1. Introduction
The postmenopausal state is associated with an increased risk of metabolic diseases such as obesity, heart disease, diabetes, and hypertension [
1]. These metabolic diseases might be caused directly by estrogen deficiency and may occur partly as secondary effects of obesity due to the orexigenic effects of estrogen deficiency [
2]. Estrogen deficiency is considered a common risk factor for developing osteoporosis.
Relief from menopausal symptoms using phytosubstances from plants as an alternative to estrogen/progesterone therapy has gained much attention. Several phytoestrogens (PEs), plant-derived chemicals with antioxidative effects in vitro, act as free radical scavengers. Isoflavones, derived from soy and soy derivatives, are the most common PEs and genistein and daidzein are the most abundant and well-studied. This class of PEs can also be found in clover.
Red clover (RC,
Trifolium pratense L.) botanical dietary supplements have received significant attention for their potential safe use in the treatment of menopausal symptoms, maintenance/improvement of cardiovascular health, and for their reported benign effects on the breast, endometrium, and neural structures [
3]. The estrogenic activity of RC is mainly due to isoflavones and, to a lesser extent, coumestans [
4]. The isoflavones formononetin, biochanin A, genistein, and daidzein are present in RC as glycosides and malonates [
5]. Isoflavones and PEs mimic estrogen [
6], and their estrogenic effects in biological systems are believed to be related to their structural similarity to estrogen.
Pomegranates (
Punica granatum L.) are commonly consumed as fresh fruit, beverages, other food products, dietary supplements, and herbal medicine ingredients [
7]. The pomegranate is one of the richest sources of flavonoid antioxidants. The main active substances in pomegranate are polyphenols, which have antioxidative, antimutagenic, anti-inflammatory, and antimicrobial properties [
8]. Recent reports showed that pomegranates contain several species of flavonoids and anthocyanidins in their seed oil and juice, and that their antioxidative activities are three times more potent than those of red wine and green tea extract [
9,
10]. The phytoestrogenic effects of pomegranate [
11] are mainly related to the phytoestrogenic effects of isoflavonoids
via antioxidant and anti-inflammatory pathways [
12,
13].
Based on the above information, we hypothesized that dried pomegranate concentrate powder (PCP) could potentiate the anti-climacteric effects of RC by increasing the diversity of bioactive isoflavonoids. Thus, we examined whether the anti-climacteric activity of RC could be enhanced by the addition of PCP in ovariectomized (OVX) rats. The anti-climacteric effects of RC, PCP, or RC + PCP were divided into three categories (estrogenic, anti-osteoporotic, and anti-obesity) and evaluated. An estrogen-deficient OVX rat model was used to evaluate the anti-climacteric effects of the mixtures. Since several symptoms of climacterium are clearly induced by ovariectomy within 4 to 6 weeks postoperatively, the rat model was chosen to investigate the mechanisms responsible for menopause-related complications in humans due to many similarities with postmenopausal climacterium symptoms in rats.
4. Discussion
Hormone therapy is often used to relieve climacteric symptoms; however, serious concerns have been raised regarding the use and safety of hormone replacement therapy over long treatment periods in relation to cardiovascular events and breast cancer [
20,
21,
22]. Consequently, many researchers have sought alternative therapies, including the use of PEs to relieve menopausal symptoms [
23,
24,
25]. Therefore, our study is meaningful because the RC:PCP 2:1 mixture we applied potently reduced climacteric symptoms (
i.e., estrogenic, anti-osteoporotic, and anti-obesity effects).
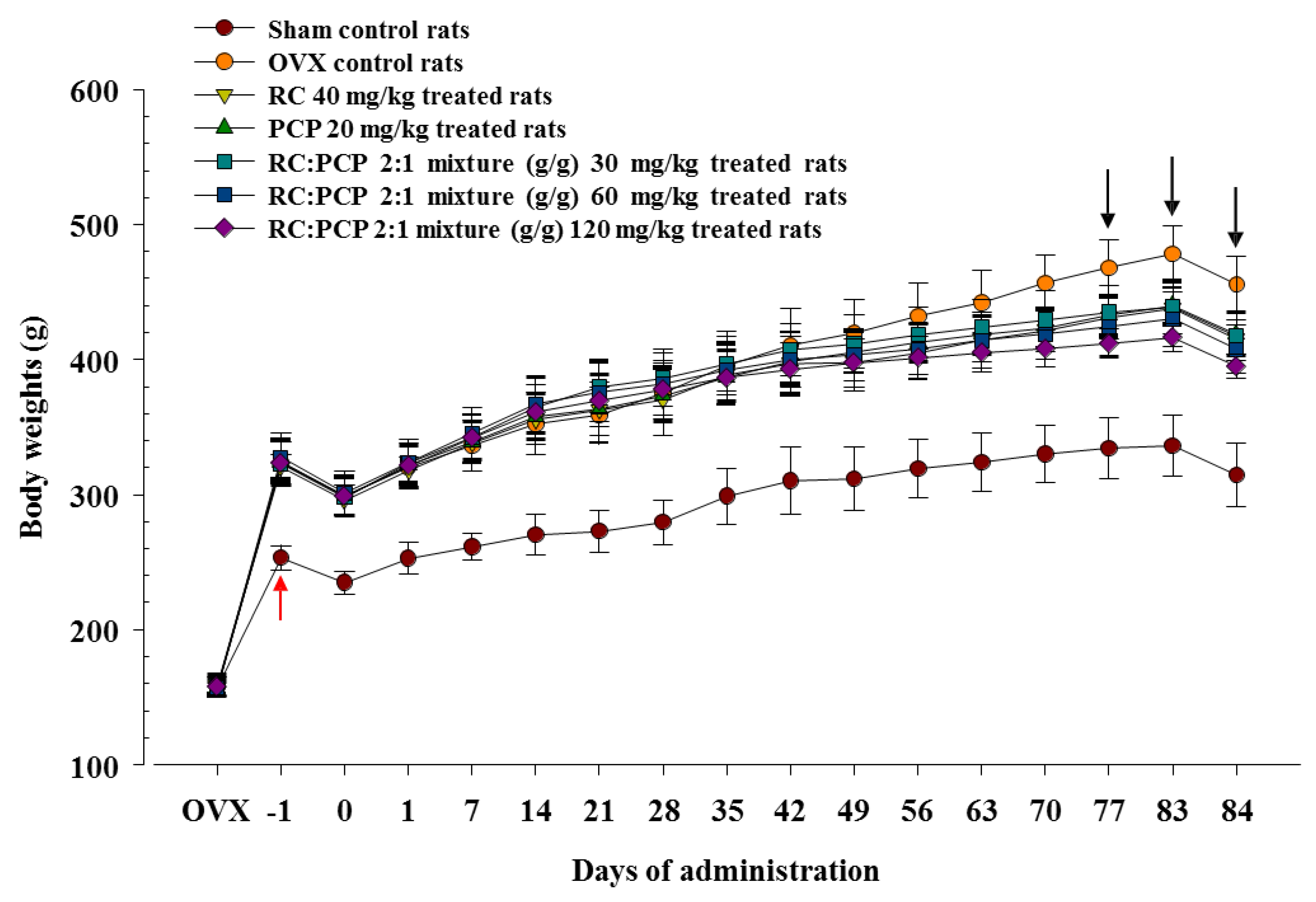
First, we observed the anti-obesity effects in OVX rats; the 120 mg/kg mixtures reduced food and water consumption compared with a single formula of RC and PCP. In addition, the 120 mg/kg mixtures caused increases in fecal and urinary excretion compared with RC or PCP treatment but did not influence food and water intake significantly. These results indicate that the addition of appropriate amounts of PCP increased the inhibitory effect in OVX-induced estrogen deficiency and related obesity. These effects are attributed to the enhancement of digestive tract motility or diuretic effects, along with estrogenic inhibition by increasing the satiating potency of digestive hormones, cholecystokinin (CCK) [
26], and glucagon [
27]. Increased digestive motility also induced increased fecal excretion and consequently induced decreased the body weight of rats [
28], similar to the decreased body weight induced by diuretics in rats [
29]. Estradiol is involved in the regulation of food consumption and body weight mainly by modulating the potency of feedback signals that control meal size [
30]. The most investigated mechanism indicates that estradiol increases the satiating potency of exogenous and endogenous CCK [
26]. A similar explanation may be applicable for glucagon because the effects of glucagon and glucagon antibodies on meal size control are amplified by estradiol in OVX rats [
27]. The absence of estradiol leads to a temporary increase in food consumption and a sustained increase in body weight [
31]. This phenomenon is of clinical relevance because estradiol levels decrease in postmenopausal females; importantly, postmenopausal females constitute a high percentage of the obese population [
32]. Estrogen depletion caused by ovariectomy in rats induced severe increases in food and water intake, but not urinary and fecal excretion, and it facilitated body fat deposition, especially in the abdominal cavity as stated by other investigators [
32,
33]. The accumulation of fat or increase in fat deposition in the body is a major characteristic of obesity, and cellular hypertrophy appears to be the major mode of expansion of intra-abdominal adipose tissue in rodents [
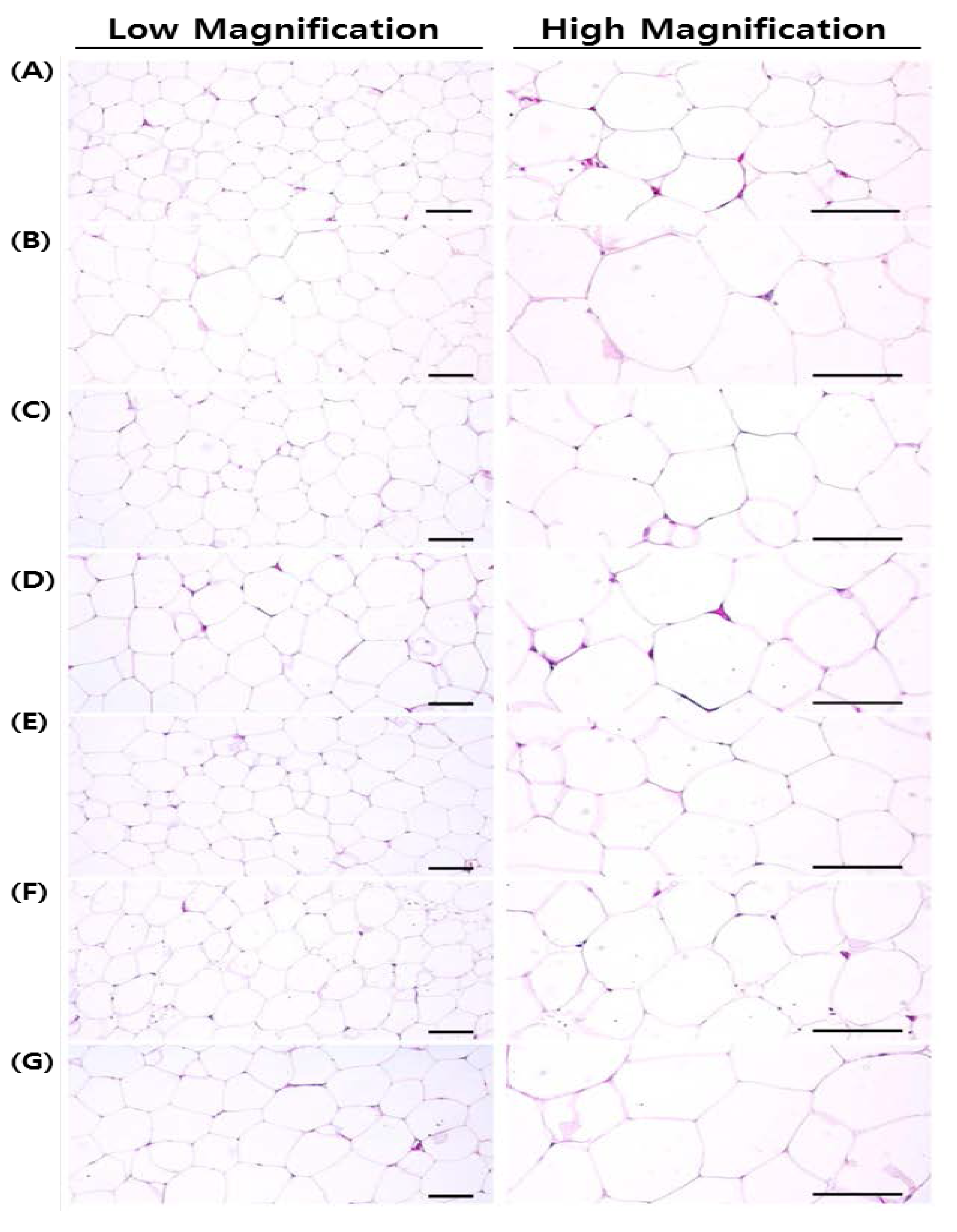
18].
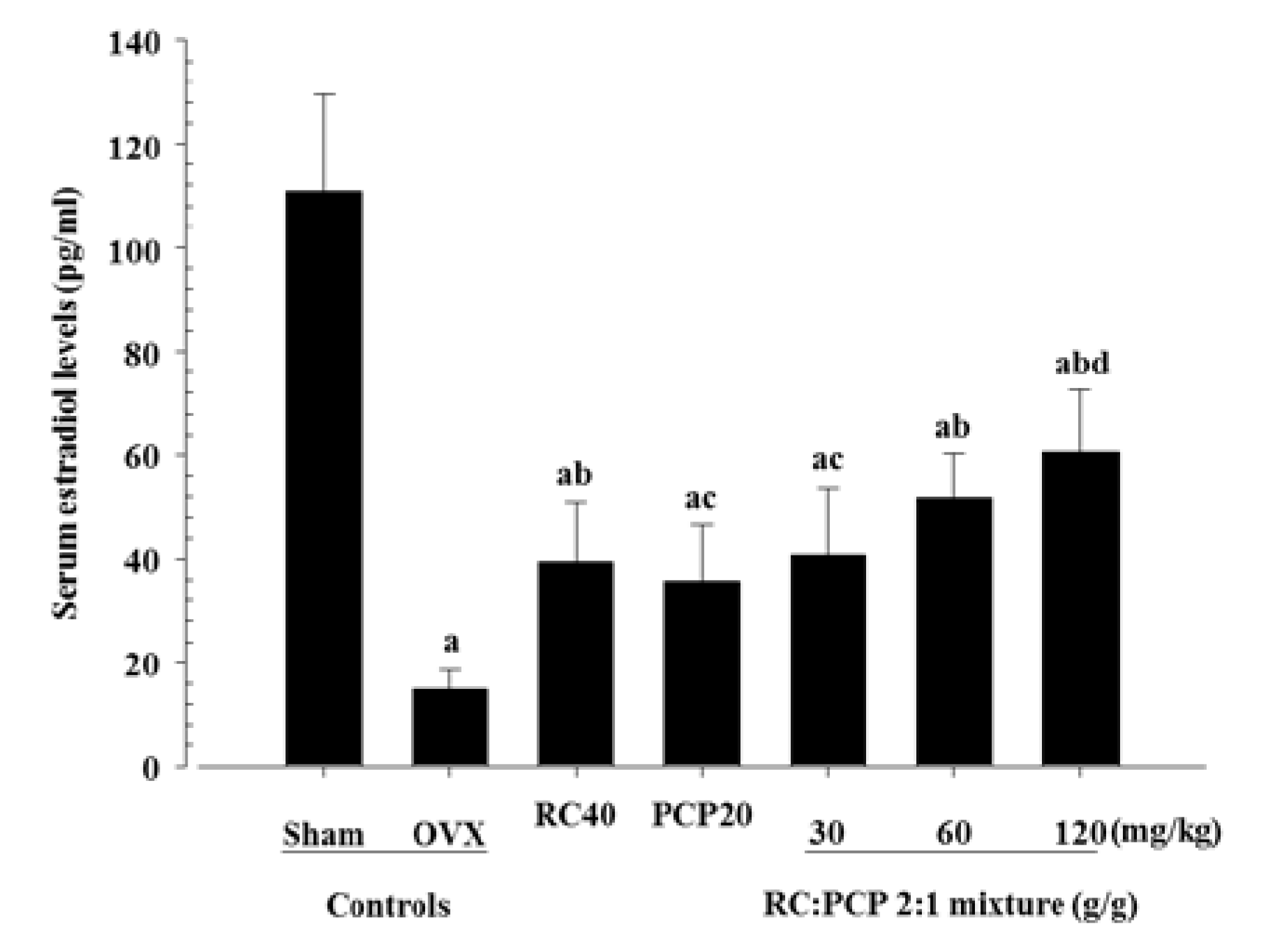
Next, we found that the 120 mg/kg mixtures exhibited stronger inhibitory effects against ovariectomy-induced estradiol depletion and related uterine atrophic changes compared with a single formula of PCP. RC and PCP and their combinations are considered to exert protective effects against ovariectomy-induced uterine atrophy. The increase in uterine mass is mainly attributed to uterine water inhibition and/or cell proliferation [
34] and is mediated through estrogen receptor (ER)-α [
35]. PEs can mimic estrogen [
6] in biological systems due to their structural similarity to estrogen. PEs and isoflavonoids can have a weak estrogen-like effect by binding to both ER-α and -β in various tissues [
35]. Estrogen deficiency is accompanied by marked atrophy of organs such as the uterus and vagina [
33].
Lastly, we determined the anti-osteoporotic effect using various data, including BMD, bALP, and trabecular and cortical bones. As a result, estrogen-deficient osteoporosis was significantly inhibited in RC, PCP, and mixture-treated OVX rats. Specifically, the 120 mg/kg mixtures showed a stronger inhibitory effect against estrogen-deficient osteoporosis compared with a single formula of RC and PCP. Osteoporosis, a metabolic bone disease, results from a disturbance in normal bone remodeling, changing the balance to bone resorption over formation. Osteoporosis is caused by an imbalance between bone resorption and bone formation and results in bone loss and fractures after mineral flux [
36]. Estrogen-deficient OVX osteoporosis animal models have been used to evaluate osteoporotic drugs [
37]. Serum osteocalcin levels are generally considered to be a marker of bone turnover and serum bALP levels to be a marker of bone formation [
38]. As progression of OVX related osteoporosis, serum osteocalcin levels were generally increased along the increases of bone turn over, but serum bALP contents were decreased along inhibition of bone formations [
38,
39]. On the contrary, the serum osteocalcin levels were decreased, but serum bALP levels were increased in present study, which suggested that the mixture treatment activated the osteoblast differentiation and inhibited the bone mineralization and turn over [
40]. BMD provides information regarding the efficacy of anti-osteoporotic agents [
41]. Microscopic observations of bones also provide valuable information regarding bone morphology [
42]. In osteoporotic animals, the histological profiles are clearly changed compared with sham controls regardless of the cause, especially in the trabecular and cortical bones. The efficacy of various anti-osteoporotic agents has been evaluated using bone histology [
43]. Namely, some histomorphometrical indices for bone mass and bone formation are decreased concomitantly with increased bone resorption, and this data can help predict the efficacy of anti-osteoporotic agents [
43].
Many studies have shown that pomegranate juice and pomegranate polyphenol extracts can prevent many types of cancer, cardiovascular disease, diabetes, Alzheimer’s disease, arthritis, and colitis [
8,
44,
45]. Recent reports have shown that pomegranate seed oil and pomegranate juice contain several species of flavonoids and anthocyanidins, and that their antioxidant activity is three times more potent than that in red wine or green tea extract [
9,
10]. Recently, RC botanical dietary supplements have received significant attention for their potential use in the treatment of menopausal symptoms. The estrogenic activity of RC is mainly due to isoflavones and, to a lesser extent, coumestans [
4]. Isoflavones have various biological activities and can improve metabolic symptoms [
46] and bone-protective effects [
47] in menopause. Isoflavonoids exert a weak estrogen-like effect by binding to ER-α and -β in various tissues [
6,
35,
48]. Furthermore, flavonoids have the ability to interact with estrogen receptors and to control the activity of CYP19, an important enzyme in estrogen biosynthesis, and/or steroid dehydrogenases, (e.g., 11β-hydroxysteroid dehydrogenase) [
49]. These effects induce various alterations causing a change in the overall hormonal balance, resulting in protection against bone loss and reducing osteoporotic effects and other menopausal symptoms [
50]. Conversely, estrogen-mediated repression of phase 2 enzyme activities may increase cellular oxidative DNA damage that can result ultimately in the formation of cancer [
51]. This DNA damage is inhibited by superoxide dismutase 3 induced by antioxidants [
52]. These findings indicate that the beneficial effects of RC [
3,
4,
25] and PCP [
10,
11] on postmenopausal symptoms are mainly due to the phytoestrogenic effects of isoflavonoids and related antioxidant pathways [
12,
13]. Therefore, appropriate mixtures consisting of RC and PCP may show greater favorable anti-climacteric effects by combination of isoflavonoids and ellagic acid [
53,
54]. The RC and PCP mixture may enhance direct free radical scavenging and indirectly induce antioxidative enzymes.
Our results show that RC:PCP 2:1 mixture relieves climacteric symptoms through anti-osteoporotic and anti-obesity activities, but, there are some limitations in this study. The estrogen-deficient OVX model is useful for evaluating anti-climacteric effects. Since several symptoms of climacterium are clearly induced by ovariectomy within 4 to 6 weeks postoperatively, the rat model was chosen to investigate the mechanisms responsible for menopause-related complications in humans due to many similarities with postmenopausal climacterium symptoms in rats. However, menopause is related with aging and postmenopausal symptoms occurring in elderly females. A limitation of this study is that the anti-climacterium effects were evaluated in young and not older female rats; the effects of RC and PCP against climacterium may be different in older animal models. Anticlimacterium effects of RC:PCP mixture were assumed to be related with the major active components of RC and PCP, however, their principal phytochemical constituents weren’t measured in plasma or the respective tissues. In addition, characterization of the phenolic composition of RC and PCP weren’t fully analyzed; only 4 types of isoflavone (genestin, biochain A, formononetin, and daidzein) were clarified in RC and ellagic acid was measured in PCP. Thus, these effects of RC:PCP mixture weren’t discussed precisely. To clearly confirm the anticlimaterium effects of RC:PCP 2:1 mixture, further studies should analyze characterization of the full phenolic composition of each material and evaluate their principal phytochemical constituents in plasma or the respective tissues.