Bee Pollen Improves Muscle Protein and Energy Metabolism in Malnourished Old Rats through Interfering with the Mtor Signaling Pathway and Mitochondrial Activity
Abstract
:1. Introduction
2. Material and Methods
2.1. Animals
2.2. Experimental Procedures
| g/100 g | Standard Diet | 5% Pollen Diet | 10% Pollen Diet |
|---|---|---|---|
| Casein | 14 | 14 | 14 |
| Cornstarch | 46 | 41 | 36 |
| Sucrose | 28 | 28 | 28 |
| Soybean oil | 4 | 4 | 4 |
| Cellulose | 4 | 4 | 4 |
| Vitamins and minerals mix | 4 | 4 | 4 |
| Bee pollen | 0 | 5 | 10 |
| Fresh Bee Pollen Formula | |
|---|---|
| Proteins (g/100 g) | 16.2 |
| EAA (g/100 g) | 4.8 |
| BCAA (g/100 g) | 2.3 |
| SAA (g/100 g) | 0.6 |
| Carbohydrates (g/100 g) | 57.7 |
| Lipids (g/100 g) | 7.3 |
| Saturated FA (% of total FA) | 43.1 |
| Monounsaturated FA (% of total FA) | 16.1 |
| Polyunsaturated FA (% of total FA) | 40.8 |
| Fiber (g/100 g) | 11.4 |
| Vitamin B1 (mg/100 g) | 0.65 |
| Vitamin B2 (mg/100 g) | 0.76 |
| Vitamin PP(mg/100 g) | 7.05 |
| Vitamin B5 (mg/100 g) | 0.77 |
| Vitamin B6 (mg/100 g) | 2.38 |
| Vitamin B9 (mg/100 g) | 0.81 |
| Vitamin C (mg/100 g) | 42.7 |
| Vitamin E (mg/10 0g) | 12.9 |
| Calories (kcal/100 g) | 315.8 |
| Antioxidant value | |
| FRAP (mmol/kg eq ascorbic acid) | 25.8 |
| ORAC (mmol/100 g eq trolox) | 5.6 |
2.3. Blood Biochemistry
2.4. In Vivo Protein Synthesis Measurement
2.5. Western Blot Analysis
2.6. Mitochondrial Enzyme Activities
2.7. Statistical Analysis
3. Results
3.1. Body and Tissue Weights
| Controls | Restricted | Refed Standard | Refed 5% Pollen | Refed 10% Pollen | |
|---|---|---|---|---|---|
| Body weight (g) | 629 ± 5 a | 418 ± 13 b | 570 ± 19 a | 556 ± 20 a | 560 ± 13 a |
| Fat mass (g) | 151 ± 16 a | 13 ± 3 b | 84 ± 11 c | 78 ± 9 c | 87 ± 5 c |
| Fat-free mass (g) | 491 ± 16 a | 385 ± 4 b | 493 ± 11 a | 487 ± 14 a | 482 ± 12 a |
| Hindlimb muscle mass (g) | 11.5 ± 0.7 | 10.8 ± 0.7 | 11.8 ± 1.1 | 12.2 ± 0.4 | 12.7 ± 0.6 |
| Plantaris (mg) | 335 ± 36 a | 317 ± 22 a | 349 ± 27 a,b | 380 ± 14 b | 389 ± 28 b |
| Soleus (mg) | 204 ± 13 | 199 ± 15 | 198 ± 18 | 203 ± 10 | 213 ± 9 |
| Gastrocnemius (g) | 1.71 ± 0.16 a | 1.47 ± 0.10 b | 1.67 ± 0.18 a,b | 1.80 ± 0.07 a | 1.79 ± 0.11 a |
| Visceral adipose tissue (g) | 15.7 ± 3.3 a | 1.8 ± 0.5 b | 11.1 ± 1.4 a | 10.0 ± 4.4 a | 11.3 ± 0.9 a |
| Subcutaneous adipose tissue (g) | 18.8 ± 7.7 a | 3.1 ± 0.8 b | 7.6 ± 1.2 b,c | 7.3 ± 1.2 b,c | 8.3 ± 0.9 c |
| Liver (g) | 14.4 ± 1.4 a | 8.9 ± 0.4 b | 14.0 ± 0.6 a | 12.6 ± 0.5 a | 12.8 ± 0.4 a |
| Heart (g) | 1.77 ± 0.9 a | 1.51 ± 0.05 b | 1.88 ± 0.04 a | 1.82 ± 0.07 a | 1.86 ± 0.06 a |
| Kidney (g) | 1.70 ± 0.15 a | 1.42 ± 0.05 b | 1.79 ± 0.08 a | 1.69 ± 0.09 a | 1.76 ± 0.08 a |
3.2. Biochemical and Cytokine Plasma Profiles
3.3. Muscle Mitochondrial Activity
| Controls | Restricted | Refed Standard | Refed 5% Pollen | Refed 10% Pollen | |
|---|---|---|---|---|---|
| Insulin (ng/mL) | 2.93 ± 1.22 | 1.36 ± 0.61 | 1.70 ± 0.41 | 1.66 ± 0.25 | 1.45 ± 0.13 |
| Leptin (pg/mL) | 2481 ± 555 a | 551 ± 89 b | 1061 ± 212 b | 1207 ± 201 b | 1087 ± 123 b |
| Adiponectin (µg/mL) | 12.9 ± 1.2 a | 23.0 ± 1.4 b | 15.0 ± 1.5 a | 14.3 ± 2.4 a | 15.7 ± 0.9 a |
| Resistin (ng/mL) | 23.6 ± 4.1 a | 36.7 ± 3.9 b | 27.2 ± 1.3 a | 24.4 ± 1.9 a | 22.0 ± 2.1 a |
| Alpha2-macroglobuline (µg/mL) | 14.0 ± 4.2 a | 10.1 ± 1.1 b | 11.8 ± 3.0 b | 11.2 ± 1.5 b | 14.8 ± 0.5 a |
| Orosomucoid (µg/mL) | 56 ± 7 a | 108 ± 14 b | 77 ± 8 a | 73 ± 6 a | 71 ± 9 a |
| TNFα (pg/mL) | 13.9 ± 0.5 | 12.9 ± 0.4 | 13.3 ± 0.3 | 12.9 ± 0.6 | 13.9 ± 0.4 |
| sTNF-R1 (pg/mL) | 128 ± 7 a | 94 ± 4 b | 122 ± 7 a | 129 ± 5 a | 130 ± 6 a |
| sTNF-R2 (pg/mL) | 1145 ± 75 a | 814 ± 30 b | 1212 ± 143 a | 1088 ± 51 a | 1105 ± 82 a |
| IL1β (pg/mL) | 38.4 ± 4.4 | 43.6 ± 13.1 | 37.1 ± 3.2 | 38.9 ± 13.8 | 31.4 ± 2.7 |
| IL6 (pg/mL) | 40.5 ± 7.7 | 32.6 ± 7.1 | 31.7 ± 3.2 | 44.6 ± 3.0 | 39.4 ± 6.3 |
| Controls | Restricted | Refed Standard | Refed 5% Pollen | Refed 10% Pollen | |
|---|---|---|---|---|---|
| CS (µmol/min/mg proteins) | 0.346 ± 0.029 a | 0.243 ± 0.028 b | 0.228 ± 0.032 b | 0.294 ± 0.031 a,b | 0.256 ± 0.034 a,b |
| HAD (nmol/min/mg proteins) | 8.45 ± 0.56 | 7.67 ± 0.92 | 7.56 ± 0.91 | 8.32 ± 1.44 | 8.73 ± 0.95 |
| Complex II (nmol/min/mg proteins) | 21.0 ± 1.3 a | 17.5 ± 0.8 a,b | 17.1 ± 1.3 b | 17.7 ± 2.4 a,b | 18.6 ± 1.5 a,b |
| Complex III (nmol/min/mg proteins) | 2.99 ± 0.64 | 2.61 ± 0.63 | 2.52 ± 0.39 | 1.98 ± 0.59 | 2.02 ± 0.45 |
| Complex IV (nmol/min/mg proteins) | 16.8 ± 1.5 a | 12.7 ± 1.7 b | 12.9 ± 1.4 b | 12.8 ± 0.9 b | 16.0 ± 2.3 a |
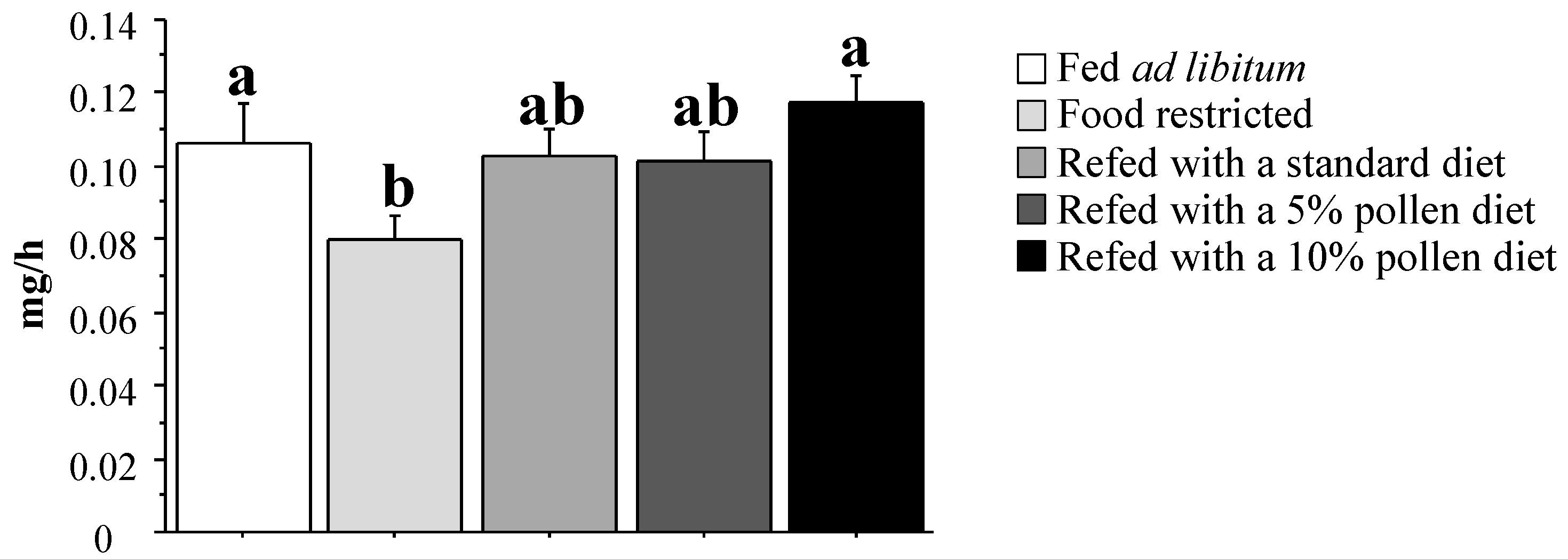
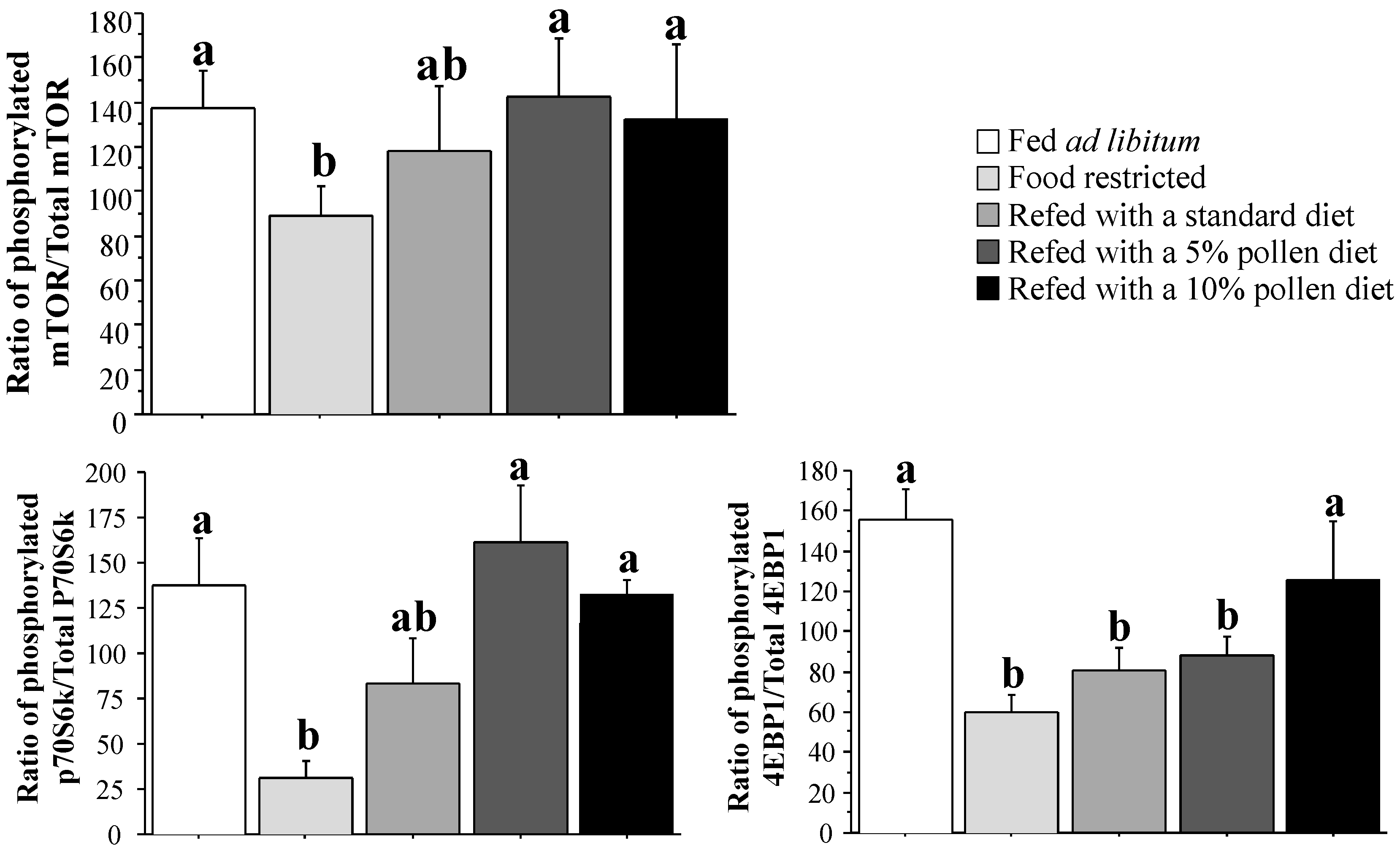
3.4. Muscle Protein Synthesis Rate and Regulation
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kroyer, G.; Hegedus, N. Evaluation of bioactive properties of pollen extracts as functional dietary food supplement. Innov. Food Sci. Emerg. Technol. 2001, 2, 171–174. [Google Scholar]
- Campos, M.G.; Webby, R.F.; Markham, K.R.; Mitchell, K.A.; da Cunha, A.P. Age-induced diminution of free radical scavenging capacity in bee pollens and the contribution of constituent flavonoids. J. Agric. Food Chem. 2003, 51, 742–745. [Google Scholar] [CrossRef]
- Almaraz-Abarca, N.; Campos, M.G.; Avila-Reyes, J.A.J.A.; Naranjo-Jimenez, N.; Corral, J.H.; Gonzalez-Valdez, L.S. Antioxidant activity of polyphenolic extract of monofloral honey bee collected pollen from mesquite (Prosopis juliflora, Leguminosae). J. Food Compos. Anal. 2007, 20, 119–124. [Google Scholar] [CrossRef]
- Linskens, H.F.; Jorde, W. Pollen as food and medicine-a review. Economic. Bot. 1997, 51, 78–87. [Google Scholar] [CrossRef]
- Gibbs, A.; Green, C.; Doctor, V.M. Isolation and anticoagulant properties of polysaccharides of Typha Augustata and Daemonorops species. Thromb. Res. 1983, 32, 97–108. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Zhang, C.Y.; Xu, D.M.; Huang, G.Q.; Xu, Y.L.; Wang, Z.Y.; Fang, S.D.; Chen, Y.; Gu, Y.L. The antiatherogenic effects of components isolated from pollen typhae. Thromb. Res. 1990, 57, 957–966. [Google Scholar] [CrossRef]
- Chung, S.; Park, S.; Yang, C.H. Unsaturated fatty acids bind Myc-Max transcription factor and inhibit Myc-Max-DNA complex formation. Cancer Lett. 2002, 188, 153–162. [Google Scholar] [CrossRef] [PubMed]
- Qin, F.; Sun, H.X. Immunosuppressive activity of Pollen Typhae ethanol extract on the immune responses in mice. J. Ethnopharmacol. 2005, 102, 424–429. [Google Scholar] [CrossRef] [PubMed]
- Burke, L.M.; Castell, L.M.; Stear, S.J.; Rogers, P.J.; Blomstrand, E.; Gurr, S.; Mitchell, N.; Stephens, F.B.; Greenhaff, P.L. BJSM reviews: A–Z of nutritional supplements: Dietary supplements, sports nutrition foods and ergogenic aids for health and performance Part 4. Br. J. Sports Med. 2009, 43, 1088–1090. [Google Scholar] [CrossRef] [PubMed]
- Zawada, E.T., Jr. Malnutrition in the elderly. Is it simply a matter of not eating enough? Postgrad. Med. 1996, 100, 207–208, 211–214, 220–222. [Google Scholar] [CrossRef]
- Russell, R.M. Changes in gastrointestinal function attributed to aging. Am. J. Clin. Nutr. 1992, 55, 1203S–1207S. [Google Scholar]
- Beaufrere, B.; Boirie, Y. Aging and protein metabolism. Curr. Opin. Clin. Nutr. Metab. Care 1998, 1, 85–89. [Google Scholar] [PubMed]
- Chandra, R.K. Impact of nutritional status and nutrient supplements on immune responses and incidence of infection in older individuals. Ageing Res. Rev. 2004, 3, 91–104. [Google Scholar] [CrossRef] [PubMed]
- Walrand, S.; Chambon-Savanovitch, C.; Felgines, C.; Chassagne, J.; Raul, F.; Normand, B.; Farges, M.C.; Beaufrere, B.; Vasson, M.P.; Cynober, L. Aging: A barrier to renutrition? Nutritional and immunologic evidence in rats. Am. J. Clin. Nutr. 2000, 72, 816–824. [Google Scholar] [PubMed]
- Walrand, S.; Moreau, K.; Caldefie, F.; Tridon, A.; Chassagne, J.; Portefaix, G.; Cynober, L.; Beaufrere, B.; Vasson, M.P.; Boirie, Y. Specific and nonspecific immune responses to fasting and refeeding differ in healthy young adult and elderly persons. Am. J. Clin. Nutr. 2001, 74, 670–678. [Google Scholar] [PubMed]
- Castaneda, C.; Charnley, J.M.; Evans, W.J.; Crim, M.C. Elderly women accommodate to a low-protein diet with losses of body cell mass, muscle function, and immune response. Am. J. Clin. Nutr. 1995, 62, 30–39. [Google Scholar] [PubMed]
- Chambon-Savanovitch, C.; Felgines, C.; Farges, M.C.; Pernet, P.; Cezard, J.; Raul, F.; Cynober, L.; Vasson, M.P. Severe dietary restriction initiated in aged rats: Evidence for poor adaptation in terms of protein metabolism and intestinal functions. Eur. J. Clin. Invest. 1999, 29, 504–511. [Google Scholar] [CrossRef] [PubMed]
- Felgines, C.; Savanovitch, C.; Farges, M.C.; Cynober, L.; Vasson, M.P. Protein metabolism in rats during long-term dietary restriction: Influence of aging. JPEN J. Parenter. Enter. Nutr. 1999, 23, 32–37. [Google Scholar] [CrossRef]
- Hebuterne, X.; Broussard, J.F.; Rampal, P. Acute renutrition by cyclic enteral nutrition in elderly and younger patients. JAMA J. Am. Med. Assoc. 1995, 273, 638–643. [Google Scholar] [CrossRef]
- Hebuterne, X.; Schneider, S.; Peroux, J.L.; Rampal, P. Effects of refeeding by cyclic enteral nutrition on body composition: Comparative study of elderly and younger patients. Clin. Nutr. 1997, 16, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Chambon-Savanovitch, C.; Felgines, C.; Walrand, S.; Raul, F.; Zarrabian, S.; Meunier, M.T.; Farges, M.C.; Cynober, L.; Vasson, M.P. A pancreatic extract-enriched diet improves the nutritional status of aged rats. J. Nutr. 2001, 131, 813–819. [Google Scholar]
- Osowska, S.; Duchemann, T.; Walrand, S.; Paillard, A.; Boirie, Y.; Cynober, L.; Moinard, C. Citrulline modulates muscle protein metabolism in old malnourished rats. Am. J. Physiol. Endocrinol. Metab. 2006, 291, E582–E586. [Google Scholar] [CrossRef] [PubMed]
- Farges, M.C.; Vasson, M.P.; Davot, P.; Villie, F.; Meunier, M.T.; Marcollet, M.; Cynober, L. Supplementation of oral nutrition with pancreatic enzymes improves the nutritional status of aged endotoxemic rats. Nutrition 1996, 12, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Walrand, S.; Guillet, C.; Salles, J.; Cano, N.; Boirie, Y. Physiopathological mechanism of sarcopenia. Clin. Geriatr. Med. 2011, 27, 365–385. [Google Scholar] [CrossRef] [PubMed]
- Walrand, S.; Short, K.R.; Heemstra, L.A.; Novak, C.M.; Levine, J.A.; Coenen-Schimke, J.M.; Nair, K.S. Altered regulation of energy homeostasis in older rats in response to thyroid hormone administration. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2014, 28, 1499–1510. [Google Scholar]
- Zangarelli, A.; Chanseaume, E.; Morio, B.; Brugere, C.; Mosoni, L.; Rousset, P.; Giraudet, C.; Patrac, V.; Gachon, P.; Boirie, Y.; et al. Synergistic effects of caloric restriction with maintained protein intake on skeletal muscle performance in 21-month-old rats: A mitochondria-mediated pathway. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2006, 20, 2439–2450. [Google Scholar]
- Vellas, B.; Lauque, S.; Andrieu, S.; Nourhashemi, F.; Rolland, Y.; Baumgartner, R.; Garry, P. Nutrition assessment in the elderly. Curr. Opin. Clin. Nutr. Metab. Care 2001, 4, 5–8. [Google Scholar] [CrossRef] [PubMed]
- Whitehead, C.; Finucane, P. Malnutrition in elderly people. Aust. N. Zeal. J. Med. 1997, 27, 68–74. [Google Scholar] [CrossRef]
- Dey, D.K.; Rothenberg, E.; Sundh, V.; Bosaeus, I.; Steen, B. Body mass index, weight change and mortality in the elderly. A 15 years longitudinal population study of 70 years olds. Eur. J. Clin. Nutr. 2001, 55, 482–492. [Google Scholar] [CrossRef] [PubMed]
- Payette, H.; Coulombe, C.; Boutier, V.; Gray-Donald, K. Nutrition risk factors for institutionalization in a free-living functionally dependent elderly population. J. Clin. Epidemiol. 2000, 53, 579–587. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, C.S.; Locher, J.L.; Roth, D.L.; McVie, T.; Sawyer, P.; Allman, R. Unintentional weight loss predicts decline in activities of daily living function and life-space mobility over 4 years among community-dwelling older adults. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2008, 63, 67–75. [Google Scholar] [CrossRef]
- Elmstahl, S.; Persson, M.; Andren, M.; Blabolil, V. Malnutrition in geriatric patients: A neglected problem? J. Adv. Nurs. 1997, 26, 851–855. [Google Scholar] [CrossRef] [PubMed]
- Hall, K.; Whiting, S.J.; Comfort, B. Low nutrient intake contributes to adverse clinical outcomes in hospitalized elderly patients. Nutr. Rev. 2000, 58, 214–217. [Google Scholar] [CrossRef] [PubMed]
- Perier, C.; Triouleyre, P.; Terrat, C.; Chomette, M.C.; Beauchet, O.; Gonthier, R. Energy and nutrient intake of elderly hospitalized patients in a steady metabolic status versus catabolic status. J. Nutr. Health Aging 2004, 8, 518–520. [Google Scholar] [PubMed]
- Rudman, D.; Feller, A.G. Protein-calorie undernutrition in the nursing home. J. Am. Geriatr. Soc. 1989, 37, 173–183. [Google Scholar] [PubMed]
- Salva, A.; Coll-Planas, L.; Bruce, S.; de Groot, L.; Andrieu, S.; Abellan, G.; Vellas, B.; Bartorelli, L.; Berner, Y.N.; Corman, B.; et al. Nutritional assessment of residents in long-term care facilities (LTCFs): Recommendations of the task force on nutrition and ageing of the IAGG European region and the IANA. J. Nutr. Health Aging 2009, 13, 475–483. [Google Scholar] [CrossRef] [PubMed]
- Schneider, S.M.; Al-Jaouni, R.; Pivot, X.; Braulio, V.B.; Rampal, P.; Hebuterne, X. Lack of adaptation to severe malnutrition in elderly patients. Clin. Nutr. 2002, 21, 499–504. [Google Scholar] [CrossRef]
- Cao, H. Adipocytokines in obesity and metabolic disease. J. Endocrinol. 2014, 220, T47–T59. [Google Scholar] [CrossRef] [PubMed]
- Park, H.K.; Ahima, R.S. Resistin in rodents and humans. Diabetes Metab. J. 2013, 37, 404–414. [Google Scholar] [CrossRef] [PubMed]
- Proenca, A.R.; Sertie, R.A.; Oliveira, A.C.; Campaaa, A.B.; Caminhotto, R.O.; Chimin, P.; Lima, F.B. New concepts in white adipose tissue physiology. Braz. J. Med. Biol. Res. 2014, 47, 192–205. [Google Scholar] [CrossRef] [PubMed]
- Jeejeebhoy, K.N. Malnutrition, fatigue, frailty, vulnerability, sarcopenia and cachexia: Overlap of clinical features. Curr. Opin. Clin. Nutr. Metab. Care 2012, 15, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Roubenoff, R. Molecular basis of inflammation: Relationships between catabolic cytokines, hormones, energy balance, and muscle. JPEN J. Parenter. Enter. Nutr. 2008, 32, 630–632. [Google Scholar] [CrossRef]
- Greiwe, J.S.; Cheng, B.; Rubin, D.C.; Yarasheski, K.E.; Semenkovich, C.F. Resistance exercise decreases skeletal muscle tumor necrosis factor alpha in frail elderly humans. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2001, 15, 475–482. [Google Scholar]
- Chandra, R.K. Nutrition and the immune system from birth to old age. Eur. J. Clin. Nutr. 2002, 56, S73–S76. [Google Scholar] [CrossRef] [PubMed]
- Lanza, I.R.; Zabielski, P.; Klaus, K.A.; Morse, D.M.; Heppelmann, C.J.; Bergen, H.R., III; Dasari, S.; Walrand, S.; Short, K.R.; Johnson, M.L.; et al. Chronic caloric restriction preserves mitochondrial function in senescence without increasing mitochondrial biogenesis. Cell Metab. 2012, 16, 777–788. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, E.; Ferguson, M.; Banks, M.; Batterham, M.; Bauer, J.; Capra, S.; Isenring, E. Malnutrition and poor food intake are associated with prolonged hospital stay, frequent readmissions, and greater in-hospital mortality: Results from the Nutrition Care Day Survey 2010. Clin. Nutr. 2013, 32, 737–745. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agarwal, E.; Miller, M.; Yaxley, A.; Isenring, E. Malnutrition in the elderly: A narrative review. Maturitas 2013, 76, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Bauer, J.; Biolo, G.; Cederholm, T.; Cesari, M.; Cruz-Jentoft, A.J.; Morley, J.E.; Phillips, S.; Sieber, C.; Stehle, P.; Teta, D.; et al. Evidence-based recommendations for optimal dietary protein intake in older people: A position paper from the PROT-AGE Study Group. J. Am. Med. Dir. Assoc. 2013, 14, 542–559. [Google Scholar] [CrossRef] [PubMed]
- Crozier, S.J.; Kimball, S.R.; Emmert, S.W.; Anthony, J.C.; Jefferson, L.S. Oral leucine administration stimulates protein synthesis in rat skeletal muscle. J. Nutr. 2005, 135, 376–382. [Google Scholar] [PubMed]
- Le Plenier, S.; Walrand, S.; Noirt, R.; Cynober, L.; Moinard, C. Effects of leucine and citrulline versus non-essential amino acids on muscle protein synthesis in fasted rat: A common activation pathway? Amino Acids 2012, 43, 1171–1178. [Google Scholar]
- Pascoal, A.; Rodrigues, S.; Teixeira, A.; Feas, X.; Estevinho, L.M. Biological activities of commercial bee pollens: Antimicrobial, antimutagenic, antioxidant and anti-inflammatory. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2014, 63, 233–239. [Google Scholar] [CrossRef]
- Marzani, B.; Balage, M.; Venien, A.; Astruc, T.; Papet, I.; Dardevet, D.; Mosoni, L. Antioxidant supplementation restores defective leucine stimulation of protein synthesis in skeletal muscle from old rats. J. Nutr. 2008, 138, 2205–2211. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.T.; Wang, T.Z.; Chen, Y.; Liu, J.B.; Liu, Y.; Wang, W.J. Pollen Typhae total flavone improves insulin-induced glucose uptake through the beta-arrestin-2-mediated signaling in C2C12 myotubes. Int. J. Mol. Med. 2012, 30, 914–922. [Google Scholar]
- Biolo, G.; Fleming, R.Y.D.; Wolfe, R.R. Physiologic hyperinsulinemia stimulates protein synthesis and enhances transport of selected amino acids in human skeletal muscle. J. Clin. Investig. 1995, 95, 811–819. [Google Scholar] [CrossRef] [PubMed]
- Short, K.R.; Bigelow, M.L.; Kahl, J.; Singh, R.; Coenen-Schimke, J.; Raghavakaimal, S.; Nair, K.S. Decline in skeletal muscle mitochondrial function with aging in humans. Proc. Natl. Acad. Sci. USA 2005, 102, 5618–5623. [Google Scholar] [CrossRef] [PubMed]
- Gojda, J.; Patkova, J.; Jacek, M.; Potockova, J.; Trnka, J.; Kraml, P.; Andel, M. Higher insulin sensitivity in vegans is not associated with higher mitochondrial density. Eur. J. Clin. Nutr. 2013, 67, 1310–1315. [Google Scholar] [CrossRef] [PubMed]
- Huttemann, M.; Lee, I.; Grossman, L.I.; Doan, J.W.; Sanderson, T.H. Phosphorylation of mammalian cytochrome c and cytochrome c oxidase in the regulation of cell destiny: Respiration, apoptosis, and human disease. Adv. Exp. Med. Biol. 2012, 748, 237–264. [Google Scholar] [PubMed]
- Gueguen, N.; Lefaucheur, L.; Fillaut, M.; Herpin, P. Muscle fiber contractile type influences the regulation of mitochondrial function. Mol. Cell. Biochem. 2005, 276, 15–20. [Google Scholar] [CrossRef] [PubMed]
- Gueguen, N.; Lefaucheur, L.; Fillaut, M.; Vincent, A.; Herpin, P. Control of skeletal muscle mitochondria respiration by adenine nucleotides: Differential effect of ADP and ATP according to muscle contractile type in pigs. Comp. Biochem. Physiol. Part B Biochem. Mol. Biol. 2005, 140, 287–297. [Google Scholar] [CrossRef]
- Saric, A.; Balog, T.; Sobocanec, S.; Kusic, B.; Sverko, V.; Rusak, G.; Likic, S.; Bubalo, D.; Pinto, B.; Reali, D.; et al. Antioxidant effects of flavonoid from Croatian Cystus incanus L. rich bee pollen. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2009, 47, 547–554. [Google Scholar] [CrossRef]
- Yildiz, O.; Can, Z.; Saral, O.; Yulug, E.; Ozturk, F.; Aliyazicioglu, R.; Canpolat, S.; Kolayli, S. Hepatoprotective potential of chestnut bee pollen on carbon tetrachloride-induced hepatic damages in rats. Evid. Based Complement. Alternat. Med. ECAM 2013, 2013, 461478. [Google Scholar] [CrossRef]
- Gredilla, R.; Phaneuf, S.; Selman, C.; Kendaiah, S.; Leeuwenburgh, C.; Barja, G. Short-term caloric restriction and sites of oxygen radical generation in kidney and skeletal muscle mitochondria. Ann. NY Acad. Sci. 2004, 1019, 333–342. [Google Scholar] [CrossRef] [PubMed]
- Guillet, C.; Boirie, Y. Insulin resistance: A contributing factor to age-related muscle mass loss? Diabetes Metab. 2005, 31, 5S20–5S26. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, L.M.; Jensen, E.L.; Bustos, R.I.; Arguelloa, G.; Gutierrez-Garcia, R.; Gonzalez, M.; Hernandez, C.; Paredes, R.; Simon, F.; Riedel, C.; et al. Adaptive responses of mitochondria to mild copper deprivation involve changes in morphology, OXPHOS remodeling and bioenergetics. J. Cell. Physiol. 2014, 229, 607–619. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salles, J.; Cardinault, N.; Patrac, V.; Berry, A.; Giraudet, C.; Collin, M.-L.; Chanet, A.; Tagliaferri, C.; Denis, P.; Pouyet, C.; et al. Bee Pollen Improves Muscle Protein and Energy Metabolism in Malnourished Old Rats through Interfering with the Mtor Signaling Pathway and Mitochondrial Activity. Nutrients 2014, 6, 5500-5516. https://doi.org/10.3390/nu6125500
Salles J, Cardinault N, Patrac V, Berry A, Giraudet C, Collin M-L, Chanet A, Tagliaferri C, Denis P, Pouyet C, et al. Bee Pollen Improves Muscle Protein and Energy Metabolism in Malnourished Old Rats through Interfering with the Mtor Signaling Pathway and Mitochondrial Activity. Nutrients. 2014; 6(12):5500-5516. https://doi.org/10.3390/nu6125500
Chicago/Turabian StyleSalles, Jérôme, Nicolas Cardinault, Véronique Patrac, Alexandre Berry, Christophe Giraudet, Marie-Laure Collin, Audrey Chanet, Camille Tagliaferri, Philippe Denis, Corinne Pouyet, and et al. 2014. "Bee Pollen Improves Muscle Protein and Energy Metabolism in Malnourished Old Rats through Interfering with the Mtor Signaling Pathway and Mitochondrial Activity" Nutrients 6, no. 12: 5500-5516. https://doi.org/10.3390/nu6125500