Marine Oil Supplements for Arthritis Pain: A Systematic Review and Meta-Analysis of Randomized Trials
Abstract
:1. Introduction
2. Materials
2.1. Protocol
2.2. Data Sources and Searches
2.3. Study Selection
2.4. Data Extraction and Quality Assessment
2.5. Data Synthesis and Analysis
2.6. Additional Analyses
3. Results
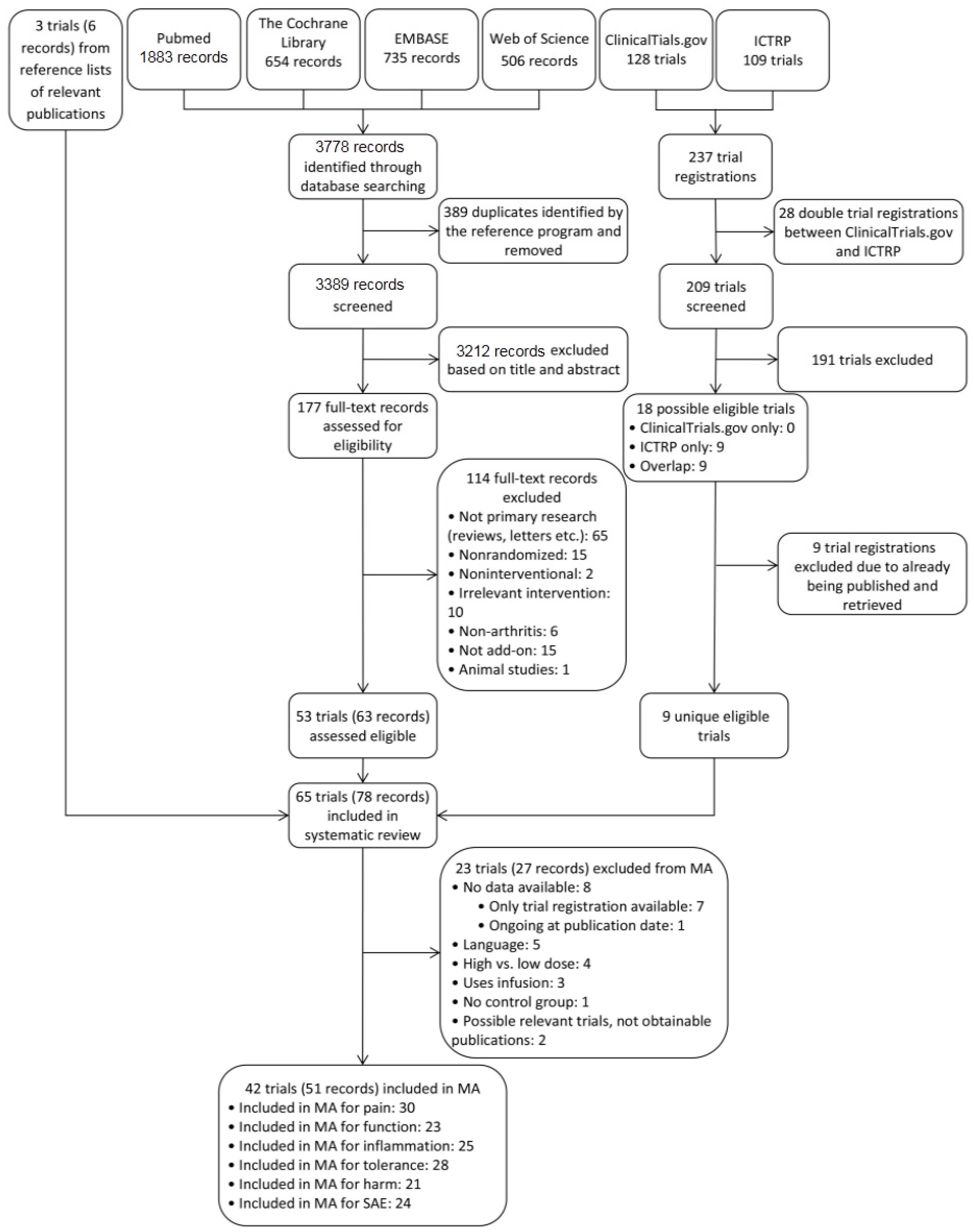
3.1. Study Selection
3.2. Study Characteristics
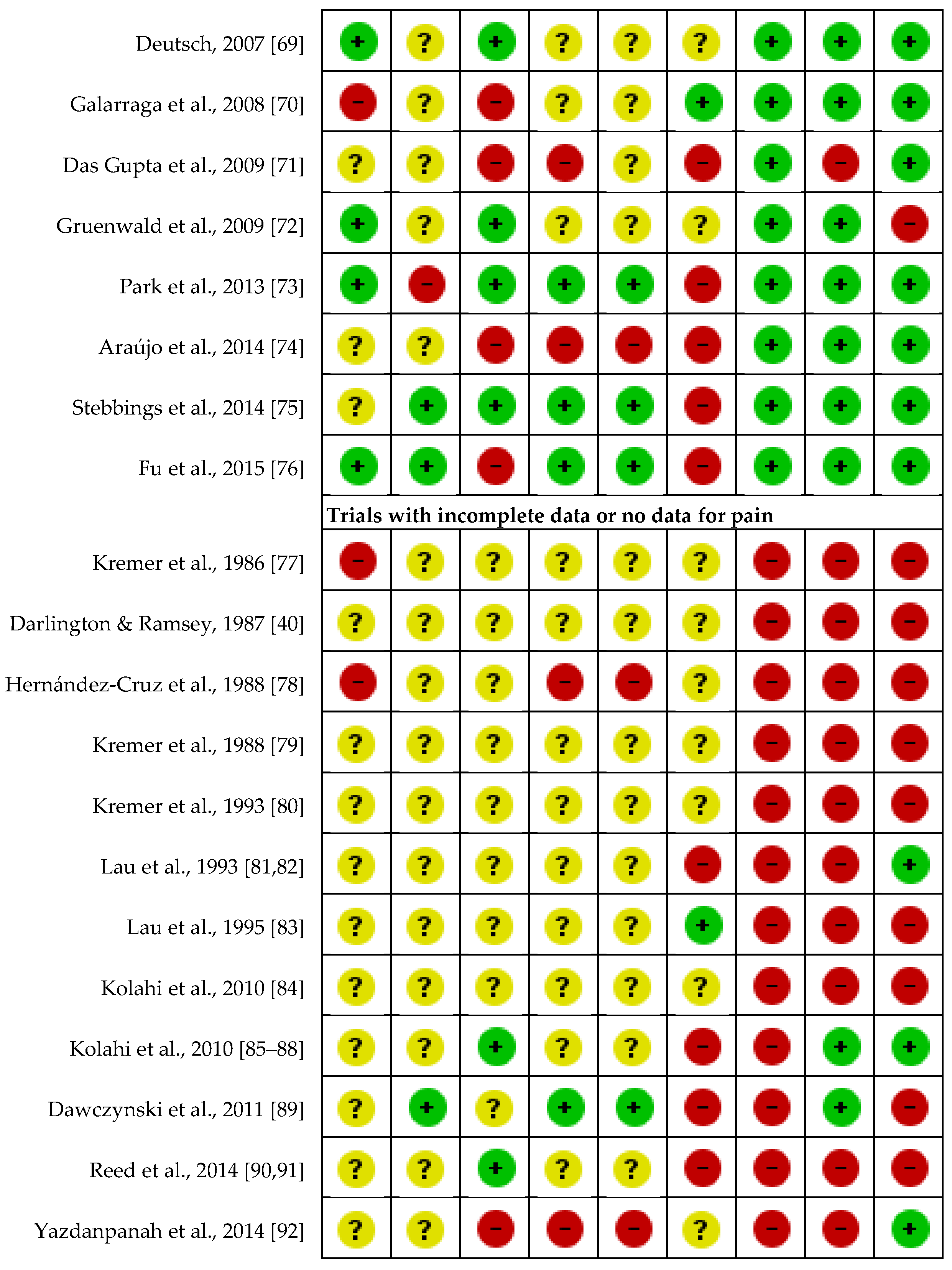
3.3. Risk of Bias in Included Studies
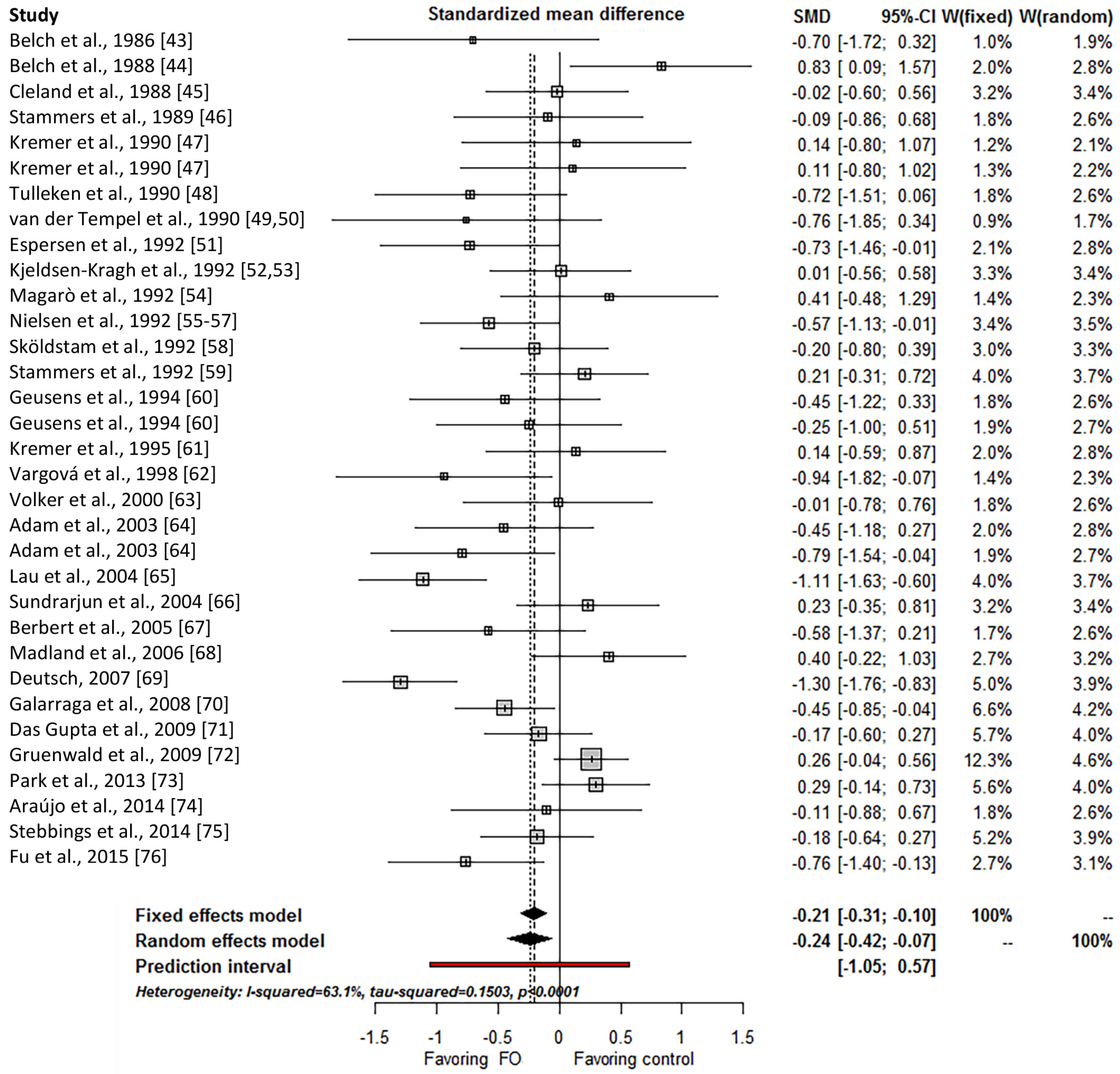
3.4. Primary Outcome: Pain
3.5. Secondary Outcomes: Inflammation and Function
3.6. Tolerance and Safety
3.7. Assessment of Reporting Bias
4. Discussion
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Dieppe, P.A.; Lohmander, L.S. Pathogenesis and management of pain in osteoarthritis. Lancet 2005, 365, 965–973. [Google Scholar] [CrossRef]
- McInnes, I.B.; Schett, G. The pathogenesis of rheumatoid arthritis. N. Engl. J. Med. 2011, 365, 2205–2219. [Google Scholar] [CrossRef] [PubMed]
- Barbour, K.E.; Helmick, C.G.; Theis, K.A.; Murphy, L.B.; Hootman, J.M.; Brady, T.J.; Cheng, Y.J. Prevalence of doctor-diagnosed arthritis and arthritis-attributable activity limitation-united states, 2010–2012. Morb. Mortal. Wkly. Rep. 2013, 62, 869–873. [Google Scholar]
- Sokolove, J.; Lepus, C.M. Role of inflammation in the pathogenesis of osteoarthritis: Latest findings and interpretations. Ther. Adv. Musculoskelet. Dis. 2013, 5, 77–94. [Google Scholar] [CrossRef] [PubMed]
- Scanzello, C.R.; Goldring, S.R. The role of synovitis in osteoarthritis pathogenesis. Bone 2012, 51, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Walsh, D.A.; McWilliams, D.F. Pain in rheumatoid arthritis. Curr. Pain Headache Rep. 2012, 16, 509–517. [Google Scholar] [CrossRef] [PubMed]
- Heiberg, T.; Kvien, T.K. Preferences for improved health examined in 1024 patients with rheumatoid arthritis: Pain has highest priority. Arthritis Rheum. 2002, 47, 391–397. [Google Scholar] [CrossRef] [PubMed]
- Desai, R.J.; Agarwal, S.J.; Aparasu, R.R. Drug use trends for arthritis and other rheumatic conditions and effect of patient’s age on treatment choice. N. C. Med. J. 2011, 72, 432–438. [Google Scholar] [PubMed]
- Wolfe, F.; Zhao, S.; Lane, N. Preference for nonsteroidal antiinflammatory drugs over acetaminophen by rheumatic disease patients: A survey of 1799 patients with osteoarthritis, rheumatoid arthritis, and fibromyalgia. Arthritis Rheumatol. 2000, 43, 378–385. [Google Scholar] [CrossRef]
- Bhala, N.; Emberson, J.; Merhi, A.; Abramson, S.; Arber, N.; Baron, J.A.; Bombardier, C.; Cannon, C.; Farkouh, M.E.; FitzGerald, G.A.; et al. Vascular and upper gastrointestinal effects of non-steroidal anti-inflammatory drugs: Meta-analyses of individual participant data from randomised trials. Lancet 2013, 382, 769–779. [Google Scholar] [PubMed]
- Fine, M. Quantifying the impact of nsaid-associated adverse events. Am. J. Manag. Care 2013, 19, s267–s272. [Google Scholar] [PubMed]
- Serhan, C.N. Pro-resolving lipid mediators are leads for resolution physiology. Nature 2014, 510, 92–101. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Marine omega-3 fatty acids and inflammatory processes: Effects, mechanisms and clinical relevance. Biochim. Biophys. Acta 2015, 1851, 469–484. [Google Scholar] [CrossRef] [PubMed]
- Fortin, P.R.; Lew, R.A.; Liang, M.H.; Wright, E.A.; Beckett, L.A.; Chalmers, T.C.; Sperling, R.I. Validation of a meta-analysis: The effects of fish oil in rheumatoid arthritis. J. Clin. Epidemiol. 1995, 48, 1379–1390. [Google Scholar] [CrossRef]
- Goldberg, R.J.; Katz, J. A meta-analysis of the analgesic effects of omega-3 polyunsaturated fatty acid supplementation for inflammatory joint pain. Pain 2007, 129, 210–223. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.H.; Bae, S.C.; Song, G.G. Omega-3 polyunsaturated fatty acids and the treatment of rheumatoid arthritis: A meta-analysis. Arch. Med. Res. 2012, 43, 356–362. [Google Scholar] [CrossRef] [PubMed]
- MacLean, C.H.; Mojica, W.A.; Morton, S.C.; Pencharz, J.; Hasenfeld Garland, R.; Tu, W.; Newberry, S.J.; Jungvig, L.K.; Grossman, J.; Khanna, P.; et al. Effects of omega-3 fatty acids on lipids and glycemic control in type ii diabetes and the metabolic syndrome and on inflammatory bowel disease, rheumatoid arthritis, renal disease, systemic lupus erythematosus, and osteoporosis. Evid. Rep. Technol. Assess. 2004, 89, 1–4. [Google Scholar]
- Lane, K.; Derbyshire, E.; Li, W.; Brennan, C. Bioavailability and potential uses of vegetarian sources of omega-3 fatty acids: A review of the literature. Crit. Rev. Food Sci. Nutr. 2014, 54, 572–579. [Google Scholar] [CrossRef] [PubMed]
- Gerster, H. Can adults adequately convert alpha-linolenic acid (18:3 n-3) to eicosapentaenoic acid (20:5 n-3) and docosahexaenoic acid (22:6 n-3)? Int. J. Vitam. Nutr. Res. 1998, 68, 159–173. [Google Scholar] [PubMed]
- Herman, C.J.; Allen, P.; Hunt, W.C.; Prasad, A.; Brady, T.J. Use of complementary therapies among primary care clinic patients with arthritis. Prev. Chronic Dis. 2004, 1, A12. [Google Scholar] [PubMed]
- Christensen, R.; Bliddal, H.; Henriksen, M. Enhancing the reporting and transparency of rheumatology research: A guide to reporting guidelines. Arthritis Res. Ther. 2013, 15, 109. [Google Scholar] [CrossRef] [PubMed]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gotzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The prisma statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. Ann. Intern. Med. 2009, 151, W65–W94. [Google Scholar] [CrossRef] [PubMed]
- Ghogomu, E.A.; Maxwell, L.J.; Buchbinder, R.; Rader, T.; Pardo, P.J.; Johnston, R.V.; Christensen, R.; Rutjes, A.W.; Winzenberg, T.M.; Singh, J.A.; et al. Updated method guidelines for cochrane musculoskeletal group systematic reviews and metaanalyses. J. Rheumatol. 2014, 41, 194–205. [Google Scholar] [CrossRef] [PubMed]
- Tugwell, P.S.; Maxwell, L.J.; Beaton, D.E.; Busse, J.W.; Christensen, R.; Conaghan, P.G.; Simon, L.S.; Terwee, C.; Tovey, D.; Wells, G.A.; et al. Deliberative dialogue on developing consensus on measurement and presentation of patient important outcomes in systematic reviews: A preconference meeting at omeract 12. J. Rheumatol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Maxwell, L.J.; Wells, G.A.; Simon, L.S.; Conaghan, P.G.; Grosskleg, S.; Scrivens, K.; Beaton, D.E.; Bingham, C.O., III; Busse, J.W.; Christensen, R.; et al. Current state of reporting pain outcomes in cochrane reviews of chronic musculoskeletal pain conditions and considerations for an omeract research agenda. J. Rheumatol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Juni, P.; Altman, D.G.; Egger, M. Systematic reviews in health care: Assessing the quality of controlled clinical trials. BMJ 2001, 323, 42–46. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Altman, D.G.; Gotzsche, P.C.; Juni, P.; Moher, D.; Oxman, A.D.; Savovic, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A. The cochrane collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dwan, K.; Altman, D.G.; Arnaiz, J.A.; Bloom, J.; Chan, A.W.; Cronin, E.; Decullier, E.; Easterbrook, P.J.; Von, E.E.; Gamble, C.; et al. Systematic review of the empirical evidence of study publication bias and outcome reporting bias. PLoS ONE 2008, 3, e3081. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dwan, K.; Gamble, C.; Kolamunnage-Dona, R.; Mohammed, S.; Powell, C.; Williamson, P.R. Assessing the potential for outcome reporting bias in a review: A tutorial. Trials 2010, 11, 52. [Google Scholar] [CrossRef] [PubMed]
- Hedges, L.V. Distribution theory for glass’s estimator of effect size and related estimators. J. Educ. Behav. Stat. 1981, 6, 107–128. [Google Scholar] [CrossRef]
- Normand, S.L. Meta-analysis: Formulating, evaluating, combining, and reporting. Stat. Med. 1999, 18, 321–359. [Google Scholar] [CrossRef]
- Bliddal, H.; Christensen, R. The treatment and prevention of knee osteoarthritis: A tool for clinical decision-making. Expert Opin. Pharmacother. 2009, 10, 1793–1804. [Google Scholar] [CrossRef] [PubMed]
- Tubach, F.; Ravaud, P.; Martin-Mola, E.; Awada, H.; Bellamy, N.; Bombardier, C.; Felson, D.T.; Hajjaj-Hassouni, N.; Hochberg, M.; Logeart, I.; et al. Minimum clinically important improvement and patient acceptable symptom state in pain and function in rheumatoid arthritis, ankylosing spondylitis, chronic back pain, hand osteoarthritis, and hip and knee osteoarthritis: Results from a prospective multinational study. Arthritis Care Res. 2012, 64, 1699–1707. [Google Scholar]
- Higgins, J.P.; Thompson, S.G. Quantifying heterogeneity in a meta-analysis. Stat. Med. 2002, 21, 1539–1558. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Thompson, S.G.; Deeks, J.J.; Altman, D.G. Measuring inconsistency in meta-analyses. BMJ 2003, 327, 557–560. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johansen, M.; Bahrt, H.; Altman, R.D.; Bartels, E.M.; Juhl, C.B.; Bliddal, H.; Lund, H.; Christensen, R. Exploring reasons for the observed inconsistent trial reports on intra-articular injections with hyaluronic acid in the treatment of osteoarthritis: Meta-regression analyses of randomized trials. Semin. Arthritis Rheum. 2016, 46, 34–48. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Egger, M.; Davey, S.G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef] [PubMed]
- R Development Core Team. A Language and Environment for Statistical Computing, 3.1.2, R Foundation for Statistical Computing: Vienna, Austria, 2014.
- Guyatt, G.H.; Oxman, A.D.; Kunz, R.; Vist, G.E.; Falck-Ytter, Y.; Schunemann, H.J. What is “quality of evidence” and why is it important to clinicians? BMJ 2008, 336, 995–998. [Google Scholar] [CrossRef] [PubMed]
- Darlington, L.G.; Ramsey, N.W. Olive oil for rheumatoid patients? Br. J. Rheumatol. 1987, 26, 129. [Google Scholar]
- Tulleken, J.E.; Limburg, P.C.; Muskiet, F.A.J.; Kazemier, K.M.; Van Rijswijk, M.H. A Comparison between the Effects of Fish Oil Ethyl Esters and Fish Oil Triglycerides in Rheumatoid Arthritis, Long Chain Omega-3 Polyunsaturated Fatty Acids in Rheumatoid Arthritis. Thesis, Rijksuniversiteit Groningen, Groningen, The Netherlands, 1991. [Google Scholar]
- Tulleken, J.E.; Limburg, P.C.; Wassenaar, W.; Van Rijswijk, M.H. Nonsteroidal Anti-Inflammatory Drug Demand during Fish Oil Treatment in Rheumatoid Arthritis, Long Chain Omega-3 Polyunsaturated Fatty Acids in Rheumatoid Arthritis. Thesis, Rijksuniversiteit Groningen, Groningen, The Netherlands, 1991. [Google Scholar]
- Belch, J.J.F.; Ansell, D.; Madhok, R.; Sturrock, R.D. The effect of evening primrose oil epo and epo-fish oil combination on rheumatoid arthritis a double blind study. Br. J. Rheumatol. 1986, 25. [Google Scholar]
- Belch, J.J.; Ansell, D.; Madhok, R.; O’Dowd, A.; Sturrock, R.D. Effects of altering dietary essential fatty acids on requirements for non-steroidal anti-inflammatory drugs in patients with rheumatoid arthritis: A double blind placebo controlled study. Ann. Rheum. Dis. 1988, 47, 96–104. [Google Scholar] [CrossRef] [PubMed]
- Cleland, L.G.; French, J.K.; Betts, W.H.; Murphy, G.A.; Elliott, M.J. Clinical and biochemical effects of dietary fish oil supplements in rheumatoid arthritis. J. Rheumatol. 1988, 15, 1471–1475. [Google Scholar] [PubMed]
- Stammers, T.; Sibbald, B.; Freeling, P. Fish oil in osteoarthritis. Lancet 1989, 2, 503. [Google Scholar] [CrossRef]
- Kremer, J.M.; Lawrence, D.A.; Jubiz, W.; DiGiacomo, R.; Rynes, R.; Bartholomew, L.E.; Sherman, M. Dietary fish oil and olive oil supplementation in patients with rheumatoid arthritis. Clinical and immunologic effects. Arthritis Rheum. 1990, 33, 810–820. [Google Scholar] [CrossRef] [PubMed]
- Tulleken, J.E.; Limburg, P.C.; Muskiet, F.A.; van Rijswijk, M.H. Vitamin e status during dietary fish oil supplementation in rheumatoid arthritis. Arthritis Rheum. 1990, 33, 1416–1419. [Google Scholar] [CrossRef] [PubMed]
- van der Tempel, H.; Tulleken, J.E.; Limburg, P.C.; Muskiet, F.A.; van Rijswijk, M.H. Effects of fish oil supplementation in rheumatoid arthritis. Ann. Rheum. Dis. 1990, 49, 76–80. [Google Scholar] [CrossRef] [PubMed]
- Tulleken, J.E.; Limburg, P.; Vanrijswijk, M.H. Fish oil and plasma-fibrinogen. BMJ 1988, 297, 615. [Google Scholar]
- Espersen, G.T.; Grunnet, N.; Lervang, H.H.; Nielsen, G.L.; Thomsen, B.S.; Faarvang, K.L.; Dyerberg, J.; Ernst, E. Decreased interleukin-1 beta levels in plasma from rheumatoid arthritis patients after dietary supplementation with n-3 polyunsaturated fatty acids. Clin. Rheumatol. 1992, 11, 393–395. [Google Scholar] [CrossRef] [PubMed]
- Kjeldsen-Kragh, J.; Lund, J.A.; Riise, T.; Finnanger, B.; Haaland, K.; Finstad, R.; Mikkelsen, K.; Forre, O. Dietary omega-3 fatty acid supplementation and naproxen treatment in patients with rheumatoid arthritis. J. Rheumatol. 1992, 19, 1531–1536. [Google Scholar] [PubMed]
- Kjeldsen-Kragh, J. Dietary treatment of rheumatoid arthritis. Scand. J. Rheumatol. 1996, 25, 63. [Google Scholar] [CrossRef]
- Magarò, M.; Zoli, A.; Altomonte, L.; Mirone, L.; De Sole, P.; Di Mario, G.; De Leo, E. Effect of fish oil on neutrophil chemiluminescence induced by different stimuli in patients with rheumatoid arthritis. Ann. Rheum. Dis. 1992, 51, 877–880. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, G.L.; Faarvang, K.L.; Thomsen, B.S.; Teglbjaerg, K.L.; Jensen, L.T.; Hansen, T.M.; Lervang, H.H.; Schmidt, E.B.; Dyerberg, J.; Ernst, E. The effects of dietary supplementation with n-3 polyunsaturated fatty acids in patients with rheumatoid arthritis: A randomized, double blind trial. Eur. J. Clin. Invest. 1992, 22, 687–691. [Google Scholar] [CrossRef] [PubMed]
- Faarvang, K.L.; Nielsen, G.L.; Thomsen, B.S.; Teglbjaerg, K.L.; Hansen, T.M.; Lervang, H.H.; Schmidt, E.B.; Dyerberg, J.; Ernst, E. Fish oils and rheumatoid arthritis. A randomized and double—blind study. Ugeskr. Laeger 1994, 156, 3495–3498. [Google Scholar] [PubMed]
- Nielsen, G.L.; Faarvang, K.L.; Thomsen, B.S.; Teglibjaerg, K.L.; Ernst, E. Effects of supplementation with n-3 fatty acids on clinical disease variables in patients with rheumatoid arthritis. Eur. J. Clin. Invest. 1991, 21, 67. [Google Scholar]
- Sköldstam, L.; Börjesson, O.; Kjällman, A.; Seiving, B.; Åkesson, B. Effect of six months of fish oil supplementation in stable rheumatoid arthritis. A double-blind, controlled study. Scand. J. Rheumatol. 1992, 21, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Stammers, T.; Sibbald, B.; Freeling, P. Efficacy of cod liver oil as an adjunct to non-steroidal anti-inflammatory drug treatment in the management of osteoarthritis in general practice. Ann. Rheum. Dis. 1992, 51, 128–129. [Google Scholar] [CrossRef] [PubMed]
- Geusens, P.; Wouters, C.; Nijs, J.; Jiang, Y.; Dequeker, J. Long-term effect of omega-3 fatty acid supplementation in active rheumatoid arthritis. A 12-month, double-blind, controlled study. Arthritis Rheum. 1994, 37, 824–829. [Google Scholar] [CrossRef] [PubMed]
- Kremer, J.M.; Lawrence, D.A.; Petrillo, G.F.; Litts, L.L.; Mullaly, P.M.; Rynes, R.I.; Stocker, R.P.; Parhami, N.; Greenstein, N.S.; Fuchs, B.R.; et al. Effects of high-dose fish oil on rheumatoid arthritis after stopping nonsteroidal antiinflammatory drugs. Clinical and immune correlates. Arthritis Rheum. 1995, 38, 1107–1114. [Google Scholar] [CrossRef] [PubMed]
- Vargová, V.; Vesely, R.; Sasinka, M.; Torok, C. Will administration of omega-3 unsaturated fatty acids reduce the use of nonsteroidal antirheumatic agents in children with chronic juvenile arthritis? Cas. Lek. Cesk. 1998, 137, 651–653. [Google Scholar] [PubMed]
- Volker, D.; Fitzgerald, P.; Major, G.; Garg, M. Efficacy of fish oil concentrate in the treatment of rheumatoid arthritis. J. Rheumatol. 2000, 27, 2343–2346. [Google Scholar] [PubMed]
- Adam, O.; Beringer, C.; Kless, T.; Lemmen, C.; Adam, A.; Wiseman, M.; Adam, P.; Klimmek, R.; Forth, W. Anti-inflammatory effects of a low arachidonic acid diet and fish oil in patients with rheumatoid arthritis. Rheumatol. Int. 2003, 23, 27–36. [Google Scholar] [PubMed]
- Lau, C.S.; Chiu, P.K.Y.; Chu, E.M.Y.; Cheng, I.Y.W.; Tang, W.M.; Man, R.Y.K.; Halpern, G.M. Treatment of knee osteoarthritis with lyprinol, lipid extract of the green-lipped mussel-a double-blind placebo-controlled study. Prog. Nutr. 2004, 6, 17–31. [Google Scholar]
- Sundrarjun, T.; Komindr, S.; Archararit, N.; Dahlan, W.; Puchaiwatananon, O.; Angthararak, S.; Udomsuppayakul, U.; Chuncharunee, S. Effects of n-3 fatty acids on serum interleukin-6, tumour necrosis factor-alpha and soluble tumour necrosis factor receptor p55 in active rheumatoid arthritis. J. Int. Med. Res. 2004, 32, 443–454. [Google Scholar] [CrossRef] [PubMed]
- Berbert, A.A.; Kondo, C.R.; Almendra, C.L.; Matsuo, T.; Dichi, I. Supplementation of fish oil and olive oil in patients with rheumatoid arthritis. Nutrition 2005, 21, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Madland, T.M.; Bjorkkjaer, T.; Brunborg, L.A.; Froyland, L.; Berstad, A.; Brun, J.G. Subjective improvement in patients with psoriatic arthritis after short-term oral treatment with seal oil. A pilot study with double blind comparison to soy oil. J. Rheumatol. 2006, 33, 307–310. [Google Scholar] [PubMed]
- Deutsch, L. Evaluation of the effect of Neptune krill oil on chronic inflammation and arthritic symptoms. J. Am. Coll. Nutr. 2007, 26, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Galarraga, B.; Ho, M.; Youssef, H.M.; Hill, A.; McMahon, H.; Hall, C.; Ogston, S.; Nuki, G.; Belch, J.J. Cod liver oil (n-3 fatty acids) as an non-steroidal anti-inflammatory drug sparing agent in rheumatoid arthritis. Rheumatology (Oxf., Engl.) 2008, 47, 665–669. [Google Scholar] [CrossRef] [PubMed]
- Das Gupta, A.B.; Hossain, A.K.; Islam, M.H.; Dey, S.R.; Khan, A.L. Role of omega-3 fatty acid supplementation with indomethacin in suppression of disease activity in rheumatoid arthritis. Bangladesh Med. Res. Counc. Bull. 2009, 35, 63–68. [Google Scholar] [PubMed]
- Gruenwald, J.; Petzold, E.; Busch, R.; Petzold, H.P.; Graubaum, H.J. Effect of glucosamine sulfate with or without omega-3 fatty acids in patients with osteoarthritis. Adv. Ther. 2009, 26, 858–871. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.; Lee, A.; Shim, S.C.; Lee, J.H.; Choe, J.Y.; Ahn, H.; Choi, C.B.; Sung, Y.K.; Bae, S.C. Effect of n-3 polyunsaturated fatty acid supplementation in patients with rheumatoid arthritis: A 16-week randomized, double-blind, placebo-controlled, parallel-design multicenter study in korea. J. Nutr. Biochem. 2013, 24, 1367–1372. [Google Scholar] [CrossRef] [PubMed]
- Araújo, C.A.; Moraes-Fontes, M.F.; Santos, L.; Riso, N. Omega-3 fatty acids and mediterranean diet as complimentary therapies for rheumatoid arthritis. Arthritis Rheumatol. 2014, 66, S1050. [Google Scholar]
- Stebbings, S.; Gray, A.; Schneiders, A.; Sansom, A. A novel green-lipped mussel lipid extract (biolex) for the treatment of pain in osteoarthritis of the hip and knee: A randomized double-blind, placebo controlled trial. Ann. Rheum. Dis. 2014, 73. [Google Scholar] [CrossRef]
- Fu, Y.; Li, G.; Zhang, X.; Xing, G.; Hu, X.; Yang, L.; Li, D. Lipid extract from hard-shelled mussel (mytilus coruscus) improves clinical conditions of patients with rheumatoid arthritis: A randomized controlled trial. Nutrients 2015, 7, 625–645. [Google Scholar] [CrossRef]
- Kremer, J.M.; Rynes, R.I.; Bartholomew, L.E.; Michalek, A.; Jubiz, W. A double-blinded placebo controlled crossover study of eicosapentaenoic-acid supplementation in active rheumatoid arthritis. Arthritis Rheum. 1986, 29, S11. [Google Scholar]
- Hernández-Cruz, B.; Alcocer-Varela, J.; Cardiel, M.H. Omega-3 fatty acids supplementation in Mexican patients with rheumatoid arthritis with standard treatment. A blinded, randomized, placebo controlled, one year, clinical trial. Arthritis Rheum. 1998, 41, S155. [Google Scholar]
- Kremer, J.; Larence, D.; Jubiz, W.; Digarcomo, R.; Rynes, R.; Bartholomew, L. Different doses of fish-oil fatty acid supplementation in rheumatoid arthritis ra a prospective double-blinded randomized study. Arthritis Rheum. 1988, 31, S303. [Google Scholar]
- Kremer, J.M.; Lawrence, D.A.; Stocker, R.S.; Rynes, R.I.; Parhami, N.; Greenstein, N.S.; Petrillo, G.F.; Simon, L.S.; Robinson, D.W. Clinical, immunological and nsaid sparing effects of fish-oil (fo) in patients with active rheumatoid-arthritis (ra)-a long-term prospective randomized double-blind-study. Arthritis Rheum. 1993, 36, S56. [Google Scholar]
- Lau, C.S.; Morley, K.D.; Belch, J.J.F. Effects of fish-oil supplementation on nonsteroidal antiinflammatory drug requirement in patients with mild rheumatoid-arthritis-a double-blind placebo-controlled study. Br. J. Rheumatol. 1993, 32, 982–989. [Google Scholar] [CrossRef] [PubMed]
- Lau, C.S.; McMahon, H.; Morley, K.D.; Belch, J.J.F. Effects of maxepa on non-steroidal anti-inflammatory drug usage in patients with mild rheumatoid arthritis. Br. J. Rheumatol. 1991, 30, 137. [Google Scholar]
- Lau, C.S.; McLaren, M.; Belch, J.J. Effects of fish oil on plasma fibrinolysis in patients with mild rheumatoid arthritis. Clin. Exp. Rheumatol. 1995, 13, 87–90. [Google Scholar] [PubMed]
- Kolahi, S.O.; Hejasi, J.A.; Mohtadinia, A. The effect of fish oil and vitamin e supplement over lipid peroxidation and antioxidant levels of plasma in patient with rheumatoid arthritis. Int. J. Rheum. Dis. 2010, 13, 108. [Google Scholar]
- Kolahi, S.; Ghorbanihaghjo, A.; Alizadeh, S.; Rashtchizadeh, N.; Argani, H.; Khabazzi, A.R.; Hajialilo, M.; Bahreini, E. Fish oil supplementation decreases serum soluble receptor activator of nuclear factor-kappa b ligand/osteoprotegerin ratio in female patients with rheumatoid arthritis. Clin. Biochem. 2010, 43, 576–580. [Google Scholar] [CrossRef] [PubMed]
- Ghorbanihaghjo, A.; Kolahi, S.; Seifirad, S.; Rashtchizadeh, N.; Argani, H.; Hajialilo, M.; Khabazi, A.; Alizadeh, S.; Bahreini, E. Effect of fish oil supplements on serum paraoxonase activity in female patients with rheumatoid arthritis: A double-blind randomized controlled trial. Arch. Iran. Med. 2012, 15, 549–552. [Google Scholar] [PubMed]
- Kolahi, S.; Haghjoo, A.G. Effect of fish oil supplements on serum paraoxonase activity in patients with rheumatoid arthritis. Rheumatology (Oxf., Engl.) 2012, 51, i31. [Google Scholar]
- Alizadeh, S.; Ghorbanihaghjo, A.; Kolahi, S.; Bahreini, E. Effect of omega-3 fatty acids supplementation on circulating osteoprotegerin ligand to osteoprotegerin ratio in female patients with rheumatoid arthritis. Clin. Biochem. 2011, 1, S142. [Google Scholar] [CrossRef]
- Dawczynski, C.; Hackermeier, U.; Viehweger, M.; Stange, R.; Springer, M.; Jahreis, G. Incorporation of n-3 pufa and gamma-linolenic acid in blood lipids and red blood cell lipids together with their influence on disease activity in patients with chronic inflammatory arthritis—A randomized controlled human intervention trial. Lipids Health Dis. 2011, 10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reed, G.W.; Leung, K.; Rossetti, R.G.; Vanbuskirk, S.; Sharp, J.T.; Zurier, R.B. Treatment of rheumatoid arthritis with marine and botanical oils: An 18-month, randomized, and double-blind trial. Evid. Based Complement. Altern. Med. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Olendzki, B.C.; Leung, K.; Van Buskirk, S.; Reed, G.; Zurier, R.B. Treatment of rheumatoid arthritis with marine and botanical oils: Influence on serum lipids. Evid. Based Complement. Altern. Med. 2011, 2011, 827286. [Google Scholar] [CrossRef] [PubMed]
- Yazdanpanah, P.; Mohamadi, H.; Sadeghi, H.; Ghaffarian Shirazi, H.; Arjmand, A. Survey of omega-3 effects on morning stiffness in osteoarthritis of the knee. Pain Pract. 2014, 14, 126. [Google Scholar]
- Singh, G.; Chandra, R.K. Fish oils in rheumatoid arthritis. Ann. Intern. Med. 1988, 108, 904–905. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Omega-3 polyunsaturated fatty acids and inflammatory processes: Nutrition or pharmacology? Br. J. Clin. Pharmacol. 2013, 75, 645–662. [Google Scholar] [CrossRef] [PubMed]
- Stamp, L.K.; James, M.J.; Cleland, L.G. Diet and rheumatoid arthritis: A review of the literature. Semin. Arthritis Rheum. 2005, 35, 77–94. [Google Scholar] [CrossRef] [PubMed]
- Villani, A.M.; Crotty, M.; Cleland, L.G.; James, M.J.; Fraser, R.J.; Cobiac, L.; Miller, M.D. Fish oil administration in older adults: Is there potential for adverse events? A systematic review of the literature. BMC Geriatr. 2013, 13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences, 2nd ed.; Lawrence Erlbaum Associates: Hillsdale, NJ, USA, 1988. [Google Scholar]
 : adequate methodology,
: adequate methodology,  : unclear methodology,
: unclear methodology,  : inadequate methodology. Risk of outcome reporting bias was not assessed for Vargová et al. (1998) [49], since only the abstract was available in English, and the rest of the article was not in English. Therefore, it was not considered appropriate to assess outcome reporting bias based only on the abstract of a full article.
: inadequate methodology. Risk of outcome reporting bias was not assessed for Vargová et al. (1998) [49], since only the abstract was available in English, and the rest of the article was not in English. Therefore, it was not considered appropriate to assess outcome reporting bias based only on the abstract of a full article.
 : adequate methodology,
: adequate methodology,  : unclear methodology,
: unclear methodology,  : inadequate methodology. Risk of outcome reporting bias was not assessed for Vargová et al. (1998) [49], since only the abstract was available in English, and the rest of the article was not in English. Therefore, it was not considered appropriate to assess outcome reporting bias based only on the abstract of a full article.
: inadequate methodology. Risk of outcome reporting bias was not assessed for Vargová et al. (1998) [49], since only the abstract was available in English, and the rest of the article was not in English. Therefore, it was not considered appropriate to assess outcome reporting bias based only on the abstract of a full article.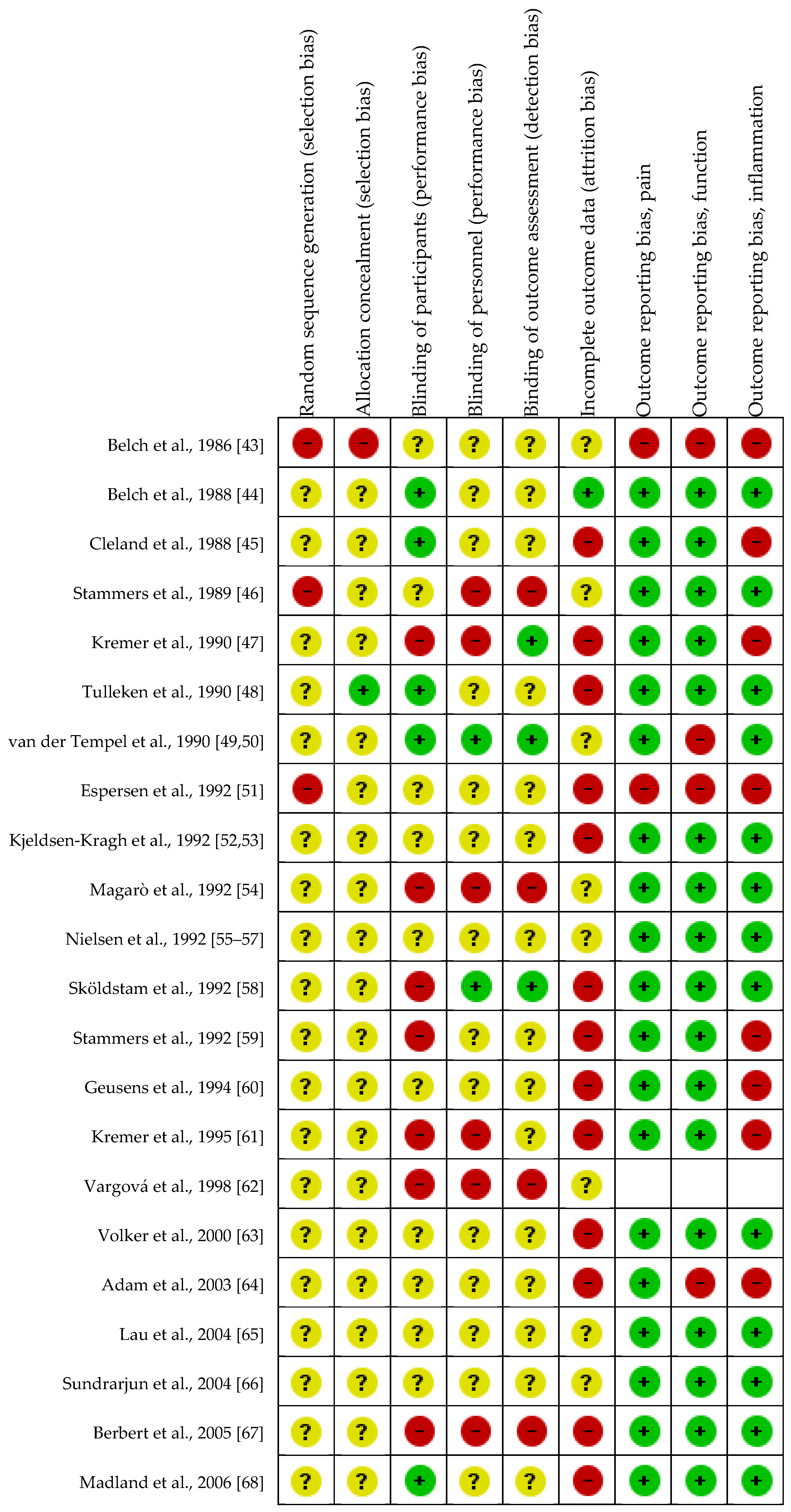
| Author, Year [Reference Number], (Trial Registry Number) | N Total | Study Design | Duration of Intervention (Weeks) | Diagnosis | Mean Age (Years) | % Females | Mean Disease Duration (Years) | No. of Patients Receiving Intervention | Intervention | Dose of EPA + DHA (g/Day) | No. of Patients Receiving Control | Control Treatment |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Trials with complete data for pain | ||||||||||||
| Belch et al. 1986 [43] | 34 | Three-arm, parallel | 52 | RA | na | na | na | 11 c | MO (whole fish), and EPO (480 mg GLA) | 0.24 + na | 12 c | Oil (unspecified) k |
| 11 c | EPO (540 mg GLA) | |||||||||||
| Belch et al. 1988 [44] | 49 | Three-arm, parallel | 52 | RA | 49.0 | 29.3 | 5.0 | 15 | MO (whole fish), and EPO (450 mg GLA) | 0.24 + na | 18 | Paraffin k |
| 16 | EPO (540 mg GLA) | |||||||||||
| Cleland et al. 1988 [45] | 60 | Two-arm, parallel | 12 | RA | 50.5 | 53.3 | 8.3 | 30 | MO (whole fish) | 3.20 + 2.00 | 30 | Olive oil |
| Stammers et al. 1989 [46] | 26 | Two-arm, parallel | 26 | OA | 52–84 | 80.8 | na | na | MO (unspecified) | na | na | Oil (unspecified) |
| Kremer et al. 1990 [47] | 64 | Three-arm, parallel c | 24 | RA | 58.3 | 51.6 | 13.8 | 19 | MO (whole fish), high dose | 4.05 e + 2.70 e | 23 | Olive oil j |
| 22 | MO (whole fish), low dose | 2.03 e + 1.35 e | ||||||||||
| Tulleken et al. 1990 [48] | 28 | Two-arm, parallel | 13 | RA | 55.0 | 85.7 | 18.0 | 14 | MO (whole fish) | 2.04 + 1.32 | 14 | Coconut oil w/fish flavor |
| van der Tempel et al. 1990 [49,50] | 16 | Cross-over, no WO | 12 a | RA | 53.0 | 56.3 | 12.0 | 16 | MO (whole fish) | 2.04 + 1.32 | 16 | Coconut oil w/fish aroma |
| Espersen et al. 1992 [51] | 32 | Two-arm parallel | 12 | RA | na | na | na | 18 c | MO (whole fish) | 2.00 + 1.20 | 14 c | Mix (38% MUFA and 21% PUFA) |
| Kjeldsen-Kragh et al. 1992 [52,53] | 79 | Three-arm, parallel | 16 | RA | 57.3 | 21.5 | 8.5 | 26 | MO (whole fish), and declining NSAID | 3.78 + 1.96 | 28 | Corn oil and declining NSAID |
| 25 | MO (whole fish), and NSAID k | 3.78 + 1.96 | ||||||||||
| Magarò et al. 1992 [54] | 20 | Two-arm, parallel | 6 | RA | 25–45 | 100.0 | na | 10 | MO (whole fish) | 1.60 + 1.10 | 10 | None |
| Nielsen et al. 1992 [55,56,57] | 57 | Two-arm, parallel | 12 | RA | 61.0 b | na | 5.0 b | 29 | MO (whole fish) | 2.00 + 1.20 | 28 | Mix of FA |
| Sköldstam et al. 1992 [58] | 46 | Two-arm, parallel | 26 | RA | 57.0 | 73.9 | 18.0 | 23 | MO (whole fish) | 1.80 + 1.20 | 23 | Maize oil, olive oil and peppermint oil |
| Stammers et al. 1992 [59] | 86 | Two-arm, parallel | 24 | OA | 68.0 | 72.1 | 15.5 | 44 | MO (cod liver oil) | 0.79 + 0.70 g | 42 | Olive oil |
| Geusens et al. 1994 [60] | 90 | Three-arm, parallel | 52 | RA | 57.3 | 52.2 | 10.1 | 30 | MO (whole fish), high dose | 1.68 + 0.36 | 30 | Olive oil j |
| 30 | MO (whole fish), low dose | 0.84 + 0.18 | ||||||||||
| Kremer et al. 1995 [61] | 66 | Four-arm, parallel | 26 or 30 | RA | 57.5 | 40.9 | 10.5 | 33 c | MO (whole fish), diclofenac, and diclofenac placebo | 4.29 e + 2.34 e | 33 c | Corn oil, diclofenac, and diclofenac placebo |
| Vargová et al. 1998 [62] | 23 | Two-arm, parallel | 21 | JCA | 10.1 | na | na | 13 | “diet with increased content of omega-3 PUFA” | na | 10 | Unspecified |
| Volker et al. 2000 [63] | 50 | Two-arm, parallel | 15 | RA | 57.0 | n | 13.5 | 25 | MO (whole fish) | 0.90 e,g +0.63 e,g | 25 | 50:50 corn oil and olive oil |
| Adam et al. 2003 [64] | 68 | Two parallel cross-over studies, 8.7 weeks. WO | 13 a | RA | 56.8 | 41.2 | 9.6 | 34 | MO (whole fish), and WD | 0.80 e + 0.60 e | 34 | Corn oil, and WD |
| 58.0 | 41.2 | 9.5 | 34 | MO (whole fish), and AID | 0.84 e + 0.63 e | 34 | Corn oil, and AID | |||||
| Lau et al. 2004 [65] | 80 | Two-arm, parallel | 24 | Knee OA | 62.5 | 86.3 | 8.9 | 40 | MO (GLM extract) | na | 40 | Olive oil |
| Sundrarjun et al. 2004 [66] | 60 | Three-arm, parallel | 12 | RA | 46.9 | 85.0 | 4.4 | 23 | MO (whole fish) | 1.88 + 1.48 | 23 | Unspecified |
| na | 14 | None k | ||||||||||
| Berbert et al. 2005 [67] | 55 | Three-arm, parallel | 24 | RA | 49.0 | 61.8 | 15.3 | 18 | MO (whole fish) | 1.80 + 1.20 | 17 | Soy oil |
| 20 | MO (whole fish), and olive oil k | 1.80 + 1.20 | ||||||||||
| Madland et al. 2006 [68] | 43 | Two-arm, parallel | 2 | p.a. PsA | 55.0 | 51.2 | 13.0 b | 22 | MO (seal oil) | 2.40 + 2.60 | 21 | Soy oil (bottle) |
| Deutsch 2007 [69] | 90 | Two-arm, parallel | 4 | RA/CVD/OA | 55.0 | 47.8 | na | 45 | MO (krill oil) | 0.05 + 0.03 | 45 | Microcrystalline cellulose |
| Galarraga et al. 2008 [70] | 97 | Two-arm, parallel | 36 | RA | 59.5 | 71.1 | 13.0 | 49 | MO (mix of oil from cod liver and whole fish) | 1.50 + 0.70 | 48 | Air filled capsule |
| Das Gupta et al. 2009 [71] | 100 | Two-arm, parallel | 12 | RA | 47.3 | na | na | 50 | MO (whole fish), and indomethacin | 1.80 g + 1.20 g | 50 | Indomethacin |
| Gruenwald et al. 2009 [72] (EUCTR200500366918DE) | 177 | Two-arm, parallel | 26 | Knee/hip OA | 62.3 | 63.8 | na | 90 | MO (mix of oil from cod liver and whole fish), and glucosamine sulfate | 0.60 i | 87 | Mix of oils, and glucosamine sulfate |
| Park et al. 2013 [73] (NCT01618019) | 109 | Two-arm, parallel | 16 | RA | 48.4 | 34.4 | 8.4 | 55 | MO (whole fish) | 2.09 + 1.17 | 54 | Sunflower oil w/oleic acid |
| Araújo et al. 2014 [74] | 37 | Three-arm, parallel | 26 | RA | na | na | na | 11 c | MO (unspecified) | na | 15 c | na |
| 8 | Mediterranean diet k | na | ||||||||||
| Stebbings et al. 2014 [75] | 80 | Two-arm, parallel | 12 | Knee or hip OA | 66.4 | 55.0 | na | 39 | MO (GLM) | 0.01 +0.01 | 41 | Corn oil |
| Fu et al. 2015 [76] (NCT02173587) | 50 | Two-arm, parallel | 26 | RA | 57.5 | 60.0 | 7.6 | 25 | 50:50 MO (HMLE) and corn oil | 0.07 f + 0.10 f | 25 | Corn oil |
| Trials with incomplete data or no data for pain | ||||||||||||
| Kremer et al. 1986 [77] | 36 | Cross-over, 4 weeks WO | 14 a | RA | na | na | na | 36 | MO (whole fish) | 2.70 + na | 36 | Unspecified |
| Darlington & Ramsey 1987 [40] | 35 | Two-arm, parallel | 12 | RA | na | na | na | (17) d | MO (whole fish) | 3.24 + 2.16 | (18) d | Olive oil |
| Hernández-Cruz et al. 1988 [78] | 90 | Two-arm, parallel | 52 | RA | 43.2 | 89.0 | 3.4 | 45 | MO (whole fish) | 1.50 + na | 45 | Sunflower oil |
| Kremer et al. 1988 [79] | 55 | Three-arm, parallel | 24 | RA | na | na | na | (18) d | MO (whole fish), high dose | 4.05 e + 2.70 e | (19) d | Olive oil j |
| (18) d | MO (whole fish), low dose | 2.03 e + 1.35 e | ||||||||||
| Kremer et al. 1993 [80] | 50 | Two-arm, parallel | 26 or 30 | RA | na | na | na | (25) d | MO (whole fish), diclofenac, and diclofenac placebo | 9.75 e,h | (25) d | Corn oil, diclofenac, and diclofenac placebo |
| Lau et al. 1993 [81,82] | 64 | Two-arm, parallel | 52 | RA | 51.4 | 70.3 | 4.2 | 32 | MO (whole fish) | 1.71 + 1.14 | 32 | Air-filled capsule |
| Lau et al. 1995 [83] | 45 | Two-arm, parallel | 26 | RA | 51.0 b | 71.1 | 2.3 b | 25 | MO (whole fish) | 1.70 + 1.10 | 20 | Air-filled capsule |
| Kolahi et al. 2010 [84] | 90 | Three-arm, parallel | na | RA | na | Na | na | (30) d | MO (whole fish), and vitamin E-placebo | na | (30) d | MO placebo (unspecified) and vitamin E placebo |
| (30) d | MO (whole fish), and vitamin E k | |||||||||||
| Kolahi et al. 2010 [85,86,87,88] (IRCT138902073812N1) | 90 | Two-arm, parallel | 13 | RA | 50.0 b | 92.2 | 4.6 b | 45 | MO (whole fish) | 0.18 + 0.12 | 45 | Paraffin |
| Dawczynski et al. 2011 [89] (NCT01179971) | 60 | Four-arm, parallel | 12 | RA (or PsA) | 56.2 | 71.7 | na | 15 | MO (unspecified) | 3.00 l | 15 | Olive oil |
| 15 | MO (unspecified), and GLA (1800 mg) | 1.58 l | 15 | GLA (3150 mg) | ||||||||
| Reed et al. 2014 [90,91] (NCT00072982) | 150 | Three-arm, parallel | 78 | RA | 59.2 | 81.3 | 8.6 b | 53 | MO (whole fish), and borage seed oil | 2.10 + 1.40 | 52 | Borage seed oil, and sunflower seed oil |
| 45 | MO (whole fish), and sunflower seed oil k | 2.10 + 1.40 | ||||||||||
| Yazdanpanah et al. 2014 [92] (IRCT2012102610799N2) | 114 | Six-arm, parallel | 6 | Knee OA | na | na | na | 19 | Omega-3, and acetaminophen and naproxen | 1.00 h | 19 | Acetaminophen and naproxen |
| 19 | Omega-3 and acetaminophen | 1.00 h | 19 | Acetaminophen | ||||||||
| 19 | Omega-3 and naproxen | 1.00 h | 19 | Naproxen | ||||||||
| Variable | Total Trials, k | SMD for Pain | 95% CI | τ2 | I2 | p-Value for Interaction |
|---|---|---|---|---|---|---|
| All trials | 30 | −0.24 | (−0.42 to −0.07) | 0.150 | 63% | n.a. |
| Diagnoses | 0.147 | 60% | 0.024 a | |||
| RA | 22 | −0.21 | (−0.42 to −0.00) a | |||
| OA | 5 | −0.16 | (−0.57 to 0.24) | |||
| Other | 3 | −0.63 | (−1.20 to −0.06) a | |||
| Supplementation type | 0.137 | 60% | 0.009 a | |||
| Capsule | 23 | −0.25 | (−0.44 to −0.06) a | |||
| Bottle | 3 | 0.19 | (−0.37 to 0.75) | |||
| Unclear | 4 | −0.61 | (−1.16 to −0.05) a | |||
| Type of control | 0.148 | 60% | 0.051 | |||
| PUFA w/o EPA and DHA | 11 | −0.12 | (−0.41 to 0.17) | |||
| Non-PUFA oils | 10 | −0.30 | (−0.59 to −0.01) a | |||
| Non-oils | 3 | −0.63 | (−1.13 to −0.12) a | |||
| Unclear | 3 | −0.01 | (−0.59 to 0.56) | |||
| None | 3 | −0.21 | (−0.87 to 0.44) | |||
| Duration | 0.165 | 63% | 0.074 | |||
| <12 weeks | 3 | −0.30 | (−0.89 to 0.30) | |||
| ≥12 weeks and <24 weeks | 12 | −0.25 | (−0.53 to 0.03) | |||
| ≥24 weeks | 15 | −0.23 | (−0.48 to 0.03) | |||
| Unspecified | 0 | |||||
| Ratio of EPA/DHA | 0.153 | 61% | 0.031 a | |||
| Ratio of EPA/DHA ≤1.5 | 12 | −0.12 | (−0.40 to 0.15) | |||
| Ratio of EPA/DHA of >1.5 | 11 | −0.38 | (−0.67 to −0.10) a | |||
| Unspecified | 7 | −0.23 | (−0.62 to 0.17) | |||
| Dosage of EPA plus DHA | 0.138 | 58% | 0.016 a | |||
| <2.6 g/day | 8 | −0.45 | (−0.75 to −0.15) a | |||
| ≥2.6 g/day and <3.6 g/day | 11 | −0.21 | (−0.51 to 0.09) | |||
| ≥3.6 g/day | 4 | 0.13 | (−0.32 to 0.57) | |||
| Unspecified | 7 | −0.22 | (−0.60 to 0.16) | |||
| Source of marine oil | 0.130 | 57% | 0.011 a | |||
| Whole fish | 19 | −0.17 | (−0.38 to 0.05) | |||
| Mussel | 2 | −0.95 | (−1.60 to −0.31) a | |||
| Other | 6 | −0.19 | (−1.53 to 0.16) | |||
| Unspecified | 3 | −0.35 | (−0.97 to 0.26) | |||
| Bias domains | ||||||
| Random sequence generation (selection bias) | 0.160 | 61% | 0.043 a | |||
| Adequate | 4 | −0.33 | (−0.78 to 0.13) | |||
| Unclear | 22 | −0.19 | (−0.40 to 0.02) | |||
| Inadequate | 4 | −0.48 | (−1.00 to 0.05) | |||
| Allocation concealment (selection bias) | 0.155 | 61% | 0.039 a | |||
| Adequate | 3 | −0.52 | (−1.09 to 0.05) | |||
| Unclear | 25 | −0.22 | (−0.42 to −0.03) a | |||
| Inadequate | 2 | −0.03 | (−0.76 to 0.70) | |||
| Blinding of participants (performance bias) | 0.149 | 61% | 0.026 a | |||
| Adequate | 9 | −0.10 | (−0.42 to 0.22) | |||
| Unclear | 10 | −0.41 | (−0.70 to −0.11) a | |||
| Inadequate | 11 | −0.20 | (−0.49 to 0.10) | |||
| Blinding of personnel (performance bias) | 0.163 | 63% | 0.060 | |||
| Adequate | 5 | −0.25 | (−0.70 to 0.20) | |||
| Unclear | 17 | −0.29 | (−0.52 to −0.05) a | |||
| Inadequate | 8 | −0.13 | (−0.50 to 0.24) | |||
| Incomplete outcome data (attrition bias) | 0.152 | 60% | 0.020 a | |||
| Adequate | 2 | 0.06 | (−0.61 to 0.73) | |||
| Unclear | 10 | −0.44 | (−0.77 to −0.12) a | |||
| Inadequate | 18 | −0.18 | (−0.40 to 0.04) | |||
| Outcome reporting (outcome reporting bias for pain) | 0.145 | 61% | 0.016 a | |||
| Adequate | 27 | −0.20 | (−0.38 to −0.02) a | |||
| Unclear | 1 | −0.94 | (−2.10 to 0.21) | |||
| Inadequate | 2 | −0.72 | (−1.52 to 0.08) | |||
| Funding source | 0.149 | 61% | 0.031 a | |||
| Industry only | 8 | −0.10 | (−0.41 to 0.21) | |||
| Mixed | 8 | −0.10 | (−0.44 to 0.25) | |||
| Nonprofit only | 4 | −0.41 | (−0.87 to 0.05) | |||
| Not reported | 7 | −0.31 | (−0.70 to 0.08) | |||
| Unclear | 3 | −0.80 | (−1.42 to −0.16) a | |||
| Quality Assessment | No. of Patients | Effect | Quality | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| No. of Trials | Risk of Bias | Inconsistency | Indirectness | Imprecision | Other Considerations | MO | Non-MO | Relative (95% CI) | Absolute (95% CI) | |
| Pain | ||||||||||
| 30 | Serious (−1) a | Serious (−1) b | Not serious | Not serious | None | 781 | 728 | - | SMD 0.24 lower (0.42 lower to 0.07 lower) | ⨁⨁LOW |
| RA 22 | Serious (−1) a | Not serious | Not serious | Not serious | None | 499 | 457 | - | SMD 0.21 lower (0.42 lower to −0.004 lower) | ⨁⨁⨁MODERATE |
| OA 5 | Serious (−1) a | Serious (−1) b | Not serious | Serious (−1) d | None | 205 | 198 | - | SMD 0.16 lower (0.57 lower to 0.24 higher) | ⨁VERY LOW |
| Other 3 | Serious (−1) a | Serious (−1) b | Serious (−1) c | Serious (−1) d | None | 77 | 73 | - | SMD 0.63 lower (1.20 lower to −0.06 lower) † | ⨁VERY LOW |
| Function (assessed with functional tests) | ||||||||||
| 23 | Serious (−1) a | Not serious | Not serious | Not serious | None | 666 | 611 | - | SMD 0.01 lower (0.19 lower to 0.18 higher) | ⨁⨁⨁MODERATE |
| Inflammation | ||||||||||
| 25 | Serious (−1) a | Serious (−1) b | Not serious | Not serious | None | 581 | 573 | - | SMD 0.28 lower (0.51 lower to 0.06 lower) | ⨁⨁LOW |
| Tolerance | ||||||||||
| 28 | Serious (−1) a | Not serious | Not serious | Not serious | None | 951 | 899 | RR 1.00 (0.96 to 1.03) | 3 fewer per 1000 (from 27 fewer to 22 more) e | ⨁⨁⨁MODERATE |
| Number of withdrawals due to adverse events | ||||||||||
| 21 | Serious (−1) a | Not serious | Not serious | Not serious | None | 751 | 691 | RR 0.82 (0.57 to 1.17) | 16 fewer per 1000 (from 36 fewer to 15 more) e | ⨁⨁⨁MODERATE |
| Serious adverse events | ||||||||||
| 24 | Serious (−1) a | Not serious | Not serious | Serious (−1) d | None | 890 | 839 | RR 0.75 (0.43 to 1.30) | 8 fewer per 1000 (from 19 fewer to 10 more) e | ⨁⨁LOW |
© 2017 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Senftleber, N.K.; Nielsen, S.M.; Andersen, J.R.; Bliddal, H.; Tarp, S.; Lauritzen, L.; Furst, D.E.; Suarez-Almazor, M.E.; Lyddiatt, A.; Christensen, R. Marine Oil Supplements for Arthritis Pain: A Systematic Review and Meta-Analysis of Randomized Trials. Nutrients 2017, 9, 42. https://doi.org/10.3390/nu9010042
Senftleber NK, Nielsen SM, Andersen JR, Bliddal H, Tarp S, Lauritzen L, Furst DE, Suarez-Almazor ME, Lyddiatt A, Christensen R. Marine Oil Supplements for Arthritis Pain: A Systematic Review and Meta-Analysis of Randomized Trials. Nutrients. 2017; 9(1):42. https://doi.org/10.3390/nu9010042
Chicago/Turabian StyleSenftleber, Ninna K., Sabrina M. Nielsen, Jens R. Andersen, Henning Bliddal, Simon Tarp, Lotte Lauritzen, Daniel E. Furst, Maria E. Suarez-Almazor, Anne Lyddiatt, and Robin Christensen. 2017. "Marine Oil Supplements for Arthritis Pain: A Systematic Review and Meta-Analysis of Randomized Trials" Nutrients 9, no. 1: 42. https://doi.org/10.3390/nu9010042
APA StyleSenftleber, N. K., Nielsen, S. M., Andersen, J. R., Bliddal, H., Tarp, S., Lauritzen, L., Furst, D. E., Suarez-Almazor, M. E., Lyddiatt, A., & Christensen, R. (2017). Marine Oil Supplements for Arthritis Pain: A Systematic Review and Meta-Analysis of Randomized Trials. Nutrients, 9(1), 42. https://doi.org/10.3390/nu9010042





