A Review of Cyanophage–Host Relationships: Highlighting Cyanophages as a Potential Cyanobacteria Control Strategy
Abstract
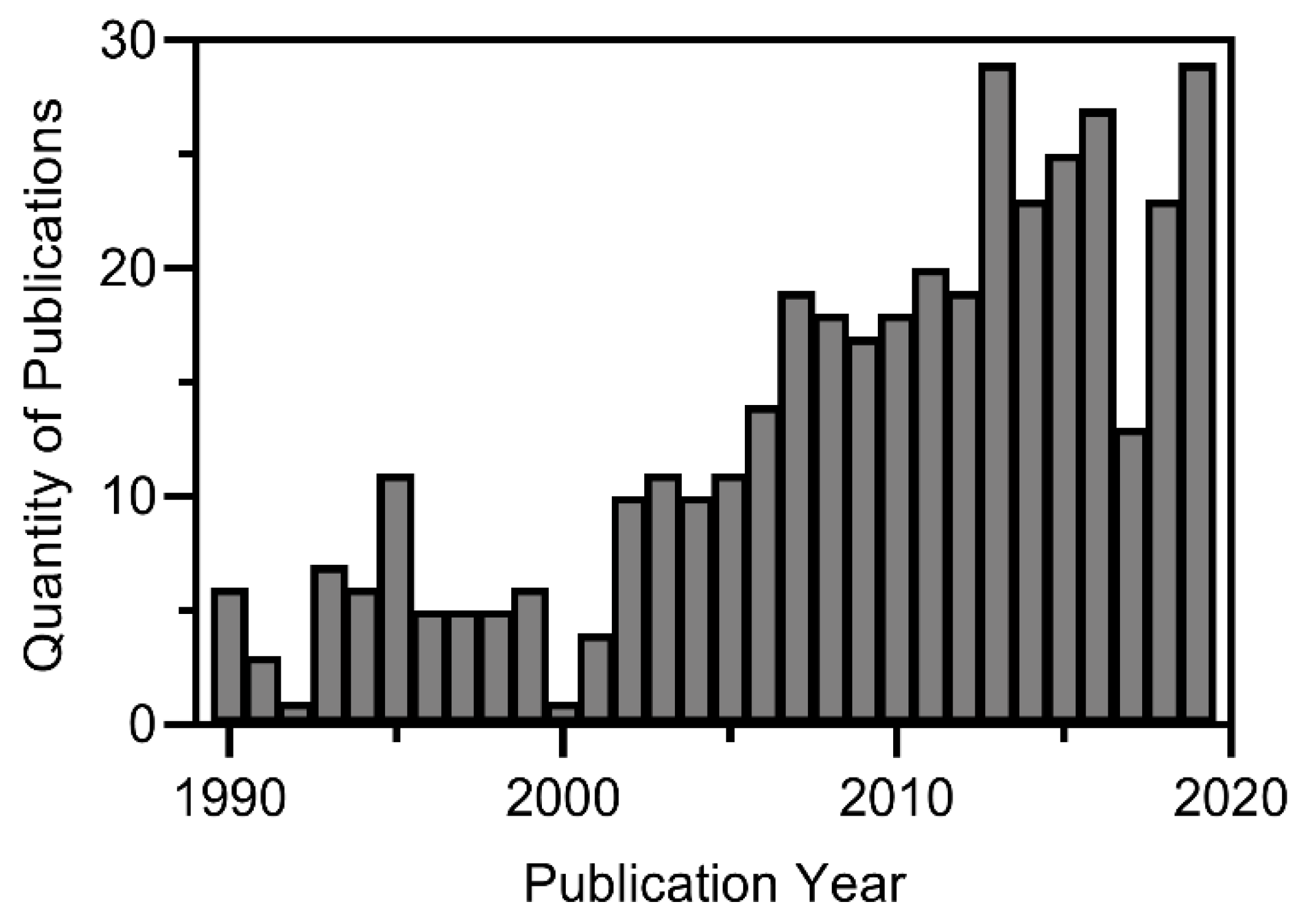
:1. Introduction
2. Cyanophages
2.1. Life Cycle
2.2. Diversity and Specificity
3. Factors Influencing Cyanophage Infectivity
3.1. Temperature
3.2. Nutrients
3.2.1. Phosphorous
3.2.2. Nitrogen
3.2.3. Carbon Dioxide
3.3. Irradiance
3.4. Cyanobacterial Extracellular Substances
3.5. Summary of Environmental Factors and Their Impact on Infectivity
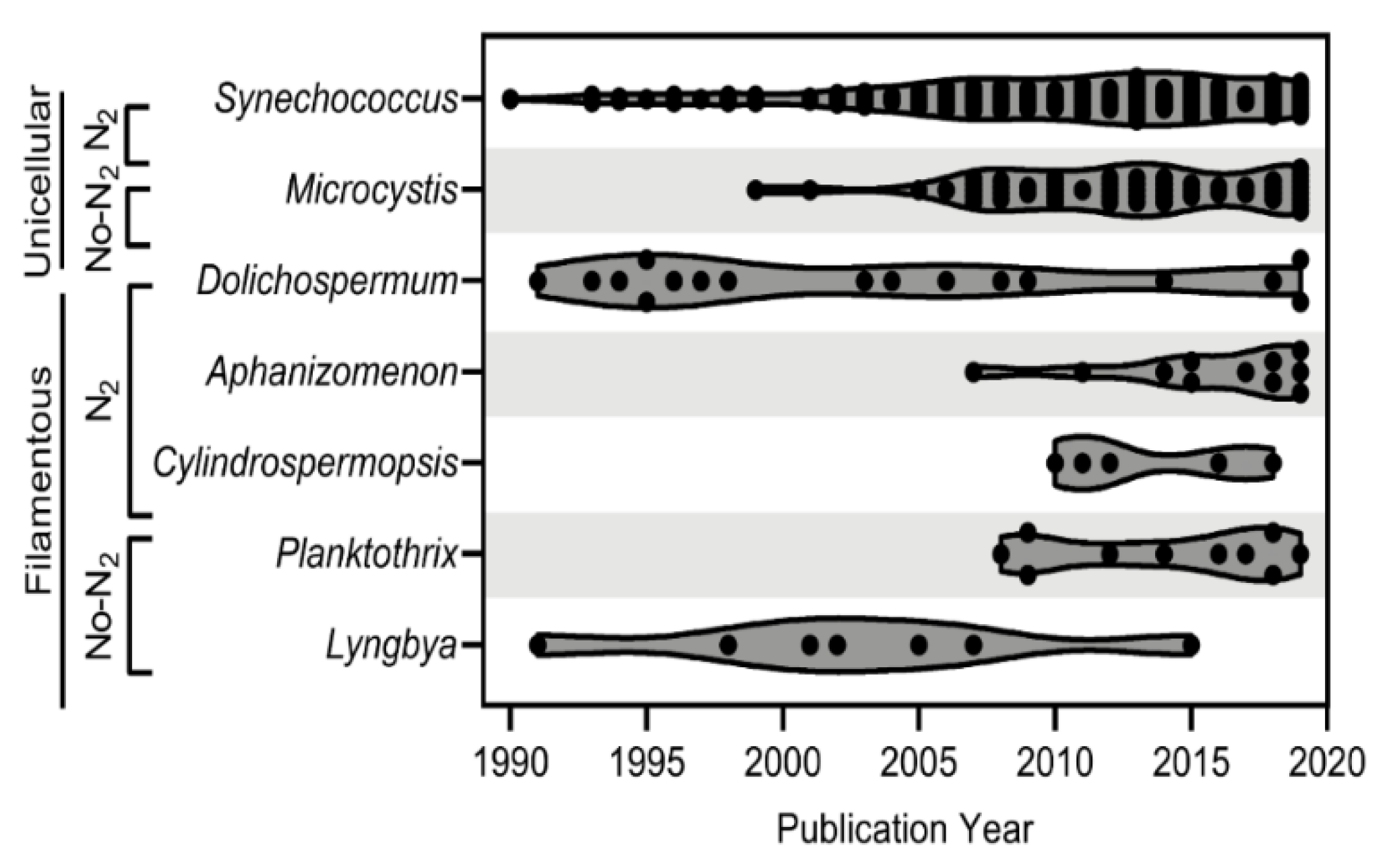
4. Cyanophage-Host Relationships
4.1. Unicellular/Colonial
4.1.1. Synechococcus
4.1.2. Microcystis
4.2. Filamentous Nitrogen-Fixers
4.2.1. Dolichospermum
4.2.2. Aphanizomenon
4.2.3. Cylindrospermopsis
4.3. Filamentous Non-Nitrogen-Fixers
4.3.1. Planktothrix
4.3.2. Lyngbya
5. Cyanophages as a Control Strategy
5.1. Efficacy Considerations
5.2. Optimization
5.3. Scalability
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Carmichael, W.W. Health Effects of Toxin-Producing Cyanobacteria: “The CyanoHABs”. Hum. Ecol. Risk Assess. Int. J. 2001, 7, 1393–1407. [Google Scholar] [CrossRef]
- Paerl, W.H.; Otten, T.G. Harmful cyanobacterial blooms: Causes, consequences, and controls. Environ. Microbiol. 2013, 65, 995–1010. [Google Scholar] [CrossRef]
- Ko, S.-R.; Srivastava, A.; Lee, N.; Jin, L.; Oh, H.-M.; Ahn, C.-Y. Bioremediation of eutrophic water and control of cyanobacterial bloom by attached periphyton. Int. J. Environ. Sci. Technol. 2019, 16, 4173–4180. [Google Scholar] [CrossRef]
- Funari, E.; Testai, E. Human Health Risk Assessment Related to Cyanotoxins Exposure. Crit. Rev. Toxicol. 2008, 38, 97–125. [Google Scholar] [CrossRef] [PubMed]
- Metcalf, J.S.; Codd, G.A. Cyanotoxins. In Ecology of Cyanobacteria II; Whitton, B., Ed.; Springer: Dodrecht, The Netherlands, 2012. [Google Scholar] [CrossRef]
- Suurnäkki, S.; Gomez-Saez, G.V.; Ylinen, A.H.; Jokela, J.; Fewer, D.; Sivonen, K. Identification of geosmin and 2-methylisoborneol in cyanobacteria and molecular detection methods for the producers of these compounds. Water Res. 2015, 68, 56–66. [Google Scholar] [CrossRef]
- Mooij, W.M.; Hülsmann, S.; Domis, L.N.D.S.; Nolet, B.A.; Bodelier, P.L.; Boers, P.C.; Pires, L.M.D.; Gons, H.J.; Ibelings, B.W.; Noordhuis, R.; et al. The impact of climate change on lakes in the Netherlands: A review. Aquat. Ecol. 2005, 39, 381–400. [Google Scholar] [CrossRef]
- Codd, G.A.; Morrison, L.F.; Metcalf, J. Cyanobacterial toxins: Risk management for health protection. Toxicol. Appl. Pharmacol. 2005, 203, 264–272. [Google Scholar] [CrossRef]
- Bláha, L.; Babica, P.; Maršálek, B. Toxins produced in cyanobacterial water blooms—Toxicity and risks. Interdiscip. Toxicol. 2009, 2, 36–41. [Google Scholar] [CrossRef] [Green Version]
- O’Neil, J.M.; Davis, T.W.; Burford, M.A.; Gobler, C.J. The rise of harmful cyanobacteria blooms: The potential roles of eutrophication and climate change. Harmful Algae 2012, 14, 313–334. [Google Scholar] [CrossRef]
- Huisman, J.; Codd, G.A.; Paerl, H.W.; Ibelings, B.W.; Verspagen, J.M.H.; Visser, P.M. Cyanobacterial blooms. Nat. Rev. Genet. 2018, 16, 471–483. [Google Scholar] [CrossRef]
- Carey, C.C.; Ibelings, B.W.; Hoffmann, E.P.; Hamilton, D.P.; Brookes, J.D. Eco-physiological adaptations that favour freshwater cyanobacteria in a changing climate. Water Res. 2012, 46, 1394–1407. [Google Scholar] [CrossRef] [PubMed]
- Boesch, F.D.; Anderson, D.M.; Horner, R.A.; Shumway, S.E.; Tester, P.A.; Whitledge, T.E. Harmful Algal Blooms in Coastal Waters: Options for Prevention, Control, and Mitigation; NOAA Coastal Ocean Program Decision Analysis Series No.10; NOAA Coastal Ocean Office: Silver Spring, MD, USA, 1997; p. 47. [Google Scholar]
- USEPA (United States Environmental Protection Agency). Control Measures for Cyanobacterial HABs in Surface Water. 2020. Available online: https://www.epa.gov/cyanohabs/control-measures-cyanobacterial-habs-surface-water (accessed on 14 July 2020).
- Burford, A.M.; Gobler, C.J.; Hamilton, D.P.; Visser, P.M.; Lurling, M.; Codd, G.A. Solutions for Managing Cyanobacterial Blooms: A Scientific Summary for Policy Makers; IOC/INF-1382; IOC/UNESCO: Paris, France, 2019. [Google Scholar]
- California Water Quality Monitoring Council. Algae Mitigation Technique Selection Process for Lakes. 2020. Available online: https://mywaterquality.ca.gov/habs/resources/docs/flow_chart_draft_20190515.pdf (accessed on 14 July 2020).
- ITRC (Interstate Technology and Regulatory Council). Strategies for Preventing and Managing Harmful Cyanobacterial Blooms (HCBs). 2020. Available online: https://hcb-1.itrcweb.org/ (accessed on 14 July 2020).
- NEIWPCC (New England Interstate Water Pollution Control Commission). Harmful Algal Bloom Control Methods Synopses. 2015. Available online: http://www.neiwpcc.org/neiwpcc_docs/NEIWPCC_HABControlMethodsSynopses_June2015.pdf (accessed on 14 July 2020).
- Newcombe, G.; House, J.; Ho, L.; Baker, P.; Burch, M. Management Strategies for Cyanobacteria (Blue-Green Algae): A Guide for Water Utilities; Research Report 74; Water Quality Research: Adelaide, Australia, 2010. [Google Scholar]
- Rajasekhar, P.; Fan, L.; Nguyen, T.; Roddick, F.A. A review of the use of sonication to control cyanobacterial blooms. Water Res. 2012, 46, 4319–4329. [Google Scholar] [CrossRef] [PubMed]
- Lurling, M.; Waajen, G.; de Senerpoint Domis, L.N. Evaluation of several end-of-pipe measures proposed to control cyanobacteria. Aquat. Ecol. 2016, 50, 499–519. [Google Scholar] [CrossRef] [Green Version]
- Piel, T.; Sandrini, G.; White, E.; Xu, T.; Schuurmans, J.M.; Huisman, J.; Visser, P.M. Suppressing Cyanobacteria with Hydrogen Peroxide Is More Effective at High Light Intensities. Toxins 2019, 12, 18. [Google Scholar] [CrossRef] [Green Version]
- Breda-Alves, F.; de Oliveira Fernandes, V.; Chia, M.A. Understanding the environmental roles of herbicides on cyano-bacteria, cyanotoxins, and cyanoHABs. Aquat. Ecol. 2021, 55, 347–361. [Google Scholar] [CrossRef]
- Sukenik, A.; Kaplan, A. Cyanobacterial Harmful Algal Blooms in Aquatic Ecosystems: A Comprehensive Outlook on Current and Emerging Mitigation and Control Approaches. Microorganisms 2021, 9, 1472. [Google Scholar] [CrossRef]
- Yoshida, T.; Takashima, Y.; Tomaru, Y.; Shirai, Y.; Takao, Y.; Hiroishi, S.; Nagasaki, K. Isolation and Characterization of a Cyanophage Infecting the Toxic Cyanobacterium Microcystis Aeruginosa. Appl. Environ. Microbiol. 2006, 72, 1239–1247. [Google Scholar] [CrossRef] [Green Version]
- Deng, L.; Hayes, P.K. Evidence for cyanophages active against bloom-forming freshwater cyanobacteria. Freshw. Biol. 2008, 53, 1240–1252. [Google Scholar] [CrossRef]
- Weinbauer, M.G. Ecology of prokaryotic viruses. FEMS Microbiol. Rev. 2004, 28, 127–181. [Google Scholar] [CrossRef] [Green Version]
- Singh, P.; Singh, S.S.; Srivastava, A.; Singh, A.; Mishra, A.K. Structural, functional and molecular basis of cyanophage-cyanobacterial interactions and its significance. Afr. J. Biotechnol. 2012, 11, 2591–2608. [Google Scholar] [CrossRef]
- Catalao, M.J.; Gil, F.; Moniz-Pereira, J.; São-José, C.; Pimentel, M. Diversity in bacterial lysis systems: Bacteriophages show the way. FEMS Microbiol. Rev. 2013, 37, 554–571. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ortmann, A.; Lawrence, J.; Suttle, C. Lysogeny and Lytic Viral Production during a Bloom of the Cyanobacterium Synechococcus spp. Microb. Ecol. 2002, 43, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Jassim, S.A.A.; Limoges, R.G. Impact of external forces on cyanophage–host interactions in aquatic ecosystems. World J. Microbiol. Biotechnol. 2013, 29, 1751–1762. [Google Scholar] [CrossRef] [PubMed]
- Dorigo, U.; Jacquet, S.; Humbert, J.-F. Cyanophage diversity, inferred from g20 gene analyses, in the largest natural lake in France, Lake Bourget. Appl. Environ. Microbiol. 2004, 70, 1017–1022. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, E.-B.; Huang, Y.; Ning, D. Metabolic Genes within Cyanophage Genomes: Implications for Diversity and Evolution. Genes 2016, 7, 80. [Google Scholar] [CrossRef]
- Finke, J.F.; Suttle, C.A. The Environment and Cyanophage Diversity: Insights from Environmental Sequencing of DNA Polymerase. Front. Microbiol. 2019, 10, 167. [Google Scholar] [CrossRef] [Green Version]
- Safferman, R.; Cannon, R.; Desjardins, P.; Gromov, B.; Haselkorn, R.; Sherman, L.; Shilo, M. Classification and Nomenclature of Viruses of Cyanobacteria. Intervirology 1983, 19, 61–66. [Google Scholar] [CrossRef]
- Safferman, R.S.; Morris, M.-E. Growth characteristics of the blue-green algal virus LPP-1. J. Bacteriol. 1964, 88, 771–775. [Google Scholar] [CrossRef] [Green Version]
- Padan, E.; Shilo, M. Cyanophages-viruses attacking blue-green algae. Bacteriol. Rev. 1973, 37, 343–370. [Google Scholar] [CrossRef]
- Xia, H.; Li, T.; Deng, F.; Hu, Z. Freshwater cyanophages. Virol. Sin. 2013, 28, 253–259. [Google Scholar] [CrossRef]
- Yoshida, M.; Yoshida, T.; Kashima, A.; Takashima, Y.; Hosoda, N.; Nagasaki, K.; Hiroishi, S. Ecological Dynamics of the Toxic Bloom-Forming Cyanobacterium Microcystis Aeruginosa and Its Cyanophages in Freshwater. Appl. Environ. Microbiol. 2008, 74, 3269–3273. [Google Scholar] [CrossRef] [Green Version]
- Morimoto, D.; Tominaga, K.; Nishimura, Y.; Yoshida, N.; Kimura, S.; Sako, Y.; Yoshida, T. Coocurrence of broad- and narrow-host-range viruses infecting the bloom-forming toxic cyanobacterium Microcystis Aeruginosa. Appl. Environ. Microbiol. 2019, 85, e01170-19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sullivan, M.B.; Coleman, M.; Weigele, P.; Rohwer, F.; Chisholm, S.W. Three Prochlorococcus Cyanophage Genomes: Signature Features and Ecological Interpretations. PLoS Biol. 2005, 3, e144. [Google Scholar] [CrossRef] [Green Version]
- Yoshida, T.; Nagasaki, K.; Takashima, Y.; Shirai, Y.; Tomaru, Y.; Takao, Y.; Sakamoto, S.; Hiroishi, S.; Ogata, H. Ma-LMM01 Infecting Toxic Microcystis Aeruginosa Illuminates Diverse Cyanophage Genome Strategies. J. Bacteriol. 2008, 190, 1762–1772. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wilson, W.H.; Joint, I.R.; Carr, N.G.; Mann, N.H. Isolation and Molecular Characterization of Five Marine Cyanophages Propagated on Synechococcus sp. Strain WH7803. Appl. Environ. Microbiol. 1993, 59, 3736–3743. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, K.; Chen, F. Genetic diversity and population dynamics of cyanophage communities in the Chesapeake Bay. Aquat. Microb. Ecol. 2004, 34, 105–116. [Google Scholar] [CrossRef] [Green Version]
- Jakulska, A.; Mankiewicz-Boczek, J. Cyanophages specific to cyanobacteria from the genus Microcystis. Int. J. Ecohydrol. Hydrobiol. 2020, 20, 83–90. [Google Scholar] [CrossRef]
- Miskiewicz, E.; Ivanov, A.G.; Williams, J.P.; Khan, M.U.; Falk, S.; Huner, N.P. Photosynthetic acclimation of the filamentous cyanobacterium, Plectonema boryanum UTEX 485, to temperature and light. Plant Cell Physiol. 2000, 41, 767–775. [Google Scholar] [CrossRef]
- Paerl, H.W. Mitigating Harmful Cyanobacterial Blooms in a Human- and Climatically-Impacted World. Life 2014, 4, 988–1012. [Google Scholar] [CrossRef] [Green Version]
- Bratbak, G.; Heldal, M.; Norland, S.; Thingstad, T.F. Viruses as Partners in Spring Bloom Microbial Trophodynamics. Appl. Environ. Microbiol. 1990, 56, 1400–1405. [Google Scholar] [CrossRef] [Green Version]
- Suttle, C.A.; Chen, F. Mechanisms and Rates of Decay of Marine Viruses in Seawater. Appl. Environ. Microbiol. 1992, 58, 3721–3729. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Manage, M.P.; Kawabata, Z.; Nakano, S.-I. Dynamics of cyanophage-like particles and algicidal bacteria causing Microcystis Aeruginosa mortality. Limnology 1999, 2, 73–78. [Google Scholar] [CrossRef]
- Safferman, R.; Schneider, I.; Steere, R.; Morris, M.; Diener, T. Phycovirus SM-1: A virus infecting unicellular blue-green algae. Virology 1969, 37, 386–395. [Google Scholar] [CrossRef]
- Safferman, R.; Diener, T.; Desjardins, P.; Morris, M. Isolation and characterization of AS-1, a phycovirus infecting the blue-green algae, Anacystis nidulans and Synechococcus cedrorum. Virology 1972, 47, 105–113. [Google Scholar] [CrossRef]
- Cheng, K.; Van de Waal, D.; Niu, X.Y.; Zhao, Y.J. Combined Effects of Elevated pCO2 and Warming Facilitate Cyanophage Infections. Front. Microbiol. 2017, 8, 1096. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murray, A.; Jackson, G. Viral dynamics: A model of the effects of size shape, motion and abundance of single-celled olanktonic organisms and other particles. Mar. Ecol. Prog. Ser. 1992, 89, 103–116. [Google Scholar] [CrossRef]
- Chu, T.-C.; Murray, S.R.; Hsu, S.; Vega, Q.; Lee, L.H. Temperature-induced activation of freshwater cyanophage AS-1 prophage. Acta Histochem. 2011, 113, 294–299. [Google Scholar] [CrossRef] [Green Version]
- Pick, F.R.; Lean, D.R.S. The role of macronutrients (C, N, P) in controlling cyanobacterial dominance in temperate lakes. N. Z. J. Mar. Freshw. Res. 1987, 21, 425–434. [Google Scholar] [CrossRef]
- Parrish, J. The Role of Nitrogen and Phosphorus in the Growth, Toxicity, and Distribution of the Toxic Cyanobacteria Microcystis Aeruginosa. Master’s Projects and Capstones. 2014. Available online: https://repository.usfca.edu/capstone/8 (accessed on 4 December 2020).
- Zachary, A. An ecological study of bacteriophages of Vibrio natriegens. Can. J. Microbiol. 1978, 24, 321–324. [Google Scholar] [CrossRef]
- Gobler, C.J.; Burkholder, J.M.; Davis, T.W.; Harke, M.J.; Johengen, T.; Stow, C.; Van de Waal, D. The dual role of nitrogen supply in controlling the growth and toxicity of cyanobacterial blooms. Harmful Algae 2016, 54, 87–97. [Google Scholar] [CrossRef]
- Bulgakov, N.G.; Levich, A.P. The nitrogen: Phosphorus ratio as a factor regulating phytoplankton community structure. Fundam. Appl. Limnol. 1999, 146, 3–22. [Google Scholar] [CrossRef] [Green Version]
- Davidson, K.; Gowen, R.J.; Tett, P.; Bresnan, E.; Harrison, P.J.; McKinney, A.; Milligan, S.; Mills, D.K.; Silke, J.; Crooks, A.M. Harmful algal blooms: How strong is the evidence that nutrient ratios and forms influence their occurrence? Estuar. Coast. Shelf Sci. 2012, 115, 399–413. [Google Scholar] [CrossRef]
- Davis, W.T.; Bullerjahn, G.S.; Tuttle, T.; McKay, R.M.; Watson, S.B. Effects of increasing nitrogen and phosphorous concentrations on phytoplankton community growth and toxicity during Planktothrix blooms in Sandusky Bay, Lake Erie. Environ. Sci. Technol. 2015, 49, 7197–7207. [Google Scholar] [CrossRef] [PubMed]
- Zimmerman, A.E.; Howard-Varona, C.; Needham, D.M.; John, S.G.; Worden, A.Z.; Sullivan, M.B.; Waldbauer, J.R.; Coleman, M.L. Metabolic and biogeochemical consequences of viral infection in aquatic ecosystems. Nat. Rev. Microbiol. 2019, 18, 1–14. [Google Scholar] [CrossRef]
- Waldbauer, J.R.; Coleman, M.L.; Rizzo, A.I.; Campbell, K.L.; Lotus, J.; Zhang, L. Nitrogen sourcing during viral infection of marine cyanobacteria. Proc. Natl. Acad. Sci. USA 2019, 116, 15590–15595. [Google Scholar] [CrossRef] [Green Version]
- Wilson, W.H.; Carr, N.G.; Mann, N.H. The effect of phosphate status on the kinetics of cyanophage infection in the oceanic Cyanobacterium synechococcus sp. WH78031. J. Phycol. 1996, 32, 506–516. [Google Scholar] [CrossRef]
- Williamson, S.J.; Houchin, L.A.; McDaniel, L.; Paul, J.H. Seasonal Variation in Lysogeny as Depicted by Prophage Induction in Tampa Bay, Florida. Appl. Environ. Microbiol. 2002, 68, 4307–4314. [Google Scholar] [CrossRef] [Green Version]
- Rihtman, B. Viral Infection of Marine Picoplankton under Nutrient Depletion Conditions: Pseudolysogeny and Magic Spot Nucleotides. Ph.D. Thesis, University of Warwick, Coventry, UK, 2016. Available online: http://webcat.warwick.ac.uk/record=b3069055~S15 (accessed on 4 December 2020).
- Zeng, Q.; Chisholm, S. Marine viruses exploit their host’s two-component regulatory system in response to resource limitation. Curr. Biol. 2012, 22, 124–128. [Google Scholar] [CrossRef] [Green Version]
- Mankiewicz-Boczek, J.; Jaskulska, A.; Pawełczyk, J.; Gągała, I.; Serwecińska, L.; Dziadek, J. Cyanophages infection of Microcystis bloom in lowland dam reservoir of Sulejow, Poland. Microb. Ecol. 2016, 71, 315–325. [Google Scholar] [CrossRef]
- Cheng, K.; Frenken, T.; Brussaard, C.P.D.; Van de Waal, D. Cyanophage Propagation in the Freshwater Cyanobacterium Phormidium Is Constrained by Phosphorus Limitation and Enhanced by Elevated pCO2. Front. Microbiol. 2019, 10, 617. [Google Scholar] [CrossRef]
- McKindles, K. The Effect of Phosphorus and Nitrogen Limitation on Viral Infection in Microcystis Aeruginosa NIES298 Using the Cyanophage Ma-LMM01; Eastern Michigan University: Ypsilanti, MI, USA, 2017; Available online: https://commons.emich.edu/theses/741 (accessed on 4 December 2020).
- McDaniel, L.; Paul, J.H. Effect of nutrient addition and environmental factors on prophage induction in natural populations of marine Synechococcus species. Appl. Environ. Microbiol. 2004, 71, 842–850. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, Q.; Gao, Y.; Zhao, Y.; Cheng, K. The effect of elevated carbon dioxide concentration on cyanophage PP multiplication and photoreactivation induced by a wild host cyanobacterium. Acta Ecol. Sin. 2015, 35, 11–15. [Google Scholar] [CrossRef]
- Verschoor, M.J.; Powe, C.R.; McQuay, E.; Schiff, S.L.; Venkiteswaran, J.J.; Li, J.; Molot, L.A. Internal iron loading and warm temperatures are preconditions for cyanobacterial dominance in embayments along Georgian Bay, Great Lakes. Can. J. Fish. Aquat. Sci. 2017, 74, 1439–1453. [Google Scholar] [CrossRef]
- Benson, R.; Martin, E. Physicochemical characterization of cyanophage SM-2. Arch. Microbiol. 1984, 140, 212–214. [Google Scholar] [CrossRef]
- Traving, S.J.; Clokie, M.R.; Middelboe, M. Increased acidification has a profound effect on the interactions between the cyanobacterium Synechococcus sp. WH7803 and its viruses. FEMS Microbiol. Ecol. 2013, 87, 133–141. [Google Scholar] [CrossRef] [Green Version]
- Davis, T.W.; Berry, D.L.; Boyer, G.L.; Gobler, C.J. The effects of temperature and nutrients on the growth and dynamics of toxic and non-toxic strains of Microcystis during cyanobacteria blooms. Harmful Algae 2009, 8, 715–725. [Google Scholar] [CrossRef]
- Bozarth, C.S.; Schwartz, A.D.; Shepardson, J.W.; Colwell, F.S.; Dreher, T.W. Population Turnover in a Microcystis Bloom Results in Predominantly Nontoxigenic Variants Late in the Season. Appl. Environ. Microbiol. 2010, 76, 5207–5213. [Google Scholar] [CrossRef] [Green Version]
- Kardinaal, W.; Janse, I.; Agterveld, M.K.-V.; Meima, M.; Snoek, J.; Mur, L.; Huisman, J.; Zwart, G.; Visser, P. Microcystis genotype succession in relation to microcystin concentrations in freshwater lakes. Aquat. Microb. Ecol. 2007, 48, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Zilliges, Y.; Kehr, J.-C.; Meissner, S.; Ishida, K.; Mikkat, S.; Hagemann, M.; Kaplan, A.; Börner, T.; Dittmann, E. The Cyanobacterial Hepatotoxin Microcystin Binds to Proteins and Increases the Fitness of Microcystis under Oxidative Stress Conditions. PLoS ONE 2011, 6, e17615. [Google Scholar] [CrossRef] [Green Version]
- Suttle, C.A. 6—Ecological, evolutionary, and geochemical consequences of viral infection of cyanobacteria and eukaryotic algae. In Viral Ecology; Hurst, C.J., Ed.; Academic Press: Cambridge, MA, USA, 2000; pp. 247–296. ISBN 9780123626752. [Google Scholar]
- Cleaver, J.E. IV—Photoreactivation. Adv. Radiat. Biol. 1974, 4, 1–75. [Google Scholar]
- Ni, T.; Zeng, Q. Diel Infection of Cyanobacteria by Cyanophages. Front. Mar. Sci. 2016, 2, 123. [Google Scholar] [CrossRef] [Green Version]
- Sherman, L.A. Infection of Synechococcus cedrorum by the cyanophage AS-1M. III. Cellular metabolism and phage development. Virology 1976, 71, 199–206. [Google Scholar] [CrossRef]
- Mackenzie, J.J.; Haselkorn, R. An electron microscope study of infection by the blue-green algal virus SM-1. Virology 1972, 49, 505–516. [Google Scholar] [CrossRef]
- Teklemariam, A.T.; Demeter, S.; Deak, Z.; Suryani, G.; Borebely, G. AS-1 cyanophage infection inhibits the photosynthetic electron flow of photosystem II in Synechococcus sp. PCC 6301, a cyanobacterium. FEBS Lett. 1990, 270, 211–215. [Google Scholar] [CrossRef] [Green Version]
- Yoshida-Takashima, Y.; Yoshida, M.; Ogata, H.; Nagasaki, K.; Hiroishi, S.; Yoshida, T. Cyanophage Infection in the Bloom-Forming Cyanobacteria Microcystis Aeruginosa in Surface Freshwater. Microbes Environ. 2012, 27, 350–355. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nakamura, G.; Kimura, S.; Sako, Y.; Yoshida, T. Genetic diversity of Microcystis cyanophages in two different freshwater environments. Arch. Microbiol. 2014, 196, 401–409. [Google Scholar] [CrossRef]
- De Philippis, R.; Sili, C.; Paperi, R.; Vincenzini, M. Exopolysaccharide-producing cyanobacteria and their possible exploitation: A review. J. Appl. Phycol. 2001, 13, 293–299. [Google Scholar] [CrossRef]
- De Philippis, R.; Colica, G.; Micheletti, E. Exopolysaccharide-producing cyanobacteria in heavy metal removal from water: Molecular basis and practical applicability of the biosorption process. Appl. Microbiol. Biotechnol. 2011, 92, 697–708. [Google Scholar] [CrossRef]
- Kehr, J.-C.; Dittmann, E. Biosynthesis and Function of Extracellular Glycans in Cyanobacteria. Life 2015, 5, 164–180. [Google Scholar] [CrossRef] [Green Version]
- Baulina, O.I.; Titel, K.; Gorelova, O.A.; Malai, O.V.; Ehwald, R. Permeability of cyanobacterial mucous surface structures for macromolecules. Microbiology 2008, 77, 198–205. [Google Scholar] [CrossRef]
- Abedon, S.T. Phage “delay” towards enhancing bacterial escape from biofilms: A more comprehensive way of viewing resistance to bacteriophages. AIMS Microbiol. 2017, 3, 186–226. [Google Scholar] [CrossRef]
- Abedon, S. Bacteriophage exploitation of bacterial biofilms: Phage preference for less mature targets? FEMS Microbiol. Lett. 2016, 363, fnv246. [Google Scholar] [CrossRef] [PubMed]
- Hughes, K.; Sutherland, I.; Clark, J.; Jones, M. Bacteriophage and associated polysaccharide depolymerases—Novel tools for study of bacterial biofilms. J. Appl. Microbiol. 1998, 85, 583–590. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Ou, T.; Zhang, Q. Two virus-like particles that cause lytic infections in freshwater cyanobacteria. Virol. Sin. 2013, 28, 303–305. [Google Scholar] [CrossRef]
- Jiang, X.; Ha, C.; Lee, S.; Kwon, J.; Cho, H.; Gorham, T.; Lee, J. Characterization of Cyanophages in Lake Erie: Interaction Mechanisms and Structural Damage of Toxic Cyanobacteria. Toxins 2019, 11, 444. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Coello-Camba, A.; Diaz-Rua, R.; Duarte, C.M.; Irigoien, X.; Pearman, J.K.; Alam, I.S.; Agusti, S. Picocyanobacteria Community and Cyanophage Infection Responses to Nutrient Enrichment in a Mesocosms Experiment in Oligotrophic Waters. Front. Microbiol. 2020, 11, 1153. [Google Scholar] [CrossRef]
- Safferman, R.S.; Morris, M.E. Algal virus: Isolation. Science 1963, 140, 679–680. [Google Scholar] [CrossRef]
- World Health Organization. Cyanobacterial Toxins: Microcystin-LR in Drinking Water; World Health Organization: Geneva, Switzerland, 2003. [Google Scholar]
- Phlips, E.J.; Zeman, C.; Hansen, P. Growth, photosynthesis, nitrogen fixation and carbohydrate production by a unicellular cyanobacterium, Synechococcus sp. (Cyanophyta). J. Appl. Phycol. 1989, 1, 137–145. [Google Scholar] [CrossRef]
- Huang, W.-J.; Cheng, Y.-L.; Cheng, B.-L. Ozonation By-products and Determination of Extracellular Release in Freshwater Algae and Cyanobacteria. Environ. Eng. Sci. 2008, 25, 139–152. [Google Scholar] [CrossRef]
- Beversdorf, L.J.; Miller, T.R.; McMahon, K.D. The Role of Nitrogen Fixation in Cyanobacterial Bloom Toxicity in a Temperate, Eutrophic Lake. PLoS ONE 2013, 8, e56103. [Google Scholar] [CrossRef] [Green Version]
- Rolff, C.; Almesjo, L.; Elmgren, R. Nitrogen fixation and abundance of the diazotrophic cyanobacterium Aphanizomenon sp. in the Baltic Proper. MEPS 2007, 332, 107–118. [Google Scholar] [CrossRef] [Green Version]
- Karlson, A.M.L.; Duberg, J.; Motwani, N.H.; Hogfors, H.; Klawonn, I.; Ploug, H.; Svedén, J.B.; Garbaras, A.; Sundelin, B.; Hajdu, S.; et al. Nitrogen fixation by cyanobacteria stimulates production in Baltic food webs. Ambio 2015, 44, 413–426. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Willis, A.; Chuang, A.W.; Woodhouse, J.N.; Neilan, B.A.; Burford, M.A. Intraspecific variation in growth, morphology and toxin quotas for the cyanobacterium, Cylindrospermopsis raciborskii. Toxicon 2016, 119, 307–310. [Google Scholar] [CrossRef] [PubMed]
- Omoregie, O.E.; Crumbliss, L.L.; Bebout, B.M.; Zehr, J.P. Determination of nitrogen-fixing phylotypes in Lyngbya sp. and Microcoleus chthonoplastes cyanobacterial mats from Guerrero Negro, Baja California, Mexico. Appl. Environ. Microbiol. 2004, 70, 2119–2128. [Google Scholar] [CrossRef] [Green Version]
- Jones, A.C.; Monroe, E.A.; Podell, S.; Hess, W.R.; Klages, S.; Esquenazi, E.; Niessen, S.; Hoover, H.; Rothmann, M.; Lasken, R.S. Genomic insights into the physiology and ecology of the marine filamentous cyanobacterium Lyngbya majuscule. Proc. Natl. Acad. Sci. USA 2011, 108, 8815–8820. [Google Scholar] [CrossRef] [Green Version]
- Pancrace, C.; Jokela, J.; Sassoon, N.; Ganneau, C.; Desnos-Ollivier, M.; Wahlsten, M.; Humisto, A.; Calteau, A.; Bay, S.; Fewer, D.P.; et al. Rearranged biosynthetic gene cluster and synthesis of Hassalladin E in Planktothrix serta PCC 8927. ACS Chem. Biol. 2017, 12, 1796–1804. [Google Scholar] [CrossRef] [Green Version]
- Fox, J.A.; Booth, S.; Martin, E. Cyanophage SM-2: A new blue-green algal virus. Pathol. Microbiol. 1976, 73, 557–560. [Google Scholar] [CrossRef]
- Tucker, S.; Pollard, P. Identification of Cyanophage Ma-LBP and Infection of the Cyanobacterium Microcystis Aeruginosa from an Australian Subtropical Lake by the Virus. Appl. Environ. Microbiol. 2005, 71, 629–635. [Google Scholar] [CrossRef] [Green Version]
- Hargreaves, K.R.; Anderson, N.J.; Clokie, M.R. Recovery of viable cyanophages from the sediments of a eutrophic lake at decadal timescales. FEMS Microbiol. Ecol. 2012, 83, 450–456. [Google Scholar] [CrossRef]
- Ou, T.; Li, S.; Liao, X.; Zhang, Q. Cultivation and characterization of the MaMV-DC cyanophage that infects bloom-forming cyanobacterium Microcystis Aeruginosa. Virol. Sin. 2013, 28, 266–271. [Google Scholar] [CrossRef]
- Wang, J.; Bai, P.; Li, Q.; Lin, Y.; Huo, D.; Ke, F.; Zhang, Q.; Li, T.; Zhao, J. Interaction between cyanophage MaMV-DC and eight Microcystis strains, revealed by genetic defense systems. Harmful Algae 2019, 85, 101699. [Google Scholar] [CrossRef]
- Currier, T.C.; Wolk, C.P. Characteristics of Anabaena variabilis influencing plaque formation by cyanophage N-1. J. Bacteriol. 1979, 139, 88–92. [Google Scholar] [CrossRef] [Green Version]
- Koz’yakov, S.Y. Cyanophages of the series A(L) specific for the blue-green alga Anabaena variabilis. Exp. Algol. Biol. Sci. Res. 1977, 25, 151–175. [Google Scholar]
- Hu, N.-T.; Thiel, T.; Giddings, T.H.; Wolk, C. New Anabaena and Nostoc cyanophages from sewage settling ponds. Virology 1981, 114, 236–246. [Google Scholar] [CrossRef]
- Monegue, R.L.; Phlips, E.J. The effect of cyanophages on the growth and survival of Lyngbya wollei, Anabaena flos-aquae, and Anabaena circinalis. J. Aquat. Plant Manag. 1991, 29, 88–93. [Google Scholar]
- Granhall, U. Aphanizomenon flos-aquae: Infection by Cyanophages. Physiol. Plant. 1972, 26, 332–337. [Google Scholar] [CrossRef]
- Coulombe, A.C.; Robinson, G.G.C. Collapsing Aphanizomenon flos-aquae blooms: Possible contributions of photo-oxidation, O2 toxicity, and cyanophages. Can. J. Bot. 1981, 59, 1277–1284. [Google Scholar] [CrossRef]
- Šulčius, S.; Alzbutas, G.; Kvederavičiūtė, K.; Koreivienė, J.; Zakrys, L.; Lubys, A.; Paškauskas, R. Draft genome sequence of the cyanobacterium Aphanizomenon flos-aquae strain 2012/KM1/DE isolated from the Curonian Lagoon (Baltic Sea). Genome Announc. 2015, 3, e01392-14. [Google Scholar] [CrossRef] [Green Version]
- Šulčius, S.; Slavuckytė, K.; Paškauskas, R. The predation paradox: Synergistic and antagonistic interactions between grazing by crustacean predator and infection by cyanophages promotes bloom formation in filamentous cyanobacteria. Limnol. Oceanogr. 2017, 62, 2189–2199. [Google Scholar] [CrossRef] [Green Version]
- Šulčius, S.; Šimoliūnas, E.; Alzbutas, G.; Gasiūnas, G.; Jauniškis, V.; Kuznecova, J.; Miettinen, S.; Nilsson, E.; Meškys, R.; Roine, E.; et al. Genomic characterization of cyanophage vB_AphaS-CL131 infecting filamentous diazotrophic cyanobacterium Aphanizomenon flos-aquae reveals novel insights into virus-bacterium interactions. Appl. Environ. Microbiol. 2018, 85, e01311-18. [Google Scholar] [CrossRef] [Green Version]
- Singh, R.N.; Singh, P.K.S.R.N. Isolation of Cyanophages from India. Nature 1967, 216, 1020–1021. [Google Scholar] [CrossRef]
- Pollard, P.; Young, L.M. Lake viruses lyse cyanobacteria, Cylindrospermopsis raciborskii, enhances filamentous-host dispersal in Australia. Acta Oecologica Int. J. Ecol. 2010, 36, 114–119. [Google Scholar] [CrossRef]
- Steenhauer, M.L.; Wierenga, J.; Carreira, C.; Limpens, R.W.A.L.; Koster, A.J. Isolation of cyanophage CrV infecting Cylin-drospermopsis raciborskii and the influence of temperature and irradiance on CrV proliferation. Aquat. Microb. Ecol. 2016, 78, 11–23. [Google Scholar] [CrossRef] [Green Version]
- Gao, E.; Yuan, X.; Li, R.; Zhang, Q. Isolation of a novel cyanophage infectious to the filamentous cyanobacterium Planktothrix agardhii (Cyanophyceae) from Lake Donghu, China. Aquat. Microb. Ecol. 2009, 54, 163–170. [Google Scholar] [CrossRef]
- Watkins, C.S.; Smith, J.R.; Hayes, P.K.; Watts, J.E.M. Characterisation of host growth after infection with a broad-range freshwater cyanopodophage. PLoS ONE 2014, 9, e87339. [Google Scholar] [CrossRef]
- Hewson, I.; O’Neil, J.M.; Dennison, W.C. Virus-like particles associated with Lyngbya icocyano (Cyanophyta; Oscillatoriacea) bloom decline in Moreton Bay, Australia. Aquat. Microb. Ecol. 2001, 25, 207–213. [Google Scholar] [CrossRef] [Green Version]
- Fahnensteil, G.L.; Carrick, H.J. Physiological characteristics and food-web dynamics of Synechococcus in Lakes Huron and Michigan. Limnol. Oceanogr. 1991, 36, 219–234. [Google Scholar] [CrossRef] [Green Version]
- Scanlan, J.D.; West, N.J. Molecular ecology of the marine cyanobacterial genera Prochlorococcus and Synechococcus. FEMS Microbiol. Ecol. 2002, 40, 1–12. [Google Scholar] [CrossRef]
- Callieri, C.; Cronberg, G.; Stockner, J. Freshwater icocyanobacterial: Single cells, microcolonies and colonial forms. In Ecology of Cyanobacteria II: Their Diversity in Time and Space; Whitton, B., Ed.; Springer: Berlin/Heidelberg, Germany, 2012; pp. 229–271. [Google Scholar]
- Mitsui, A.; Cao, S.; Takahashi, A.; Arai, T. Growth synchrony and cellular parameters of the unicellular nitrogen-fixing marine cyanobacterium, Synechococcus sp. strain Miami BG 043511 under continuous illumination. Physiol. Plant. 1987, 69, 1–8. [Google Scholar] [CrossRef]
- Steunou, A.-S.; Bhaya, D.; Bateson, M.M.; Melendrez, M.C.; Ward, D.M.; Brecht, E.; Peters, J.W.; Kühl, M.; Grossman, A.R. In situ analysis of nitrogen fixation and metabolic switching in unicellular thermophilic cyanobacteria inhabiting hot spring microbial mats. Proc. Natl. Acad. Sci. USA 2006, 103, 2398–2403. [Google Scholar] [CrossRef] [Green Version]
- Blaha, L.; Marsalek, B. Microcystin production and toxicity of picocyanobacterial as risk factor for drinking water treatment plants. Algol. Stud. 1999, 92, 95–108. [Google Scholar]
- Carmichael, W.W.; Li, R. Cyanobacteria toxins in the Salton Sea. Aquat. Biosyst. 2006, 2, 5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Furtado, A.L.F.F.; Calijuri, M.D.C.; Lorenzi, A.S.; Honda, R.Y.; Genuário, D.B.; Fiore, M.F. Morphological and molecular characterization of cyanobacteria from a Brazilian facultative wastewater stabilization pond and evaluation of microcystin production. Hydrobiologia 2009, 627, 195–209. [Google Scholar] [CrossRef]
- Kao, C.C.; Green, S.; Stein, B.; Golden, S.S. Diel Infection of a Cyanobacterium by a Contractile Bacteriophage. Appl. Environ. Microbiol. 2005, 71, 4276–4279. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sherman, L.A.; Connelly, M.; Sherman, D.M. Infection of Synechococcus cedrorum by the cyanophage AS-1M. I. Ultrastructure of infection and phage assembly. Virology 1976, 71, 1–16. [Google Scholar] [CrossRef]
- Sherman, L.A.; Pauw, P. Infection of Synechococcus cedrorum by the cyanophage AS-1M. II. Protein and DNA synthesis. Virology 1976, 71, 17–27. [Google Scholar] [CrossRef]
- Kim, M.; Choi, Y.-K. A New Synechococcus Cyanophage from a Reservoir in Korea. Virology 1994, 204, 338–342. [Google Scholar] [CrossRef]
- Park, G.J.; Kim, M.; Choi, Y.K.; Yoon, S.N. Restriction pattern of the nucleic acid of Synechococcus sp. cyanophage. J. Microbiol. 1996, 34, 1–6. [Google Scholar]
- Dreher, T.W.; Brown, N.; Bozarth, C.S.; Schwartz, A.D.; Riscoe, E.; Thrash, C.; Bennett, S.E.; Tzeng, S.-C.; Maier, C.S. A freshwater cyanophage whose genome indicates close relationships to photosynthetic marine cyanomyophages. Environ. Microbiol. 2011, 13, 1858–1874. [Google Scholar] [CrossRef] [Green Version]
- Chénard, C.; Chan, A.M.; Vincent, W.; Suttle, A.C. Polar freshwater cyanophage S-EIV1 represents a new widespread evolutionary lineage of phages. ISME J. 2015, 9, 2046–2058. [Google Scholar] [CrossRef] [Green Version]
- Zhong, K.X.; Suttle, C.A.; Baudoux, A.-C.; Derelle, E.; Colombet, J.; Cho, A.; Caleta, J.; Six, C.; Jacquet, S. A New Freshwater Cyanosiphovirus Harboring Integrase. Front. Microbiol. 2018, 9, 2204. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yamamoto, Y.; Shiah, F.-K.; Chen, Y.-L. Importance of large colony formation in bloom-forming cyanobacteria to dominate in eutrophic ponds. Ann. Limnol. Int. J. Limnol. 2011, 47, 167–173. [Google Scholar] [CrossRef] [Green Version]
- Eldridge, S.L.C.; Wood, T.M.; Echols, K.R. Spatial and Temporal Dynamics of Cyanotoxins and Their Relation to Other Water Quality Variables in Upper Klamath Lake, Oregon, 2007–2009; U.S. Geological Survey Scientific Investigations Report, Series Number 2012-5069; US Department of the Interior: Washington, DC, USA, 2012. [CrossRef]
- Welker, M.; von Dohren, H. Cyanobacterial peptides—Nature’s own combinatorial synthesis. FEMS Microbiol. Rev. 2006, 30, 530–563. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Otten, G.T.; Paerl, H.W. Health effects of toxic cyanobacteria in US drinking and recreational waters: Our current understanding and proposed direction. Water Health 2015, 2, 75–84. [Google Scholar]
- Takashima, Y.; Yoshida, T.; Yoshida, M.; Shirai, Y.; Tomaru, Y.; Takao, Y.; Hiroishi, S.; Nagasaki, K. Development and Application of Quantitative Detection of Cyanophages Phylogenetically Related to Cyanophage Ma-LMM01 Infecting Microcystis Aeruginosa in Fresh Water. Microbes Environ. 2007, 22, 207–213. [Google Scholar] [CrossRef] [Green Version]
- Kimura-Sakai, S.; Sako, Y.; Yoshida, T. Development of a real-time PCR assay for the quantification of Ma-LMM01-type Microcystis cyanophages in a natural pond. Lett. Appl. Microbiol. 2015, 60, 400–408. [Google Scholar] [CrossRef]
- Kimura, S.; Yoshida, T.; Hosoda, N.; Honda, T.; Kuno, S.; Kamiji, R.; Hashimoto, R.; Sako, Y. Diurnal infection patterns and impact of Microcystis cyanophages in a Japanese pond. Appl. Environ. Microbiol. 2012, 78, 5805–5811. [Google Scholar] [CrossRef] [Green Version]
- Morimoto, D.; Kimura, S.; Sako, Y.; Yoshida, T. Transcriptome Analysis of a Bloom-Forming Cyanobacterium Microcystis Aeruginosa during Ma-LMM01 Phage Infection. Front. Microbiol. 2018, 9, 2. [Google Scholar] [CrossRef] [Green Version]
- Ou, T.; Gao, X.-C.; Li, S.-H.; Zhang, Q.-Y. Genome analysis and gene nblA identification of Microcystis Aeruginosa myovirus (MaMV-DC) reveal the evidence for horizontal gene transfer events between cyanomyovirus and host. J. Gen. Virol. 2015, 96, 3681–3697. [Google Scholar] [CrossRef]
- Komarek, J.; Kovacik, L. Trichome structure of four Aphanizomenon taxa (Cyanophyceae) from Czechoslovakia, with notes on the taxonomy and delimitation of the genus. Plant Syst. Evol. 1989, 164, 47–64. [Google Scholar] [CrossRef]
- Gugger, M.; Lyra, C.; Henriksen, P.; Couté, A.; Humbert, J.-F.; Sivonen, K. Phylogenetic comparison of the cyanobacterial genera Anabaena and Aphanizomenon. Int. J. Syst. Evol. Microbiol. 2002, 52, 1867–1880. [Google Scholar] [CrossRef] [PubMed]
- Kipp, R.M. Cylindrospermopsis Raciborskii Factsheet. 2006. Available online: https://www.glerl.noaa.gov/res/HABs_and_Hypoxia/cylindro_factsheet.html (accessed on 3 April 2021).
- Sivonen, K.; Börner, T. Bioactive compounds produced by cyanobacteria. In The Cyanobacteria: Molecular Biology, Genomics and Evolution; Herrero, A., Flores, E., Eds.; Caister Academic Press: Norfolk, UK, 2008; pp. 159–197. [Google Scholar]
- Bancroft, I.; Smith, R.J. The isolation of genomic DNA from cyanophage infecting Nostoc and Anabaena species of cyanobacteria. New Phytol. 1988, 110, 233–239. [Google Scholar] [CrossRef]
- Bancroft, I.; Smith, R.J. An analysis of restriction endonuclease sites in cyanophages infecting the heterocystous cyanobacteria Anabaena and Nostoc. J. Gen. Virol. 1988, 69, 739–743. [Google Scholar] [CrossRef]
- Bancroft, I.; Wolk, C.P.; Oren, E.V. Physical and genetic maps of the genome of the heterocyst-forming cyanobacterium Anabaena sp. strain PCC 7120. J. Bacteriol. 1989, 171, 5940–5948. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baker, A.C.; Goddard, V.J.; Davy, J.; Schroeder, D.C.; Adams, D.G.; Wilson, W.H. Identification of a Diagnostic Marker To Detect Freshwater Cyanophages of Filamentous Cyanobacteria. Appl. Environ. Microbiol. 2006, 72, 5713–5719. [Google Scholar] [CrossRef] [Green Version]
- Xiong, Z.; Wang, Y.; Dong, Y.; Zhang, Q.; Xu, X. Cyanophage A-1(L) adsorbs to lipopolysaccharides of Anabaena sp. strain PCC7120 via the tail protein lipopolysaccharide-interacting protein (ORF36). J. Bacteriol. 2019, 201, e00516. [Google Scholar] [CrossRef] [Green Version]
- Wu, W.; Zhu, Q.; Liu, X.; An, C.; Wang, J. Isolation of a freshwater cyanophage (F1) capable of infecting Anabaena flos-aquae and its potentials in the control of water bloom. Int. J. Environ. Pollut. 2009, 38, 212–221. [Google Scholar] [CrossRef]
- Salam, E.A.; Shabana, E.T.; Din, A.M. Isolation and characterization of two cyanophages infecting some Anabaena spp. Egypt. J. Biol. Pest Control. 2014, 24, 23–28. [Google Scholar]
- Kurmayer, R.; Deng, L.; Entfellner, E. Role of toxic and bioactive secondary metabolites in colonization and bloom formation by filamentous cyanobacteria Planktothrix. Harmful Algae 2016, 54, 69–86. [Google Scholar] [CrossRef] [Green Version]
- Sharp, K.; Arthur, K.E.; Gu, L.; Ross, C.; Harrison, G.; Gunasekera, S.P.; Meickle, T.; Matthew, S.; Luesch, H.; Thacker, R.W.; et al. Phylogenetic and Chemical Diversity of Three Chemotypes of Bloom-Forming Lyngbya Species (Cyanobacteria: Oscillatoriales) from Reefs of Southeastern Florida. Appl. Environ. Microbiol. 2009, 75, 2879–2888. [Google Scholar] [CrossRef] [Green Version]
- Paul, J.V.; Cruz-Rivera, E.; Thacker, R.W. Chemical mediation of macroalgal-herbivore interactions: Ecological and evolutionary perspectives. In Marine Chemical Ecology; McClintock, J., Baker, B., Eds.; CRC Press, LLC: Boca Raton, FL, USA, 2001; pp. 227–265. [Google Scholar]
- Suda, S.; Watanabe, M.M.; Otsuka, S.; Mahakahant, A.; Yongmanitchai, W.; Nopartnaraporn, N.; Liu, Y.; Day, J.G. Taxonomic revision of water-bloom-forming species of oscillatorioid cyanobacteria. Int. J. Syst. Evol. Microbiol. 2002, 52, 1577–1595. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bratbak, G.; Jacobsen, A.; Heldal, M.; Nagasaki, K.; Thingstad, F. Virus production in Phaeocystis pouchetii and its relation to host cell growth and nutrition. Aquat. Microb. Ecol. 1998, 16, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Zborowsky, S.; Lindell, D. Resistance in marine cyanobacteria differs against specialist and generalist cyanophages. Proc. Natl. Acad. Sci. USA 2019, 116, 16899–16908. [Google Scholar] [CrossRef] [Green Version]
- Jia, Y.; Shan, J.; Millard, A.; Clokie, M.R.; Mann, N.H. Light-dependent adsorption of photosynthetic cyanophages to Synechococcus sp. WH7803. FEMS Microbiol. Lett. 2010, 310, 120–126. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mangan, N.M.; Flamholz, A.; Hood, R.D.; Milo, R.; Savage, D.F. pH determines the energetic efficiency of the cyanobacterial CO 2 concentrating mechanism. Proc. Natl. Acad. Sci. USA 2016, 113, E5354–E5362. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stanier, R.Y.; Kunisawa, R.; Mandel, M.; Cohen-Bazire, G. Purification and properties of unicellular blue-green algae (order Chroococcales). Bacteriol. Rev. 1971, 35, 171–205. [Google Scholar] [CrossRef] [PubMed]
- Waechter, C.; Aligata, A.; Zhang, Y. Viral Treatment of Harmful Algal Blooms; Science and Technology Program; ST-2019-0157-1; Bureau of Reclamation, Research and Development Office: Boulder, CO, USA, 2019.

| Virus Family | Morphology | Examples |
|---|---|---|
| Myoviridae | An isometric head separated by a neck from a long complex tail with a contractile sheath and central tube | Cyanomyovirus |
| Podoviridae | An isometric head with a short tail (without a neck), generally less than half the diameter of the widest head dimension | Cyanopodovirus |
| Siphoviridae | An isometric head with a noncontractile tail as long or longer than the diameter of the widest head dimension | Cyanosiphovirus (formerly Cyanostylovirus) |
| Cyanophage class | Groups | Known Target Cyanobacteria | Unique Cyanobacteria Traits |
|---|---|---|---|
| Class 1 | LPP | Lyngbya Phormidium Plectonema | Filamentous, non-heterocystous |
| Class 2 | A | Dolichospermum | Filamentous, both heterocystous and non-heterocystous |
| N | Dolichospermum | ||
| AN | Dolichospermum Nostoc | ||
| NP | Nostoc Plectonema | ||
| Class 3 | AS | Anacystis Synechococcus Microcystis | Unicellular, colonial |
| SM | Anacystis Synechococcus Microcystis |
| Temperature | Nutrients | EPS | Irradiance | References | |
|---|---|---|---|---|---|
| Burst size | Increased with temperature. | Decreased under P-limitation. Inconsistent findings with elevated pCO2. | [55,65,67,70] | ||
| Latent period | Decreased with temperature. | Increased under P-limitation. Decreased under elevated pCO2 | [55,65,76] | ||
| Infectivity | Increased in warmer waters (up to 40 °C to 45 °C). | Decreased under P-limitation. | Decreased with greater EPS production. | Decreased with high light owing to dimer formation. Light-dependent for some cyanophages. | [53,81,83,84,93] |
| Adsorption | Increased with temperature (e.g., shift from 24 °C to 35 °C). | Increased with elevated pCO2. Decreased under N-limitation. | Decreased with physical impedance of cyanophage diffusion. | Light-dependent as cyanophage adsorption derives much if its energy from host photosynthesis. | [55,71,84,91] |
| Life cycle | Driven toward lytic with increasing temperature. | Driven toward lysogenic under P- and N-limitation. | Driven toward lytic with increasing irradiance for some cyanophages. | [55,65,66,72,97] | |
| Abundance | Increased with temperature. | Increased free cyanophage in heightened P conditions. Increased production with elevated pCO2. No change in replication within host. Decreased under N-limitation. | Decreased due to inactivation from extended exposure to germicidal UV wavelengths. | [48,49,50,69,70,71,81,98] |
| Cyanobacteria Genus | Identified Cyanophages | Range of Specificity | Candidate for Broad HAB Control | References |
|---|---|---|---|---|
| Synechococcus | SM-1, SM-2, NRC-1, AS-1, AS-1M, S-CRM01, S-EIV1, S-LBS1 | Strain-to-Species-Level | Poor; primarily strain-specific | [51,81,85,99,110] |
| Microcystis | Ma-LBP, Ma-LMM01, MaMV-DC, ΦMHI42 | Strain-to-Genus-Level | Fair; some phages infectious across multiple species | [25,96,111,112,113,114] |
| Dolichospermum | N-1, A-1L, A-4L, AC-1, AF-1, AN-10, AN-13, AN-23, M-CP1/2/3, A-CP1/2/3/4, A-CP6/7, A-CM1/2, A-CF1, A-CS1/2 | Strain-to-Class-Level | Good; multiple phages infectious in multiple genera | [26,115,116,117,118] |
| Aphanizomenon | Vb_AphaS-CL131 | Strain-Level | Poor; strain-specific | [119,120,121,122,123] |
| Cylindrospermopsis | AR-1, CrV | Strain-Level | Poor; strain-specific | [124,125,126] |
| Planktothrix | PaV-LD, M-CP5/6, A-CP1/4, P-Z1 through P-Z12 | Strain-to-Class-Level | Fair; some phages infectious in Dolichospermum spp. | [26,127,128] |
| Lyngbya | LPP-1, LW-1 | Strain-to-Class-Level | Good; LPP-1 infectious in multiple filamentous genera | [99,118,129] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Grasso, C.R.; Pokrzywinski, K.L.; Waechter, C.; Rycroft, T.; Zhang, Y.; Aligata, A.; Kramer, M.; Lamsal, A. A Review of Cyanophage–Host Relationships: Highlighting Cyanophages as a Potential Cyanobacteria Control Strategy. Toxins 2022, 14, 385. https://doi.org/10.3390/toxins14060385
Grasso CR, Pokrzywinski KL, Waechter C, Rycroft T, Zhang Y, Aligata A, Kramer M, Lamsal A. A Review of Cyanophage–Host Relationships: Highlighting Cyanophages as a Potential Cyanobacteria Control Strategy. Toxins. 2022; 14(6):385. https://doi.org/10.3390/toxins14060385
Chicago/Turabian StyleGrasso, Christopher R., Kaytee L. Pokrzywinski, Christopher Waechter, Taylor Rycroft, Yanyan Zhang, Alyssa Aligata, Michael Kramer, and Anisha Lamsal. 2022. "A Review of Cyanophage–Host Relationships: Highlighting Cyanophages as a Potential Cyanobacteria Control Strategy" Toxins 14, no. 6: 385. https://doi.org/10.3390/toxins14060385





