Trichothecene Mycotoxins Inhibit Mitochondrial Translation—Implication for the Mechanism of Toxicity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Yeast Strains
2.2. Trichothecene Isolation
2.3. Growth Assay
2.4. Analysis of Total Translation
2.5. Analysis of Mitochondrial Translation
2.6. Mitochondria Isolation from Yeast
2.7. Mitochondrial in Organello Translation Assay
2.8. Staining, Microscopy & Image Analysis
2.9. Flow Cytometry
2.10. Data Analysis & Graphing
3. Results
3.1. Mitochondria Are Important for Sensitivity to Type A Trichothecenes
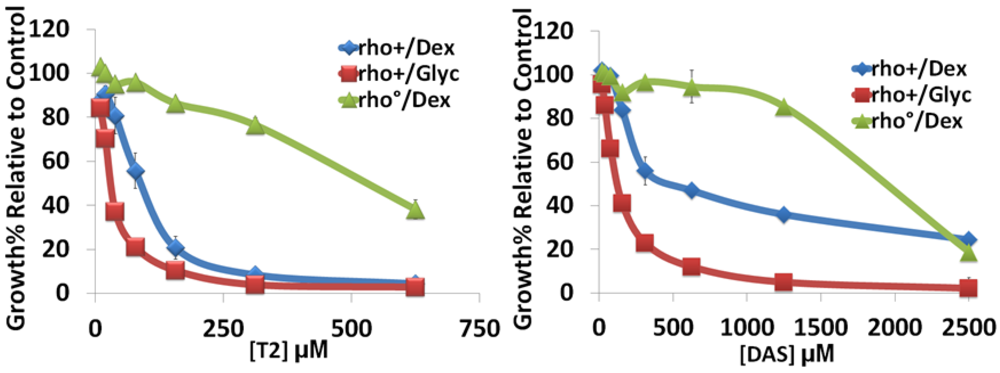
| rho+/Dex 1 | rho+/Glyc 1 | rho0/Dex 1 | |
|---|---|---|---|
| T-2 | 95 µM | 37 µM | 367 µM |
| DAS | 300 µM | 139 µM | 2400 µM |
| Tcin 1 | 2.5 µM | 0.75 µM | 17 µM |
3.2. Trichothecenes Have a Direct Effect on Mitochondrial Translation
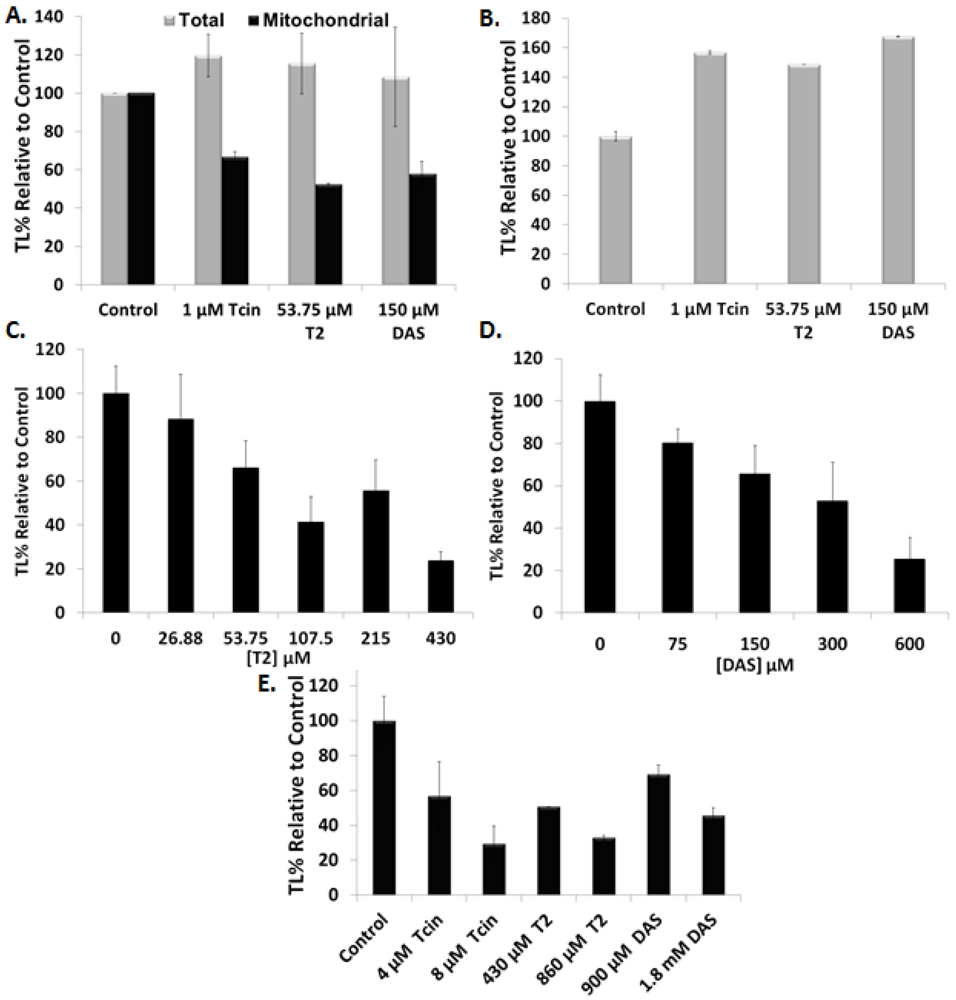
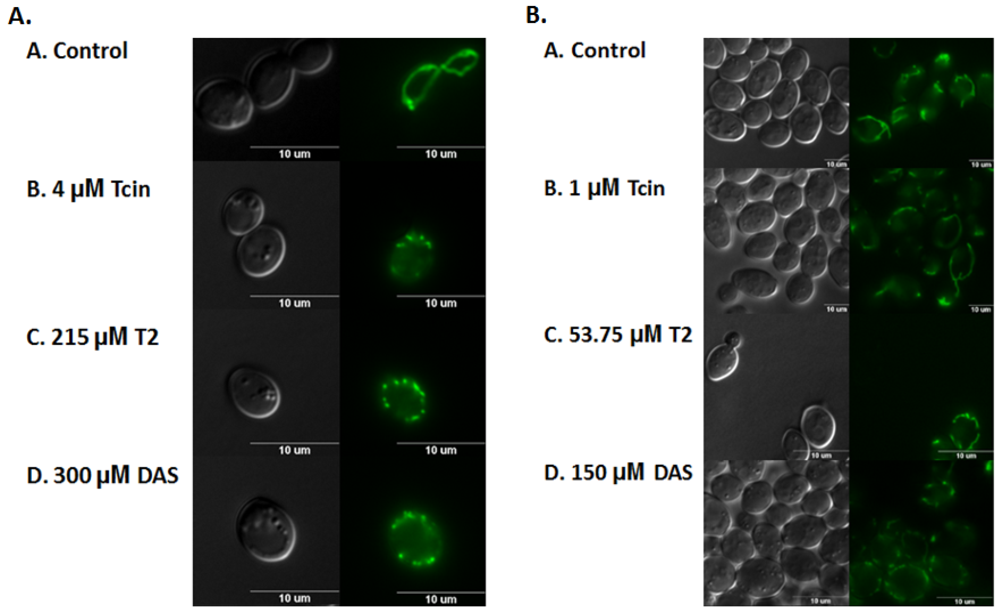
3.3. Trichothecenes Cause Dose-Dependent Alteration of Mitochondrial Membrane Morphology
3.4. Trichothecenes Disrupt Mitochondrial Membrane Potential and Generation of ROS in a Dose and Time-Dependent Manner
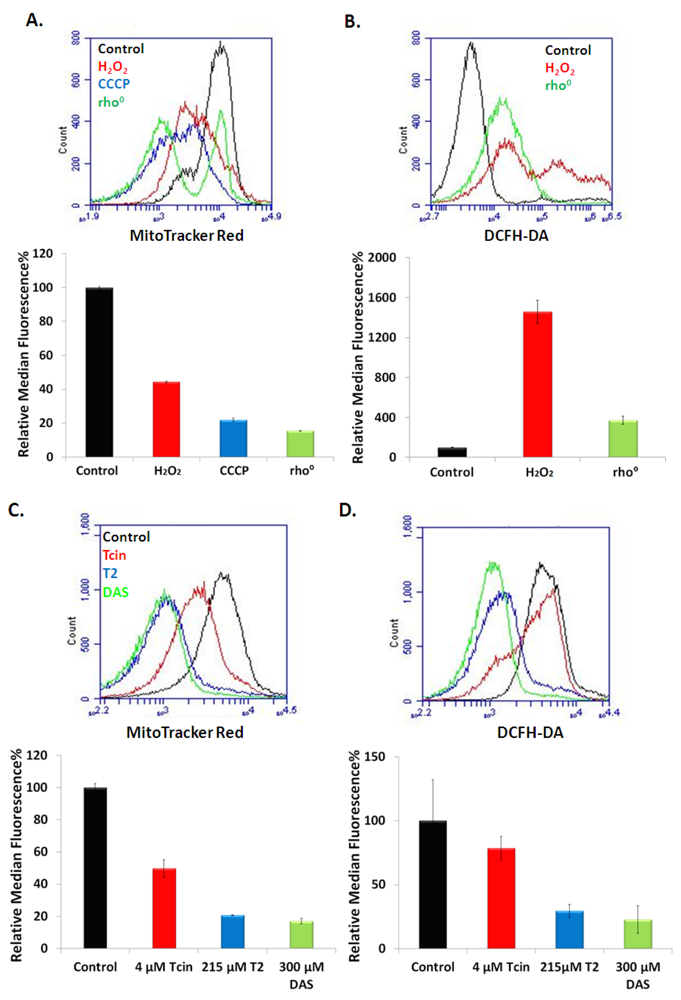

4. Discussion
4.1. Inhibition of Mitochondrial Translation by Trichothecenes Is Not a Secondary Effect of the Inhibition of Cytosolic Translation
4.2. Inhibition of Mitochondrial Translation by Trichothecenes Is Not Due to Mitochondrial Membrane Damage
4.3. Mitochondrial Membrane Damage by Trichothecenes Is Not a Secondary Effect of the Inhibition of Total Translation
5. Conclusions
Acknowledgments
Conflict of Interest
References
- Desjardins, A.E.; Hohn, T.M.; McCormick, S.P. Trichothecene biosynthesis in Fusarium species: Chemistry, genetics, and significance. Microbiol. Rev. 1993, 57, 595–604. [Google Scholar] [PubMed]
- McMullen, M.; Jones, R.; Gallenberg, D. Scab of wheat and barley: A re-emerging disease of devastating impact. Plant Dis. 1997, 81, 1340–1348. [Google Scholar]
- Windels, C.E. Economic and social impacts of Fusarium head blight: Changing farms and rural communities in the Northern Great Plains. Phytopathology 2000, 90, 17–21. [Google Scholar]
- Pestka, J.J. Mechanisms of deoxynivalenol-induced gene expression and apoptosis. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2008, 25, 1128–1140. [Google Scholar] [PubMed]
- McCormick, S.P.; Stanley, A.M.; Stover, N.A.; Alexander, N.J. Trichothecenes: From simple to complex mycotoxins. Toxins 2011, 3, 802–814. [Google Scholar]
- Fried, H.M.; Warner, J.R. Cloning of yeast gene for trichodermin resistance and ribosomal protein L3. Proc. Natl. Acad. Sci. USA 1981, 78, 238–242. [Google Scholar]
- Rocha, O.; Ansari, K.; Doohan, F.M. Effects of trichothecene mycotoxins on eukaryotic cells: A review. Food Addit. Contam. 2005, 22, 369–378. [Google Scholar]
- Cundliffe, E.; Cannon, M.; Davies, J. Mechanism of inhibition of eukaryotic protein synthesis by trichothecene fungal toxins. Proc. Natl. Acad. Sci. USA 1974, 71, 30–34. [Google Scholar]
- Cundliffe, E.; Davies, J.E. Inhibition of initiation, elongation, and termination of eukaryotic protein synthesis by trichothecene fungal toxin. Antimicrob. Agents. Chemother. 1977, 11, 491–499. [Google Scholar]
- Shifrin, V.I.; Anderson, P. Trichothecene mycotoxins trigger a ribotoxic stress response that activates c-Jun N-terminal kinase and p38 mitogen-activated protein kinase and induces apoptosis. J. Biol. Chem. 1999, 274, 13985–13992. [Google Scholar] [CrossRef] [PubMed]
- Iordanov, M.S.; Pribnow, D.; Magun, J.L.; Dinh, T.H.; Pearson, J.A.; Chen, S.L.; Magun, B.E. Ribotoxic stress response: Activation of the stress-activated protein kinase JNK1 by inhibitors of the peptidyl transferase reaction and by sequence-specific RNA damage to the alpha-sarcin/ricin loop in the 28S rRNA. Mol. Cell. Biol. 1997, 17, 3373–3381. [Google Scholar]
- Li, M.; Pestka, J.J. Comparative induction of 28S ribosomal RNA cleavage by ricin and the trichothecenes deoxynivalenol and T-2 toxin in the macrophage. Toxicol. Sci. 2008, 105, 67–78. [Google Scholar]
- Pace, J.G. Effect of T-2 mycotoxin on rat liver mitochondria electron transport system. Toxicon 1983, 21, 675–680. [Google Scholar]
- Schappert, K.T.; Khachatourians, G.G. Effects of T-2 toxin on induction of petite mutants and mitochondrial function in Saccharomyces cerevisiae. Curr. Genet. 1986, 10, 671–676. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.H.; Li, S.; Pestka, J.J. Down-regulation of the endoplasmic reticulum chaperone GRP78/BiP by vomitoxin (Deoxynivalenol). Toxicol. Appl. Pharmacol. 2000, 162, 207–217. [Google Scholar]
- Bushnell, W.; Perkins-Veazie, P.; Russo, V.; Collins, J.; Seeland, T. Effects of Deoxynivalenol on Content of Chloroplast Pigments in Barley Leaf Tissues. Phytopathology 2010, 100, 33–41. [Google Scholar]
- Bouaziz, C.; Martel, C. Fusarial toxin-induced toxicity in cultured cells and in isolated mitochondria involves PTPC-dependent activation of the mitochondrial pathway of apoptosis. Toxicol. Sci. 2009, 110, 363–375. [Google Scholar] [CrossRef] [PubMed]
- Jossé, L.; Li, X.; Coker, R.D.; Gourlay, C.W.; Evans, I.H. Transcriptomic and phenotypic analysis of the effects of T 2 toxin on Saccharomyces cerevisiae: Evidence of mitochondrial involvement. FEMS Yeast Res. 2010, 1, 133–150. [Google Scholar]
- Iwahashi, Y.; Kitagawa, E.; Iwahashi, H. Analysis of mechanisms of T-2 toxin toxicity using yeast DNA microarrays. Int. J. Mol. Sci. 2008, 9, 2585–2600. [Google Scholar]
- McLaughlin, J.E.; Bin-Umer, M.A.; Tortora, A.; Mendez, N.; McCormick, S.; Tumer, N.E. A genome-wide screen in Saccharomyces cerevisiae reveals a critical role for the mitochondria in the toxicity of a trichothecene mycotoxin. Proc. Natl. Acad. Sci. USA 2009, 106, 21883–21888. [Google Scholar]
- Hohn, T.M.; McCormick, S.P.; Desjardins, A.E. Evidence for a gene cluster involving trichothecene-pathway biosynthetic genes in Fusarium sporotrichioides. Curr. Genet. 1993, 24, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Barrientos, A.; Korr, D.; Tzagoloff, A. Shy1p is necessary for full expression of mitochondrial COX1 in the yeast model of Leigh’s syndrome. EMBO J. 2002, 21, 43–52. [Google Scholar]
- Diekert, K.; IPM, K. Isolation and subfractionation of mitochondria from the yeast Saccharomyces cerevisiae. Methods Cell Biol. 2001, 65, 37–51. [Google Scholar] [CrossRef] [PubMed]
- Kruger, N. The Bradford Method for Protein Quantitation. In The Protein Protocols Handbook; Humana Press: Totowa, NJ, USA, 2002; pp. 15–21. [Google Scholar]
- Westermann, B.; Neupert, W. Mitochondria targeted green fluorescent proteins: Convenient tools for the study of organelle biogenesis in Saccharomyces cerevisiae. Yeast 2000, 16, 1421–1427. [Google Scholar] [CrossRef] [PubMed]
- Visconti, A.; Minervini, F.; Lucivero, G.; Gambatesa, V. Cytotoxic and immunotoxic effects of Fusarium mycotoxins using a rapid colorimetric bioassay. Mycopathologia 1991, 113, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Funes, S.; Herrmann, J.M. Analysis of mitochondrial protein synthesis in yeast. Methods Mol. Biol. 2007, 372, 255–263. [Google Scholar]
- Chen, H.; Detmer, S.A.; Ewald, A.J.; Griffin, E.E.; Fraser, S.E.; Chan, D.C. Mitofusins Mfn1 and Mfn2 coordinately regulate mitochondrial fusion and are essential for embryonic development. J. Cell Biol. 2003, 160, 189–200. [Google Scholar]
- Chen, H.; Chomyn, A.; Chan, D.C. Disruption of fusion results in mitochondrial heterogeneity and dysfunction. J. Biol. Chem. 2005, 280, 26185–26189. [Google Scholar]
- Gong, D.W.; He, Y.; Karas, M.; Reitman, M. Uncoupling protein-3 is a mediator of thermogenesis regulated by thyroid hormone, 3-adrenergic agonists, and leptin. J. Biol Chem. 1997, 272, 24129–24132. [Google Scholar] [PubMed]
- Machida, K.; Tanaka, T.; Fujita, K.; Taniguchi, M. Farnesol-induced generation of reactive oxygen species via indirect inhibition of the mitochondrial electron transport chain in the yeast Saccharomyces cerevisiae. J. Bacteriol. 1998, 180, 4460–4465. [Google Scholar] [PubMed]
- Chaudhari, M.; Jayaraj, R.; Bhaskar, A.; Lakshmana Rao, P. Oxidative stress induction by T-2 toxin causes DNA damage and triggers apoptosis via caspase pathway in human cervical cancer cells. Toxicology 2009, 262, 153–161. [Google Scholar]
- Ludovico, P.; Rodrigues, F.; Almeida, A.; Silva, M.T.; Barrientos, A.; Corte-Real, M. Cytochrome c release and mitochondria involvement in programmed cell death induced by acetic acid in Saccharomyces cerevisiae. Mol. Biol. Cell 2002, 13, 2598–2606. [Google Scholar] [CrossRef] [PubMed]
- Ludovico, P.; Sousa, M.J.; Silva, M.T.; Leao, C.; Corte-Real, M. Saccharomyces cerevisiae commits to a programmed cell death process in response to acetic acid. Microbiology 2001, 147, 2409–2415. [Google Scholar] [PubMed]
- Whittemore, E.; Loo, D.; Watt, J.; Cotmans, C. A detailed analysis of hydrogen peroxide-induced cell death in primary neuronal culture. Neuroscience 1995, 67, 921–932. [Google Scholar]
- Dumont, A.; Hehner, S.P.; Hofmann, T.G.; Ueffing, M.; Droege, W.; Schmitz, M.L. Hydrogen peroxide-induced apoptosis is CD95-independent, requires the release of mitochondria-derived reactive oxygen species and the activation of NF-kappaB. Oncogene 1999, 18, 747–757. [Google Scholar]
- Pozniakovsky, A.I.; Knorre, D.A.; Markova, O.V.; Hyman, A.A.; Skulachev, V.P.; Severin, F.F. Role of mitochondria in the pheromone-and amiodarone-induced programmed death of yeast. J. Cell Biol. 2005, 168, 257–269. [Google Scholar]
- Eisenberg, T.; Büttner, S.; Kroemer, G.; Madeo, F. The mitochondrial pathway in yeast apoptosis. Apoptosis 2007, 12, 1011–1023. [Google Scholar]
- Ishihara, N.; Jofuku, A.; Eura, Y.; Mihara, K. Regulation of mitochondrial morphology by membrane potential, and DRP1-dependent division and FZO1-dependent fusion reaction in mammalian cells. Biochem. Biophys. Res. Commun. 2003, 301, 891–898. [Google Scholar]
- Chen, H.; Chomyn, A.; Chan, D.C. Disruption of fusion results in mitochondrial heterogeneity and dysfunction. J. Biol Chem. 2005, 280, 26185–26192. [Google Scholar]
- Legros, F.; Lombes, A.; Frachon, P.; Rojo, M. Mitochondrial fusion in human cells is efficient, requires the inner membrane potential, and is mediated by mitofusins. Mol. Biol. Cell. 2002, 13, 4343–4354. [Google Scholar] [CrossRef] [PubMed]
- Gasperi-Campani, A.; Brognara, I.; Baiocchi, D.; Roncuzzi, L. Mitochondrial DNA D-loop as a new target of Saporin 6 nuclease activity. Toxicon 2005, 45, 475–480. [Google Scholar]
- Tonshin, A.A.; Teplova, V.V.; Andersson, M.A.; Salkinoja-Salonen, M.S. The Fusarium mycotoxins enniatins and beauvericin cause mitochondrial dysfunction by affecting the mitochondrial volume regulation, oxidative phosphorylation and ion homeostasis. Toxicology 2010, 1, 49–57. [Google Scholar]
- Domijan, A.M.; Abramov, A.Y. Fumonisin B1 inhibits mitochondrial respiration and deregulates calcium homeostasis-Implication to mechanism of cell toxicity. Int. J. Biochem. Cell Biol. 2011, 43, 897–904. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bin-Umer, M.A.; McLaughlin, J.E.; Basu, D.; McCormick, S.; Tumer, N.E. Trichothecene Mycotoxins Inhibit Mitochondrial Translation—Implication for the Mechanism of Toxicity. Toxins 2011, 3, 1484-1501. https://doi.org/10.3390/toxins3121484
Bin-Umer MA, McLaughlin JE, Basu D, McCormick S, Tumer NE. Trichothecene Mycotoxins Inhibit Mitochondrial Translation—Implication for the Mechanism of Toxicity. Toxins. 2011; 3(12):1484-1501. https://doi.org/10.3390/toxins3121484
Chicago/Turabian StyleBin-Umer, Mohamed Anwar, John E. McLaughlin, Debaleena Basu, Susan McCormick, and Nilgun E. Tumer. 2011. "Trichothecene Mycotoxins Inhibit Mitochondrial Translation—Implication for the Mechanism of Toxicity" Toxins 3, no. 12: 1484-1501. https://doi.org/10.3390/toxins3121484





