrating Lymphocytes in Laryngeal Squamous Cell Carcinoma. Clin. Cancer Res. 2016, 22, 704–713. [Google Scholar] [CrossRef] [PubMed]
- Taube, J.M.; Anders, R.A.; Young, G.D.; Xu, H.; Sharma, R.; McMiller, T.L.; Chen, S.; Klein, A.P.; Pardoll, D.M.; Topalian, S.L.; et al. Colocalization of inflammatory response with B7-h1 expression in human melanocytic lesions supports an adaptive resistance mechanism of immune escape. Sci. Transl. Med. 2012, 4, 127ra137. [Google Scholar] [CrossRef] [PubMed]
- Deng, L.; Liang, H.; Burnette, B.; Beckett, M.; Darga, T.; Weichselbaum, R.R.; Fu, Y.X. Irradiation and anti-PD-L1 treatment synergistically promote antitumor immunity in mice. J. Clin. Investig. 2014, 124, 687–695. [Google Scholar] [CrossRef] [PubMed]
- Hecht, M.; Buttner-Herold, M.; Erlenbach-Wunsch, K.; Haderlein, M.; Croner, R.; Grutzmann, R.; Hartmann, A.; Fietkau, R.; Distel, L.V. PD-L1 is upregulated by radiochemotherapy in rectal adenocarcinoma patients and associated with a favourable prognosis. Eur. J. Cancer 2016, 65, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.W.M.; Lydiatt, W.M.; Colevas, A.D.; Clastonbury, C.M.; Le, Q.T.X.; O’Sullivan, B.; Weber, R.S.; Shah, J.P. Nasopharynx. In AJCC Cancer Staging Manual, 8th ed.; Amin, M.B., Edge, S., Greene, F., Byrd, D.R., Brookland, R.K., Washington, M.K., Gershenwald, J.E., Compton, C.C., Hess, K.R., Sullivan, D.C., et al., Eds.; Springer International Publishing AG: Basel, Switzerland, 2017; pp. 103–111. ISBN 978-3-319-40617-6. [Google Scholar]
| Characteristic | Total (n = 208) | PD-L1 on TCs | p-Value | PD-L1 on ICs | p-Value | ||
|---|---|---|---|---|---|---|---|
| Low (n = 101) | High (n = 107) | Low (n = 77) | High (n = 131) | ||||
| Pre-treatment | |||||||
| Age | |||||||
| Mean ± SD | 49 ± 11 | 50 ± 11 | 48 ± 11 | 0.22 | 49 ± 11 | 49 ± 11 | 0.86 |
| Median (min; max) | 49 (20; 84) | 49 (25; 84) | 48 (20; 73) | 49 (21; 81) | 49 (20; 84) | ||
| Gender | |||||||
| Male | 146 (70) | 70 (69) | 76 (71) | 0.79 | 53 (69) | 93 (71) | 0.74 |
| Female | 62 (30) | 31 (31) | 31 (29) | 24 (31) | 38 (29) | ||
| Smoking | |||||||
| Yes | 109 (52) | 58 (57) | 51 (48) | 0.16 | 37 (48) | 72 (55) | 0.33 |
| No | 99 (48) | 43 (43) | 56 (52) | 40 (52) | 59 (45) | ||
| Alcohol | |||||||
| Yes | 56 (27) | 29 (29) | 27 (25) | 0.57 | 23 (30) | 33 (25) | 0.46 |
| No | 152 (73) | 72 (71) | 80 (75) | 54 (70) | 98 (75) | ||
| Betel quid | |||||||
| Yes | 40 (19) | 23 (23) | 17 (16) | 0.21 | 19 (25) | 21 (16) | 0.13 |
| No | 168 (81) | 78 (77) | 90 (84) | 58 (75) | 110 (84) | ||
| T category | |||||||
| T1-2 | 104 (50) | 50 (50) | 54 (50) | 0.89 | 36 (47) | 68 (52) | 0.47 |
| T3-4 | 104 (50) | 51 (50) | 53 (50) | 41 (53) | 63 (48) | ||
| N category | |||||||
| N0-1 | 101 (49) | 49 (49) | 52 (49) | 0.99 | 39 (51) | 62 (47) | 0.64 |
| N2-3 | 107 (51) | 52 (51) | 55 (51) | 38 (49) | 69 (53) | ||
| M category | |||||||
| M0 | 205 (99) | 101 (100) | 104 (97) | 0.27 | 76 (99) | 129 (99) | 1.0 |
| M1 | 3 (1) | 0 (0) | 3 (3) | 1 (1) | 2 (1) | ||
| AJCC Stage | |||||||
| I–II | 55 (26) | 29 (29) | 26 (24) | 0.47 | 22 (29) | 33 (25) | 0.59 |
| III–IV | 153 (74) | 72 (71) | 81 (76) | 55 (71) | 98 (75) | ||
| Treatment | |||||||
| Chemotherapy | |||||||
| Yes | 203 (98) | 97 (96) | 106 (99) | 0.33 | 75 (97) | 128 (98) | 1.0 |
| No | 5 (2) | 4 (4) | 1 (1) | 2 (3) | 3 (2) | ||
| Post-treatment | |||||||
| Local recurrence | |||||||
| Yes | 32 (15) | 22 (22) | 10 (9) | 0.013 * | 20 (26) | 12 (9) | 0.0012 * |
| No | 176 (85) | 79 (78) | 97 (91) | 57 (74) | 119 (91) | ||
| Neck recurrence | |||||||
| Yes | 34 (16) | 21 (21) | 13 (12) | 0.09 | 15 (20) | 19 (15) | 0.35 |
| No | 174 (84) | 80 (79) | 94 (88) | 62 (80) | 112 (85) | ||
| Distant metastasis | |||||||
| Yes | 35 (17) | 20 (20) | 15 (14) | 0.27 | 18 (23) | 17 (13) | 0.053 |
| No | 173 (83) | 81 (80) | 92 (86) | 59 (77) | 114 (87) | ||
| Factor | Hazard Ratio | 95% CI | p-Value | |||
|---|---|---|---|---|---|---|
| Local recurrence-free survival | ||||||
| Univariate analysis | ||||||
| PD-L1 (ICs-low vs. ICs-high) | 2.99 | 1.46–6.12 | 0.0028 * | |||
| PD-L1 (TCs-low vs. TCs-high) | 2.34 | 1.11–4.95 | 0.026 * | |||
| PD-L1 (ICs/TCs both low vs. others) | 3.48 | 1.74–6.98 | 0.00045 * | |||
| PD-L1 (ICs/TCs both high vs. others) | 0.33 | 0.13–0.86 | 0.023 * | |||
| Age (≥50 years vs. <50 years) | 1.28 | 0.64–2.57 | 0.48 | |||
| Gender (male vs. female) | 0.54 | 0.27–1.09 | 0.085 | |||
| Smoking (yes vs. no) | 0.70 | 0.35–1.40 | 0.31 | |||
| Alcohol drinking (yes vs. no) | 0.87 | 0.39–1.93 | 0.73 | |||
| Betel quid chewing (yes vs. no) | 1.00 | 0.41–2.42 | 1.00 | |||
| AJCC stage (III–IV vs. I–II) | 1.78 | 0.73–4.32 | 0.21 | |||
| T category (T3–4 vs. T1–2) | 1.60 | 0.79–3.23 | 0.20 | |||
| N category (N2–3 vs. N0–1) | 1.53 | 0.76–3.10 | 0.24 | |||
| M category (M1 vs. M0) | 0.05 | 0–2.0 × 105 | 0.70 | |||
| Chemotherapy (yes vs. no) | 0.40 | 0.10–1.66 | 0.21 | |||
| Multivariate analysis | ||||||
| PD-L1 (ICs-low vs. ICs-high) | 2.74 | 1.33–5.63 | 0.0062 * | |||
| PD-L1 (TCs-low vs. TCs-high) | 2.07 | 0.98–4.40 | 0.058 | |||
| Disease-free survival | ||||||
| Univariate analysis | ||||||
| PD-L1 (ICs-low vs. ICs-high) | 1.93 | 1.21–3.07 | 0.0054 * | |||
| PD-L1 (TCs-low vs. TCs-high) | 1.55 | 0.97–2.48 | 0.067 | |||
| PD-L1 (ICs/TCs both low vs. others) | 2.04 | 1.25–3.33 | 0.0045 * | |||
| PD-L1 (ICs/TCs both high vs. others) | 0.53 | 0.31–0.91 | 0.022 * | |||
| Age (≥50 years vs. <50 years) | 0.99 | 0.62–1.58 | 0.97 | |||
| Gender (male vs. female) | 0.77 | 0.48–1.25 | 0.29 | |||
| Smoking (yes vs. no) | 0.78 | 0.49–1.24 | 0.29 | |||
| Alcohol drinking (yes vs. no) | 1.40 | 0.86–2.29 | 0.18 | |||
| Betel quid chewing (yes vs. no) | 1.91 | 1.14–3.21 | 0.014 * | |||
| AJCC stage (III–IV vs. I–II) | 2.40 | 1.26–4.55 | 0.0079 * | |||
| T category (T3–4 vs. T1–2) | 1.60 | 1.00–2.56 | 0.050 | |||
| N category (N2–3 vs. N0–1) | 1.84 | 1.14–2.96 | 0.013 * | |||
| M category (M1 vs. M0) | 0.05 | 0–617 | 0.53 | |||
| Chemotherapy (yes vs. no) | 0.96 | 0.24–3.91 | 0.95 | |||
| Multivariate analysis | ||||||
| PD-L1 (ICs-low vs. ICs-high) | 1.88 | 1.18–3.00 | 0.0080 * | |||
| Betel quid chewing (yes vs. no) | 1.68 | 0.99–2.83 | 0.051 | |||
| AJCC stage (III–IV vs. I–II) | 2.40 | 1.26–4.58 | 0.0077 * | |||
| Overall survival | ||||||
| Univariate analysis | ||||||
| PD-L1 (ICs-low vs. ICs-high) | 1.51 | 0.90–2.55 | 0.12 | |||
| PD-L1 (TCs-low vs. TCs-high) | 1.48 | 0.87–2.51 | 0.15 | |||
| PD-L1 (ICs/TCs both low vs. others) | 1.28 | 0.72–2.28 | 0.41 | |||
| PD-L1 (ICs/TCs both high vs. others) | 0.46 | 0.24–0.88 | 0.020 * | |||
| Age (≥50 years vs. <50 years) | 1.78 | 1.05–3.01 | 0.033 * | |||
| Gender (male vs. female) | 0.71 | 0.42–1.22 | 0.22 | |||
| Smoking (yes vs. no) | 0.87 | 0.52–1.47 | 0.60 | |||
| Alcohol drinking (yes vs. no) | 0.88 | 0.49–1.59 | 0.68 | |||
| Betel quid chewing (yes vs. no) | 1.19 | 0.63–2.25 | 0.60 | |||
| AJCC stage (III–IV vs. I–II) | 1.83 | 0.95–3.54 | 0.073 | |||
| T category (T3–4 vs. T1–2) | 1.72 | 1.01–2.93 | 0.045 * | |||
| N category (N2–3 vs. N0–1) | 2.16 | 1.24–3.74 | 0.0063 * | |||
| M category (M1 vs. M0) | 2.41 | 0.33–17.5 | 0.39 | |||
| Chemotherapy (yes vs. no) | 0.77 | 0.19–3.14 | 0.71 | |||
| Multivariate analysis | ||||||
| PD-L1 (ICs/TCs both high vs. others) | 0.46 | 0.24–0.89 | 0.022 * | |||
| Age (≥50 years vs. <50 years) | 1.80 | 1.06–3.07 | 0.030 * | |||
| T category (T3–4 vs. T1–2) | 1.53 | 0.89–2.62 | 0.12 | |||
| N category (N2–3 vs. N0–1) | 2.22 | 1.27–3.89 | 0.0052 * | |||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Y.-J.; Tsang, N.-M.; Hsueh, C.; Yeh, C.-J.; Ueng, S.-H.; Wang, T.-H.; Chuang, W.-Y. Low PD-L1 Expression Strongly Correlates with Local Recurrence in Epstein-Barr Virus-Positive Nasopharyngeal Carcinoma after Radiation-Based Therapy. Cancers 2018, 10, 374. https://doi.org/10.3390/cancers10100374
Liu Y-J, Tsang N-M, Hsueh C, Yeh C-J, Ueng S-H, Wang T-H, Chuang W-Y. Low PD-L1 Expression Strongly Correlates with Local Recurrence in Epstein-Barr Virus-Positive Nasopharyngeal Carcinoma after Radiation-Based Therapy. Cancers. 2018; 10(10):374. https://doi.org/10.3390/cancers10100374
Chicago/Turabian StyleLiu, Yu-Jen, Ngan-Ming Tsang, Chuen Hsueh, Chi-Ju Yeh, Shir-Hwa Ueng, Tong-Hong Wang, and Wen-Yu Chuang. 2018. "Low PD-L1 Expression Strongly Correlates with Local Recurrence in Epstein-Barr Virus-Positive Nasopharyngeal Carcinoma after Radiation-Based Therapy" Cancers 10, no. 10: 374. https://doi.org/10.3390/cancers10100374





