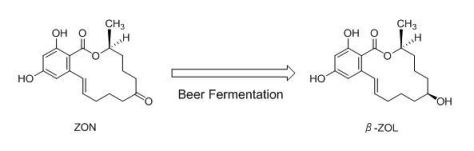
Metabolism of Zearalenone in the Course of Beer Fermentation
Abstract
:1. Introduction

2. Experimental
2.1. Materials
2.2. LC/MS/MS Conditions
| ZONs | Precursor ions (m/z) | Product Ions (m/z) | |
|---|---|---|---|
| For Quantification m/z | For Qualification m/z | ||
| (Collision Energy, eV) | (Collision Energy, eV) | ||
| ZON | 317 | 131(28) | 175(18), 273(15) |
| α-ZOL | 319 | 174(22) | 160(28), 275(18) |
| β-ZOL | 319 | 275(18) | 160(24), 174(26) |
| α-ZAL | 321 | 277(22) | 303(18), 259(20) |
| β-ZAL | 321 | 277(22) | 303(20), 259(22) |
| ZAN (I.S.) | 319 | 275(18) | 205(20) |
2.3. Sample Preparation
2.4. Fermentation Test
| Fermentation Time | Day: | 1 | 2 | 3 | 4 | 7 | 10 | 14 | 18 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hour: | 0 | 1 | 2 | 4 | 6 | 8 | 24 | 48 | 72 | 96 | 168 | 240 | 336 | 432 | |
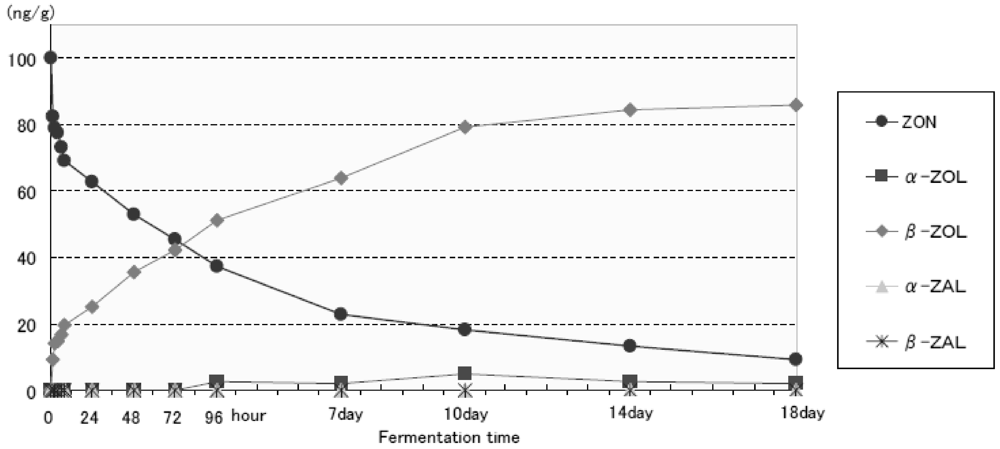
| ZON | ng/g | 100 | 82.4 | 78.7 | 77.5 | 73.0 | 69.1 | 62.7 | 52.8 | 45.4 | 37.4 | 22.8 | 18.1 | 13.4 | 9.3 |
| α-ZOL | 0 | - | - | - | - | - | - | - | - | 2.5 | 2.0 | 4.9 | 2.5 | 2.0 | |
| β-ZOL | 0 | 9.2 | 14.1 | 14.7 | 16.9 | 19.7 | 25.0 | 35.6 | 42.3 | 51.2 | 63.9 | 79.0 | 84.3 | 85.9 | |
| α-ZAL | 0 | - | - | - | - | - | - | - | - | - | - | - | - | - | |
| β-ZAL | 0 | - | - | - | - | - | - | - | - | - | - | - | - | - | |
| Total ZONs | ng/g | 100 | 91.6 | 92.8 | 92.2 | 89.9 | 88.8 | 87.7 | 88.4 | 87.7 | 91.1 | 88.7 | 102 | 100.2 | 97.2 |
3. Results and Discussion
3.1. LC/MS/MS Conditions
3.2. Validation Tests

| ZONs | ZON | α-ZOL | β-ZOL | α-ZAL | β-ZAL |
|---|---|---|---|---|---|
| Recovery (%) | 110.4 | 111.9 | 115.8 | 113.9 | 119.7 |
| 10 ng/g, n = 5 | |||||
| RSD (%) | 4.0 | 3.0 | 5.4 | 4.3 | 8.1 |
| 10 ng/g, n = 5 | |||||
| R | 0.9999 | 0.9999 | 0.9999 | 0.9998 | 0.9997 |
| 2–200 ng/g |
| ZONs | ZON | α-ZOL | β-ZOL | α-ZAL | β-ZAL |
|---|---|---|---|---|---|
| Recovery (%) | 102.3 | 104.8 | 101.1 | 111.3 | 102.8 |
| 10 ng/g, n = 5 | |||||
| RSD (%) | 2.2 | 3.4 | 7.9 | 7.2 | 5.1 |
| 10 ng/g, n = 5 | |||||
| R | 1.0000 | 1.0000 | 1.0000 | 1.0000 | 1.0000 |
| 2–200 ng/g |
3.3. Fermentation Test
4. Conclusions
References
- Betina, V. Mycotoxins Chemical, Biological and Environmental Aspects; Betina, V., Ed.; Elsevier: Amsterdam, The Netherlands, 1989; Volume 9. [Google Scholar]
- Hagler, W.M.; Mirocha, C.J. Biosynthesis of [14C] Zearalenone from [1-14C]Acetate by Fusarium reseum ‘Gibbosum’. Appl. Environ. Microbiol. 1980, 39, 668–670. [Google Scholar]
- Mirocha, C.J.; Schauerhamer, B.; Christensen, C.M.; Niku-Paavola, M.L.; Nummi, M. Incidence of Zearalenol (Fusarium Mycotoxin) in Animal Feed. Appl. Environ. Microbiol. 1979, 38, 749–750. [Google Scholar] [PubMed]
- Okoye, Z.S. Stability of zearalenone in naturally contaminated corn during Nigerian traditional brewing. Food Addit. Contam. 1987, 4, 57–59. [Google Scholar]
- Scott, P.M. Mycotoxins transmitted into beer from contaminated grains during brewing. J. Assoc. Off. Anal. Chem. Int. 1996, 79, 875–882. [Google Scholar]
- Shim, W.B.; Kim, J.C.; Seo, J.A.; Lee, Y.W. Natural occurrence of trichothecenes and zearalenone in Korean and imported beers. Food Addit. Contam. 1997, 14, 1–5. [Google Scholar]
- Erasmuson, A.F.; Scahill, B.G.; West, D.M. Natural zeranol (α-zearalnol) in the urine of pasture-fed animals. J. Agric. Food Chem. 1994, 42, 2721–2725. [Google Scholar] [CrossRef]
- Jodlbauer, J.; Zöllner, P.; Lindner, W. Determination of zeranol, taleranol, zearalenone, α-and β-zearalenol in urine and tissue by high-performance liquid chromatography-tandem mass spectrometry. Chromatographia 2000, 51, 681–687. [Google Scholar] [CrossRef]
- World Health Organization, Some Naturally Occurring Substances: Food Items and Constituents, Heterocyclic Aromatic Amines and Mycotoxins; WHO Press: Geneva, Switzerland, 1993; pp. 523–524.
- Shier, W.T.; Shier, A.C.; Xie, W.; Mirocha, C.J. Structure-activity relationships for human estrogenic activity in zearalenone mycotoxins. Toxicon 2001, 39, 1435–1438. [Google Scholar]
- Scott, P.M.; Kanhere, S.R.; Daley, E.F.; Farber, J.M. Fermentation of wort containing deoxynivalenol and zearalenone. Mycotoxin Res. 1992, 8, 58–66. [Google Scholar]
- Zöllner, P.; Jodlbauer, J.; Lindner, W. Determination of zearalenone in grains by high-performance liquid chromatography-tandem mass spectrometry after solid-phase extraction with RP-18 columns or immunoaffinity columns. J. Chromatogr. A 1999, 858, 167–174. [Google Scholar]
- Zöllner, P.; Berner, D.; Jodlbauer, J.; Lindner, W. Determination of zearalenone and its metabolites α- and β-zearalenol in beer samples by high-performance liquid chromatography-tandem mass spectrometry. J. Chromatogr. B 2000, 738, 233–241. [Google Scholar] [CrossRef]
- Rosenberg, E.; Krska, R.; Wissiack, R.; Kmetov, V.; Josephs, R.; Razzazi, E.; Grasserbauer, M. High-performance liquid chromatography-atmospheric-pressure chemical ionization mass spectrometry as a new tool for the determination of the mycotoxin zearalenone in food and feed. J. Chromatogr. A 1998, 819, 277–288. [Google Scholar]
- Mochizuki, N.; Sugai, T.; Ohta, H. Biochemical reduction of 3-oxoalkanoic esters by a bottom-fermentation yeast, Saccharomyces cerevisiae IFO 0565. Biosci. Biotech. Biochem. 1994, 58, 1666–1670. [Google Scholar]
- Fitzpatrick, D.W.; Picken, C.A.; Murphy, L.C.; Buhr, M.M. Measurement of the relative binding affinity of zearalenone, α-zearalenol and β-zearalenol for uterine and oviduct estrogen receptors in swine, rats and chickens: An indicator for estrogenic potencies. Comp. Biochem. Physiol. 1989, 94C, 691–694. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mizutani, K.; Nagatomi, Y.; Mochizuki, N. Metabolism of Zearalenone in the Course of Beer Fermentation. Toxins 2011, 3, 134-141. https://doi.org/10.3390/toxins3020134
Mizutani K, Nagatomi Y, Mochizuki N. Metabolism of Zearalenone in the Course of Beer Fermentation. Toxins. 2011; 3(2):134-141. https://doi.org/10.3390/toxins3020134
Chicago/Turabian StyleMizutani, Kohei, Yasushi Nagatomi, and Naoki Mochizuki. 2011. "Metabolism of Zearalenone in the Course of Beer Fermentation" Toxins 3, no. 2: 134-141. https://doi.org/10.3390/toxins3020134
APA StyleMizutani, K., Nagatomi, Y., & Mochizuki, N. (2011). Metabolism of Zearalenone in the Course of Beer Fermentation. Toxins, 3(2), 134-141. https://doi.org/10.3390/toxins3020134