Anti-Inflammatory and Anti-Apoptotic Effects of Acer Palmatum Thumb. Extract, KIOM-2015EW, in a Hyperosmolar-Stress-Induced In Vitro Dry Eye Model
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
2.2. Preparation of Herbal Extract, KIOM-2015EW
2.3. HCEC Culture
2.4. Cell-Viability Assay
2.5. ELISA
2.6. RNA Isolation and Reverse Transcription–Polymerase Chain Reaction (RT-PCR) Analysis
2.7. Cellular Protein Extraction and Western Blotting
2.8. Immunofluorescence Staining
2.9. Flow Cytometry
2.10. Cytoplasmic and Nuclear Protein Extraction
2.11. High Performance Liquid Chromatography (HPLC) Analysis
2.12. Statistical Analysis
3. Results
3.1. Effects of Hyperosmotic Medium and KIOM-2015EW on HCECs
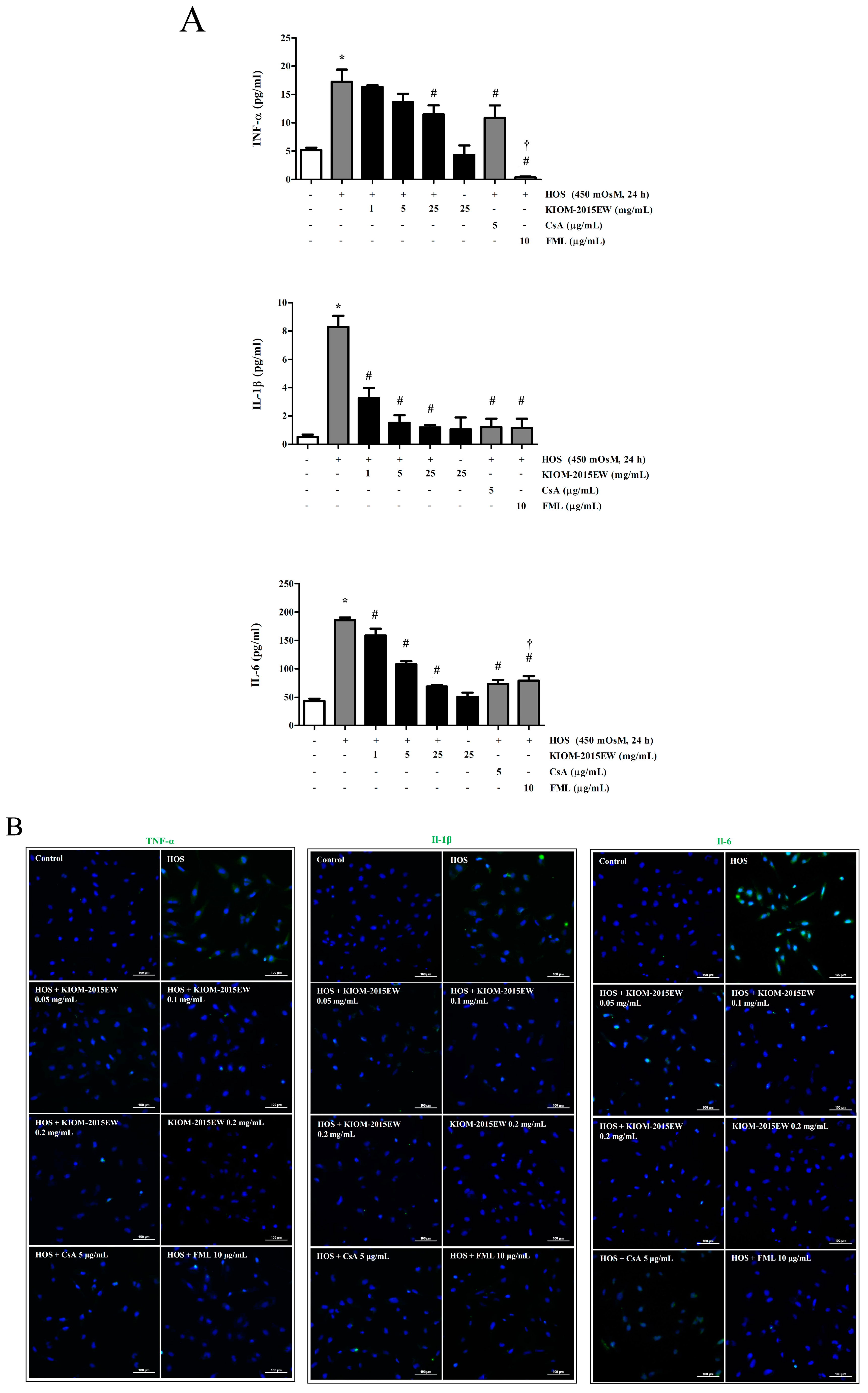
3.2. KIOM-2015EW Significantly Decreased Pro-Inflammatory Cytokine Expression in HOS-Exposed HCECs
3.3. KIOM-2015EW Blocked Apoptosis Due to HOS-Induced Cytotoxicity in HCECs
3.4. KIOM-2015EW Downregulates HOS-Mediated p38, ERK, and JNK Activation in HCECs
3.5. MAPK Inhibition by KIOM-2015EW Blocks HOS-Induced Apoptosis
3.6. KIOM-2015EW Suppresses HOS-Induced NF-κB Activation through p65 Nuclear Translocation Blockage in HCECs
3.7. Identification of the Main Components in KIOM-2015EW Using HPLC
3.8. Orientin, Isoorientin and Vitexin Reduced Level of HOS-Induced Pro-Inflammatory Cytokines
4. Discussion
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- The International Dry Eye Workshop. The definition and classification of dry eye disease: Report of the definition and classification subcommittee of the International Dry Eye Workshop (2007). Ocul. Surf. 2007, 5, 75–92. [Google Scholar]
- Brignole, F.; Pisella, P.J.; Goldschild, M.; De Saint Jean, M.; Goguel, A.; Baudouin, C. Flow cytometric analysis of inflammatory markers in conjunctival epithelial cells of patients with dry eyes. Investig. Ophthalmol. Vis. Sci. 2000, 41, 1356–1363. [Google Scholar]
- Gao, J.; Morgan, G.; Tieu, D.; Schwalb, T.A.; Luo, J.Y.; Wheeler, L.A.; Stern, M.E. ICAM-1 expression predisposes ocular tissues to immune-based inflammation in dry eye patients and Sjögrens syndrome-like MRL/lpr mice. Exp. Eye Res. 2004, 78, 823–835. [Google Scholar] [CrossRef] [PubMed]
- Li, D.Q.; Chen, Z.; Song, X.J.; Luo, L.; Pflugfelder, S.C. Stimulation of matrix metalloproteinases by hyperosmolarity via a JNK pathway in human corneal epithelial cells. Investig. Ophthalmol. Vis. Sci. 2004, 45, 4302–4311. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.E.; Murphy, P.J. Changes in the tear film and ocular surface from dry eye syndrome. Prog. Retin. Eye Res. 2004, 23, 449–474. [Google Scholar] [CrossRef] [PubMed]
- Zoukhri, D. Effect of inflammation on lacrimal gland function. Exp. Eye Res. 2006, 82, 885–898. [Google Scholar] [CrossRef] [PubMed]
- Zoukhri, D.; Macari, E.; Kublin, C.L. A single injection of interleukin-1 induces reversible aqueous-tear deficiency, lacrimal gland inflammation, and acinar and ductal cell proliferation. Exp. Eye Res. 2007, 84, 894–904. [Google Scholar] [CrossRef] [PubMed]
- Solomon, A.; Dursun, D.; Liu, Z.; Xie, Y.; Macri, A.; Pflugfelder, S.C. Pro- and anti-inflammatory forms of interleukin-1 in the tear fluid and conjunctiva of patients with dry-eye disease. Investig. Ophthalmol. Vis. Sci. 2001, 42, 2283–2292. [Google Scholar]
- Li, S.; Nikulina, K.; DeVoss, J.; Wu, A.J.; Strauss, E.C.; Anderson, M.S.; McNamara, N.A. Small proline-rich protein 1b (sprr1b) is a biomarker for squamous metaplasia in dry eye disease. Investig. Ophthalmol. Vis. Sci. 2008, 49, 34–41. [Google Scholar] [CrossRef] [PubMed]
- De Paiva, C.S.; Villarreal, A.L.; Corrales, R.M.; Rahman, H.T.; Chang, V.Y.; Farley, W.J.; Stern, M.E.; Niederkorn, J.Y.; Li, D.Q.; Pflugfelder, S.C. Dry eye-induced conjunctival epithelial squamous metaplasia is modulated by interferon-γ. Investig. Ophthalmol. Vis. Sci. 2007, 48, 2553–2560. [Google Scholar] [CrossRef] [PubMed]
- De Paiva, C.S.; Chotikavanich, S.; Pangelinan, S.B.; Pitcher, J.D., 3rd; Fang, B.; Zheng, X.; Ma, P.; Farley, W.J.; Siemasko, K.F.; Niederkorn, J.Y.; et al. IL-17 disrupts corneal barrier following desiccating stress. Mucosal Immunol. 2009, 2, 243–253. [Google Scholar] [CrossRef] [PubMed]
- Luo, L.; Li, D.Q.; Pflugfelder, S.C. Hyperosmolarity-induced apoptosis in human corneal epithelial cells is mediated by cytochrome c and MAPK pathways. Cornea 2007, 26, 452–460. [Google Scholar] [CrossRef] [PubMed]
- Li, D.Q.; Luo, L.; Chen, Z.; Kim, H.S.; Song, X.J.; Pflugfelder, S.C. JNK and ERK MAP kinases mediate induction of IL-1beta, TNF-alpha and IL-8 following hyperosmolar stress in human limbal epithelial cells. Exp. Eye Res. 2006, 82, 588–596. [Google Scholar] [CrossRef] [PubMed]
- Corrales, R.M.; Luo, L.; Chang, E.Y.; Pflugfelder, S.C. Effects of osmoprotectants on hyperosmolar stress in cultured human corneal epithelial cells. Cornea 2008, 27, 574–579. [Google Scholar] [CrossRef] [PubMed]
- Foulks, G.N. Dews report: A mission completed. Ocular Surf. 2007, 5, 65–66. [Google Scholar] [CrossRef]
- Baudouin, C.; Aragona, P.; Messmer, E.M.; Tomlinson, A.; Calonge, M.; Boboridis, K.G.; Akova, Y.A.; Geerling, G.; Labetoulle, M.; Rolando, M. Role of hyperosmolarity in the pathogenesis and management of dry eye disease: Proceedings of the ocean group meeting. Ocul. Surf. 2013, 11, 246–258. [Google Scholar] [CrossRef] [PubMed]
- Lemp, M.A.; Bron, A.J.; Baudouin, C.; Benitez Del Castillo, J.M.; Geffen, D.; Tauber, J.; Foulks, G.N.; Pepose, J.S.; Sullivan, B.D. Tear osmolarity in the diagnosis and management of dry eye disease. Am. J. Ophthalmol. 2011, 151, 792–798.e1. [Google Scholar] [CrossRef] [PubMed]
- Julio, G.; Lluch, S.; Pujol, P.; Merindano, M.D. Effects of tear hyperosmolarity on conjunctival cells in mild to moderate dry eye. Ophthalmic Physiol. Opt. 2012, 32, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Luo, L.; Li, D.Q.; Doshi, A.; Farley, W.; Corrales, R.M.; Pflugfelder, S.C. Experimental dry eye stimulates production of inflammatory cytokines and MMP-9 and activates MAPK signaling pathways on the ocular surface. Investig. Ophthalmol. Vis. Sci. 2004, 45, 4293–4301. [Google Scholar] [CrossRef] [PubMed]
- Luo, L.; Li, D.Q.; Corrales, R.M.; Pflugfelder, S.C. Hyperosmolar saline is a proinflammatory stress on the mouse ocular surface. Eye Contact Lens 2005, 31, 186–193. [Google Scholar] [CrossRef] [PubMed]
- Corrales, R.M.; Stern, M.E.; De Paiva, C.S.; Welch, J.; Li, D.Q.; Pflugfelder, S.C. Desiccating stress stimulates expression of matrix metalloproteinases by the corneal epithelium. Investig. Ophthalmol. Vis. Sci. 2006, 47, 3293–3302. [Google Scholar] [CrossRef] [PubMed]
- Pflugfelder, S.C.; de Paiva, C.S.; Li, D.Q.; Stern, M.E. Epithelial-immune cell interaction in dry eye. Cornea 2008, 27 (Suppl. 1), S9–S11. [Google Scholar] [CrossRef]
- Gao, J.; Schwalb, T.A.; Addeo, J.V.; Ghosn, C.R.; Stern, M.E. The role of apoptosis in the pathogenesis of canine keratoconjunctivitis sicca: The effect of topical Cyclosporin A therapy. Cornea 1998, 17, 654–663. [Google Scholar] [CrossRef] [PubMed]
- Brignole, F.; De Saint-Jean, M.; Goldschild, M.; Becquet, F.; Goguel, A.; Baudouin, C. Expression of Fas-Fas ligand antigens and apoptotic marker APO2.7 by the human conjunctival epithelium. Positive correlation with class II HLA DR expression in inflammatory ocular surface disorders. Exp. Eye Res. 1998, 67, 687–697. [Google Scholar] [CrossRef] [PubMed]
- Yeh, S.; Song, X.J.; Farley, W.; Li, D.Q.; Stern, M.E.; Pflugfelder, S.C. Apoptosis of ocular surface cells in experimentally induced dry eye. Investig. Ophthalmol. Vis. Sci. 2003, 44, 124–129. [Google Scholar] [CrossRef]
- Strong, B.; Farley, W.; Stern, M.E.; Pflugfelder, S.C. Topical cyclosporine inhibits conjunctival epithelial apoptosis in experimental murine keratoconjunctivitis sicca. Cornea 2005, 24, 80–85. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Dai, W.; Lu, L. Hyperosmotic stress-induced corneal epithelial cell death through activation of Polo-like kinase 3 and c-Jun. Investig. Ophthalmol. Vis. Sci. 2011, 52, 3200–3206. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.; Tunnacliffe, A. Response of human cells to desiccation: Comparison with hyperosmotic stress response. J. Physiol. 2004, 558, 181–191. [Google Scholar] [CrossRef] [PubMed]
- Lang, F.; Busch, G.L.; Ritter, M.; Volkl, H.; Waldegger, S.; Gulbins, E.; Haussinger, D. Functional significance of cell volume regulatory mechanisms. Physiol. Rev. 1998, 78, 247–306. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.P.; Choi, D.W. Ions, cell volume, and apoptosis. Proc. Natl. Acad. Sci. USA 2000, 97, 9360–9362. [Google Scholar] [CrossRef] [PubMed]
- Deutsch, C.; Slater, L.; Goldstein, P. Volume regulation of human peripheral blood lymphocytes and stimulated proliferation of volume-adapted cells. Biochim. Biophys. Acta 1982, 721, 262–267. [Google Scholar] [CrossRef]
- Burg, M.B.; Ferraris, J.D.; Dmitrieva, N.I. Cellular response to hyperosmotic stresses. Physiol. Rev. 2007, 87, 1441–1474. [Google Scholar] [CrossRef] [PubMed]
- Sheikh-Hamad, D.; Gustin, M.C. MAP kinases and the adaptive response to hypertonicity: Functional preservation from yeast to mammals. Am. J. Physiol. Renal Physiol. 2004, 287, F1102–F1110. [Google Scholar] [CrossRef] [PubMed]
- Westfall, P.J.; Patterson, J.C.; Chen, R.E.; Thorner, J. Stress resistance and signal fidelity independent of nuclear MAPK function. Proc. Natl. Acad. Sci. USA 2008, 105, 12212–12217. [Google Scholar] [CrossRef] [PubMed]
- Lunn, J.A.; Jacamo, R.; Rozengurt, E. Preferential phosphorylation of focal adhesion kinase tyrosine 861 is critical for mediating an anti-apoptotic response to hyperosmotic stress. J. Biol. Chem. 2007, 282, 10370–10379. [Google Scholar] [CrossRef] [PubMed]
- Galcheva-Gargova, Z.; Derijard, B.; Wu, I.H.; Davis, R.J. An osmosensing signal transduction pathway in mammalian cells. Science 1994, 265, 806–808. [Google Scholar] [CrossRef] [PubMed]
- Kyriakis, J.M.; Banerjee, P.; Nikolakaki, E.; Dai, T.; Rubie, E.A.; Ahmad, M.F.; Avruch, J.; Woodgett, J.R. The stress-activated protein kinase subfamily of c-Jun kinases. Nature 1994, 369, 156–160. [Google Scholar] [CrossRef] [PubMed]
- Rosette, C.; Karin, M. Ultraviolet light and osmotic stress: Activation of the JNK cascade through multiple growth factor and cytokine receptors. Science 1996, 274, 1194–1197. [Google Scholar] [CrossRef] [PubMed]
- Barchowsky, A.; Frleta, D.; Vincenti, M.P. Integration of the NF-kappaB and mitogen-activated protein kinase/AP-1 pathways at the collagenase-1 promoter: Divergence of IL-1 and TNF-dependent signal transduction in rabbit primary synovial fibroblasts. Cytokine 2000, 12, 1469–1479. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Barrett, T.; Whitmarsh, A.J.; Cavanagh, J.; Sluss, H.K.; Derijard, B.; Davis, R.J. Selective interaction of JNK protein kinase isoforms with transcription factors. EMBO J. 1996, 15, 2760–2770. [Google Scholar] [PubMed]
- The International Dry Eye Workshop. Management and therapy of dry eye disease: Report of the management and therapy subcommittee of the International Dry Eye Workshop (2007). Ocul. Surf. 2007, 5, 163–178. [Google Scholar]
- Carnahan, M.C.; Goldstein, D.A. Ocular complications of topical, peri-ocular, and systemic corticosteroids. Curr. Opin. Ophthalmol. 2000, 11, 478–483. [Google Scholar] [CrossRef] [PubMed]
- Sall, K.; Stevenson, O.D.; Mundorf, T.K.; Reis, B.L. Two multicenter, randomized studies of the efficacy and safety of cyclosporine ophthalmic emulsion in moderate to severe dry eye disease. CsA Phase 3 Study Group. Ophthalmology 2000, 107, 631–639. [Google Scholar] [PubMed]
- Bi, W.; Gao, Y.; Shen, J.; He, C.; Liu, H.; Peng, Y.; Zhang, C.; Xiao, P. Traditional uses, phytochemistry, and pharmacology of the genus Acer (maple): A review. J. Ethnopharmacol. 2016, 189, 31–60. [Google Scholar] [CrossRef] [PubMed]
- Park, H.R.; Lee, H.; Park, H.; Jeon, J.W.; Cho, W.K.; Ma, J.Y. Neuroprotective effects of Liriope platyphylla extract against hydrogen peroxide-induced cytotoxicity in human neuroblastoma SH-SY5Y cells. BMC Complement. Altern. Med. 2015, 15, 171. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Tong, L.; Li, Z.; Yoon, K.C.; Qi, H.; Farley, W.; Li, D.Q.; Pflugfelder, S.C. Hyperosmolarity-induced cornification of human corneal epithelial cells is regulated by JNK MAPK. Investig. Ophthalmol. Vis. Sci. 2008, 49, 539–549. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Shi, X.; Chen, X.; Wu, H.; Zhang, H.; Xie, J.; Yang, X.; Gou, Z.; Ye, J. 17-β-estradiol inhibits hyperosmolarity-induced proinflammatory cytokine elevation via the p38 MAPK pathway in human corneal epithelial cells. Mol. Vis. 2012, 18, 1115–1122. [Google Scholar] [PubMed]
- Lawrence, T. The nuclear factor NF-kappaB pathway in inflammation. Cold Spring Harb. Perspect. Biol. 2009, 1, a001651. [Google Scholar] [CrossRef] [PubMed]
- Eisner, V.; Quiroga, C.; Criollo, A.; Eltit, J.M.; Chiong, M.; Parra, V.; Hidalgo, K.; Toro, B.; Diaz-Araya, G.; Lavandero, S. Hyperosmotic stress activates p65/RelB NFkappaB in cultured cardiomyocytes with dichotomic actions on caspase activation and cell death. FEBS Lett. 2006, 580, 3469–3476. [Google Scholar] [CrossRef] [PubMed]
- Bron, A.J.; Yokoi, N.; Gafney, E.; Tiffany, J.M. Predicted phenotypes of dry eye: Proposed consequences of its natural history. Ocul. Surf. 2009, 7, 78–92. [Google Scholar] [CrossRef]
- Jones, D.T.; Monroy, D.; Ji, Z.; Atherton, S.S.; Pflugfelder, S.C. Sjogren’s syndrome: Cytokine and epstein-barr viral gene expression within the conjunctival epithelium. Investig. Ophthalmol. Vis. Sci. 1994, 35, 3493–3504. [Google Scholar]
- Yoon, K.C.; Jeong, I.Y.; Park, Y.G.; Yang, S.Y. Interleukin-6 and tumor necrosis factor-alpha levels in tears of patients with dry eye syndrome. Cornea 2007, 26, 431–437. [Google Scholar] [CrossRef] [PubMed]
- De Paiva, C.S.; Pflugfelder, S.C. Rationale for anti-inflammatory therapy in dry eye syndrome. Arq. Bras. Oftalmol. 2008, 71, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Kymionis, G.D.; Bouzoukis, D.I.; Diakonis, V.F.; Siganos, C. Treatment of chronic dry eye: Focus on cyclosporine. Clin. Ophthalmol. 2008, 2, 829–836. [Google Scholar] [CrossRef] [PubMed]
- McCabe, E.; Narayanan, S. Advancements in anti-inflammatory therapy for dry eye syndrome. Optometry 2009, 80, 555–566. [Google Scholar] [CrossRef] [PubMed]
- Cavet, M.E.; Harrington, K.L.; Vollmer, T.R.; Ward, K.W.; Zhang, J.Z. Anti-inflammatory and anti-oxidative effects of the green tea polyphenol epigallocatechin gallate in human corneal epithelial cells. Mol. Vis. 2011, 17, 533–542. [Google Scholar] [PubMed]
- Li, J.; Ruzhi, D.; Hua, X.; Zhang, L.; Lu, F.; Coursey, T.G.; Pflugfelder, S.C.; Li, D.Q. Blueberry component pterostilbene protects corneal epithelial cells from inflammation via anti-oxidative pathway. Sci. Rep. 2016, 6, 19408. [Google Scholar] [CrossRef] [PubMed]
- Pescosolido, N.; Giannotti, R.; Plateroti, A.M.; Pascarella, A.; Nebbioso, M. Curcumin: Therapeutical potential in ophthalmology. Planta Med. 2014, 80, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Abengozar-Vela, A.; Calonge, M.; Stern, M.E.; Gonzalez-Garcia, M.J.; Enriquez-De-Salamanca, A. Quercetin and resveratrol decrease the inflammatory and oxidative responses in human ocular surface epithelial cells. Investig. Ophthalmol. Vis. Sci. 2015, 56, 2709–2719. [Google Scholar] [CrossRef] [PubMed]
- Robinson, M.J.; Cobb, M.H. Mitogen-activated protein kinase pathways. Curr. Opin. Cell Biol. 1997, 9, 180–186. [Google Scholar] [CrossRef]
- Berra, A.; Berra, M. Hyperosmolarity induce nuclear translocation of NF-KB in human conjunctival epithelial cells. Investig. Ophthalmol. Vis. Sci. 2005, 46, 4402. [Google Scholar]
- De Bosscher, K.; Vanden Berghe, W.; Haegeman, G. The interplay between the glucocorticoid receptor and nuclear factor-kappaB or activator protein-1: Molecular mechanisms for gene repression. Endocr. Rev. 2003, 24, 488–522. [Google Scholar] [CrossRef] [PubMed]
- Seo, C.S.; Lee, M.Y.; Shin, I.S.; Lee, J.A.; Ha, H.; Shin, H.K. Spirodela polyrhiza (L.) Sch. ethanolic extract inhibits LPS-induced inflammation in RAW264.7 cells. Immunopharmacol. Immunotoxicol. 2012, 34, 794–802. [Google Scholar] [CrossRef] [PubMed]
- Nam, T.G.; Lim, T.G.; Lee, B.H.; Lim, S.; Kang, H.; Eom, S.H.; Yoo, M.; Jang, H.W.; Kim, D.O. Comparison of anti-inflammatory effects of flavonoid-rich common and tartary buckwheat sprout extracts in lipopolysaccharide-stimulated RAW264.7 and peritoneal macrophages. Oxid. Med. Cell. Longev. 2017, 2017, 9658030. [Google Scholar] [CrossRef] [PubMed]
- Gou, K.J.; Zeng, R.; Dong, Y.; Hu, Q.Q.; Hu, H.W.; Maffucci, K.G.; Dou, Q.L.; Yang, Q.B.; Qin, X.H.; Qu, Y. Anti-inflammatory and analgesic effects of Polygonum orientale L. Extracts Front. Pharmacol. 2017, 8, 562. [Google Scholar] [CrossRef] [PubMed]
- Yuan, L.; Wei, S.; Wang, J.; Liu, X. Isoorientin induces apoptosis and autophagy simultaneously by reactive oxygen species (ROS)-related p53, PI3K/Akt, JNK, and p38 signaling pathways in HepG2 cancer cells. J. Agric. Food Chem. 2014, 62, 5390–5400. [Google Scholar] [CrossRef] [PubMed]
- Kim, A.; Im, M.; Gu, M.J.; Ma, J.Y. Ethanol extract of Lophatheri herba exhibits anti-cancer activity in human cancer cells by suppression of metastatic and angiogenic potential. Sci. Rep. 2016, 6, 36277. [Google Scholar] [CrossRef] [PubMed]
- An, F.; Wang, S.; Tian, Q.; Zhu, D. Effects of orientin and vitexin from Trollius chinensis on the growth and apoptosis of esophageal cancer EC-109 cells. Oncol. Lett. 2015, 10, 2627–2633. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.H.; Liao, P.H.; Pan, Y.F.; Chen, S.L.; Chou, S.S.; Chou, M.Y. The novel p53-dependent metastatic and apoptotic pathway induced by vitexin in human oral cancer OC2 cells. Phytother. Res. 2013, 27, 1154–1161. [Google Scholar] [CrossRef] [PubMed]
- Flores, G.; Dastmalchi, K.; Dabo, A.J.; Whalen, K.; Pedraza-Penalosa, P.; Foronjy, R.F.; D’Armiento, J.M.; Kennelly, E.J. Antioxidants of therapeutic relevance in COPD from the neotropical blueberry anthopterus wardii. Food Chem. 2012, 131, 119–125. [Google Scholar] [CrossRef] [PubMed]







| Primers | ||||
|---|---|---|---|---|
| Gene Name | Sequence | Tm. (°C) | Cycles | |
| TNF-α | F | 5′-GCGGTGCTTGTTCCTCAG-3′ | 58 | 40 |
| R | 5′-CTGGCAGGGGCTCTTGAT-3′ | |||
| IL-1β | F | 5′-TCAGCACCTCTCAAGCAGAA-3′ | 58 | 30 |
| R | 5′-ATCAGAATGTGGGAGCGAATG-3′ | |||
| IL-6 | F | 5′-CAATCTGGATTCAATGAGGAGACTT-3′ | 58 | 30 |
| R | 5′-CCATTAACAACAACAATCTGAGGTG-3′ | |||
| GAPDH | F | 5′-GAAGGTGAAGGTCGGAGT-3′ | 56 | 35 |
| R | 5′-GAAGATGGTGATGGGATTTC-3′ | |||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, Y.-H.; Oh, T.W.; Park, E.; Yim, N.-H.; Park, K.I.; Cho, W.K.; Ma, J.Y. Anti-Inflammatory and Anti-Apoptotic Effects of Acer Palmatum Thumb. Extract, KIOM-2015EW, in a Hyperosmolar-Stress-Induced In Vitro Dry Eye Model. Nutrients 2018, 10, 282. https://doi.org/10.3390/nu10030282
Kim Y-H, Oh TW, Park E, Yim N-H, Park KI, Cho WK, Ma JY. Anti-Inflammatory and Anti-Apoptotic Effects of Acer Palmatum Thumb. Extract, KIOM-2015EW, in a Hyperosmolar-Stress-Induced In Vitro Dry Eye Model. Nutrients. 2018; 10(3):282. https://doi.org/10.3390/nu10030282
Chicago/Turabian StyleKim, Yeoun-Hee, Tae Woo Oh, Eunhee Park, Nam-Hui Yim, Kwang Il Park, Won Kyung Cho, and Jin Yeul Ma. 2018. "Anti-Inflammatory and Anti-Apoptotic Effects of Acer Palmatum Thumb. Extract, KIOM-2015EW, in a Hyperosmolar-Stress-Induced In Vitro Dry Eye Model" Nutrients 10, no. 3: 282. https://doi.org/10.3390/nu10030282