Vegetable and Fruit Intakes Are Associated with hs-CRP Levels in Pre-Pubertal Girls
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Data Collection
2.3. Anthropometric Variables
2.4. Biochemical Data
2.5. Food and Nutritional Data
2.6. Statistical Analysis
3. Results
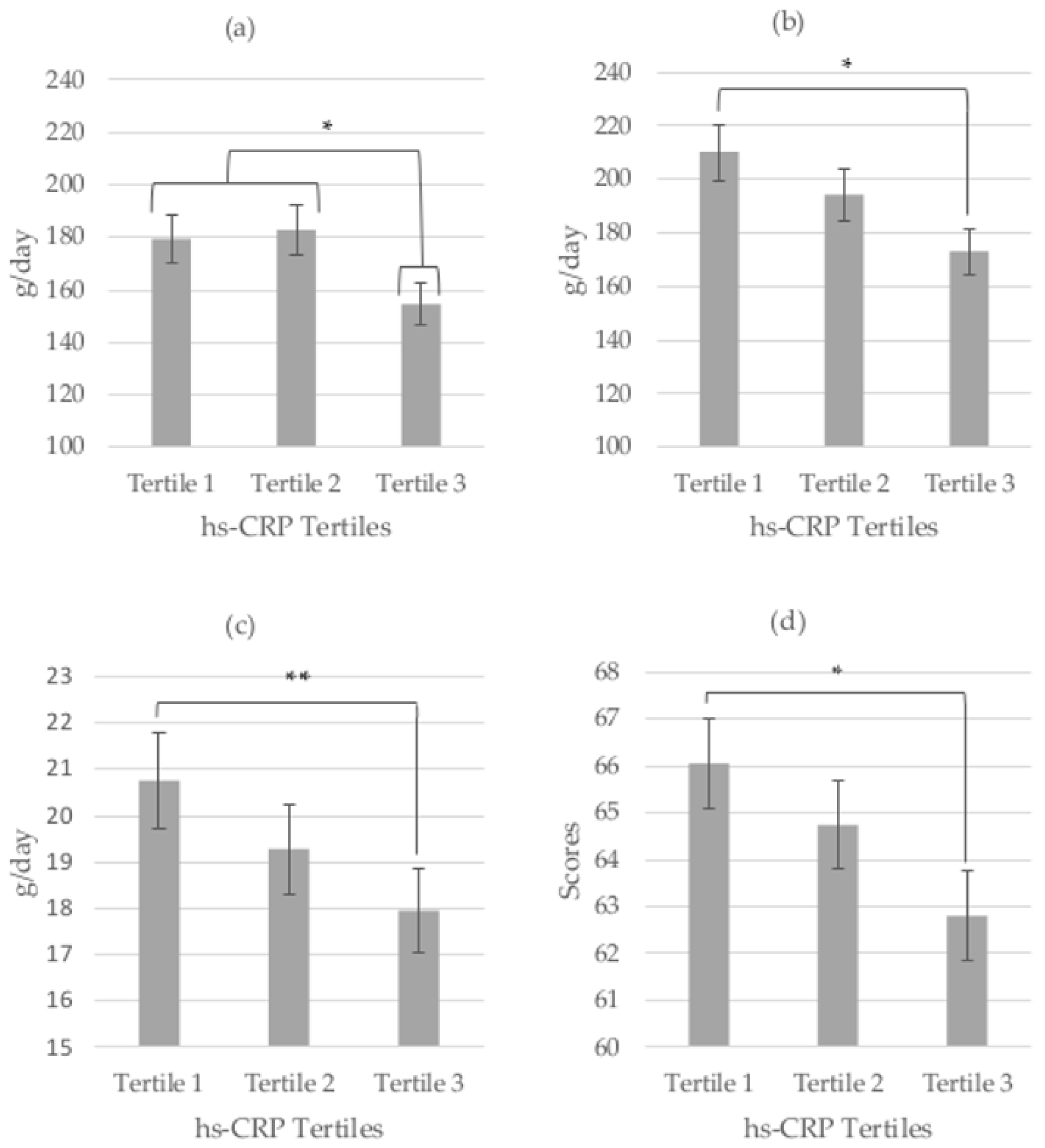
3.1. Nutrient Intake and hs-CRP Levels
3.2. Food Consumption and hs-CRP Levels
3.3. Dietary Patterns and hs-CRP Levels
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kapiotis, S.; Holzer, G.; Schaller, G.; Haumer, M.; Widhalm, H.; Weghuber, D.; Jilma, B.; Röggla, G.; Wolzt, M.; Widhalm, K.; et al. A proinflammatory state is detectable in obese children and is accompanied by functional and morphological vascular changes. Arterioscler. Thromb. Vasc. Biol. 2006, 26, 2541–2546. [Google Scholar] [CrossRef] [PubMed]
- Brooks, G.C.; Blaha, M.J.; Blumenthal, R.S. Relation of C-Reactive Protein to Abdominal Adiposity. Am. J. Cardiol. 2010, 106, 56–61. [Google Scholar] [CrossRef] [PubMed]
- Berenson, G.S.; Wattigney, W.A.; Tracy, R.E.; Newman, W.P.; Srinivasan, S.R.; Webber, L.S.; Dalferes, E.R.; Strong, J.P. Atherosclerosis of the aorta and coronary arteries and cardiovascular risk factors in persons aged 6 to 30 years and studied at necropsy (The Bogalusa Heart Study). Am. J. Cardiol. 1992, 70, 851–858. [Google Scholar] [CrossRef]
- Tuzcu, E.M.; Kapadia, S.R.; Tutar, E.; Ziada, K.M.; Hobbs, R.E.; McCarthy, P.M.; Young, J.B.; Nissen, S.E. High Prevalence of Coronary Atherosclerosis in Asymptomatic Teenagers and Young Adults. Circulation 2001, 103, 2705–2710. [Google Scholar] [CrossRef] [PubMed]
- Yeh, E.T. H. High-sensitivity C-reactive protein as a risk assessment tool for cardiovascular disease. Clin. Cardiol. 2005, 28, 408–412. [Google Scholar] [CrossRef] [PubMed]
- Minihane, A.M.; Vinoy, S.; Russell, W.R.; Baka, A.; Roche, H.M.; Tuohy, K.M.; Teeling, J.L.; Blaak, E.E.; Fenech, M.; Vauzour, D.; et al. Low-grade inflammation, diet composition and health: Current research evidence and its translation. Br. J. Nutr. 2015, 114, 999–1012. [Google Scholar] [CrossRef] [PubMed]
- Giugliano, D.; Ceriello, A.; Esposito, K. The Effects of Diet on Inflammation. Emphasis on the Metabolic Syndrome. J. Am. Coll. Cardiol. 2006, 48, 677–685. [Google Scholar] [CrossRef] [PubMed]
- Santos, S.; Oliveira, A.; Lopes, C. Systematic review of saturated fatty acids on inflammation and circulating levels of adipokines. Nutr. Res. 2013, 33, 687–695. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, S.; Inoue, N.; Ohashi, Y.; Terashima, M.; Matsui, K.; Mori, T.; Fujita, H.; Awano, K.; Kobayashi, K.; Azumi, H.; et al. Interaction of oxidative stress and inflammatory response in coronary plaque instability: Important role of C-reactive protein. Arterioscler. Thromb. Vasc. Biol. 2003, 23, 1398–1404. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, A.; Rodríguez-Artalejo, F.; Lopes, C. The association of fruits, vegetables, antioxidant vitamins and fibre intake with high-sensitivity C-reactive protein: Sex and body mass index interactions. Eur. J. Clin. Nutr. 2009, 63, 1345–1352. [Google Scholar] [CrossRef] [PubMed]
- Navarro, S.L.; Kantor, E.D.; Song, X.; Milne, G.L.; Lampe, J.W.; Kratz, M.; White, E. Factors Associated with Multiple Biomarkers of Systemic Inflammation. Cancer Epidemiol. Biomark. Prev. 2016, 25, 521–532. [Google Scholar] [CrossRef] [PubMed]
- Esmaillzadeh, A.; Kimiagar, M.; Mehrabi, Y.; Azadbakht, L.; Hu, F.B.; Willett, W.C. Fruit and vegetable intakes, C-reactive protein, and the metabolic syndrome. Am. J. Clin. Nutr. 2006, 84, 1489–1497. [Google Scholar] [PubMed]
- Wannamethee, S.G.; Lowe, G.D. O.; Rumley, A.; Bruckdorfer, K.R.; Whincup, P.H. Associations of vitamin C status, fruit and vegetable intakes, and markers of inflammation and hemostasis. Am. J. Clin. Nutr. 2006, 83, 567–574. [Google Scholar] [PubMed]
- Qureshi, M.M.; Singer, M.R.; Moore, L.L. A cross-sectional study of food group intake and C-reactive protein among children. Nutr. Metab. (Lond). 2009, 6, 40. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Gil, E.M.; Santabarbara, J.; Russo, P.; Ahrens, W.; Claessens, M.; Lissner, L.; Brnhorst, C.; Krogh, V.; Iacoviello, L.; Molnar, D.; et al. Food intake and inflammation in European children: The IDEFICS study. Eur. J. Nutr. 2016, 55, 2459–2468. [Google Scholar] [CrossRef] [PubMed]
- Buyken, A.E.; Goletzke, J.; Joslowski, G.; Felbick, A.; Cheng, G.; Herder, C.; Brand-Miller, J.C. Association between carbohydrate quality and inflammatory markers: Systematic review of observational and interventional studies. Am. J. Clin. Nutr. 2014, 99, 813–833. [Google Scholar] [CrossRef] [PubMed]
- Barbaresko, J.; Koch, M.; Schulze, M.B.; Nöthlings, U. Dietary pattern analysis and biomarkers of low-grade inflammation: A systematic literature review. Nutr. Rev. 2013, 71, 511–527. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Kang, D.; Lee, S.-A. Effect of dietary patterns on serum C-reactive protein level. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 1004–1011. [Google Scholar] [CrossRef] [PubMed]
- Estruch, R. Anti-inflammatory effects of the Mediterranean diet: The experience of the PREDIMED study. Proc. Nutr. Soc. 2010, 69, 333–340. [Google Scholar] [CrossRef] [PubMed]
- Bédard, A.; Lamarche, B.; Corneau, L.; Dodin, S.; Lemieux, S. Sex differences in the impact of the Mediterranean diet on systemic inflammation. Nutr. J. 2015, 14, 46. [Google Scholar] [CrossRef] [PubMed]
- Royo-Bordonada, M.A.; Gorgojo, L.; Martín-Moreno, J.M.; Garcés, C.; Rodríguez-Artalejo, F.; Benavente, M.; Mangas, A.; de Oya, M. Spanish children’s diet: Compliance with nutrient and food intake guidelines. Eur. J. Clin. Nutr. 2003, 57, 930–939. [Google Scholar] [CrossRef] [PubMed]
- Moreiras, O.; Carbajal, Á.; Cabrera, L.; Cuadrado, C. Tablas de Composición de Alimentos Guía de Prácticas, 18th ed.; Pirámide: Málaga, Spain, 2016. [Google Scholar]
- Gorgojo, L.; Guallar, E.; Martín-Moreno, J.M.; López-Nomdedeu, C.; Vázquez, C.; Martí-Henneberg, C.; Serrano-Ríos, M. Encuestas alimentarias en los niños españoles de edad escolar: Análisis del período 1984–1994. Med. Clin. 1999, 112, 368–374. [Google Scholar]
- Kennedy, E.T.; Ohls, J.; Carlson, S.; Fleming, K. The Healthy Eating Index. Design and Applications. J. Am. Diet. Assoc. 1995, 95, 1103–1108. [Google Scholar] [CrossRef]
- Steckhan, N.; Hohmann, C.-D.; Kessler, C.; Dobos, G.; Michalsen, A.; Cramer, H. Effects of different dietary approaches on inflammatory markers in patients with metabolic syndrome: A systematic review and meta-analysis. Nutrition 2016, 32, 338–348. [Google Scholar] [CrossRef] [PubMed]
- Aeberli, I.; Molinari, L.; Spinas, G.; Lehmann, R.; L’Allemand, D.; Zimmermann, M.B. Dietary intakes of fat and antioxidant vitamins are predictors of subclinical inflammation in overweight Swiss children. Am. J. Clin. Nutr. 2006, 84, 748–755. [Google Scholar] [PubMed]
- Del Mar Bibiloni, M.; Maffeis, C.; Llompart, I.; Pons, A.; Tur, J.A. Dietary factors associated with subclinical inflammation among girls. Eur. J. Clin. Nutr. 2013, 67, 1264–1270. [Google Scholar] [CrossRef] [PubMed]
- Negele, L.; Schneider, B.; Ristl, R.; Stulnig, T.M.; Willfort-Ehringer, A.; Helk, O.; Widhalm, K. Effect of a low-fat diet enriched either with rapeseed oil or sunflower oil on plasma lipoproteins in children and adolescents with familial hypercholesterolaemia. Results of a pilot study. Eur. J. Clin. Nutr. 2015, 69, 337–343. [Google Scholar] [CrossRef] [PubMed]
- Jois, A.; Navarro, P.; Ortega-Senovilla, H.; Gavela-Pérez, T.; Soriano-Guillén, L.; Garcés, C. Relationship of high leptin levels with an adverse lipid and insulin profile in 6–8 year-old children in Spain. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 1111–1116. [Google Scholar] [CrossRef] [PubMed]
- Navarro, P.; de Dios, O.; Gavela-Pérez, T.; Jois, A.; Garcés, C.; Soriano-Guillén, L. High-Sensitivity C-Reactive Protein and Leptin Levels Related to Body Mass Index Changes Throughout Childhood. J. Pediatr. 2016, 2541–2546. [Google Scholar] [CrossRef] [PubMed]
- King, D.E. Effect of a High-Fiber Diet vs. a Fiber-Supplemented Diet on C-Reactive Protein Level. Arch. Intern. Med. 2007, 167, 502. [Google Scholar] [CrossRef] [PubMed]
- Estruch, R.; Martinez-Gonzalez, M.A.; Corella, D.; Basora-Gallisa, J.; Ruiz-Gutierrez, V.; Covas, M.I.; Fiol, M.; Gomez-Gracia, E.; Lopez-Sabater, M.C.; Escoda, R.; et al. Effects of dietary fibre intake on risk factors for cardiovascular disease in subjects at high risk. J. Epidemiol. Community Health 2009, 63, 582–588. [Google Scholar] [CrossRef] [PubMed]
- Weickert, M.O.; Pfeiffer, A.F.H. Metabolic effects of dietary fiber consumption and prevention of diabetes. J. Nutr. 2008, 138, 439–442. [Google Scholar] [PubMed]
- Monfort-Pires, M.; Folchetti, L.D.; Previdelli, A.N.; Siqueira-Catania, A.; de Barros, C.R.; Ferreira, S.R. Healthy Eating Index is associated with certain markers of inflammation and insulin resistance but not with lipid profile in individuals at cardiometabolic risk. Appl. Physiol. Nutr. Metab. 2014, 39, 497–502. [Google Scholar] [CrossRef] [PubMed]
- Haghighatdoost, F.; Sarrafzadegan, N.; Mohammadifard, N.; Sajjadi, F.; Maghroon, M.; Boshtam, M.; Alikhasi, H.; Azadbakht, L. Healthy Eating Index and Cardiovascular Risk Factors among Iranians. J. Am. Coll. Nutr. 2013, 32, 111–121. [Google Scholar] [CrossRef] [PubMed]
- Lipsky, L.M.; Cheon, K.; Nansel, T.R.; Albert, P.S. Candidate measures of whole plant food intake are related to biomarkers of nutrition and health in the US population (National Health and Nutrition Examination Survey 1999–2002). Nutr. Res. 2012, 32, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Loprinzi, P.D.; Lee, I.-M.; Andersen, R.E.; Crespo, C.J.; Smit, E. Association of Concurrent Healthy Eating and Regular Physical Activity With Cardiovascular Disease Risk Factors in U.S. Youth. Am. J. Heal. Promot. 2015, 30, 2–8. [Google Scholar] [CrossRef] [PubMed]
| Variable | Girls (n = 301) | Boys (n = 270) |
|---|---|---|
| Age (years) | 6.7 ± 0.7 | 6.7 ± 0.6 |
| Weight (kg) | 27.4 ± 5.9 | 27.5 ± 5.6 |
| Height (m) | 1.26 ± 0.07 | 1.27 ± 0.06 |
| BMI | 17.2 ± 2.7 | 17.2 ± 2.6 |
| z-Score BMI | 0.01 ± 0.96 | 0.05 ± 0.98 |
| hs-CRP (mg/L) | 1.05 ± 1.74 | 0.87 ± 1.55 * |
| Energy (Kcal) | 2090 ± 497 | 2186 ± 518 * |
| Protein energy (%) | 17.2 ± 2.4 | 17.3 ± 2.5 |
| Total fat energy (%) | 45.7 ± 4.5 | 46.4 ± 4.6 |
| Saturated-fat energy (%) | 16.4 ± 2.7 | 17.2 ± 2.8 ** |
| Monounsaturated-fat energy (%) | 18.4 ± 2.7 | 18.3 ± 2.6 |
| Polyunsaturated-fat energy (%) | 8.3 ± 1.8 | 8.2 ± 1.9 |
| Total carbohydrate energy (%) | 38.4 ± 5.8 | 37.8 ± 6.0 |
| Complex-carbohydrate energy (%) | 18.0 ± 4.5 | 17.3 ± 4.1 * |
| Simple-carbohydrate energy (%) | 21.1 ± 5.6 | 21.3 ± 6.1 |
| Fiber (g) | 19.6 ± 7.2 | 19.9 ± 7.6 |
| Nutritional Variable | Girls | Boys | ||||||
|---|---|---|---|---|---|---|---|---|
| Tertile 1 (≤0.15 mg/L) | Tertile 2 (0.16–0.61 mg/L) | Tertile 3 (≥0.62 mg/L) | p Value | Tertile 1 (≤0.15 mg/L) | Tertile 2 (0.16–0.61 mg/L) | Tertile 3 (≥0.62 mg/L) | p Value | |
| Energy (kcal) | 2083 ± 446 | 2104 ± 531 | 2021 ± 492 | NS | 2162 ± 542 | 2173 ± 510 | 2223 ± 539 | NS |
| Protein (%) | 16.9 ± 2.5 | 17.3 ± 2.7 | 17.3 ± 2.5 | NS | 16.9 ± 2.7 | 17.1 ± 2.3 | 17.5 ± 2.4 | NS |
| Total fat (%) | 45.3 ± 4.8 | 46.3 ± 4.2 | 45.7 ± 4.3 | NS | 46.0 ± 4.3 | 46.7 ± 4.5 | 45.6 ± 4.3 | NS |
| Saturated-fat (%) | 15.7 ± 2.7 | 16.6 ± 2.3 | 17.1 ± 3.0 | 1–3 ** | 17.3 ± 2.8 | 17.0 ± 2.7 | 16.9 ± 2.7 | NS |
| Monounsaturated-fat (%) | 17.9 ± 2.5 | 18.5 ± 2.8 | 18.7 ± 2.9 | NS | 18.1 ± 2,4 | 18.5 ± 2.5 | 18.0 ± 2.4 | NS |
| Polyunsaturated-fat (%) | 8.5 ± 1.9 | 8.5 ± 1.8 | 8.1 ± 1.8 | NS | 8.1 ± 1.8 | 8.5 ± 2.1 | 7.9 ± 1.8 | NS |
| Total carbohydrate (%) | 39.3 ± 6.1 | 37.7 ± 5.3 | 38.4 ± 5.8 | NS | 38.4 ± 5.9 | 37.5 ± 5.6 | 38.6 ± 5.7 | NS |
| Simple-carbohydrate (%) | 21.6 ± 5.1 | 20.8 ± 5.9 | 21.3 ± 6.5 | NS | 21.9 ± 6.1 | 21.1 ± 5.4 | 21.7 ± 5.8 | NS |
| Complex-carbohydrate (%) | 18.1 ± 4.5 | 17.8 ± 5.1 | 17.7 ± 3.9 | NS | 17.4 ± 3.9 | 17.5 ± 4.6 | 17.5 ± 4.1 | NS |
| Fiber (g) | 20.8 ± 7.3 | 19.3 ± 7.7 | 17.9 ± 6.3 | 1–3 ** | 19.9 ± 7.4 | 19.5 ± 6.2 | 20.1 ± 6.5 | NS |
| Vitamin A (μg) | 646 ± 169 | 659 ± 170 | 616 ± 206 | NS | 706 ± 207 | 693 ± 191 | 700 ± 212 | NS |
| Vitamin E (mg) | 10.8 ± 3.5 | 10.9 ± 4.3 | 9.8 ± 4.2 | 1–3 * | 10.8 ± 4.7 | 10.9 ± 3.7 | 10.6 ± 3.9 | NS |
| 2–3 * | ||||||||
| HEI score | 66.1 ± 8.9 | 64.7 ± 8.5 | 62.8 ± 10.3 | 1–3 * | 63.4 ± 8.6 | 64.3 ± 7.6 | 65.4 ± 7.9 | NS |
| Nutritional Variable | Dietary Patterns as Detected by Factor Analysis | |
|---|---|---|
| Factor 1: High Vegetable Intake Diet | Factor 2: High Fat Intake Diet | |
| Energy | 0.786 | −0.160 |
| Protein energy (%) | −0.156 | 0.163 |
| Total fat energy (%) | −0.127 | 0.921 |
| Saturated fat energy (%) | −0.446 | 0.217 |
| Monounsaturated fat energy (%) | −0.002 | 0.782 |
| Polyunsaturated fat energy (%) | −0.005 | 0.712 |
| Total carbohydrate energy (%) | 0.195 | −0.853 |
| Complex carbohydrate energy (%) | 0.009 | −0.384 |
| Simple carbohydrate energy (%) | 0.129 | −0.538 |
| Fiber | 0.814 | −0.307 |
| Vitamin A | 0.521 | 0.071 |
| Vitamin E | 0.779 | −0.002 |
| Fruit | 0.711 | −0.239 |
| Fruit and vegetables | 0.785 | −0.267 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Navarro, P.; De Dios, O.; Jois, A.; Gavela-Pérez, T.; Gorgojo, L.; Martín-Moreno, J.M.; Soriano-Guillen, L.; Garcés, C. Vegetable and Fruit Intakes Are Associated with hs-CRP Levels in Pre-Pubertal Girls. Nutrients 2017, 9, 224. https://doi.org/10.3390/nu9030224
Navarro P, De Dios O, Jois A, Gavela-Pérez T, Gorgojo L, Martín-Moreno JM, Soriano-Guillen L, Garcés C. Vegetable and Fruit Intakes Are Associated with hs-CRP Levels in Pre-Pubertal Girls. Nutrients. 2017; 9(3):224. https://doi.org/10.3390/nu9030224
Chicago/Turabian StyleNavarro, Pilar, Olaya De Dios, Asha Jois, Teresa Gavela-Pérez, Lydia Gorgojo, José M. Martín-Moreno, Leandro Soriano-Guillen, and Carmen Garcés. 2017. "Vegetable and Fruit Intakes Are Associated with hs-CRP Levels in Pre-Pubertal Girls" Nutrients 9, no. 3: 224. https://doi.org/10.3390/nu9030224
APA StyleNavarro, P., De Dios, O., Jois, A., Gavela-Pérez, T., Gorgojo, L., Martín-Moreno, J. M., Soriano-Guillen, L., & Garcés, C. (2017). Vegetable and Fruit Intakes Are Associated with hs-CRP Levels in Pre-Pubertal Girls. Nutrients, 9(3), 224. https://doi.org/10.3390/nu9030224






