Live Coral Cover Index Testing and Application with Hyperspectral Airborne Image Data
Abstract
:1. Introduction
1.1. Spectral Mixing Challenges in Coral Reef Remote Sensing
1.2. Spectral Indices and Coral Reef Mapping Potential
2. Data and Methods
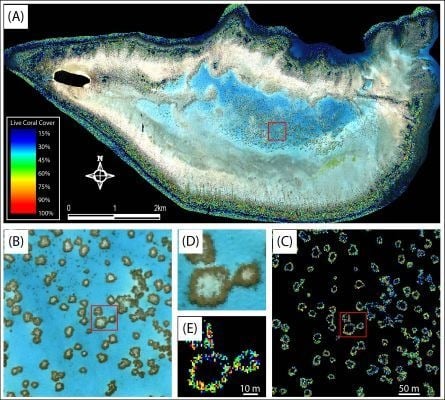
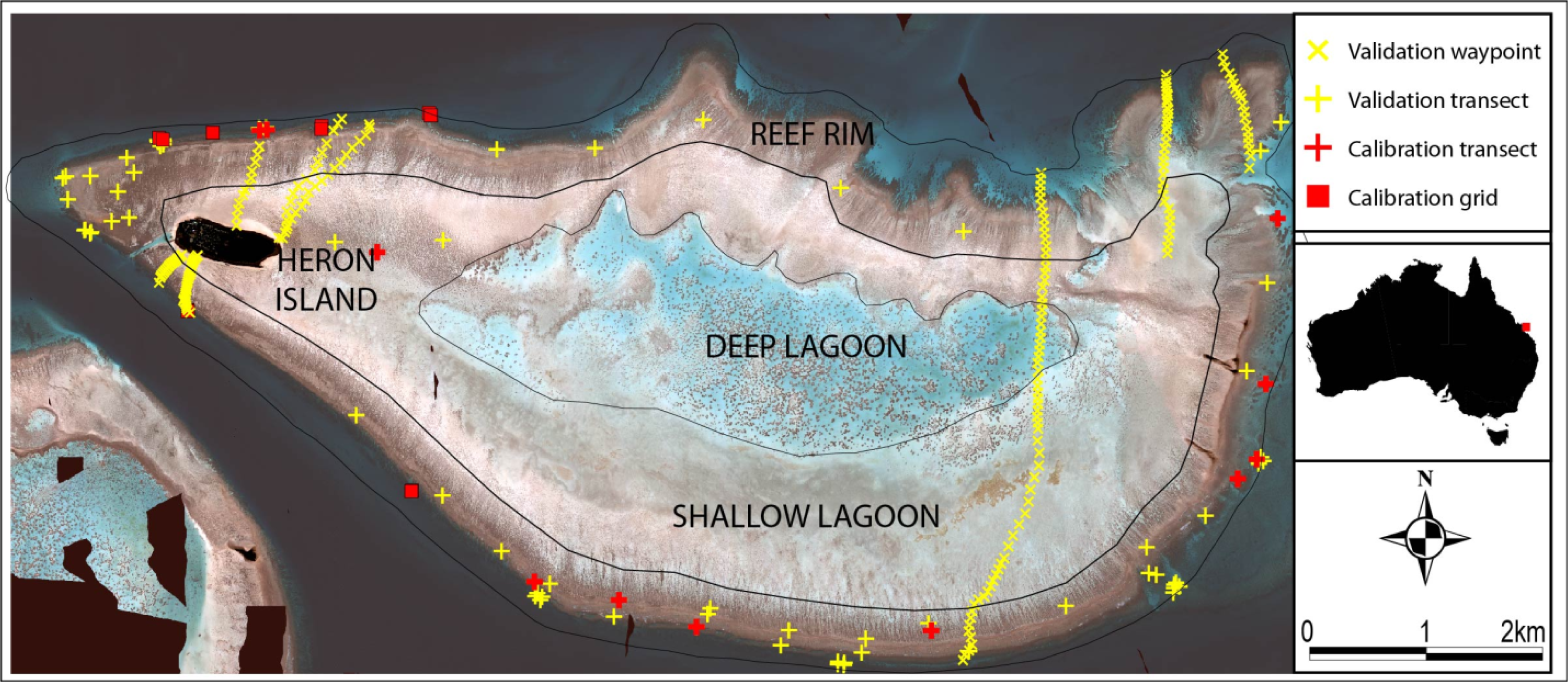
2.1. Study Area
2.2. Field Data Collection
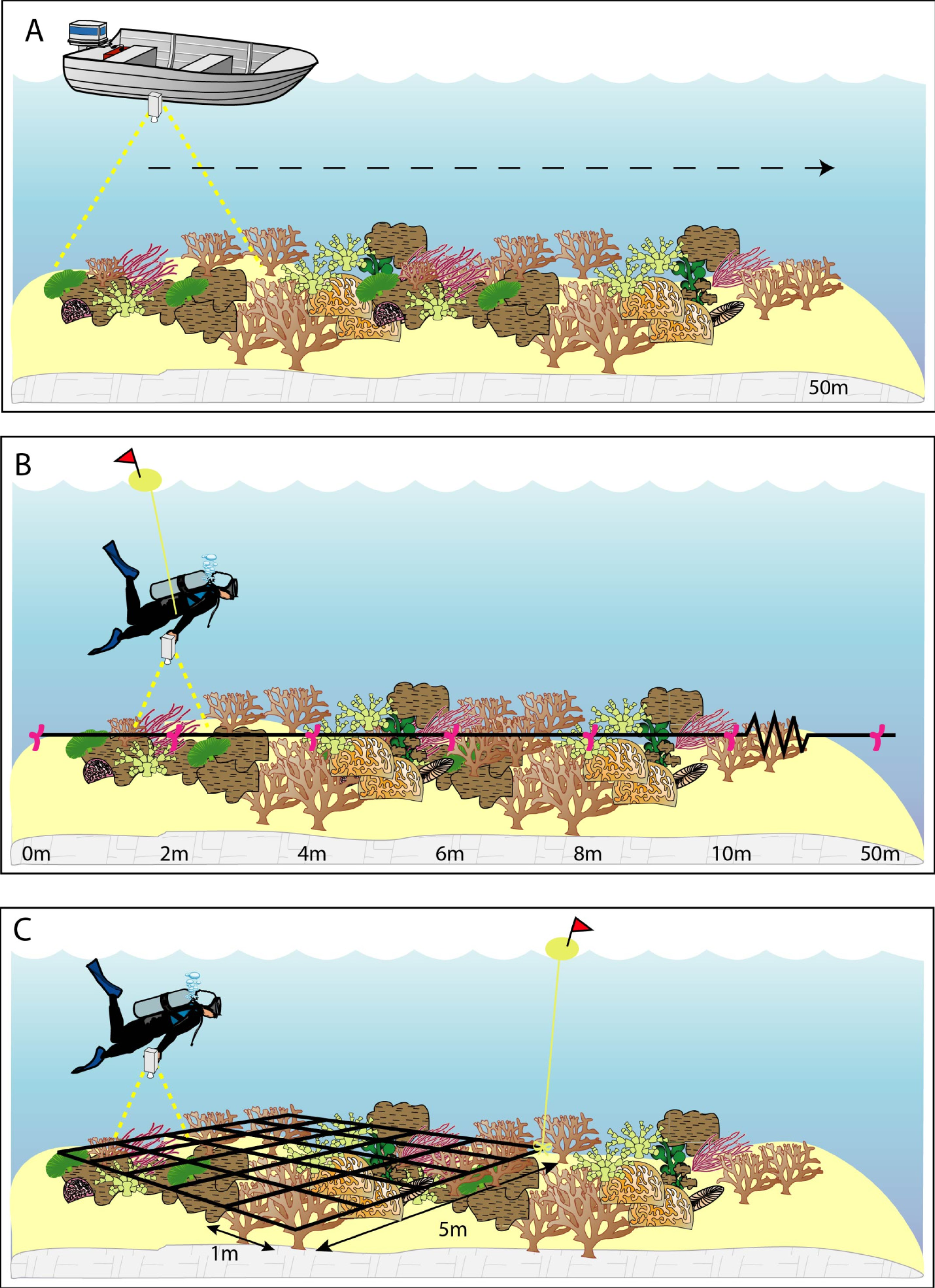
2.2.1. Long Photo Transects: 1–5 km
2.2.2. Short Photo Transects (50 m)
2.2.3. Detailed Grid Photo (5 m × 5 m)
2.3. Field Data Processing and Benthic Photo Analysis
2.4. Image Data Pre-Processing
2.5. Live Coral Cover Index calibration
2.6. Live Coral Cover Index Application
3. Results and Discussion
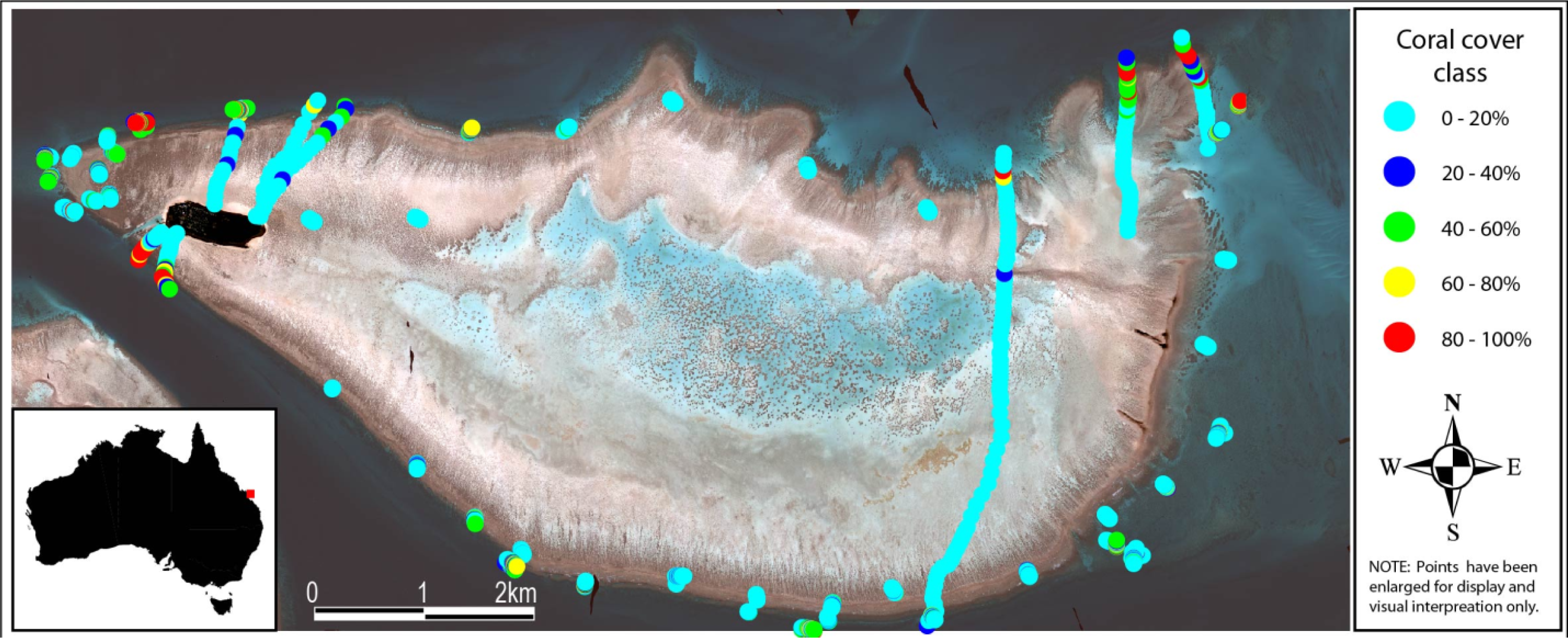
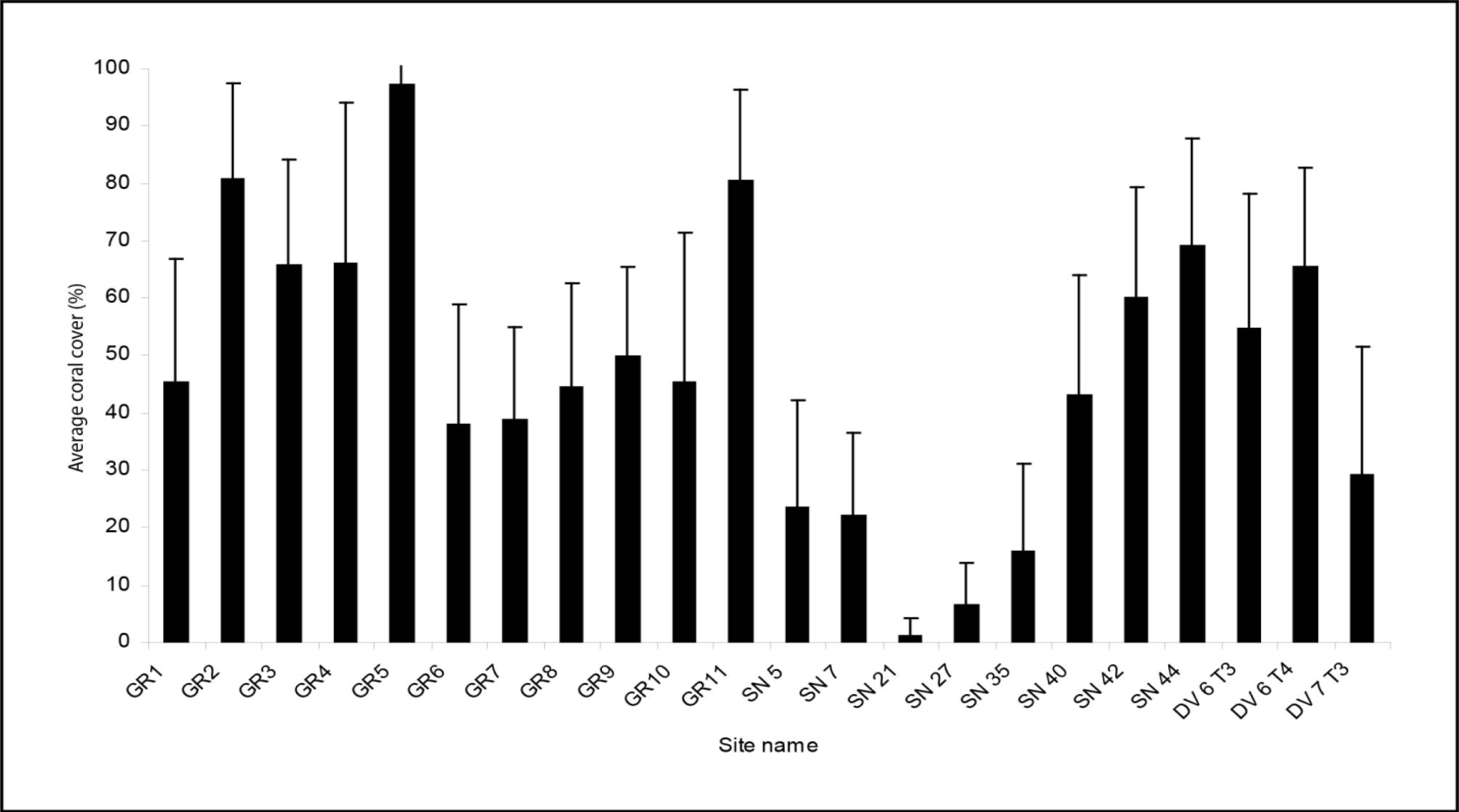
3.1. Field Data Collection and Processing
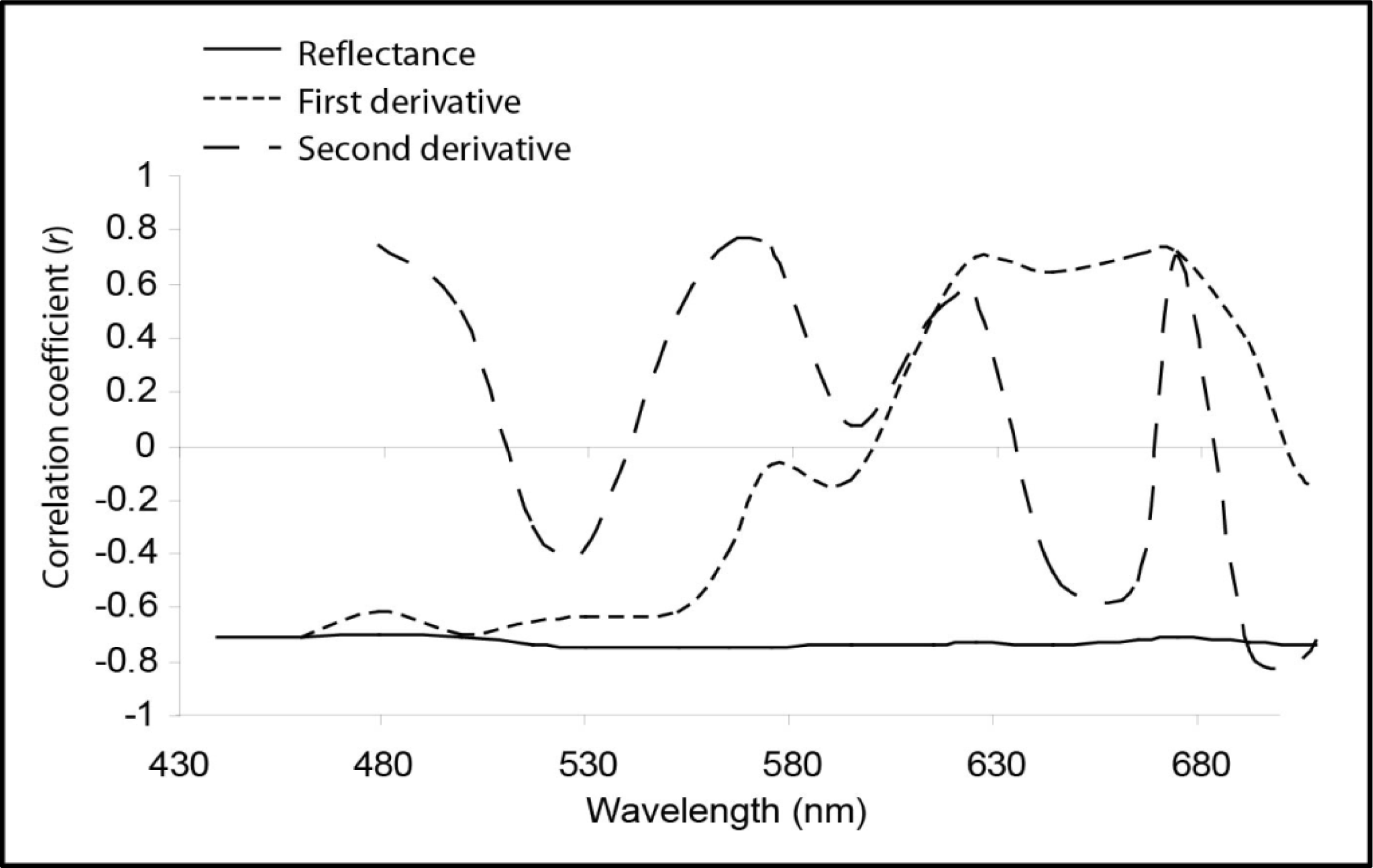
3.2. Live Coral Cover Index Calibration
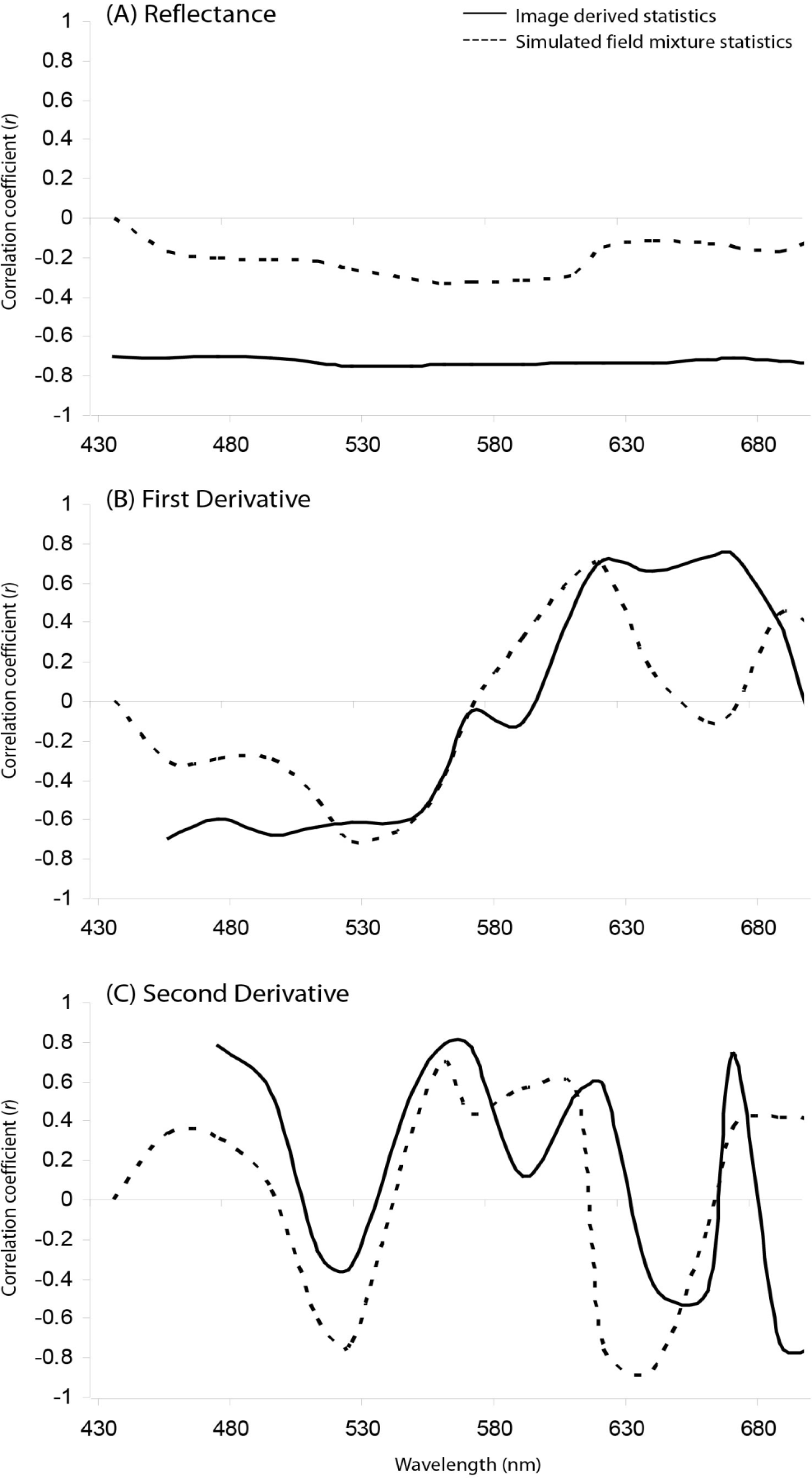
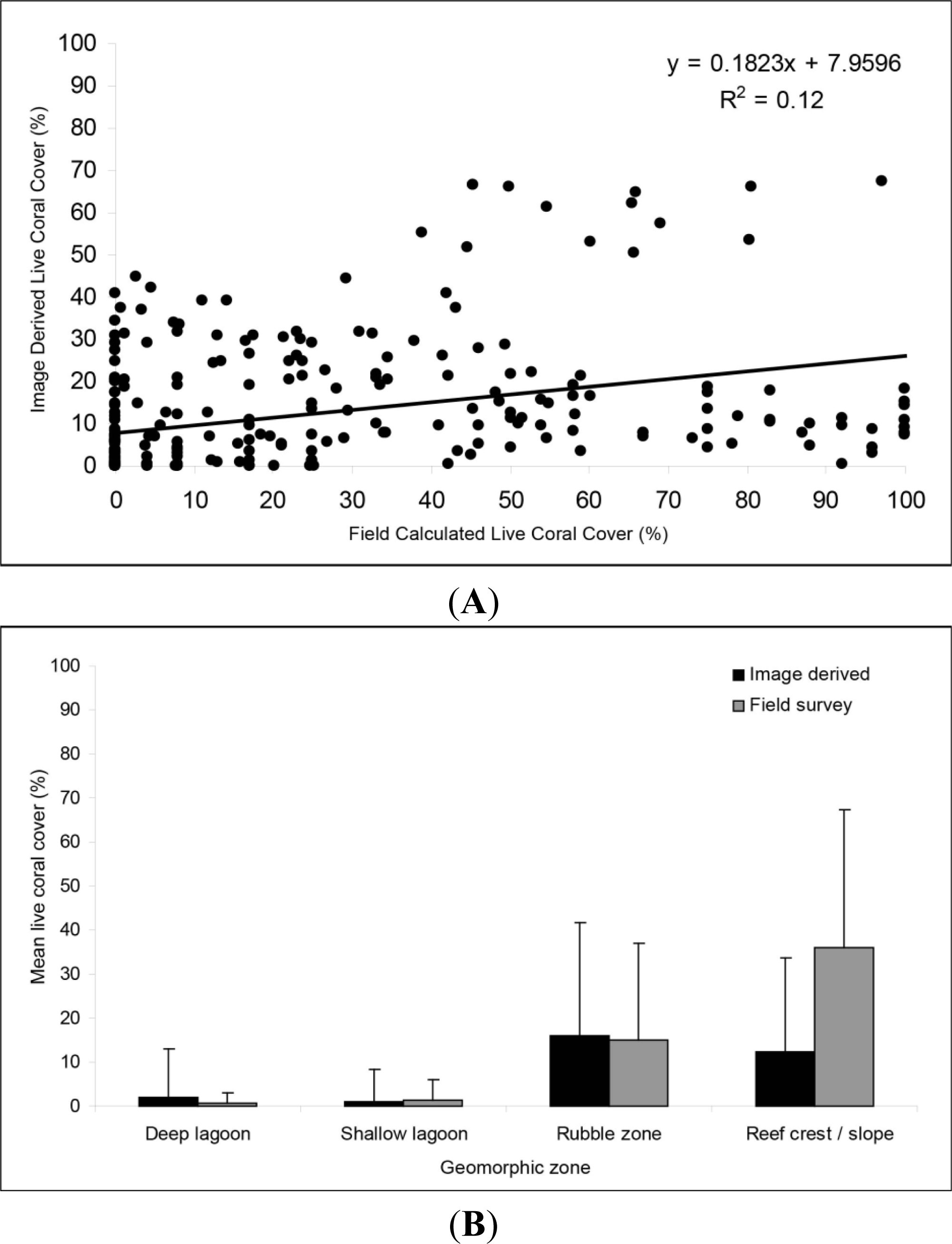
3.3. Live Coral Cover Index Application and Validation
3.4. Validity and Limitations to the Live Coral Cover Index
4. Conclusions and Future Work
Acknowledgments
Conflicts of Interest
References and Notes
- Hopley, D. Aerial Photography and Other Remote Sensing Techniques. In Coral Reefs: Research Methods; Stoddart, D.R., Johannes, R.E., Eds.; UNESCO: Paris, France, 1978; pp. 23–44. [Google Scholar]
- Jupp, D.L.B. The Application and Potential of Remote Sensing in the Great Barrier Reef Region; Great Barrier Reef Marine Park Authority: Townsville, QLD, Australia, 1986; p. 56. [Google Scholar]
- Jupp, D.L.B.; Heggen, S.J.; Mayo, K.K.; Kendal, S.W.; Bolton, J.R.; Harrison, B.A. The BRIAN Handbook: An Introduction to Landsat and the BRIAN (Barrier Reef Image Analysis) Systems for Users; Institute of Biological Resources, Commonwealth Scientific and Industrial Research Organization Australia: Melbourne, Australia, 1985. [Google Scholar]
- Kuchler, D.A. Remote sensing: What can it offer coral reef studies? Oceanus 1986, 29, 90–92. [Google Scholar]
- Kuchler, D.A.; Bina, R.T.; van R Classen, D. Remote sensing of coral reefs. Ascent 1988, 16, 28. [Google Scholar]
- Kuchler, D.A.; Jupp, D.L.B.; van R Claasen, D.B.; Bour, W. Coral reef remote sensing applications. Geocarto Int 1986, 4, 3–15. [Google Scholar]
- Andréfouët, S.; Kramer, P.; Torres-Pulliza, D.; Joyce, K.E.; Hochberg, E.J.; Garza-Perez, R.; Mumby, P.J.; Riegl, B.; Yamano, H.; White, W.H.; et al. Multi-sites evaluation of IKONOS data for classification of tropical coral reef environments. Remote Sens. Environ 2003, 88, 128–143. [Google Scholar]
- Mumby, P.J.; Clark, C.D.; Green, E.P.; Edwards, A.J. Benefits of water column correction and contextual editing for mapping coral reefs. Int. J. Remote Sens 1998, 19, 203–210. [Google Scholar]
- Mumby, P.J.; Green, E.P.; Clark, C.D.; Edwards, A.J. Digital analysis of multispectral airborne imagery of coral reefs. Coral Reefs 1998, 17, 59–69. [Google Scholar]
- Mumby, P.J.; Green, E.P.; Edwards, A.J.; Clark, C.D. Coral reef habitat-mapping: How much detail can remote sensing provide? Mar. Biol 1997, 130, 193–202. [Google Scholar]
- Mumby, P.J.; Edwards, A.J. Mapping marine environments with IKONOS imagery: Enhanced spatial resolution can deliver greater thematic accuracy. Remote Sens. Environ 2002, 82, 248–257. [Google Scholar]
- Kobryn, H.T.; Wouters, K.; Beckley, L.E.; Heege, T. Ningaloo reef: Shallow marine habitats mapped using a hyperspectral sensor. PLoS One 2013, 8, e70105. [Google Scholar]
- Roelfsema, C.; Phinn, S. Validation. In Coral Reef Remote Sensing: A Guide for Multi-Level Sensing Mapping and Assessment; Goodman, J.A., Purkis, S., Phinn, S., Eds.; Springer: New York, NY, USA, 2013. [Google Scholar]
- Phinn, S.R.; Roelfsema, C.M.; Mumby, P.J. Multi-scale, object-based image analysis for mapping geomorphic and ecological zones on coral reefs. Int. J. Remote Sens 2012, 33, 3768–3797. [Google Scholar]
- Leon, J.; Woodroffe, C.D. Improving the synoptic mapping of coral reef geomorphology using object-based image analysis. Int. J. Geogr. Inf. Sci 2011, 25, 949–969. [Google Scholar]
- Roelfsema, C.; Phinn, S.; Jupiter, S.; Comley, J.; Albert, S. Mapping coral reefs at reef to reef-system scales, 10 s–1000 s km2, using object-based image analysis. Int. J. Remote Sens 2013, 34, 6367–6388. [Google Scholar]
- Hochberg, E.J.; Atkinson, M.J. Capabilities of remote sensors to classify coral, algae, and sand as pure and mixed spectra. Remote Sens. Environ 2003, 85, 174–189. [Google Scholar]
- Holden, H.; LeDrew, E. Hyperspectral identification of coral reef features. Int. J. Remote Sens 1999, 20, 2545–2563. [Google Scholar]
- Hedley, J.D.; Mumby, P.J. A remote sensing method for resolving depth and subpixel composition of aquatic benthos. Limnol. Oceanogr 2003, 48, 480–488. [Google Scholar]
- Hedley, J.D.; Mumby, P.J.; Joyce, K.E.; Phinn, S.R. Spectral unmixing of coral reef benthos under ideal conditions. Coral Reefs 2004, 23, 60–73. [Google Scholar]
- Goodman, J.A.; Ustin, S.L. Airborne Hyperspectral Analysis of Coral Ecosystems in the Hawaiian Islands. Proceedings of 30th International Symposium for Remote Sensing of the Environment, Honolulu, HI, USA, 10–14 November 2003.
- Hamylton, S. Estimating the coverage of coral reef benthic communities from airborne hyperspectral remote sensing data: Multiple discriminant function analysis and linear spectral unmixing. Int. J. Remote Sens 2011, 32, 9673–9690. [Google Scholar]
- Hedley, J.D.; Roelfsema, C.M.; Phinn, S.R.; Mumby, P.J. Environmental and sensor limitations in optical remote sensing of coral reefs: Implications for monitoring and sensor design. Remote Sens 2012, 4, 271–302. [Google Scholar]
- Lee, Z.P.; Carder, K.L.; Mobley, C.D.; Steward, R.G.; Patch, J.S. Hyperspectral remote sensing for shallow waters. A semianalytical model. Appl. Opt 1998, 37, 6329–6338. [Google Scholar]
- Chen, Y.; Gillieson, D. Evaluation of Landsat TM vegetation indices for estimating vegetation cover on semi-arid rangelands: A case study from Australia. Can. J. Remote Sens 2009, 35, 435–446. [Google Scholar]
- Xavier, A.C.; Vettorazzi, C.A. Mapping leaf area index through spectral vegetation indices in a subtropical watershed. Int. J. Remote Sens 2004, 25, 1661–1672. [Google Scholar]
- O’Reilly, J.E.; Maritorena, S.; Mitchell, B.G.; Siegel, D.A.; Carder, K.L.; Garver, S.A.; Kahru, M.; McClain, C. Ocean color chlorophyll algorithms for SeaWiFS. J. Geophys. Res 1998, 103, 24937–24953. [Google Scholar]
- Franklin, S.E. Remote Sensing for Sustainable Forest Management; CRC Press: Boca Raton, FL, USA, 2001; p. 407. [Google Scholar]
- Mumby, P.J. Assessing Seagrass Standing Crop. In Remote Sensing Handbook for Tropical Coastal Management; Edwards, A.J., Ed.; UNESCO: Paris, France, 2000; pp. 237–244. [Google Scholar]
- Minghelli-Roman, A.; Chisholm, J.R.M.; Marchioretti, M.; Jaubert, J. Discrimination of coral reflectance spectra in the Red Sea. Coral Reefs 2002, 21, 307–314. [Google Scholar]
- Bour, W.S.; Dupont, S.; Joannot, P. Establishing a “SPOT” thematic neo-channel for the study of hard-of-access lagoon environments: Example of application on the growth area of the New Caledonian Reefs. Geocarto Int 1996, 11, 29–39. [Google Scholar]
- Collin, A.; Hench, J.L.; Planes, S. A Novel Spaceborne Proxy for Mapping Coral Cover. Proceedings of the 12th International Coral Reef Symposium, Cairns, QLD, Australia, 9–13 July 2012.
- Joyce, K.E.; Phinn, S.R. Spectral index development for mapping live coral cover. J. Appl. Remote Sens 2013. [Google Scholar] [CrossRef]
- Scopélitis, J.; Andréfouët, S.; Phinn, S.; Done, T.; Chabanet, P. Coral colonisation of a shallow reef flat in response to rising sea level: Quantification from 35 years of remote sensing data at Heron Island, Australia. Coral Reefs 2011, 30, 951–965. [Google Scholar]
- Mumby, P.J.; Hedley, J.D.; Chisholm, J.R.M.; Clark, C.D.; Ripley, H.T.; Jaubert, J. The cover of living and dead corals from airborne remote sensing. Coral Reefs 2004, 23, 171–183. [Google Scholar]
- Phinn, S.R.; Joyce, K.E.; Roelfsema, C.M. Airborne Hyperspectral Image Data of Heron Reef, Australia; University of Queensland: Brisbane, QLD, Australia, 2012. [Google Scholar]
- Flood, P. The Geomorphology of A Coral Reef. In A Coral Reef Handbook; Mather, P., Bennett, I., Eds.; Surrey Beatty & Sons: Baulkham Hills, NSW, Australia, 1993; pp. 7–15. [Google Scholar]
- Roelfsema, C.M.; Joyce, K.E.; Phinn, S.R. Evaluation of Benthic Survey Techniques for Validating Remotely Sensed Images of Coral Reefs. Proceeings of the International Coral Reef Symposium, Okinawa, Japan, 28 June–2 July 2004.
- Joyce, K.E.; Phinn, S.R.; Roelfsema, C.; Neil, D.T.; Dennison, W.C. Combining landsat ETM+ and reef check classifications for mapping coral reefs: A critical assessment from the southern great barrier reef, Australia. Coral Reefs 2004, 23, 21–25. [Google Scholar]
- Ahmad, W.; Neil, D.T. An evaluation of Landsat Thematic Mapper (TM) digital data for discriminating coral reef zonation: Heron Reef (GBR). Int. J. Remote Sens 1994, 15, 2583–2597. [Google Scholar]
- Joyce, K.E.; Phinn, S.R.; Roelfsema, C.; Neil, D.T.; Dennison, W.C. Mapping the Southern Great Barrier Reef using Landsat ETM and the Reef Check Classification Scheme. Proceedings of the 11th Australasian Remote Sensing and Photogrammetry Conference, Brisbane, QLD, Australia, 2–6 September 2002.
- Hodgson, G. A global assessment of human effects on coral reefs. Mar. Pollut. Bull 1999, 38, 345–355. [Google Scholar]
- Joyce, K.E.; Phinn, S.R.; Roelfsema, C.M. Weighing the costs and benefits of image acquisition and pre-processing for coral reef remote sensing. Position Mag 2003, 8, 76–78. [Google Scholar]
- Joyce, K.E. A. Method for Mapping Live Coral Cover Using Remote Sensing. In School of Geography, Planning and Architecture; University of Queensland: Brisbane, Australia, 2005; p. 132. [Google Scholar]
- Joyce, K.E.; Phinn, S.R. Hyperspectral analysis of chlorophyll content and photosynthetic capacity of coral reef substrates. Limnol. Oceanogr 2003, 48, 489–496. [Google Scholar]
- Hopley, D. The Geomorphology of the Great Barrier Reef: Quaternary Development of Coral Reefs; John Wiley & Sons: Brisbane, QLD, Australia, 1982. [Google Scholar]
- Hatcher, B.G. Coral reef ecosystems: How much greater is the whole than the sum of the parts? Coral Reefs 1997, 16, S77–S91. [Google Scholar]
- Hochberg, E.J.; Atkinson, M.J. Spectral discrimination of coral reef benthic communities. Coral Reefs 2000, 19, 164–171. [Google Scholar]
- Hochberg, E.J.; Atkinson, M.J.; Andréfouët, S. Spectral reflectance of coral reef bottom-types worldwide and implications for coral reef remote sensing. Remote Sens. Environ 2003, 85, 159–173. [Google Scholar]
- Holden, H.; LeDrew, E. Spectral discrimination of healthy and non-healthy corals based on cluster analysis, principal components analysis, and derivative spectroscopy. Remote Sens. Environ 1998, 65, 217–225. [Google Scholar]
- Wettle, M.; Ferrier, G.; Lawrence, A.J.; Anderson, K. Fourth derivative analysis of Red Sea coral reflectance spectra. Int. J. Remote Sens 2003. [Google Scholar] [CrossRef]
- Reichelt, R.E.; Bradbury, R.H. Spatial patterns in coral reef benthos: Multiscale analysis of sites from three oceans. Mar. Ecol. Prog. Ser 1984, 17, 251–257. [Google Scholar]
- Mayo, K.K.; Jupp, D.L.B.; Heggan, S.J.; Kendall, S.W. Heron Island: Field Notes and Data Sheets; CSIRO Div. Water & Land Res. Tech. Mem.: Canberra, ACT, Australia, 1985; p. 14. [Google Scholar]
- Bennett, I. Reef Zonation. In A Coral Reef Handbook; Mather, P., Bennett, I., Eds.; Surrey Beatty & Sons: Baulkham Hills, QLD, Australia, 1993; pp. 179–184. [Google Scholar]
- Roelfsema, C.; Phinn, S. Integrating field data with high spatial resolution multispectral satellite imagery for calibration and validation of coral reef benthic community maps. J. Appl. Remote Sens 2010. [Google Scholar] [CrossRef]




© 2013 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Joyce, K.E.; Phinn, S.R.; Roelfsema, C.M. Live Coral Cover Index Testing and Application with Hyperspectral Airborne Image Data. Remote Sens. 2013, 5, 6116-6137. https://doi.org/10.3390/rs5116116
Joyce KE, Phinn SR, Roelfsema CM. Live Coral Cover Index Testing and Application with Hyperspectral Airborne Image Data. Remote Sensing. 2013; 5(11):6116-6137. https://doi.org/10.3390/rs5116116
Chicago/Turabian StyleJoyce, Karen E., Stuart R. Phinn, and Chris M. Roelfsema. 2013. "Live Coral Cover Index Testing and Application with Hyperspectral Airborne Image Data" Remote Sensing 5, no. 11: 6116-6137. https://doi.org/10.3390/rs5116116
APA StyleJoyce, K. E., Phinn, S. R., & Roelfsema, C. M. (2013). Live Coral Cover Index Testing and Application with Hyperspectral Airborne Image Data. Remote Sensing, 5(11), 6116-6137. https://doi.org/10.3390/rs5116116