Effects of Hazelnut Consumption on Blood Lipids and Body Weight: A Systematic Review and Bayesian Meta-Analysis
Abstract
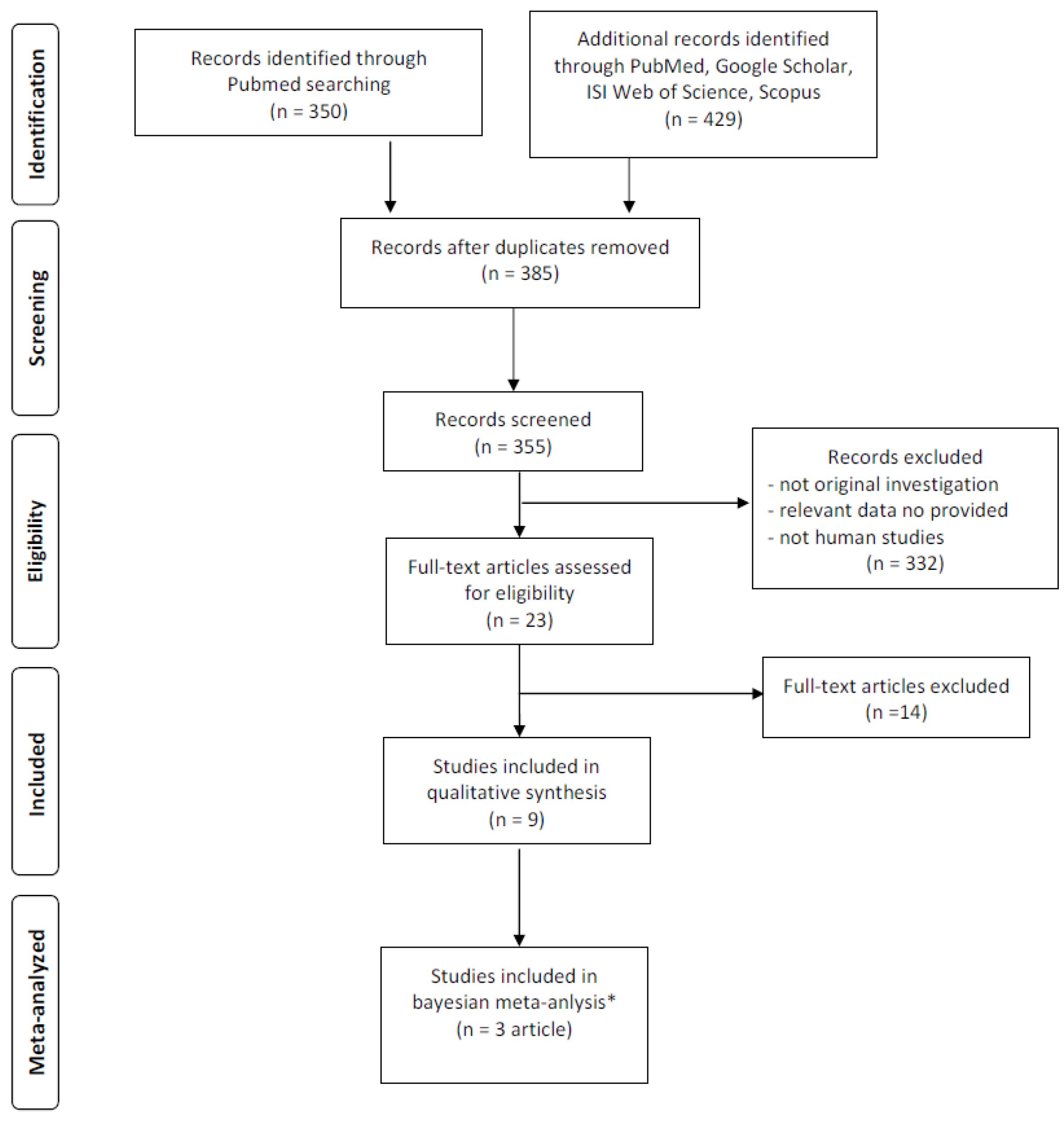
:1. Introduction
2. Experimental Section
2.1. Data Sources and Search Strategy
2.2. Inclusion and Exclusion Criteria
2.3. Data Collection
2.4. Risk of Bias in Individual Studies
2.5. Bayesian Meta-Analysis
3. Results
4. Risk of Bias
Bayesian Meta-Analysis
5. Discussion
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Appendix. Jags Code of the Statistical Model for Bayesian Meta-Analysis
Model 1: (τ2)−1 prior—Gamma(0.001, 0.001)
model{
## Likelihood
for (i in 1:k) {
# Variance of change in Treatment group (post-pre diff)
vdT[i] <- vpostT[i] + vpreT[i] - (2*rho*sqrt(vpostT[i]*vpreT[i]))
# Variance of change in Control group (post-pre diff)
vdC[i] <- vpostC[i] + vpreC[i] - (2*rho*sqrt(vpostC[i]*vpreC[i]))
# Variance of the difference between Treatment and Control (pooled variance)
diff.var[i] <- ((nT[i]-1)*vdT[i] + (nC[i]-1)*vdC[i])/( nT[i]+nC[i]-2)
w[i] <- 1/diff.var[i]
diff[i] ~ dnorm(theta[i],w[i])
theta[i] ~ dnorm(mu,prec)
}
# 4th study that have Variance of the difference between Treatment and Control
w4 <- 1/vd4
diff4 ~ dnorm(theta4,w4)
theta4 ~ dnorm(mu,prec)
## Priors
mu ~ dnorm(0.0,0.0001)
prec ~ dgamma(0.0001,0.0001)
tau2 <- 1/prec
rho ~ dunif(-1,1)
}
Model 2: τ prior ~ Uniform(0, 100)
model{
## Likelihood
for (i in 1:k) {
# Variance of change in Treatment group (post – pre diff)
vdT[i] <- vpostT[i] + vpreT[i] - (2*rho*sqrt(vpostT[i]*vpreT[i]))
# Variance of change in Control group (post – pre diff)
vdC[i] <- vpostC[i] + vpreC[i] - (2*rho*sqrt(vpostC[i]*vpreC[i]))
# Variance of the difference between Treatment and Control (pooled variance)
diff.var[i] <- ((nT[i]-1)*vdT[i] + (nC[i]-1)*vdC[i]) /(nT[i]+nC[i]-2)
w[i] <- 1/diff.var[i]
diff[i] ~ dnorm(theta[i],w[i])
theta[i] ~ dnorm(mu,prec)
}
# 4th study that have Variance of the difference between Treatment and Control
w4 <- 1/vd4
diff4 ~ dnorm(theta4,w4)
theta4 ~ dnorm(mu,prec)
## Priors
mu ~ dnorm(0.0,0.0001)
prec <- 1/(tau * tau)
rho ~ dunif(-1,1)
tau ~ dunif(0,2)
tau2 <- pow(tau,2)
}
Model 3: τ prior ~ Uniform(0, 2)
model{
## Likelihood
for (i in 1:k) {
# Variance of change in Treatment group (post-pre diff)
vdT[i] <- vpostT[i] + vpreT[i] - (2*rho*sqrt(vpostT[i]*vpreT[i]))
# Variance of change in Control group (post-pre diff)
vdC[i] <- vpostC[i] + vpreC[i] - (2*rho*sqrt(vpostC[i]*vpreC[i]))
# Variance of the difference between Treatment and Control (pooled variance)
diff.var[i] <- ((nT[i]-1)*vdT[i] + (nC[i]-1)*vdC[i]) /(nT[i] + nC[i]-2)
w[i] <- 1/diff.var[i]
diff[i] ~ dnorm(theta[i],w[i])
theta[i] ~ dnorm(mu,prec)
}
# 4th study that have Variance of the difference between Treatment and Control
w4 <- 1/vd4
diff4 ~ dnorm(theta4,w4)
theta4 ~ dnorm(mu,prec)
## Priors
mu ~ dnorm(0.0,0.0001)
prec <- 1/pow(tau,2)
rho ~ dunif(-1,1)
tau ~ dnorm(0,1)I(0,)
tau2 <- pow(tau,2)
References
- Kelly, J.H.; Sabaté, J. Nuts and coronary heart disease: An epidemiological perspective. Br. J. Nutr. 2006, 96, S61–S67. [Google Scholar] [CrossRef] [PubMed]
- Mukuddem-Petersen, J.; Oosthuizen, W.; Jerling, J.C. A systematic review of the effects of nuts on blood lipid profiles in humans. J. Nutr. 2005, 135, 2082–2089. [Google Scholar] [PubMed]
- Alphan, E.; Pala, M.; Açkurt, F.; Yilmaz, T. Nutritional composition of hazelnut and its effects on glucose and lipid metabolism. In Proceedings of the fourth international symposium on hazelnut. Acta Hortic. 1997, 445, 305–310. [Google Scholar] [CrossRef]
- Durak, I.; Köksal, I.; Kaçmaz, M.; Büyükkoçak, S.; Cimen, B.M.; Oztürk, H.S. Hazelnut supplementation enhances plasma antioxidant potential and lowers plasma cholesterol levels. Clin. Chim. Acta 1999, 284, 113–115. [Google Scholar] [CrossRef]
- Mercanligil, S.M.; Arslan, P.; Alasalvar, C.; Okut, E.; Akgül, E.; Pinar, A.; Geyik, P.O.; Tokgözoğlu, L.; Shahidi, F. Effects of hazelnut-enriched diet on plasma cholesterol and lipoprotein profiles in hypercholesterolemic adult men. Eur. J. Clin. Nutr. 2007, 61, 212–220. [Google Scholar] [CrossRef] [PubMed]
- Tey, S.L.; Brown, R.C.; Chisholm, A.W.; Delahunty, C.M.; Gray, A.R.; Williams, S.M. Effects of different forms of hazelnuts on blood lipids and α-tocopherol concentrations in mildly hypercholesterolemic individuals. Eur. J. Clin. Nutr. 2011, 65, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Tey, S.L.; Brown, R.; Gray, R.; Chisholm, A.; Delahunty, C. Nuts improve diet quality compared to other energy-dense snacks while maintaining bodyweight. J. Nutr. Metab. 2011, 2011, 350–357. [Google Scholar] [CrossRef] [PubMed]
- Tey, S.L.; Gray, A.R.; Chisholm, A.W.; Delahunty, C.M.; Brown, R.C. The dose of hazelnuts influences acceptance and diet quality but not inflammatory markers and body composition in overweight and obese individuals. J. Nutr. 2013, 143, 1254–1262. [Google Scholar] [CrossRef] [PubMed]
- Tey, S.L.; Robinson, T.; Davis, H.; Gray, A.R.; Chisholm, A.W.; Brown, R.C. The effect of regular hazelnut consumption on cardiovascular risk factors and acceptance in Māori and European. J. Nutr. 2015, 2, 39–53. [Google Scholar]
- Yücesan, F.B.; Orem, A.; Kural, B.V.; Orem, C.; Turan, I. Hazelnut consumption decreases the susceptibility of LDL to oxidation, plasma oxidized LDL level and increases the ratio of large/small LDL in normolipidemic healthy subjects. Anadolu. Kardiyol. Derg. 2010, 10, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Alasalvar, C.; Shahidi, F.; Ohshima, T.; Wanasundara, U.; Yurttas, H.C.; Liyanapathirana, C.M.; Rodrigues, F.B. Turkish Tombul hazelnut (Corylus avellana L.). 2. Lipid characteristics and oxidative stability. J. Agric. Food. Chem. 2003, 51, 3797–3805. [Google Scholar] [CrossRef] [PubMed]
- Alasalvar, C.; Shahidi, F.; Liyanapathirana, C.M.; Ohshima, T. Turkish Tombul hazelnut (Corylus avellana L.). 1. Compositional characteristics. J. Agric. Food. Chem. 2003, 51, 3790–3796. [Google Scholar] [CrossRef] [PubMed]
- Steinberg, D.; Lewis, A. Oxidative modification of LDL and atherogenesis. Circulation 1997, 95, 1062–1071. [Google Scholar] [CrossRef] [PubMed]
- Yurttas, H.C.; Schafer, H.W.; Warthesen, J.J. Antioxidant activity of nontocopherol hazelnut (Corylus spp.) phenolics. J. Food. Sci. 2000, 65, 276–280. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. Ann. Intern. Med. 2009, 151, 264–269. [Google Scholar] [CrossRef] [PubMed]
- National Library of Medicine, Bethesda, MD, USA. Available online: http://www.pubmed.com (accessed on 2 August 2016).
- Scopus. Available online: http://www.scopus.com (accessed on 2 August 2016).
- Thomson Reuters. Available online: http://www.thomsonreuters.com (accessed on 2 August 2016).
- Google Scholar. Available online: http://www.scholar.google.com (accessed on 2 August 2016).
- Higgins, J.P.; Altman, D.G.; Gøtzsche, P.C.; Jüni, P.; Moher, D.; Oxman, A.D.; Savovic, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A.; et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef] [PubMed]
- Mila, A.L.; Ngugi, H.K. A Bayesian approach to meta-analysis of plant pathology studies. Phytopathology 2011, 101, 42–51. [Google Scholar] [CrossRef] [PubMed]
- Sutton, A.J.; Abrams, K.R. Bayesian methods in meta-analysis and evidence synthesis. Stat. Methods Med. Res. 2001, 10, 277–303. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Thompson, S.G.; Spiegelhalter, D.J. A re-evaluation of random-effects meta-analysis. J. R. Stat. Soc. Ser. A Stat. Soc. 2009, 172, 137–159. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.C.; Sabavala, D.J. Bayesian Estimation and Prediction for the Beta-Binomial Model. J. Bus. Econ. Stat. 1987, 5, 357–367. [Google Scholar]
- Lee, J.C.; Lio, Y.L. A note on Bayesian estimation and prediction for the beta-binomial model. J. Stat. Comput. Simul. 1997, 63, 73–91. [Google Scholar] [CrossRef]
- Abrams, K.R.; Gillies, C.L.; Lambert, P.C. Meta-analysis of heterogeneously reported trials assessing change from baseline. Stat. Med. 2005, 24, 3823–3844. [Google Scholar] [CrossRef] [PubMed]
- Smith, B.J. boa: An R Package for MCMC Output Convergence Assessment and Posterior Inference. J. Stat. Softw. 2007, 21, 1–37. [Google Scholar] [CrossRef]
- Liu, G.C.; Sui, G.Y.; Liu, G.Y.; Zheng, Y.; Deng, Y.; Gao, Y.-Y.; Wang, L. A Bayesian meta-analysis on prevalence of hepatitis B virus infection among Chinese volunteer blood donors. PLoS ONE 2013, 8, e79203. [Google Scholar] [CrossRef] [PubMed]
- Lambert, P.C.; Sutton, A.J.; Burton, P.R.; Abrams, K.R.; Jones, D.R. How vague is vague? A simulation study of the impact of the use of vague prior distributions in MCMC using WinBUGS. Stat. Med. 2005, 24, 2401–2428. [Google Scholar] [CrossRef] [PubMed]
- Plummer, M. JAGS: A program for analysis of Bayesian graphical models using Gibbs sampling. In Proceedings of the 3rd International Workshop on Distributed Statistical Computing, Vienna, Austria, 20–22 March 2003.
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria; Available online: http://www.R-project.org/ (accessed on 3 September 2013).
- Su, Y.S.; Yajima, M. R2jags: Using R to Run “JAGS”. R Package Version 0.5–7. Available online: http://CRAN.R-project.org/package=R2jags (accessed on 2 December 2015).
- Plummer, M.; Best, N.; Cowles, K.; Vines, K. CODA: Convergence Diagnosis and Output Analysis for MCMC. R. News 2006, 6, 7–11. [Google Scholar]
- Centre for Evidence-Based Medicine. Levels of Evidence. New Levels of Evidence Table. Available online: http://www.cebm.net/index.aspx?o=5653 (accessed on 2 August 2016).
- Damavandi, R.D.; Eghtesadi, S.; Shidfar, F.; Heydari, I.; Foroushani, A.R. Effects of hazelnuts consumption on fasting blood sugar and lipoproteins in patients with type 2 diabetes. J. Res. Med. Sci. 2013, 18, 314–321. [Google Scholar] [PubMed]
- Orem, A.; Yücesan, F.B.; Orem, C.; Akcan, B.; Kural, B.V.; Alasalvar, C.; Shahidi, F. Hazelnut-enriched diet improves cardiovascular risk biomarkers beyond a lipid-lowering effect in hypercholesterolemic subjects. J. Clin. Lipidol. 2013, 7, 123–131. [Google Scholar] [CrossRef] [PubMed]
- US Department of Agriculture, Agricultural Research Service. USDA Nutrient Database for Standard Reference, Release 17. Available online: http://www.nal.usda.gov./fnic/foodcomp (accessed on 2 August 2016).
- Gebhardt, S.E.; Lemar, L.E.; Haytowitz, D.B.; Pehrsson, PR.; Lemar, L.E.; Holcomb, G.T.; Stup, M.A.; Thomas, R.G.; Exler, J.; Showell, B.A.; et al. USDA National Nutrient Database for Standard Reference, Release 21; United States Department of AgricultureAgricultural Research Service: Beltsville, MD, USA.
- US Department of Agriculture, Agricultural Research Service. USDA Nutrient Database for Standard Reference Release 28. Available online: http://www.nal.usda.gov./fnic/foodcomp (accessed on 2 August 2016).
- Piscopo, S. The Mediterranean diet as a nutrition education, health promotion and disease prevention tool. Public. Health. Nutr. 2009, 12, 1648–1655. [Google Scholar] [CrossRef] [PubMed]
- Mensink, R.P.; Katan, M.B. Effect of dietary fatty acids on serum lipids and lipoproteins. A meta-analysis of 27 trials. Arterioscler. Thromb. 1992, 12, 911–919. [Google Scholar] [CrossRef] [PubMed]
- Hegsted, D.M.; Ausman, L.M.; Johnson, J.A.; Dallal, G.E. Dietary fat and serum lipids: An evaluation of the experimental data. Am. J. Clin. Nutr. 1993, 57, 875–883. [Google Scholar] [PubMed]
- Brown, L.; Rosner, B.; Willett, W.W.; Sacks, F.M. Cholesterol-lowering effects of dietary fiber: A meta-analysis. Am. J. Clin. Nutr. 1999, 69, 30–42. [Google Scholar] [PubMed]
- Caimari, A.; Puiggròs, F.; Suárez, M.; Crescenti, A.; Laos, S.; Ruiz, J.A.; Alonso, V.; Moragas, J.; Del Bas, J.M.; Arola, L. The intake of a hazelnut skin extract improves the plasma lipid profile and reduces the lithocholic/deoxycholic bile acid faecal ratio, a risk factor for colon cancer, in hamsters fed a high-fat diet. Food. Chem. 2015, 15, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Mukuddem-Petersen, J.; Oosthuizen, W.; Jerling, J.C. A Systematic Review of the Effects of Nuts on Blood Lipid Profiles in Humans. J. Nutr. 2005, 135, 2082–2089. [Google Scholar] [PubMed]
- Banel, D.K.; Hu, F.B. Effects of walnut consumption on blood lipids and other cardiovascular risk factors: A meta-analysis and systematic review. Am. J. Clin. Nutr. 2009, 90, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Greco, T.; Landoni, G.; Biondi-Zoccai, G.; D’Ascenzo, F.; Zangrillo, A. A Bayesian Network meta-analysis for binary outcome: How to do it. Stat. Methods Med. Res. 2013, 25, 1757–1773. [Google Scholar] [CrossRef] [PubMed]
- Sabaté, J.; Cordero-MacIntyre, Z.; Siapco, G.; Torabian, S.; Haddad, E. Does regular walnut consumption lead to weight gain? Br. J. Nutr. 2005, 94, 859–864. [Google Scholar] [CrossRef] [PubMed]
| First Author and Year (Reference) | Sample Size | Age (Years) (Mean ± SD) | BMI (kg/m2) or Weight (kg) | Sample Gender Distribution | Baseline Lipid Values * (Mean ± SD or 95%CI) | Study Design (Level of Evidence) | Trial Duration (Days) | Hazelnut Diet | Control Diet |
|---|---|---|---|---|---|---|---|---|---|
| Tey et al., 2015 [9] | 39 | 21.4 ± 2.7 | BMI: 25.00 ± 1.10 Weight: 73.9 ± 1.2 | Maori: 20 males: 8 females: 12 European: 19 males: 5 females: 14 | Maori group: TC: 4.14 (3.77–4.54) HDL-C: 1.16 (1.06–1.26) LDL-C: 2.46 (2.17–2.79) TG: 1.01 (0.84–1.22) Apo A: 1.51 (1.41–1.61) Apo B: 0.71 (0.63–0.80) European group: TC: 3.96 (3.63–4.31) HDL-C: 1.16 (1.01–1.32) LDL-C: 2.28 (2.01–2.59) TG: 0.96 (0.81–1.13) Apo A: 1.51 (1.39–1.66) Apo B: 0.65 (0.57–0.74) | Randomized controlled trials (level 2) | 28 | 30 g/day | - |
| Tey et al., 2011 [6] | 46 | 49.9 ± 9.4 | BMI: 25.90 ± 3.50 Weight: 74.4 ± 13.1 | males: 19 females: 27 | TC: 5.88 ± 0.67 HDL-C: 1.21 ± 0.35 LDL-C: 4.01 ± 0.67 TG: 1.43 ± 0.63 Apo A: 1.78 ± 0.28 Apo B: 1.05 ± 0.21 | Randomized cross-over (level 2) | 84 | 30 g/day | - |
| Durak et al., 1999 [4] | 30 | 18.5 ± NR | Weight: 68.7 ± 9.2 | males: 18 females: 12 | TC: 3.40 ± 0.51 HDL-C: 1.03 ± 0.26 LDL-C: 1.96 ± 0.51 TG: 0.87 ± 0.29 | No randomized clinical trial (level 3) | 30 | 69 g/day | - |
| Damavandi et al., 2013 [35] | 48 | 55.68 ± 7.74 | Hazelnut group BMI: 28.47 ± 3.57 Weight: 72.13 ± 10.27 Control group BMI: 28.18 ± 3.55 Weight: 71.98 ± 9.58 | Hazelnut group: 23 males: 7 females: 16 Control group: 25 males: 8 females: 17 | Hazelnut group: TC: 4.12 ± 0.95 HDL-C: 1.14 ± 0.21 LDL-C: 2.18 ± 0.75 TG: 1.75 ± 0.96 Control group: TC: 3.62 ± 0.76 HDL-C: 1.04 ± 0.20 LDL-C: 1.94 ± 0.72 TG: 1.41 ± 0.65 | Randomized parallel study (level 2) | 56 | 29 g/day | sel- selected diet |
| Orem et al., 2013 [36] | 21 | 44.6 ± 10.4 | BMI: 27.40 ± 3.11 Weight: 81.0 ± 14.1 | males: 18 females: 3 | TC: 5.77 ± 0.58 HDL-C: 1.12 ± 0.17 LDL-C: 4.00 ± 0.68 TG: 1.65 (1.12–2.18) Apo A: 1.31 ± 0.00 Apo B: 1.15 ± 0.00 | Double control sandwich model (level 3) | 56 | 49–86 g/day | - |
| Mercanligil et al., 2007 [5] | 15 | 48 ± 8 | BMI: 25.90 ± 1.7 Weight: 74.2 ± 5.1 | males: 15 females: 0 | TC: 5.86 ± 0.61 HDL-C: 1.13 ± 0.29 LDL-C: 3.80 ± 0.53 TG: 2.02 ± 1.51 Apo A: 1.32 ± 0.15 Apo B: 1.28 ± 0.22 | Single group study (level 3) | 56 | 40 g/day | - |
| Yücesan et al., 2010 [10] | 21 | 28 ± 5 | BMI: 23.89 ± 3.52 Weight: 64.52 ± 11.5 | males: 8 females: 13 | TC: 4.21 ± 0.59 HDL-C: 1.38 ± 0.32 LDL-C: 2.81 ± 0.66 TG: 1.01 ± 0.67 Apo A: 1.35 ± 0.18 Apo B: 0.78 ± 0.20 | Single group study (level 3) | 30 | 49–86 g/day | - |
| Tey et al., 2013 [8] | 107 | 42.5 ± 12.4 | Hazelnut group 30 g BMI: 30.70 ± 4.70 Weight: 86.2 ± 11.8 Hazelnut group 60 g: BMI: 30.90 ± 6.0 Weight: 92.0 ± 19.6 Control group: BMI: 30.40 ± 4.50 Weight: 88.7 ± 16.7 | Hazelnut group 30 g: males: 13 females: 20 Hazelnut group 60 g: males: 17 females: 20 Control group: males: 16 females: 21 | Hazelnut group 30 g: TC: 4.92 ± 1.00 HDL-C: 1.26 ± 0.25 LDL-C: 3.07 ± 0.86 TG: 1.29 ± 0.44 Apo A: 1.60 ± 0.03 Apo B: 0.88 ± 0.04 Hazelnut group 60 g: TC: 4.93 ± 1.01 HDL-C: 1.20 ± 0.32 LDL-C: 3.05 ± 0.96 TG: 1.49 ± 0.70 Apo A: 1.56 ± 0.05 Apo B: 0.89 ± 0.04 Control group: TC: 4.93 ± 1.00 HDL-C: 1.32 ± 0.33 LDL-C: 3.03 ± 0.86 TG: 1.27 ± 0.40 Apo A: 1.67 ± 0.04 Apo B: 0.87 ± 0.04 | Randomized controlled parallel (level 2) | 84 | 30 or 60 g/day | no nuts |
| Tey et al., 2011 [7] | 61 | 37.5 ± 14 | Hazelnut group: BMI: 24.60 ± 2.80 Weight: 72.0 ± 11.1 Control group: BMI: 22.90 ± 2.80 Weight: 67.3 ± 9.5 | Hazelnut group: 32 males: 15 females: 17 Control group: 29 males: 12 females: 17 | Hazelnut and control group: TC: 4.79 ± 0.95 HDL-C: 1.32 ± 1.30 LDL-C: 2.94 ± 0.84 TG: 0.98 ± 1.48 | Randomized controlled parallel (level 2) | 84 | 42 g/day | no additional food |
| First Authors, Year (Reference) | Inclusion Criteria | Exclusion Criteria |
|---|---|---|
| Tey et al., 2015 [9] | Free-living Māori and European males and females, aged above 18 years living in Dunedin and surrounding areas. | Allergies or intolerances to nuts, medication known to affect blood lipid levels, familial or secondary hyperlipidemia, condition known to affect blood cholesterol levels, smoker, pregnant or lactating. |
| Tey et al., 2011 [6] | Healthy males or females aged between 18 and 65 years and: TC > 44.8 mmol/L and <8.0 mmol/L | Asthma, food allergies, familial hyperlipidemia, a chronic disease, or cholesterol-lowering medication or medication known to affect blood lipid concentrations. |
| Durak et al., 1999 [4] | healthy medical students aged 18 to 19 years | None |
| Damavandi et al., 2013 [36] | Previously diagnosed with type 2 diabetes based on FBS >126 mg/dL or 2-h blood sugar ≥200 mg/dL, serum TGs <400 mg/dL, body mass index (BMI) ≤35 kg/m2, Hemoglobin-A1C (HbA1C) <9%, serum LDL-C <200 mg/dL, and blood pressure ≤160/90 mmHg | Any known allergies to nuts, insulin therapy, cigarette smoking, history of stroke, heart disease or thyroid disorders, diabetic nephropathy or retinopathy, or following vegetarian or weight-loss diets up to 2 months before the study. Those patients who had consumed nuts more than 2 times/week and changed their medications (type or dosage) up to 2 months before the study were also excluded. All subjects were taking hypoglycemic agents. |
| Orem et al., 2013 [35] | Serum cholesterol level greater than 200 mg/dL with or without triacylglycerol greater than 150 mg/dL | On a medication or supplementation known to alter lipid metabolism |
| Mercanligil et al., 2007 [5] | Hypercholesterolemic (>200 mg/dL) adult males aged 33–59 years were recruited voluntarily from the staff of the Hacettepe University | Patients with TAG levels above 300 mg/dL were excluded from the study. All subjects were required not to be obese, be non-smokers and non-alcoholics, free of dietary restrictions/food allergies and not taking medications known to alter plasma lipids |
| Yücesan et al., 2010 [10] | Normolipidemic healthy subject among highly educated university staff members. | Systemic illness (diabetes mellitus, liver or kidney disease, or hypertension) or history of allergy to hazelnut. Also, individuals who consume alcohol or smoke were excluded. One of the exclusion criteria was the consumption of nuts, nut butters, or nuts oil more than once per week |
| Tey et al., 2013 [8] | Aged between 18 and 65 year inclusive, with a BMI 25 kg/m2 | Asthma, allergies, or aversion to nuts; familial hyperlipidemia; major chronic disease; or inflammatory diseases such as Crohn’s or celiac disease. Current smokers, pregnant or breastfeeding women, and people who were participating in weight-loss programs or taking medications known to affect inflammatory markers were also excluded |
| Tey et al., 2011 [7] | Healthy males or females aged between 18 and 65 years | People with BMI ≥ 30 kg/m2, people who have asthma, women who are pregnant or breastfeeding, people with a chronic disease such as cancer, heart disease, or diabetes, and people with food allergies or food aversions |
| First Author, Year (Reference) | BMI (kg/m2) Mean Change (p-Value) | Body Weight (kg) Mean Change (p-Value) | TC (mmol/L) Mean Change (p-Value) | HDL-C (mmol/L) Mean Change (p-Value) | LDL-C (mmol/L) Mean Change (p-Value) | TG (mmol/L) Mean Change (p-Value) | APO-A (g/L) Mean Change (p-Value) | APO-B (g/L) Mean Change (p-Value) |
|---|---|---|---|---|---|---|---|---|
| Tey et al., 2015 [9] | 0.00 (p = 0.938) | 0.15 (p = 0.456) | 0.00 (p = 0.983) | 0.025 (p = 0.128) | −0.035 (p = 0.556) | 0.005 (p = 0.834) | 0.035 (p = 0.114) | −0.015 (p = 0.371) |
| Tey et al., 2011 [6] | 0.01 (p = 0.822) | 0.04 (p = 0.813) | −0.19 (p < 0.001) | 0.03 (p = 0.023) | −0.22 (p < 0.001) | −0.01 (p = 0.725) | 0.01 (p = 0.749) | −0.04 (p = 0.002) |
| Durak et al., 1999 [4] | NR | 0.50 (p > 0.05) | −0.20 (p < 0.005) | 0.09 (p < 0.05) | −0.36 (p < 0.0005) | 0.21 (p < 0.001) | NR | NR |
| Damavandi et al., 2013 [36] | −0.55 (p > 0.05) | −0.66 (p > 0.05) | −0.12 (p > 0.05) | −0.06 (p > 0.05) | 0.03 (p > 0.05) | −0.30 (p > 0.05) | NR | NR |
| Orem et al, 2013 [35] | −0.50 (p < 0.05) | −1.90 (p < 0.05) | −0.47 (p < 0.05) | 0.07 (p < 0.05) | −0.25 (p < 0.05) | −0.27 (p < 0.05) | 0.15 (p < 0.05) | −0.03 (p > 0.05) |
| Mercanligil et al., 2007 [5] | −0.10 (p > 0.05) | −0.20 (p > 0.05) | 0.03 (p = 0.064) | 0.15 (p = 0.001) | 0.10 (p = 0.448) | −0.45 (p = 0.006) | 0.04 (p = 0.557) | −0.07 (p = 0.01) |
| Yücesan et al., 2010 [10] | 0.01 (p = 0.166) | 0.21 (p = 0.166) | −0.36 (p < 0.001) | 0.06 (p = 0.086) | −0.21 (p = 0.008) | −0.13 (p = 0.131) | 0.06 (p = 0.005) | −0.07 (p = 0.005) |
| Tey et al., 2013 (30 g/day) ^ [8] | 0.00 (p > 0.05) | 0.00 (p > 0.05) | −0.14 (p > 0.05) | 0.04 (p > 0.05) | −0.14 (p > 0.05) | −0.1 (p > 0.05) | 0.03 (p > 0.05) | −0.03 (p > 0.05) |
| Tey et al., 2013 (60 g/day) ^ [8] | 0.00 (p > 0.05) | 0.2 (p > 0.05) | −0.13 (p > 0.05) | 0.00 (p > 0.05) | −0.09 (p > 0.05) | −0.08 (p > 0.05) | 0.01 (p > 0.05) | −0.02 (p > 0.05) |
| Tey et al., 2011 [7] | 0.28 (p = 0.001) | 0.83 (p = 0.001) | −0.06 (p = 0.398) | 1.02 (p = 0.325) | −0.09 (p = 0.144) | 0.99 (p = 0.349) | NR | NR |
| First Author, Year (Reference) | Sequence Generation | Allocation Concealment | Blinding | Blinding of Outcome Assessment | Incomplete Outcome Data | Selective Reporting | Overall Quality |
|---|---|---|---|---|---|---|---|
| Tey et al., 2015 [9] | + | + | - | - | + | + | Good |
| Tey et al., 2011 [6] | + | + | - | - | + | + | Good |
| Durak et al., 1999 [4] | - | - | - | - | + | + | Poor |
| Damavandi et al., 2013 [36] | - | + | - | - | + | + | Fair |
| Orem et al., 2013 [35] | - | + | - | - | + | + | Fair |
| Mercanligil et al., 2007 [5] | - | + | - | - | + | + | Fair |
| Yücesan et al., 2010 [10] | - | + | - | - | + | - | Fair |
| Tey et al., 2013 [8] | + | + | - | - | + | + | Good |
| Tey et al, 2011 [7] | + | + | - | - | + | + | Good |
| First Author, Year (Reference) | Total Cholesterol (mmol/L) | HDL-C (mmol/L) | LDL-C (mmol/L) | Triglycerides (mmol/L) | BMI (kg/m2) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Hazelnut Diet | Control Diet | MD Δ-changes (SD) | Hazelnut Diet | Control Diet | MD Δ-changes (SD) | Hazelnut Diet | Control Diet | MD Δ-changes (SD) | Hazelnut Diet | Control Diet | MD Δ-changes (SD) | Hazelnut Diet | Control Diet | MD Δ-changes (SD) | |
| t0 (SD) | t0 (SD) | t0 (SD) | t0 (SD) | t0 (SD) | t0 (SD) | t0 (SD) | t0 (SD) | t0 (SD) | t0 (SD) | ||||||
| t1 (SD) | t1 (SD) | t1 (SD) | t1 (SD) | t1 (SD) | t1 (SD) | t1 (SD) | t1 (SD) | t1 (SD) | t1 (SD) | ||||||
| Δ-changes (SD) | Δ-changes (SD) | Δ-changes (SD) | Δ-changes (SD) | Δ-changes (SD) | Δ-changes (SD) | Δ-changes (SD) | Δ-changes (SD) | Δ-changes (SD) | Δ-changes (SD) | ||||||
| Damavandi et al., 2013 [36] | 4.12 (0.95) | 3.62 (0.76) | NR | 1.14 (0.21) | 1.04 (0.20) | NR | 2.18 (0.75) | 1.94 (0.72) | NR | 1.75 (0.96) | 1.41 (0.65) | NR | 28.47 (3.57) | 28.18 (3.55) | NR |
| 4.00 (0.95) | 3.50 (0.81) | 1.08 (0.17) | 0.95 (0.19) | 2.21 (0.72) | 1.90 (0.75) | 1.45 (0.80) | 1.40 (0.95) | 27.92 (3.57) | 28.05 (3.64) | ||||||
| −0.12 (NR) | −0.12 (NR) | −0.06 (NR) | −0.09 (NR) | 0.03 (NR) | −0.04 (NR) | −0.30 (NR) | −0.01 (NR) | −0.55 (NR) | −0.13 (NR) | ||||||
| Tey et al., 2011 [7] | 4.79 (0.95) | 4.79 (0.95) | NR | 1.32 (1.30) | 1.32 (1.30) | NR | 2.94 (0.84) | 2.94 (0.84) | NR | 0.98 (1.48) | 0.98 (1.48) | NR | 23.76 (2.99) | 23.76 (2.99) | NR |
| 4.73 (NR) | 4.89 (NR) | 2.34 (NR) | 2.32 (NR) | 2.85 (NR) | 3.03 (NR) | 1.97 (NR) | 2.01 (NR) | 24.04 (NA) | 23.90 (NA) | ||||||
| −0.06 (0.07) | 0.10 (0.07) | 1.02 (1.02) | 1.00 (1.02) | −0.09 (0.06) | 0.09 (0.07) | 0.99 (1.04) | 1.03 (1.05) | 0.28 (0.08) | 0.14 (0.18) | ||||||
| Tey et al., 2013 (30 g/day) [8] | 4.92 (0.17) | 4.93 (0.17) | NR | 1.26 (0.04) | 1.32 (0.05) | NR | 3.07 (0.15) | 3.03 (0.14) | NR | 1.29 (0.08) | 1.27 (0.07) | NR | 30.70 (0.81) | 30.40 (0.74) | NR |
| 4.78 (0.16) | 4.91 (0.16) | 1.30 (0.04) | 1.34 (0.06) | 2.93 (0.15) | 3.05 (0.15) | 1.19 (0.07) | 1.13 (0.05) | 30.70 (0.81) | 30.40 (0.78) | ||||||
| −0.14 (NR) | −0.02 (NR) | 0.04 (NR) | 0.02 (NR) | −0.14 (NR) | 0.02 (NR) | −0.10 (NR) | −0.14 (NR) | 0 (NA) | 0 (NR) | ||||||
| Tey et al., 2013 (60 g/day) [8] | 4.93 (0.17) | 4.93 (0.17) | NR | 1.20 (0.05) | 1.32 (0.05) | NR | 3.05 (0.16) | 3.03 (0.14) | NR | 1.49 (0.12) | 1.27 (0.07) | NR | 30.90 (0.99) | 30.40 (0.74) | NR |
| 4.80 (0.16) | 4.91 (0.16) | 1.20 (0.05) | 1.34 (0.06) | 2.96 (0.15) | 3.05 (0.15) | 1.41 (0.09) | 1.13 (0.05) | 30.90 (0.97) | 30.40 (0.78) | ||||||
| −0.13 (NR) | −0.02 (NR) | 0 (NR) | 0.02 (NR) | −0.09 (NR) | 0.02 (NR) | −0.08 (NR) | −0.14 (NR) | 0 (NR) | 0 (NR) | ||||||
| Outcome | Parameters | Model 1 * | Model 2 ** | Model 3 *** | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Median | SD | 95%HPD | MC Error | Median | SD | 95%HPD | MC Error | Median | SD | 95%HPD | MC Error | ||
| Total cholesterol | μ | −0.127 | 0.084 | −0.284; 0.014 | 0.000 | −0.127 | 0.173 | −0.474; 0.215 | 0.001 | −0.126 | 0.147 | −0.418; 0.159 | 0.001 |
| τ2 | 0.001 | 0.270 | 0.000; 0.040 | 0.001 | 0.007 | 0.310 | 0.000; 0.447 | 0.004 | 0.006 | 0.204 | 0.000; 0.264 | 0.003 | |
| ρ | 0.863 | 0.452 | −0.429; 0.999 | 0.003 | 0.756 | 0.522 | −0.646; 0.999 | 0.004 | 0.778 | 0.512 | −0.611; 0.999 | 0.005 | |
| DIC | 3.1 | 5.9 | 5.5 | ||||||||||
| HDL-C | μ | 0.002 | 0.082 | −0.140; 0.147 | 0.000 | 0.003 | 0.171 | −0.341; 0.361 | 0.001 | 0.005 | 0.150 | −0.297; 0.321 | 0.001 |
| τ 2 | 0.001 | 0.214 | 0.000; 0.044 | 0.001 | 0.007 | 0.303 | 0.000; 0.460 | 0.006 | 0.008 | 0.213 | 0.000; 0.309 | 0.006 | |
| ρ | 0.493 | 0.564 | −0.776; 0.999 | 0.003 | 0.351 | 0.580 | −0.831; 0.999 | 0.004 | 0.352 | 0.580 | −0.829; 0.999 | 0.004 | |
| DIC | 0.1 | 1.4 | 1.2 | ||||||||||
| LDL-C | μ | −0.150 | 0.088 | −0.308; −0.003 | 0.000 | −0.149 | 0.179 | −0.478; 0.210 | 0.001 | −0.150 | 0.155 | −0.437; 0.165 | 0.001 |
| τ2 | 0.001 | 0.347 | 0.000; 0.045 | 0.001 | 0.009 | 0.319 | 0.000; 0.442 | 0.004 | 0.008 | 0.228 | 0.000; 0.292 | 0.004 | |
| ρ | 0.747 | 0.490 | −0.574; 0.999 | 0.003 | 0.646 | 0.537 | −0.705; 0.999 | 0.004 | 0.648 | 0.531 | −0.689 0.999; | 0.004 | |
| DIC | 0.1 | 2.5 | 2.2 | ||||||||||
| Triglycerides | μ | 0.045 | 0.141 | −0.195; 0.269 | 0.001 | 0.040 | 0.293 | −0.590; 0.627 | 0.001 | 0.041 | 0.243 | −0.510; 0.495 | 0.001 |
| τ2 | 0.002 | 0.968 | 0.000; 0.118 | 0.004 | 0.029 | 0.537 | 0.000; 1.201 | 0.006 | 0.023 | 0.396 | 0.000; 0.708 | 0.003 | |
| ρ | 0.508 | 0.560 | −0.767; 0.999 | 0.003 | 0.375 | 0.577 | −0.821; 0.999 | 0.004 | 0.391 | 0.577 | −0.816; 0.999 | 0.004 | |
| DIC | 3.2 | 4.7 | 4.5 | ||||||||||
| BMI | μ | 0.062 | 0.245 | −0.293; 0.469 | 0.001 | 0.062 | 0.419 | −0.867; 0.979 | 0.002 | 0.062 | 0.347 | −0.665; 0.803 | 0.002 |
| τ2 | 0.003 | 2.594 | 0.000; 0.286 | 0.007 | 0.078 | 0.716 | 0.000; 2.072 | 0.006 | 0.051 | 0.543 | 0.000; 1.165 | 0.006 | |
| ρ | 0.936 | 0.381 | −0.156; 0.999 | 0.003 | 0.862 | 0.470 | −0.471; 0.999 | 0.004 | 0.884 | 0.447 | −0.405; 0.999 | 0.004 | |
| DIC | 12.1 | 15.7 | 14.8 | ||||||||||
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perna, S.; Giacosa, A.; Bonitta, G.; Bologna, C.; Isu, A.; Guido, D.; Rondanelli, M. Effects of Hazelnut Consumption on Blood Lipids and Body Weight: A Systematic Review and Bayesian Meta-Analysis. Nutrients 2016, 8, 747. https://doi.org/10.3390/nu8120747
Perna S, Giacosa A, Bonitta G, Bologna C, Isu A, Guido D, Rondanelli M. Effects of Hazelnut Consumption on Blood Lipids and Body Weight: A Systematic Review and Bayesian Meta-Analysis. Nutrients. 2016; 8(12):747. https://doi.org/10.3390/nu8120747
Chicago/Turabian StylePerna, Simone, Attilio Giacosa, Gianluca Bonitta, Chiara Bologna, Antonio Isu, Davide Guido, and Mariangela Rondanelli. 2016. "Effects of Hazelnut Consumption on Blood Lipids and Body Weight: A Systematic Review and Bayesian Meta-Analysis" Nutrients 8, no. 12: 747. https://doi.org/10.3390/nu8120747
APA StylePerna, S., Giacosa, A., Bonitta, G., Bologna, C., Isu, A., Guido, D., & Rondanelli, M. (2016). Effects of Hazelnut Consumption on Blood Lipids and Body Weight: A Systematic Review and Bayesian Meta-Analysis. Nutrients, 8(12), 747. https://doi.org/10.3390/nu8120747





