The Role of Vitamin D Level and Related Single Nucleotide Polymorphisms in Crohn’s Disease
Abstract
:1. Introduction
2. Experimental Section
2.1. Study Subjects
2.2. Vitamin D Determination
2.3. Genotyping
2.4. Statistical Analysis
3. Results
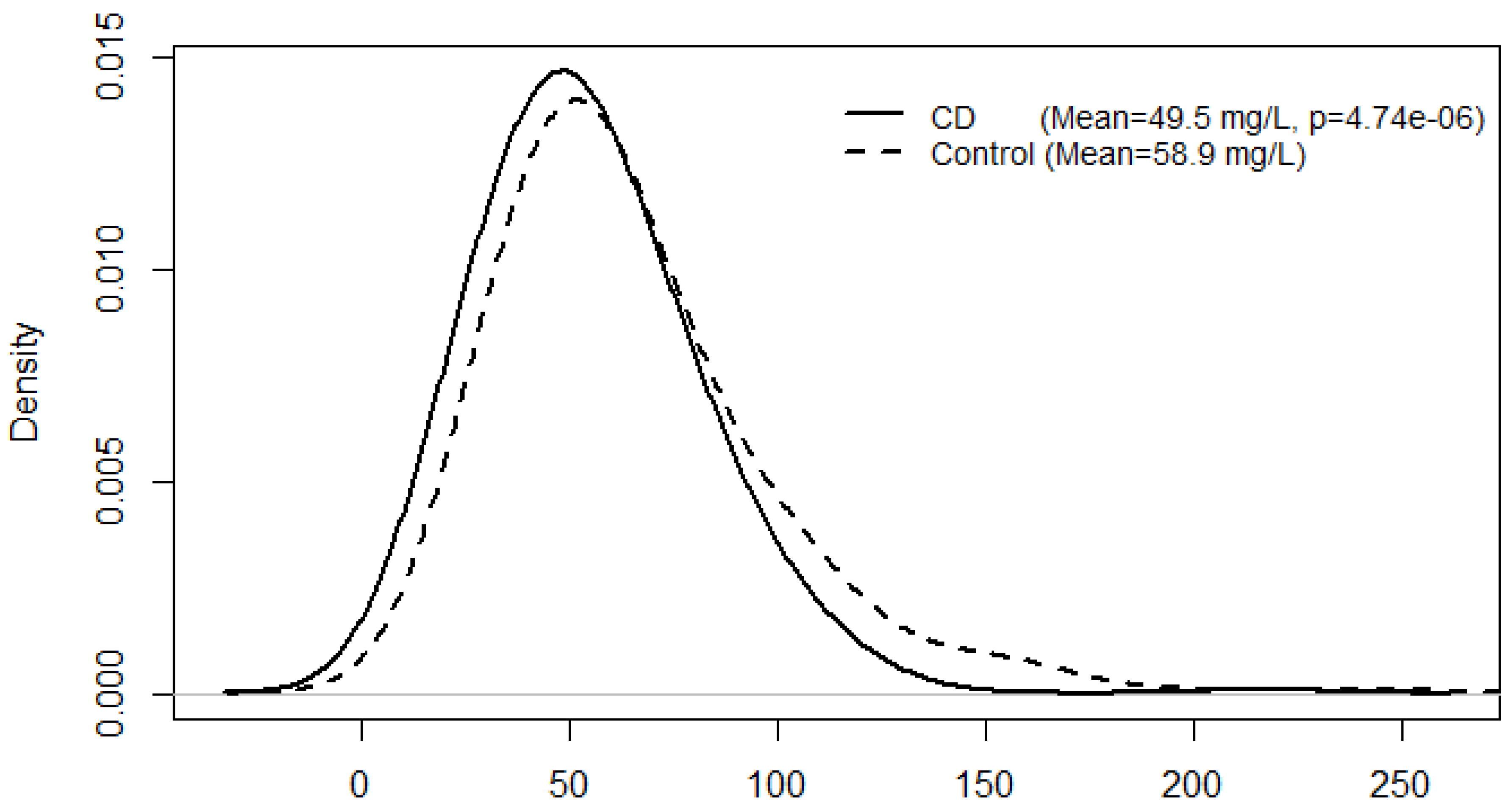
3.1. CD Status, Serum Vitamin D Levels and Multivariate Analysis
| Variable | Status | Mean (95% CI) | p |
|---|---|---|---|
| IBD | CD (n = 302) | 0.805 (0.741–0.874) | 2.97 × 10−7 |
| Control (n = 306) | 1.00 | ||
| Smoking | Yes (n = 144) | 1.040 (0.946–1.144) | 0.4165 |
| No (n = 464) | 1.00 | ||
| Seasonal variation of sunlight exposure | HSE (n = 278) | 1.138 (1.052–1.231) | 0.0013 |
| LSE (n = 330) | 1.00 | ||
| Gender | Female (n = 397) | 0.998 (0.902–1.103) | 0.9648 |
| Male (n = 211) | 1.00 | ||
| Age (mean = 48.3, range of 10–91) | 0.995 (0.992–0.997) | 4.19 × 10−5 |
| Seasonal variation of sunlight exposure | CD status | N | Mean (95% CI) | p-values for multiple comparison | HSE-Control | LSE-CD | LSE-Control |
|---|---|---|---|---|---|---|---|
| HSE | CD | 123 | 53.9 (49.7–58.5) | HSE-CD | 0.0253 | 0.0079 | 0.3507 |
| HSE | Control | 155 | 61.0 (56.8–65.6) | HSE-Control | 1.54 × 10−7 | 0.1695 | |
| LSE | CD | 179 | 46.7 (43.6–49.9) | LSE-CD | 1.31 × 10−4 | ||
| LSE | Control | 151 | 56.8 (52.8–61.1) | LSE-Control |
3.2. Gene-CD Interaction on Serum Vitamin D Levels
| Gene | SNP effect | SNP × CD Interaction effect | |||
|---|---|---|---|---|---|
| SNP | Tested | Estimate | p | ||
| Allele | (95% CI) | ||||
| VDR | rs7975232 | A | CD | 0.981 (0.904–1.066) | 0.6535 |
| Control | 1.101 (1.008–1.202) | 0.0328 | |||
| VDR | rs731236 | A | CD | 0.893 (0.825–0.966) | 0.0050 |
| Control | 1.058 (0.972–1.152) | 0.1883 | |||
| SCUBE3 | rs732594 | A | CD | 0.917 (0.847–0.992) | 0.0316 |
| Control | 1.111 (1.007–1.226) | 0.0363 | |||
| PHF-11 | rs2980 | T | CD | 1.023 (0.942–1.112) | 0.5842 |
| Control | 1.104 (1.012–1.205) | 0.0265 | |||
| PHF-11 | rs2981 | A | CD | 1.024 (0.941–1.113) | 0.5817 |
| Control | 1.107 (1.014–1.209) | 0.0229 | |||
| PPP6R3 | rs7109294 | C | CD | 1.023 (0.922–1.136) | 0.6640 |
| Control | 1.102 (0.976–1.243) | 0.1152 | |||
4. Discussion
4.1. Vitamin D Levels: Influence on Susceptibility to Crohn’s Disease
4.2. Age, Gender and Smoking Habits
4.3. Single Nucleotide Polymorphisms Influencing Crohn’s Disease and Vitamin D Levels
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Gearry, R.B.; Richardson, A.; Frampton, C.M.; Collett, J.A.; Burt, M.J.; Chapman, B.A.; Barclay, M.L. High incidence of Crohn’s disease in Canterbury, New Zealand: Results of an epidemiologic study. Inflamm. Bowel Dis. 2006, 12, 936–943. [Google Scholar] [CrossRef]
- Carbonnel, F.; Jantchou, P.; Monnet, E.; Cosnes, J. Environmental risk factors in Crohn’s disease and ulcerative colitis: An update. Gastroenterol. Clin. Biol. 2009, 33, S145–S157. [Google Scholar] [CrossRef]
- Cho, J.H. The genetics and immunopathogenesis of inflammatory bowel disease. Nat. Rev. Immunol. 2008, 8, 458–466. [Google Scholar] [CrossRef]
- Neuman, M.G.; Nanau, R.M. Inflammatory bowel disease: Role of diet, microbiota, life style. Transl. Res. 2012, 160, 29–44. [Google Scholar] [CrossRef]
- Gearry, R.B.; Richardson, A.K.; Frampton, C.M.; Dodgshun, A.J.; Barclay, M.L. Population-based cases control study of inflammatory bowel disease risk factors. J. Gastroenterol. Hepatol. 2010, 25, 325–333. [Google Scholar] [CrossRef]
- Shanahan, F. Crohn’s disease. Lancet 2002, 359, 62–69. [Google Scholar] [CrossRef]
- Rook, G.A. The changing microbial environment and chronic inflammatory disorders. Allergy Asthma Clin. Immunol. 2008, 4, 117–124. [Google Scholar] [CrossRef]
- Cantorna, M.T. Vitamin D and its role in immunology: Multiple sclerosis, and inflammatory bowel disease. Prog. Biophys. Mol. Biol. 2006, 92, 60–64. [Google Scholar] [CrossRef]
- Han, D.Y.; Fraser, A.G.; Dryland, P.; Ferguson, L.R. Environmental factors in the development of chronic inflammation: A case–control study on risk factors for Crohn’s disease within New Zealand. Mutat. Res. Fundam. Mol. Mech. Mutagen. 2010, 690, 116–122. [Google Scholar] [CrossRef]
- Munns, C.; Zacharin, M.R.; Rodda, C.P.; Batch, J.A.; Morley, R.; Cranswick, N.E.; Craig, M.E.; Cutfield, W.S.; Hofman, P.L.; Taylor, B.J.; et al. Prevention and treatment of infant and childhood vitamin D deficiency in Australia and New Zealand: A consensus statement. Med. J. Aust. 2006, 185, 268–272. [Google Scholar]
- Macdonald, H.M. Contributions of sunlight and diet to vitamin D status. Calcif. Tissue Int. 2013, 92, 163–176. [Google Scholar] [CrossRef]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M. Guidelines for preventing and treating vitamin D deficiency and insufficiency revisited. J. Clin. Endocrinol. Metab. 2012, 97, 1153–1158. [Google Scholar] [CrossRef]
- Bentley, R.W.; Keown, D.; Merriman, T.R.; Raj Krishnan, M.; Gearry, R.B.; Barclay, M.L.; Roberts, R.L.; Day, A.S. Vitamin D receptor gene polymorphism associated with inflammatory bowel disease in New Zealand males. Aliment. Pharmacol. Ther. 2011, 33, 855–856. [Google Scholar] [CrossRef]
- Fedirko, V.; Bostick, R.M.; Goodman, M.; Flanders, W.D.; Gross, M.D. Blood 25-hydroxyvitamin D3 concentrations and incident sporadic colorectal adenoma risk: A pooled case-control study. Am. J. Epidemiol. 2010, 172, 489–500. [Google Scholar] [CrossRef]
- Holick, M.F.; Chen, T.C. Vitamin D deficiency: A worldwide problem with health consequences. Am. J. Clin. Nutr. 2008, 87, 1080S–1086S. [Google Scholar]
- Vimaleswaran, K.S.; Berry, D.J.; Lu, C.; Tikkanen, E.; Pilz, S.; Hiraki, L.T.; Cooper, J.D.; Dastani, Z.; Li, R.; Houston, D.K.; et al. The genetic investigation of anthropometric traits, c. causal relationship between obesity and Vitamin D status: Bi-directional mendelian randomization analysis of multiple cohorts. PLoS Med. 2013, 10, e1001383. [Google Scholar] [CrossRef]
- Brennan, E.P.; Ehrich, M.; O’Donovan, H.; Brazil, D.P.; Crean, J.K.; Murphy, M.; Sadlier, D.M.; Martin, F.; Godson, C.; van den Boom, D.; et al. DNA methylation profiling in cell models of diabetic nephropathy. Epigenetics 2010, 5, 396–401. [Google Scholar] [CrossRef]
- Morris, M.R.; Ricketts, C.J.; Gentle, D.; McRonald, F.; Carli, N.; Khalili, H.; Brown, M.; Kishida, T.; Yao, M.; Banks, R.E.; et al. Genome-wide methylation analysis identifies epigenetically inactivated candidate tumour suppressor genes in renal cell carcinoma. Oncogene 2011, 30, 1390–1401. [Google Scholar] [CrossRef]
- Pereira, S.R.; Vasconcelos, V.M.; Antunes, A. The phosphoprotein phosphatase family of Ser/Thr phosphatases as principal targets of naturally occurring toxins. Crit. Rev. Toxicol. 2011, 41, 83–110. [Google Scholar]
- Rahman, N.; Stewart, G.; Jones, G. A role for the atopy-associated gene PHF11 in T-cell activation and viability. Immunol. Cell Biol. 2010, 88, 817–824. [Google Scholar] [CrossRef]
- Holt, R.J.; Zhang, Y.; Binia, A.; Dixon, A.L.; Vandiedonck, C.; Cookson, W.O.; Knight, J.C.; Moffatt, M.F. Allele-specific transcription of the asthma-associated PHD finger protein 11 gene (PHF11) modulated by octamer-binding transcription factor 1 (Oct-1). J. Allergy Clin. Immunol. 2011, 127, 1054–1062. [Google Scholar] [CrossRef]
- Jones, G.; Stewart, G. Association of PHF11 polymorphisms with asthma and allergy. Thorax 2010, 65, 659–660. [Google Scholar] [CrossRef]
- Sutherland, E.R.; Goleva, E.; Jackson, L.P.; Stevens, A.D.; Leung, D.Y. Vitamin D levels, lung function, and steroid response in adult asthma. Am. J. Respir. Crit. Care Med. 2010, 181, 699–704. [Google Scholar] [CrossRef]
- Brehm, J.M.; Celedon, J.C.; Soto-Quiros, M.E.; Avila, L.; Hunninghake, G.M.; Forno, E.; Laskey, D.; Sylvia, J.S.; Hollis, B.W.; Weiss, S.T.; et al. Serum vitamin D levels and markers of severity of childhood asthma in Costa Rica. Am. J. Respir. Crit. Care Med. 2009, 179, 765–771. [Google Scholar] [CrossRef]
- Gentschew, L.; Ferguson, L.R. Role of nutrition and microbiota in susceptibility to inflammatory bowel diseases. Mol. Nutr. Food Res. 2012, 56, 524–535. [Google Scholar] [CrossRef]
- Jostins, L.; Ripke, S.; Weersma, R.K.; Duerr, R.H.; McGovern, D.P.; Hui, K.Y.; Lee, J.C.; Schumm, L.P.; Sharma, Y.; Anderson, C.A.; et al. Host-microbe interactions have shaped the genetic architecture of inflammatory bowel disease. Nature 2012, 491, 119–124. [Google Scholar] [CrossRef]
- R Foundation for Statistical Computing. R: A Language and Environment for Statistical Computing. Available online: http://www.R-project.org (accessed on 3 April 2013).
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef]
- Cantorna, M. Vitamin D and autoimmunity: Is Vitamin D status an environmental factor affecting autoimmune disease prevalence? Proc. Soc. Exp. Biol. Med. 2000, 223, 230–233. [Google Scholar] [CrossRef]
- Fu, Y.T.; Chatur, N.; Cheong-Lee, C.; Salh, B. Hypovitaminosis D in adults with inflammatory bowel disease: Potential role of ethnicity. Dig. Dis. Sci. 2012, 57, 2144–2148. [Google Scholar] [CrossRef]
- Kamen, D.L.; Tangpricha, V. Vitamin D and molecular actions on the immune system: Modulation of innate and autoimmunity. J. Mol. Med. (Berl.) 2010, 88, 441–450. [Google Scholar] [CrossRef]
- Andreassen, H.; Rix, M.; Brot, C.; Eskildsen, P. Regulators of calcium homeostasis and bone mineral density in patients with Crohn’s disease. Scand. J. Gastroenterol. 1998, 33, 1087–1093. [Google Scholar] [CrossRef]
- Cantorna, M.T. Vitamin D, multiple sclerosis and inflammatory bowel disease. Arch. Biochem. Biophys. 2012, 523, 103–106. [Google Scholar] [CrossRef]
- Bruce, D.; Cantorna, M.T. Intrinsic requirement for the Vitamin D receptor in the development of CD8αα-expressing T cells. J. Immunol. 2011, 186, 2819–2825. [Google Scholar] [CrossRef]
- Kobayashi, T.; Okamoto, S.; Hisamatsu, T.; Kamada, N.; Chinen, H.; Saito, R.; Kitazume, M.T.; Nakazawa, A.; Sugita, A.; Koganei, K.; et al. IL23 differentially regulates the Th1/Th17 balance in ulcerative colitis and Crohn’s disease. Gut 2008, 57, 1682–1689. [Google Scholar] [CrossRef]
- Sakuraba, A.; Sato, T.; Kamada, N.; Kitazume, M.; Sugita, A.; Hibi, T. Th1/Th17 immune response is induced by mesenteric lymph node dendritic cells in Crohn’s disease. Gastroenterology 2009, 137, 1736–1745. [Google Scholar] [CrossRef]
- Cantorna, M.T. Mechanisms underlying the effect of vitamin D on the immune system. Proc. Nutr. Soc. 2010, 69, 286–289. [Google Scholar] [CrossRef]
- Baumgart, D.C.; Bernstein, C.N.; Abbas, Z.; Colombel, J.F.; Day, A.S.; D’Haens, G.; Dotan, I.; Goh, K.L.; Hibi, T.; Kozarek, R.A.; et al. IBD Around the world: Comparing the epidemiology, diagnosis, and treatment: Proceedings of the World Digestive Health Day 2010—Inflammatory bowel disease task force meeting. Inflamm. Bowel Dis. 2011, 17, 639–644. [Google Scholar] [CrossRef]
- Gentschew, L.; Bishop, K.S.; Han, D.Y.; Morgan, A.R.; Fraser, A.G.; Lam, W.J.; Karunasinghe, N.; Campbell, B.; Ferguson, L.R. Selenium, selenoprotein genes and Crohn’s disease in a case-control population from Auckland, New Zealand. Nutrients 2012, 4, 1247–1259. [Google Scholar] [CrossRef]
- Janssens, W.; Bouillon, R.; Claes, B.; Carremans, C.; Lehouck, A.; Buysschaert, I.; Coolen, J.; Mathieu, C.; Decramer, M.; Lambrechts, D. Vitamin D deficiency is highly prevalent in COPD and correlates with variants in the vitamin D-binding gene. Thorax 2010, 65, 215–220. [Google Scholar] [CrossRef]
- McCullough, M.L.; Weinstein, S.J.; Freedman, D.M.; Helzlsouer, K.; Flanders, W.D.; Koenig, K.; Kolonel, L.; Laden, F.; Le Marchand, L.; Purdue, M.; et al. Correlates of circulating 25-hydroxyvitamin D: Cohort consortium Vitamin D pooling project of rarer cancers. Am. J. Epidemiol. 2010, 172, 21–35. [Google Scholar] [CrossRef]
- Houghton, L.A.; Szymlek-Gay, E.A.; Gray, A.R.; Ferguson, E.L.; Deng, X.; Heath, A.-L.M. Predictors of vitamin D status and its association with parathyroid hormone in young New Zealand children. Am. J. Clin. Nutr. 2010, 92, 69–76. [Google Scholar] [CrossRef]
- Kostner, K.; Denzer, N.; Muller, C.S.L.; Klein, R.; Tilgen, W.; Reichrath, J. The relevance of Vitamin D Receptor (VDR) gene polymorphisms for cancer: A review of the literature. Anticancer Res. 2009, 29, 3511–3536. [Google Scholar]
- Vasilopoulos, Y.; Sarafidou, T.; Kotsa, K.; Papadimitriou, M.; Goutzelas, Y.; Stamatis, C.; Bagiatis, V.; Tsekmekidou, X.; Yovos, J.G.; Mamuris, Z. VDR TaqI is associated with obesity in the Greek population. Gene 2013, 512, 237–239. [Google Scholar] [CrossRef]
- Wu, Y.Y.; Peck, K.; Chang, Y.L.; Pan, S.H.; Cheng, Y.F.; Lin, J.C.; Yang, R.B.; Hong, T.M.; Yang, P.C. SCUBE3 is an endogenous TGF-β receptor ligand and regulates the epithelial-mesenchymal transition in lung cancer. Oncogene 2011, 30, 3682–3693. [Google Scholar] [CrossRef]
- Chou, C.-H.; Cheng, Y.-F.; Siow, T.; Kumar, A.; Peck, K.; Chang, C. SCUBE3 regulation of early lung cancer angiogenesis and metastatic progression. Clin. Exp. Metastasis 2013. [Google Scholar] [CrossRef]
- Walker, B.A.; Wardell, C.P.; Johnson, D.C.; Kaiser, M.F.; Begum, D.B.; Dahir, N.B.; Ross, F.M.; Davies, F.E.; Gonzalez, D.; Morgan, G.J. Characterization of IgH breakpoints in multiple myeloma indicates a subset of translocations appear to occur In pre-germinal center B cells. Blood 2013, 121, 3413–3419. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Carvalho, A.Y.O.M.; Bishop, K.S.; Han, D.Y.; Ellett, S.; Jesuthasan, A.; Lam, W.J.; Ferguson, L.R. The Role of Vitamin D Level and Related Single Nucleotide Polymorphisms in Crohn’s Disease. Nutrients 2013, 5, 3898-3909. https://doi.org/10.3390/nu5103898
Carvalho AYOM, Bishop KS, Han DY, Ellett S, Jesuthasan A, Lam WJ, Ferguson LR. The Role of Vitamin D Level and Related Single Nucleotide Polymorphisms in Crohn’s Disease. Nutrients. 2013; 5(10):3898-3909. https://doi.org/10.3390/nu5103898
Chicago/Turabian StyleCarvalho, Andre Y. O. M., Karen S. Bishop, Dug Yeo Han, Stephanie Ellett, Amalini Jesuthasan, Wen J. Lam, and Lynnette R. Ferguson. 2013. "The Role of Vitamin D Level and Related Single Nucleotide Polymorphisms in Crohn’s Disease" Nutrients 5, no. 10: 3898-3909. https://doi.org/10.3390/nu5103898
APA StyleCarvalho, A. Y. O. M., Bishop, K. S., Han, D. Y., Ellett, S., Jesuthasan, A., Lam, W. J., & Ferguson, L. R. (2013). The Role of Vitamin D Level and Related Single Nucleotide Polymorphisms in Crohn’s Disease. Nutrients, 5(10), 3898-3909. https://doi.org/10.3390/nu5103898







