Lengthening of Insect Development on Bt Zone Results in Adult Emergence Asynchrony: Does It Influence the Effectiveness of the High Dose/Refuge Zone Strategy?
Abstract
:1. Introduction
2. Materials and Methods
2.1. Mathematical Model
| Symbol | Default Value | Ref. | |
|---|---|---|---|
| Operational Parameters | |||
| Refuge zone relative size | v | 0.05 | [27] |
| Mortality of AsAs on the toxin A (AsAs mortality) | sBt | 1 | [27] |
| Field area (hectare) | 260 | [28] | |
| Plants/hectare | 67,000 | [28] | |
| Biological Parameters | |||
| Initial Ar frequency | ArFreq | 1.5 × 10−3 | [29] |
| Ar dominance | hAr | 0 | [30,31] |
| Fitness cost associated to Ar | fcost | 0.15 | [32] |
| Fitness cost dominance associated to Ar | hfc | 0 | [32] |
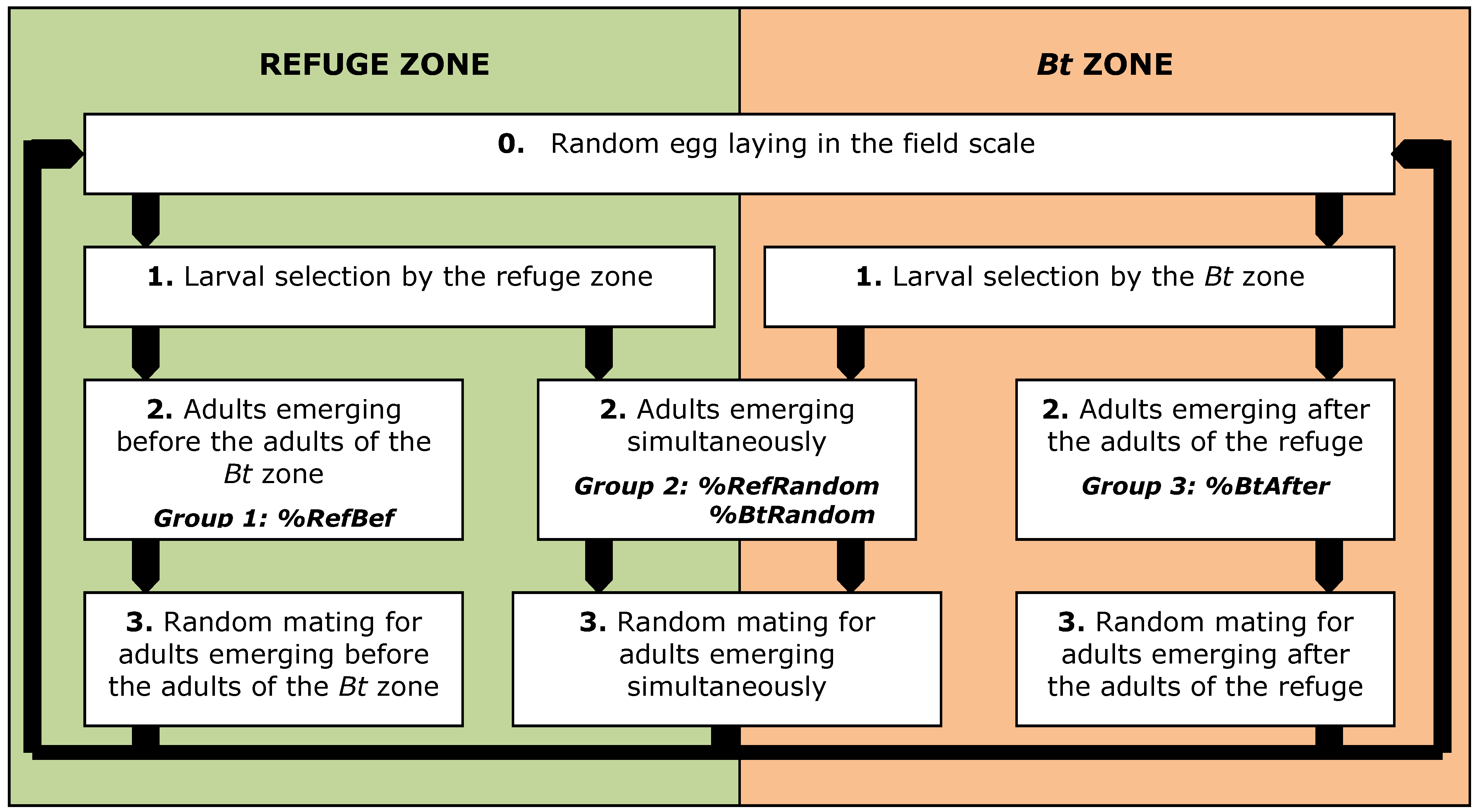
| % adults emerging from the refuge before the adults of the Bt zone, unavailable for random mating | %RefBef | 0 | [27] |
| % adults emerging from the refuge simultaneously with adults emerging from the Bt zone, available for random mating | %RefRandom | 1–%RefBef | |
| % adults emerging from the Bt zone after the adults of the refuge, unavailable for random mating | % BtAfter | 0 | [27] |
| % adults emerging from the Bt zone simultaneously with adults emerging from the refuge, available for random mating | % BtRandom | 1–% BtAfter | |
| Initial individual number/ha | nzero | 50,000 | [28] |
| Intrinsic growth rate | r | 0.15 | [18,33] |
| Carrying capacity/plant | K | 22 | [28] |
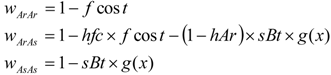
| Bt Zone | Refuge Zone | ||||||
|---|---|---|---|---|---|---|---|
| hAr | sBt | ArAr | ArAs | AsAs | ArAr | ArAs | AsAs |
| 0 | 1 | 0.85 | 0 | 0 | 0.85 | 1 | 1 |
| 0.53 | 0.5 | 0.85 | 0.765 | 0.5 | 0.85 | 1 | 1 |
| 0.23 | 0.93 | 0.85 | 0.2839 | 0.07 | 0.85 | 1 | 1 |
2.2. Model Insect and Experimental Values Introduced in the Model
2.2.1. Insect Mortality in Relation to Toxin Concentration in the Bt Plant
2.2.2. Proportion of Asynchrony in Relation to Toxin Concentration in the Bt Plant
2.3. Impact of Emergence Asynchrony (%RefBef–%BtAfter) on the Effectiveness of Resistance Management
| A. sBt 1-hAr0 | |||||
| Control | Test 1 | Test 2 | Test 3 | Test 4 | |
| Toxin concentration | - | - | - | 0.27 µg Cry1Ab/g fwt | 112 ng Cry1Fa/mg total protein |
| %RefRandom | 100 | 10 | 100 | 0 | - |
| %BtRandom | 100 | 100 | 90 | 0 | - |
| GF50Bef (adults from the refuge) | - | 45 | - | 6 | - |
| GF50Random (adults from the refuge and the Bt zones) | 47 | 43 | 13 | - | - |
| GF50After (adults from the Bt zone) | - | - | 1 | 1 | - |
| Global GF50 | 48 | 45 | 14 | 6 | - |
| %popdecrease (between generation 3 and 1) | 99.7 | 99.7 | 99.7 | 99.7 | - |
| B. sBt0.93-hAr0.23 | |||||
| Control | Test 1 | Test 2 | Test 3 | Test 4 | |
| Toxin concentration | - | - | - | 0.25 µg Cry1Ab/g fwt | 104 ng Cry1Fa/mg total protein |
| %RefRandom | 100 | 10 | 100 | 0 | - |
| %BtRandom | 100 | 100 | 90 | 0 | - |
| GF50Bef | - | 7 | - | 7 | - |
| GF50Random | 6 | 6 | 7 | 6 | - |
| GF50After | - | - | 6 | 6 | - |
| Global GF50 | 7 | 7 | 7 | 7 | - |
| %popdecrease (between generation 3 and 1) | 98 | 98 | 98 | 98 | - |
| C. sBt0.50-hAr0.53 | |||||
| Control | Test 1 | Test 2 | Test 3 | Test 4 | |
| Toxin concentration | - | - | - | 0.14 µg Cry1Ab/g fwt | 57 ng Cry1Fa/mg total protein |
| %RefRandom | 100 | 10 | 100 | 46 | 26 |
| %BtRandom | 100 | 100 | 90 | 55 | 22 |
| GF50Bef | - | 20 | - | 20 | 20 |
| GF50Random | 19 | 18 | 20 | 19 | 19 |
| GF50After | - | - | 18 | 18 | 18 |
| Global GF50 | 20 | 20 | 20 | 20 | 20 |
| %popdecrease (between generation 3 and 1) | 63 | 63 | 63 | 63 | 63 |
2.4. Observed %RefBef and %BtAfter at Lower Toxin Concentration in the Bt Plants (sBt = 0.93 and sBt = 0.50) and Their Impacts on Resistance Management
2.5. Impact of the Refuge Zone’s Relative Size
2.6. Model Output
3. Results and Discussion
3.1. Effectiveness of Resistance Management with Bt Plants Expressing a High Toxin Concentration (sBt = 1) in Relation to Emergence Asynchrony

3.2. Reducing Toxin Concentration: Impact on the Effectiveness of Resistance Management
3.3. Refuge Zone Relative Size: Impact on Resistance Management
3.3.1. sBt = 1 − hAr = 0
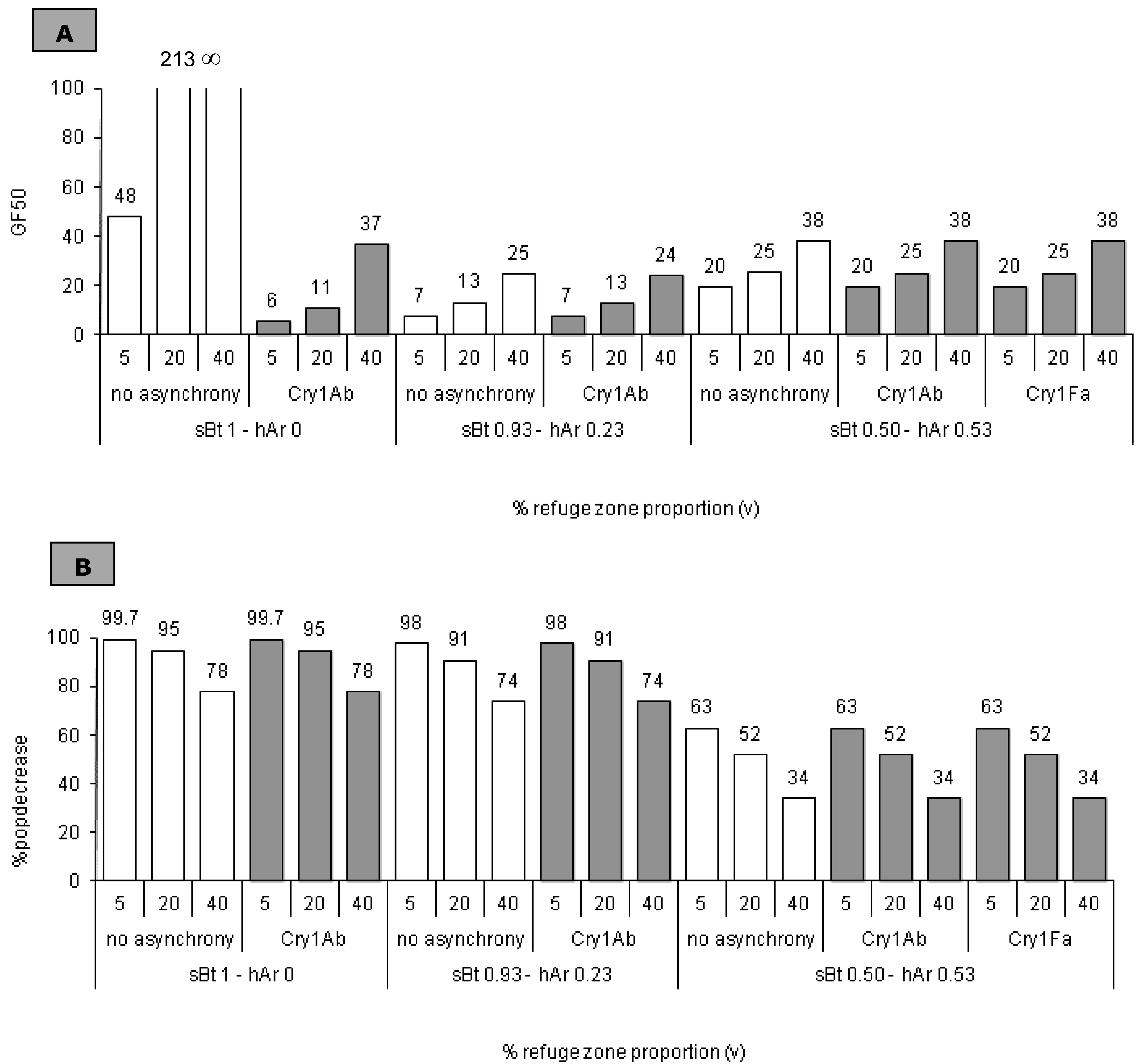
3.3.2. sBt < 1 − hAr > 0
4. Conclusions and Limitations
Acknowledgments
Conflict of Interest
References
- Gallais, A.; Ricroch, A. Plantes Transgéniques: Faits et Enjeux; Editions Quae: Versailles, France, 2006. [Google Scholar]
- Carlile, W.R. Pesticide Selectivity, Health and the Environment; Cambridge University Press: Cambridge, UK, 2006. [Google Scholar]
- Metcalf, R.L.; Luckmann, W.H. Introduction to Insect Pest Management; Wiley-IEEE: New York, NY, USA, 1994. [Google Scholar]
- Lacey, L.; Goettel, M. Current developments in microbial control of insect pests and prospects for the early 21st century. BioControl 1995, 40, 3–27. [Google Scholar]
- Hofte, H.; Whiteley, H.R. Insecticidal crystal proteins of Bacillus thuringiensis. Microbiol. Mol. Biol. Rev. 1989, 53, 242. [Google Scholar]
- Thacker, J.R.M.D. An Introduction to Arthropod Pest Control; Cambridge University Press: Cambridge, UK, 2002. [Google Scholar]
- Joung, K.-B.; Côté, J.-C. Une Analyse des Incidences Environnementales de L’insecticide Microbien Bacillus thuringiensis; Centre de Recherche et de Développement en Agriculture: Québec, Canada, 2000. [Google Scholar]
- James, C. 2010 ISAAA Report on Global Status of Biotech/GM Crops. ISAAA Brief 2012, 2012, 42. [Google Scholar]
- Jorge, F.-C. USDA Economic Research Service ERS/USDA Data—Adoption of Genetically Engineered Crops in the U.S. Available online: http://www.ers.usda.gov/data-products/adoption-of-genetically-engineered-crops-in-the-us.aspx/ (accessed on 24 September 2012).
- Bates, S.L.; Zhao, J.-Z.; Roush, R.T.; Shelton, A.M. Insect resistance management in GM crops: Past, present and future. Nat. Biotech. 2005, 23, 57–62. [Google Scholar] [CrossRef]
- Caprio, M.; Summerford, D.; Simms, S. Evaluating transgenic plants for suitability in pest and resistance management programs. In Field Manual of Techniques in Invertebrate Pathology; Lacey, L.A., Kaya, H.K., Eds.; Springer: Dordrecht, The Netherlands, 2000; pp. 805–828. [Google Scholar]
- Giles, K.L.; Hellmich, R.L.; Iverson, C.T.; Lewis, L.C. Effects of transgenic Bacillus thuringiensis maize grain on B. thuringiensis-susceptible Plodia interpunctella (Lepidoptera: Pyralidae). J. Econ. Entomol. 2000, 93, 1011–1016. [Google Scholar] [CrossRef]
- Gryspeirt, A.; Grégoire, J.-C. Effects of Two Varieties of Bacillus thuringiensis Maize on the Biology of Plodia Interpunctella. Toxins 2012, 4, 373–389. [Google Scholar] [CrossRef]
- Sedlacek, J.D.; Komaravalli, S.R.; Hanley, A.M.; Price, B.D.; Davis, P.M. Life History Attributes of Indian Meal Moth (Lepidoptera: Pyralidae) and Angoumois Grain Moth (Lepidoptera: Gelechiidae) Reared on Transgenic Corn Kernels. J. Econ. Entomol. 2001, 94, 586–592. [Google Scholar] [CrossRef]
- Liu, Y.-B.; Tabashnik, B.E.; Dennehy, T.J.; Patin, A.L.; Bartlett, A.C. Development time and resistance to Bt crops. Nature 1999, 400, 519. [Google Scholar]
- Liu, Y.-B.; Tabashnik, B.E.; Dennehy, T.J.; Patin, A.L.; Sims, M.A.; Meyer, S.K.; Carrière, Y. Effects of Bt Cotton and Cry1Ac Toxin on Survival and Development of Pink Bollworm (Lepidoptera: Gelechiidae). J. Econ. Entomol. 2001, 94, 1237–1242. [Google Scholar]
- Khalique, F.; Ahmed, K. Impact of Bacillus thuringiensis subsp. kurstaki on biology of Helicoverpa zea armigera. Pakistan J. Biol. Sci. 2003, 6, 615–621. [Google Scholar] [CrossRef]
- Bird, L.J.; Akhurst, R.J. Relative fitness of Cry1A-resistant and -susceptible Helicoverpa armigera (Lepidoptera: Noctuidae) on conventional and transgenic cotton. J. Econ. Entomol. 2004, 97, 1699–1709. [Google Scholar] [CrossRef]
- Lopez, M.D.; Sumerford, D.V.; Lewis, L.C. Nosema pyrausta and Cry1Ab-incorporated diet led to decreased survival and developmental delays in European corn borer. Entomologia Experimentalis et Applicata 2010, 134, 146–153. [Google Scholar] [CrossRef]
- Horner, T.A.; Dively, G.P.; Herbert, D.A. Development, survival and fitness performance of Helicoverpa zea (Lepidoptera: Noctuidae) in MON810 Bt field corn. J. Econ. Entomol. 2003, 96, 914–924. [Google Scholar] [CrossRef]
- Storer, N.P.; Van Duyn, J.W.; Kennedy, G.G. Life history traits of Helicoverpa zea (Lepidoptera: Noctuidae) on non-Bt and Bt transgenic corn hybrids in eastern North Carolina. J. Econ. Entomol. 2001, 94, 1268–1279. [Google Scholar] [CrossRef]
- Caprio, M.A. Evaluating Resistance Management Strategies for Multiple Toxins in the Presence of External Refuges. J. Econ. Entomol. 1998, 91, 1021–1031. [Google Scholar]
- Horner, T.A.; Dively, G.P.; Herbert, D.A. Effects of MON810 Bt field corn on adult emergence of Helicoverpa zea (Lepidoptera: Noctuidae). J. Econ. Entomol. 2003, 96, 925–930. [Google Scholar] [CrossRef]
- Peck, S.L.; Gould, F.; Ellner, S.P. Spread of resistance in spatially extended regions of transgenic cotton: Implications for management of Heliothis virescens (Lepidoptera: Noctuidae). J. Econ. Entomol. 1999, 92, 1–16. [Google Scholar]
- Fabrick, J.A.; Forlow Jech, L.; Henneberry, T.J. Novel pink bollworm resistance to the Bt toxin Cry 1Ac: Effects on mating, oviposition, larval development and survival. J. Insect Sci. 2009, 9, 24. [Google Scholar]
- Gryspeirt, A.; Grégoire, J.-C. Effectiveness of the High Dose/Refuge Strategy for Managing Pest Resistance to Bacillus thuringiensis (Bt) Plants Expressing One or Two Toxins. Toxins 2012, 4, 810–835. [Google Scholar] [CrossRef]
- US EPA. Biopesticides Registration Action Document—Bacillus thuringiensis Plant-Incorporated Protectants| Pesticides| US EPA. Available online: http://www.epa.gov/oppbppd1/biopesticides/pips/bt_brad.htm (accessed on 24 September 2012).
- Guse, C.A.; Onstad, D.W.; Buschman, L.L.; Porter, P.; Higgins, R.A.; Sloderbeck, P.E.; Cronholm, G.B.; Peairs, F.B. Modeling the development of resistance by stalk-boring Lepidoptera (Crambidae) in areas with irrigated transgenic corn. Environ. Entomol. 2002, 31, 676–685. [Google Scholar] [CrossRef]
- Andow, D.A.; Olson, D.M.; Hellmich, R.L.; Alstad, D.N.; Hutchison, W.D. Frequency of resistance to Bacillus thuringiensis toxin Cry1Ab in an Iowa population of European corn borer (Lepidoptera: Crambidae). J. Econ. Entomol. 2000, 93, 26–30. [Google Scholar]
- Tabashnik, B.E.; Patin, A.L.; Dennehy, T.J.; Liu, Y.B.; Carrière, Y.; Sims, M.A.; Antilla, L. Frequency of resistance to Bacillus thuringiensis in field populations of pink bollworm. Proc. Nat. Acad. Sci. USA 2000, 97, 12980. [Google Scholar]
- Liu, Y.B.; Tabashnik, B.E.; Meyer, S.K.; Carrière, Y.; Bartlett, A.C. Genetics of pink bollworm resistance to Bacillus thuringiensis toxin Cry1Ac. J. Econ. Entomol. 2001, 94, 248–252. [Google Scholar] [CrossRef]
- Vacher, C.; Bourguet, D.; Rousset, F.; Chevillon, C.; Hochberg, M.E. Modelling the spatial configuration of refuges for a sustainable control of pests: A case study of Bt cotton. J. Evol. Biol. 2003, 16, 378–387. [Google Scholar] [CrossRef]
- Sayyed, A.H.; Wright, D.J. Fitness costs and stability of resistance to Bacillus thuringiensis in a field population of the diamondback moth Plutella xylostella L. Ecol. Entomol. 2001, 26, 502–508. [Google Scholar] [CrossRef]
- Gould, F.; Anderson, A.; Reynolds, A.; Bumgarner, L.; Moar, W. Selection and genetic analysis of a Heliothis virescens (Lepidoptera: Noctuidae) strain with high levels of resistance to Bacillus thuringiensis toxins. J. Econ. Entomol. 1995, 88, 1545–1559. [Google Scholar]
- Gassmann, A.J.; Carrière, Y.; Tabashnik, B.E. Fitness costs of insect resistance to Bacillus thuringiensis. Annu. Rev. Entomol. 2009, 54, 147–163. [Google Scholar] [CrossRef]
- Lenormand, T.; Raymond, M. Resistance management: The stable zone strategy. Proc. Biol. Sci. 1998, 265, 1985–1990. [Google Scholar]
- Conner, J.K.; Hartl, D.L. A Primer of Ecological Genetics; Sinauer Associates: Massachussets, MA, USA, 2004. [Google Scholar]
- Sayyed, A.H.; Haward, R.; Herrero, S.; Ferre, J.; Wright, D.J. Genetic and Biochemical Approach for Characterization of Resistance to Bacillus thuringiensis Toxin Cry1Ac in a Field Population of the Diamondback Moth, Plutella xylostella. Appl. Environ. Microbiol. 2000, 66, 1509–1516. [Google Scholar] [CrossRef]
- Rawlings, P.; Davidson, G.; Sakai, R.K.; Rathor, H.R.; Aslamkhan, M.; Curtis, C.F. Field measurement of the effective dominance of an insecticide resistance in anopheline mosquitos. Bull. World Health Organ 1981, 59, 631–640. [Google Scholar]
- Bourguet, D.; Prout, M.; Raymond, M. Dominance of insecticide resistance presents a plastic response. Genetics 1996, 143, 407. [Google Scholar]
- Mallet, J.; Porter, P. Preventing insect adaptation to insect-resistant crops: Are seed mixtures or refugia the best strategy? Proc. Biol. Sci. 1992, 165–169. [Google Scholar] [CrossRef]
- Georghiou, G.P.; Taylor, C.E. Genetic and biological influences in the evolution of insecticide resistance. J. Econ. Entomol. 1977, 70, 319–323. [Google Scholar]
- Roush, R.T. Two-toxin strategies for management of insecticidal transgenic crops: Can pyramiding succeed where pesticide mixtures have not? Phil. Trans. R Soc. Lond. B Biol. Sci. 1998, 353, 1777–1786. [Google Scholar] [CrossRef]
- Lenormand, T. Gene flow and the limits to natural selection. Trends Ecol. Evol. 2002, 17, 183–189. [Google Scholar] [CrossRef]
- Lingren, P.D.; Warner, W.B.; Henneberry, T.J. Influence of Delayed Mating on Egg-Production, Egg Viability, Mating, and Longevity of Female Pink-Bollworm (Lepidoptera, Gelechiidae). Environ. Entomol. 1988, 17, 86–89. [Google Scholar]
- Huang, F.; Subramanyam, B. Effects of delayed mating on reproductive performance of Plodia interpunctella (H\übner)(Lepidoptera: Pyralidae). J. Stored Prod. Res. 2003, 39, 53–63. [Google Scholar] [CrossRef]
- Wenninger, E.J.; Averill, A.L. Effects of delayed mating on reproductive output of female oriental beetle Anomala orientalis (Coleoptera: Scarabaeidae). Agric. Forest Entomol. 2006, 8, 221–231. [Google Scholar] [CrossRef]
- Proshold, F.I. Reproductive capacity of laboratory-reared gypsy moths (Lepidoptera: Lymantriidae): Effect of age of female at time of mating. J. Econ. Entomol. 1996, 89, 337–342. [Google Scholar]
- Torres-Vila, L.M.; Rodríguez-Molina, M.C.; Stockel, J. Delayed mating reduces reproductive output of female European grapevine moth, Lobesia botrana (Lepidoptera: Tortricidae). Bull. Entomol. Res. 2002, 92, 241–249. [Google Scholar]
- Dalecky, A.; Ponsard, S.; Bailey, R.I.; Pélissier, C.; Bourguet, D. Resistance evolution to Bt crops: Predispersal mating of European corn borers. PLoS Biol. 2006, 4, e181. [Google Scholar] [CrossRef] [Green Version]
- Alyokhin, A.; Ferro, D. Relative fitness of Colorado potato beetle (Coleoptera: Chrysomelidae) resistant and susceptible to the Bacillus thuringiensis Cry3A toxin. J. Econ. Entomol. 1999, 92, 510–515. [Google Scholar]
- Groeters, F.R.; Tabashnik, B.E.; Finson, N.; Johnson, M.W. Resistance to Bacillus thuringiensis affects mating success of the diamondback moth (Lepidoptera: Plutellidae). J. Econ. Entomol. 1993, 86, 1035–1039. [Google Scholar]
- Higginson, D.M.; Morin, S.; Nyboer, M.E.; Biggs, R.W.; Tabashnik, B.E.; Carrière, Y. Evolutionary trade-offs of insect resistance to Bacillus thuringiensis crops: Fitness cost affecting paternity. Evolution 2005, 59, 915–920. [Google Scholar]
- Mangel, M.; Clark, C.W. Dynamic Modeling in Behavioral Ecology; Princeton University Press: Princeton, NJ, USA, 1988. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Gryspeirt, A.; Grégoire, J.-C. Lengthening of Insect Development on Bt Zone Results in Adult Emergence Asynchrony: Does It Influence the Effectiveness of the High Dose/Refuge Zone Strategy? Toxins 2012, 4, 1323-1342. https://doi.org/10.3390/toxins4111323
Gryspeirt A, Grégoire J-C. Lengthening of Insect Development on Bt Zone Results in Adult Emergence Asynchrony: Does It Influence the Effectiveness of the High Dose/Refuge Zone Strategy? Toxins. 2012; 4(11):1323-1342. https://doi.org/10.3390/toxins4111323
Chicago/Turabian StyleGryspeirt, Aiko, and Jean-Claude Grégoire. 2012. "Lengthening of Insect Development on Bt Zone Results in Adult Emergence Asynchrony: Does It Influence the Effectiveness of the High Dose/Refuge Zone Strategy?" Toxins 4, no. 11: 1323-1342. https://doi.org/10.3390/toxins4111323




