The State of Soil Degradation in Sub-Saharan Africa: Baselines, Trajectories, and Solutions
Abstract
:1. New Perspectives for Examining Soil Degradation in Sub-Saharan Africa
1.1. Time Horizons
1.2. Spatial Scales
| Million ha in Africa | Percent of Land in Africa * | |
|---|---|---|
| Acrisol | 87.8 | 2.9 |
| Alisols | 20.3 | 0.7 |
| Andosols | 4.0 | 0.1 |
| Arenosols | 650.3 | 21.5 |
| Chernozems | 1.0 | <0.1 |
| Calcisols | 161.0 | 5.3 |
| Cambisols | 325.4 | 10.8 |
| Durisols | 0.9 | <0.1 |
| Fluvisols | 82.2 | 2.7 |
| Ferralsols | 312.4 | 10.3 |
| Gleysols | 52.5 | 1.7 |
| Gypsisols | 37.5 | 1.2 |
| Histosols | 4.4 | 0.1 |
| Kastanozems | 2.7 | 0.1 |
| Leptosols | 530.0 | 17.5 |
| Luvisols | 105.1 | 3.5 |
| Lixisols | 126.8 | 4.2 |
| Nitisols | 60.4 | 2 |
| Phaeozems | 12.1 | 0.4 |
| Planosols | 27.7 | 0.9 |
| Plinthosols | 146.1 | 4.8 |
| Podzols | 2.9 | 0.1 |
| Regosols | 93.5 | 3.1 |
| Solonchaks | 32.6 | 1.1 |
| Solonets | 36.0 | 1.2 |
| Stagnosols | 0.5 | <0.1 |
| Technosols | 0.0 | <0.1 |
| Umbrisols | 5.6 | 0.2 |
| Vertisols | 102.0 | 3.4 |
1.3. Multiple Indicators
2. Soil Degradation in Sub-Saharan Africa
2.1. Drivers of Degradation
2.2. Types of Degradation in Sub-Saharan Africa
| Category | Specific degradation processes | State factors | Socioeconomic drivers | |
|---|---|---|---|---|
| Parent material and topography | Climate | |||
| Physical | Soil erosion by water | Slope | Humid to semi-arid regions | Tillage agriculture, deforestation and improper grazing |
| Soil erosion by wind | Less vegetation | Semi-arid to arid regions | Disturbance of soil, vegetation or bio-crust by agricultural tillage and poorly-managed grazing | |
| Soil erosion by tillage | Hilly landscapes | Continuous cultivation, especially with tillage | ||
| Surface sealing | Low organic matter sandy or silty soils | Urbanization, compaction, tillage | ||
| Soil compaction | Clayey soils | Humid regions | Heavy machinery, grazing | |
| Reduced capacity to store water | Low organic matter | Compaction, erosion, removal of mulch or residue | ||
| Chemical | Nutrient depletion | Low inherent fertility | Low input agriculture, grazing, excessive forest harvest | |
| Acidification | Old, weathered soils | Humid regions | Excessive N fertilization, leaching, sulfur and nitrogen oxidation | |
| Dispersion/alkalization | Excessive monovalent ions, exposure and incorporation of calcareous subsoil material into surface horizon | Poor quality irrigation water, loss of perennial vegetation, tillage | ||
| Salinization | Shallow water table | Arid to semi-arid regions | Excessive irrigation | |
| Toxic Contamination | Urbanization, mining, industrial waste spillage or disposal | |||
| Biological | Depletion of soil organic matter | Sandy texture, steep slopes, deep water table | High temperatures, limited rainfall | Degradation of vegetation, excessive tillage, lack of sufficient organic amendments and plant residues; excessive biomass removal by harvest, grazing or fire; erosion of sloping surface soil by tillage, wind and water |
| Loss of soil biological diversity | Sandy texture, steep slopes, root limiting subsoil layers (fragipans, cemented layers, aluminum toxicity, calcic horizons) | High temperatures | Mono-cropping, deforestation and poorly managed grazing | |
| Loss of plant, animal and microbial biomass | Side slopes, shallow bedrock, root limiting subsoil layers (fragipans, cemented layers, aluminum toxicity, calcic horizons) | Reduced plant growth and subsequent addition of litter, roots and exudates limits carbon fuel for food web; exposure to extremes of dryness and temperature by removal of plant litter; destruction of macropores, aggregates and other habitat by tillage, compaction and erosion. | ||
| Soil Constraint | Modifier | Million ha in SSA | Percent of Land in SSA * |
|---|---|---|---|
| Low nutrient capital reserves | k | 942.06 | 39.94 |
| Al toxicity | a | 588.27 | 24.94 |
| High P fixation | i | 200.35 | 8.49 |
| Steep sloped (>30%) | s | 55.62 | 2.36 |
| Poor drainage | g | 159.95 | 6.78 |
| High leaching potential | e | 425.05 | 18.02 |
| Calcareous reaction | b | 158.11 | 6.70 |
| Salinity | s | 19.09 | 0.81 |
| Alkalinity | n | 52.06 | 2.21 |
| Allophane | x | 2.83 | 0.12 |
| Shrink-swell | v | 132.65 | 5.62 |
| Total area | 2358.79 |
3. Synthesis of Knowledge
- (1)
- A temporal dimension, as degradation is a dynamic process;
- (2)
- A spatial scale of analysis that is meaningful both for assessing degradation and for providing soil management recommendation for smallholder farmers; and
- (3)
- Multiple criteria of assessment that reflect the use of the soil because degradation results from a complex set of processes and cannot be captured in a single measure.
3.1. Methods for Data Synthesis
| Reference | Study Type | Select Indicators of Degradation | Temporal scale | Spatial scale | Baseline (Reference) | Depth | Region | Trajectory | |
|---|---|---|---|---|---|---|---|---|---|
| Quantitative | Qualitative | ||||||||
| [93] | Chrono | Particle size, Water holding capacity, SOM, Exch. Ca, Exch. K, Exch. Mg, total N, Ext. P, pH, and CEC | NA | 15 years | Landscape | 0–20 cm | Nigeria | Downward | |
| [94] | Chrono | Soil spectra, total C, Exch. Mg, Exch. Ca, Exch. K, total N, pH, ECEC, Clay, Silt, and Sand | NA | 100 years | Landscape | Humid tropical forest | 0–20 cm | Kenya | Downward |
| [95] | Chrono | Total N, pH, SOM, Sand, Silt, Clay, Bulk density, Tree density, Tree species | NA | 50 years | Landscape | Tropical dry Afro-montane forest (deforested/heavy harvesting) | 0–100 cm | Ethiopia | Downward |
| [16] | Long | Soil erosion (water-induced), Sediment flux, River discharge, and Coral Ba/Ca | NA | 300 years | River basin (66,800 km2) | None | NA | Kenya | Downward |
| [78] | Long; Integ | Land use and land cover. Trees in fields, CEC, Exch. Ca, Exch. K, Exch. Mg, total N, Ext. P, pH, and SOC | Farmer mgmt, perception of change, veg cover | 15 years (imagery);8 years (soils) | Multi-scale (Landscape and farm field) | 1981—imagery; 1988—soils | 0–20 cm | Burkina Faso | Minimal change to upward (field scale), Possibly downward (landscape scale) |
| [96] | Long | Exch. Ca, Exch. Mg, ECEC, SOC, pH, bulk density, maize grain yield | NA | 13 years | Landscape | Tropical forest | 0–15 cm | Nigeria | Mixed dependent on management strategies: Decline without fallow or addition of organic input |
| [97] | Chrono | Total N, Ext. P, SOM, Maize biomass, Plant tissue (N, P, K, Ca, Mg, Mn, Cu and Zn), Socioeconomic survey | Crop yield, Indicator plants, Soil softness and Soil color | 57 years | Landscape | Tropical dry Afromontane forest (deforested/heavy harvesting) | 0–20 cm | Ethiopia | Downward (maize biomass) |
| [98] | Chrono | CEC (effective and potential), pH, SOC, Grain and stover yield, Plant tissue: N, P, K, Ca, and Mg | NA | 100 years | Landscape | Humid tropical forest | 0–10 cm | Kenya | Downward (non-linear) |
| [99] | Long | Land cover classes, Precipitation, Socioeconomic survey, Soil chemical properties | Incidence of soil erosion | 40 years | Landscape | Baseline (1966) | NA | Tanzania | Spatially heterogeneous (Downward in some zones) |
| [100] | Long | CEC, Exch. Ca, Exch. K, Exch. Mg, pH, total N, Ext. P, SOC, Bulk density, Infiltration, Penetrometer resistance, Soil moisture retention, Water stable aggregates, and Yield | NA | 8 years | Farm field (Field trial) | 0–20 cm | Nigeria | Downward (dependent on management) | |
| [79,101] | Chrono | Soil depth, Base Saturation, % of CEC, C:N, Exch. Ca, Exch. K, Exch. Na, Total N, Ext. P, pH, SOC, Bulk density, Particle size analysis, Pore space, 13C and 15N, carbon fractions | Qualitative land evaluation for maize | 53 years | Landscape | Tropical dry Afro-montane forest (deforested/heavy harvesting) | 0–20 cm; 60–70 cm, 90–100 cm | Ethiopia | Downward (C-exponential) in topsoil, C & N increase in subsoil |
| [102] | Chrono | Active C, CEC, Exch. Ca, EC, Exch. K, Exch. Mg, pH, Total N, Ext. P, S, SOM, Zn, Sand, Silt, Clay, Water stable aggregation (WSA), Available water capacity (AWC), Penetrometer resistance, Crop yield | NA | 77 years | Landscape | Humid tropical forest | 0–15 cm, 0–45 cm | Kenya | Downward in most properties, slope of trajectory less severe with better soil management |
| [103] | Chrono | Mineral N, P fractions, P sorption capacity, Fertilizer recovery, Maize yield, Maize nutrient concentration | NA | 100 years | Landscape | Humid tropical forest | 0–10 cm | Kenya | Downward trend in soil fertility; yield increased dependent on nutrient additions |
| [104] | Chrono | Soil C & N concentration, Isotopic signature of soil C, Infiltrability, Bulk density, Proportion of macro and micro-aggregates in soil | Crop yield estimates | 120 years | Landscape | Humid tropical forest | 0–15 cm | Kenya | Downward |
| [105] | Long | EC, Exch. K & Exch. Mg, Ext. P, pH, SOM, and Plant tissue analysis (N, P, K, Ca, Mg, S, Zn, B, Mn, Fe, Cu and Al) | NA | 7 years | Sub-national | Baseline (1991) | 0–15 cm | Gambia | Minimal change |
| [106] | Chrono | 13C, Near-edge X-ray absorption fine structure, SOC, | NA | 103 years (Kenya); 90 years (South Africa) | Landscape | Humid tropical forest (Kenya); Subtropical grassland (South Africa) | 0–10 cm (Kenya; 0–20 cm (South Africa) | Kenya; South Africa | Downward (exponential) |
| [41] | Chrono; Integ | N, P, K, SOC, Woody and herbaceous species, Land cover change | Soil properties Livestock Yield, Pests, Trees | 50 years (soil); 15 years (imagery) | Landscape | Grass strips adjacent to fields | NA | Botswana and Swaziland | Downward |
| [107] | Chrono | CEC, Exch. Ca, Exch. K, Exch. Mg, pH, total N, Ext. P, SOC, Clay, Silt, SFI, Surface reflectance, Soil spectra | Soil quality - poor, average, good | 50 years | Landscape | Rainforest | 0–20 cm | Madagascar | Downward |
3.2. Longitudinal Studies
3.3. Chronosequences (Space-for-Time)
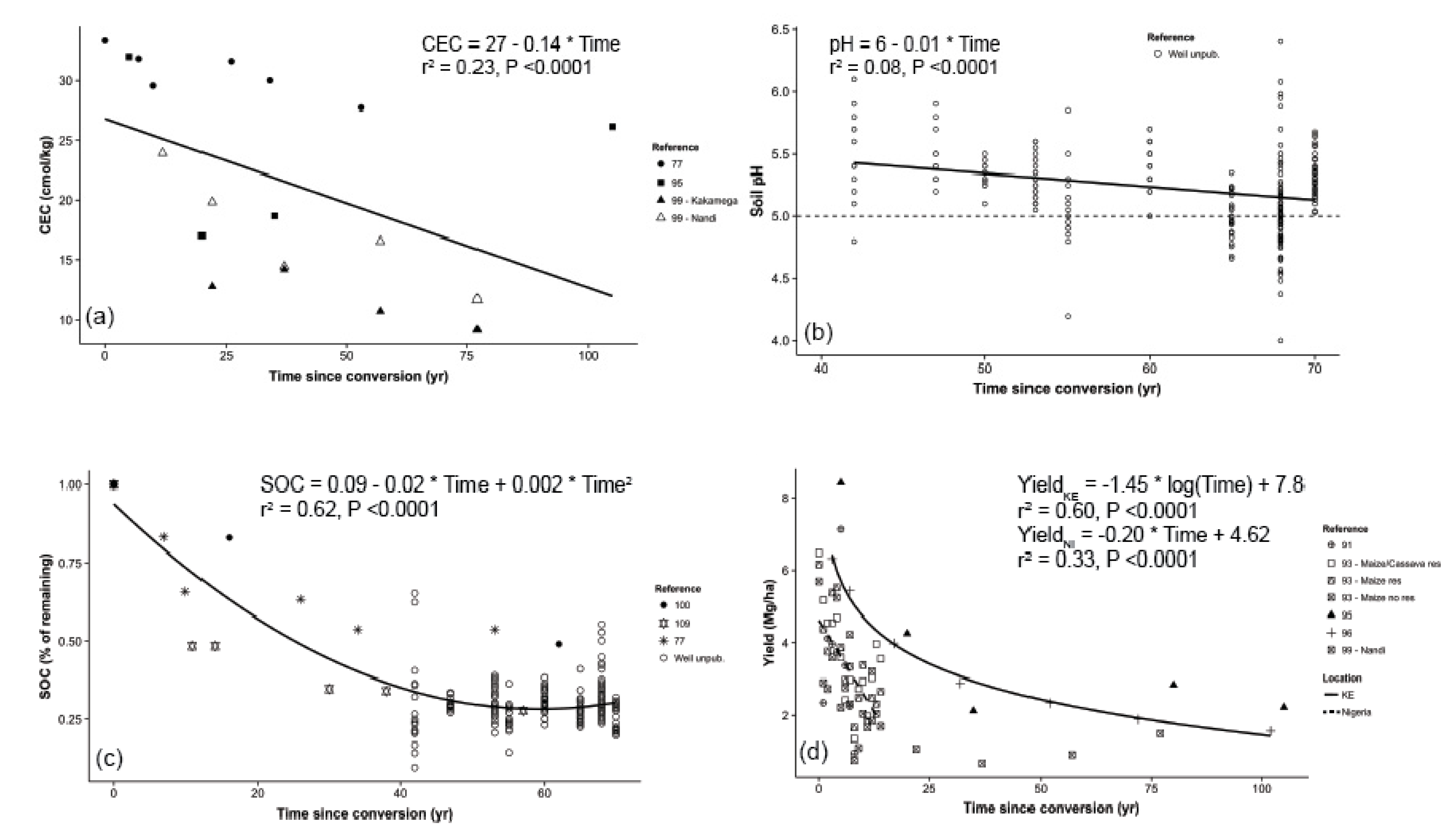
3.4. Integrated Assessments
3.5. Synthesis Summary
4. Methods for Monitoring Soil Degradation in Sub-Saharan Africa
4.1. Visual Indicators

4.2. Management Indicators

4.3. Physical Indicators
4.4. Chemical Indicators
4.5. Biological Indicators
5. Positive Trajectories and Conclusions
Acknowledgments
Author Contributions
Appendix: Methods Used by R. Weil for Collecting Thirty-Year Trends on Soil Properties in Red Soils near Holetta Research Center, Ethiopia
Conflicts of Interest
References
- Sanchez, P.A.; Swaminathan, M.S. Hunger in Africa: The link between unhealthy people and unhealthy soils. Lancet 2005, 365, 442–444. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, P.A. Soil fertility and hunger in Africa. Science 2002, 295, 2019–2020. [Google Scholar] [CrossRef] [PubMed]
- Koning, N.; Smaling, E. Environmental crisis or “lie of the land?” The debate on soil degradation in Africa. Land Use Policy 2005, 22, 3–11. [Google Scholar] [CrossRef]
- Sanchez, P.; Palm, C.; Sachs, J.; Denning, G.; Flor, R.; Harawa, R.; Jama, B.; Kiflemariam, T.; Konecky, B.; Kozar, R.; et al. The African Millennium Villages. Proc. Natl. Acad. Sci. USA 2007, 104, 16775–16780. [Google Scholar] [CrossRef] [PubMed]
- Nziguheba, G.; Palm, C.A.; Berhe, T.; Denning, G.; Dicko, A.; Diouf, O.; Diru, W.; Flor, R.; Frimpong, F.; Harawa, R.; et al. The African Green Revolution. In Advances in Agronomy; Elsevier: Amsterdam, The Netherlands, 2010; Volume 109, pp. 75–115. [Google Scholar]
- Barrett, C.B.; Reardon, T.; Webb, P. Nonfarm income diversification and household livelihood strategies in rural Africa: Concepts, dynamics, and policy implications. Food Policy 2001, 26, 315–331. [Google Scholar] [CrossRef]
- United Nations Human Settlements Programme (UN-Habitat). The State of African Cities: Re-Imagining Sustainable Urban Transitions; UN-Habitat: Nairobi, Kenya, 2014. [Google Scholar]
- World Commission of Environment and Development. Our Common Future: Report of the World Commission on Environment and Development; Oxford University Press: Oxford, UK, 1987; pp. 1–300. [Google Scholar]
- Vandecar, K.L.; Lawrence, D.; Wood, T.; Oberbauer, S.F.; Das, R.; Tully, K.; Schwendenmann, L. Biotic and abiotic controls on diurnal fluctuations in labile soil phosphorus of a wet tropical forest. Ecology 2009, 90, 2547–2555. [Google Scholar] [CrossRef] [PubMed]
- McGrath, D.A.; Comerford, N.B.; Duryea, M.L. Litter dynamics and monthly fluctuations in soil phosphorus availability in an Amazonian agroforest. Forest Ecol. Manag. 2000, 131, 167–181. [Google Scholar] [CrossRef]
- Crews, T.E.; Kitayama, K.; Fownes, J.H.; Riley, R.H.; Herbert, D.A.; Mueller-Dombois, D.; Vitousek, P.M. Changes in Soil Phosphorus Fractions and Ecosystem Dynamics across a Long Chronosequence in Hawaii. Ecology 1995, 76, 1407–1424. [Google Scholar] [CrossRef]
- Walker, T.W.; Syers, J.K. The fate of phosphorus during pedogenesis. Geoderma 1976, 15, 1–19. [Google Scholar] [CrossRef]
- Zingore, S.; Murwira, H.K.; Delve, R.J.; Giller, K.E. Influence of nutrient management strategies on variability of soil fertility, crop yields and nutrient balances on smallholder farms in Zimbabwe. Agric. Ecosyst. Environ. 2007, 119, 112–126. [Google Scholar] [CrossRef]
- Tully, K.L.; Wood, S.A.; Almaraz, M.; Neill, C.; Palm, C.A. The effect of mineral and organic nutrient inputs on yields and nitrogen balances in western Kenya. Agric. Syst. 2015. submitted for publication. [Google Scholar]
- Hamilton, A.C.; Taylor, D. History of climate and forests in tropical Africa during the last 8 million years. Clim. Chang. 1991, 19, 65–78. [Google Scholar] [CrossRef]
- Fleitmann, D.; Dunbar, R.B.; McCulloch, M.; Mudelsee, M.; Vuille, M.; McClanahan, T.R.; Cole, J.E.; Eggins, S. East African soil erosion recorded in a 300 year old coral colony from Kenya. Geophys. Res. Lett. 2007, 34, L04401. [Google Scholar] [CrossRef]
- Scheffer, M.; Carpenter, S.; Foley, J.A.; Folke, C.; Walker, B. Catastrophic shifts in ecosystems. Nature 2001, 413, 591–596. [Google Scholar] [CrossRef] [PubMed]
- Holling, C.S. Resilience and Stability of Ecological Systems. Annu. Rev. Ecol. Syst. 1973, 4, 1–23. [Google Scholar] [CrossRef]
- Folke, C. Resilience: The emergence of a perspective for social—Ecological systems analyses. Global Environ. Chang. 2006, 16, 253–267. [Google Scholar] [CrossRef]
- Bayon, G.; Dennielou, B.; Etoubleau, J.; Ponzevera, E.; Toucanne, S.; Bermell, S. Intensifying Weathering and Land Use in Iron Age Central Africa. Science 2012, 335, 1219–1222. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, S.; Walker, B.; Anderies, J.M.; Abel, N. From Metaphor to Measurement: Resilience of What to What? Ecosystems 2001, 4, 765–781. [Google Scholar] [CrossRef]
- Levin, S.A. Fragile Dominion: Complexity and the Commons; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1999. [Google Scholar]
- Warren, A. Land degradation is contextual. Land Degrad. Dev. 2002, 13, 449–459. [Google Scholar] [CrossRef]
- Dewitte, O.; Jones, A.; Spaargaren, O.; Breuning-Madsen, H.; Brossard, M.; Dampha, A.; Deckers, J.; Gallali, T.; Hallett, S.; Jones, R.; et al. Harmonisation of the soil map of Africa at the continental scale. Geoderma 2013, 211–212, 138–153. [Google Scholar] [CrossRef] [Green Version]
- Sanchez, P.A.; Palm, C.A.; Buol, S.W. Fertility capability soil classification: A tool to help assess soil quality in the tropics. Geoderma 2003, 114, 157–185. [Google Scholar] [CrossRef]
- Lohse, K.A.; Matson, P. Consequences of nitrogen additions for soil losses from wet tropical forests. Ecol. Appl. 2005, 15, 1629–1648. [Google Scholar] [CrossRef]
- Kinjo, T.; Pratt, P.F. In some acid soils of Mexico and South America. II. In competition with chloride, sulfate and phosphate. III. Desorption, movement and distribution in Andepts. Soil Sci. Soc. Am. J. 1971, 35, 722–732. [Google Scholar] [CrossRef]
- Tittonell, P.; Muriuki, A.; Klapwijk, C.J.; Shepherd, K.D.; Coe, R.; Vanlauwe, B. Soil Heterogeneity and Soil Fertility Gradients in Smallholder Farms of the East African Highlands. Soil Sci. Soc. Am. J. 2013. [Google Scholar] [CrossRef]
- Almaraz, M.; Tully, K.L.; Neill, C.; Palm, C.A.; Porder, S. Nitrogen dynamics in soil profiles from intensifying agricultural fields in sub-Saharan Africa: The role of soil type. 2015, unpublished work. [Google Scholar]
- Scoones, I. Wetlands in drylands: Key resources for agricultural and pastoral production in Africa. Ambio 1991, 20, 366–371. [Google Scholar]
- Mekonnen, K.; Buresh, R.J.; Jama, B. Root and inorganic nitrogen distributions in sesbania fallow, natural fallow and maize fields. Plant Soil 1997, 188, 319–327. [Google Scholar] [CrossRef]
- Chintu, R.; Mafongoya, P.L.; Chirwa, T.S.; Mwale, M.; Matibini, J. Subsoil nitrogen dynamics as affected by planted coppicing tree legume fallows in eastern Zambia. Ex. Agric. 2004, 40, 327–340. [Google Scholar] [CrossRef]
- Vanlauwe, B.; Kihara, J.; Chivenge, P.; Pypers, P.; Coe, R.; Six, J. Agronomic use efficiency of N fertilizer in maize-based systems in sub-Saharan Africa within the context of integrated soil fertility management. Plant Soil 2010, 339, 35–50. [Google Scholar] [CrossRef]
- Tittonell, P.; Vanlauwe, B.; Leffelaar, P.A.; Rowe, E.C.; Giller, K.E. Exploring diversity in soil fertility management of smallholder farms in western Kenya. Agric. Ecosyst. Environ. 2005, 110, 149–165. [Google Scholar] [CrossRef]
- Palm, C.A.; Smukler, S.M.; Sullivan, C.C.; Mutuo, P.K.; Nyadzi, G.I.; Walsh, M.G. Identifying potential synergies and trade-offs for meeting food security and climate change objectives in sub-Saharan Africa. Proc. Natl. Acad. Sci. USA 2010, 107, 19661–19666. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, K.D.; Shepherd, G.; Walsh, M.G. Land health surveillance and response: A framework for evidence-informed land management. Agric. Syst. 2015, 132, 93–106. [Google Scholar] [CrossRef]
- Letey, J.; Sojka, R.E.; Upchurch, D.R. Deficiencies in the soil quality concept and its application. J. Soil Water Conserv. 2003, 58, 180–187. [Google Scholar]
- Karlen, D.L.; Mausbach, M.J.; Doran, J.W. Soil quality: A concept, definition, and framework for evaluation. Soil Sci. Soc. Am. J. 1997, 61, 4–10. [Google Scholar] [CrossRef]
- Wyland, L.J.; Jackson, L.E.; Schulbach, K.F. Soil-plant nitrogen dynamics following incorporation of a mature rye cover crop in a lettuce production system. J. Agric. Sci. 1995, 124, 17–25. [Google Scholar] [CrossRef]
- Dean, J.E.; Weil, R.R. Brassica Cover Crops for Nitrogen Retention in the Mid-Atlantic Coastal Plain. J. Environ. Qual. 2009, 38, 520–528. [Google Scholar] [CrossRef] [PubMed]
- Stringer, L.C.; Reed, M.S. Land degradation assessment in Southern Africa: Integrating local and scientific knowledge bases. Land Degrad. Dev. 2007, 18, 99–116. [Google Scholar] [CrossRef]
- Stoorvogel, J.J.; Smaling, E.M. A. Assessment of Soil Nutrient Depletion in Sub-Saharan Africa,1983–2000; Winand Staarting Center-DLO: Wageningen, The Netherlands, 1990. [Google Scholar]
- Lal, R. Erosion-Crop Productivity Relationships for Soils of Africa. Soil Sci. Soc. Am. J. 1995, 59, 661–667. [Google Scholar] [CrossRef]
- Oldeman, L.R.; Hakkeling, R.T.A.; Sombroek, W.G. World Map of the Status of Human-Induced Soil Degradation; ISRIC: Wageningen, The Netherlands; FAO: Nairobi, Kenya, 1991; pp. 1–35. [Google Scholar]
- Mazzucato, V.; Niemeijer, D. Rethinking Soil and Water Conservation in a Changing Society: A Case Study from Burkina Faso. Ph.D. Thesis, Wageningen University, Wageningen, The Netherlands, 20 June 2000; pp. 1–421. [Google Scholar]
- Scherr, S.J. Economic factors in farmer adoption of agroforestry: Patterns observed in Western Kenya. World Dev. 1995, 23, 787–804. [Google Scholar] [CrossRef]
- Forsyth, T. Science, myth and knowledge: Testing himalayan environmental degradation in Thailand. Geoforum 1996, 27, 375–392. [Google Scholar] [CrossRef]
- Tiffen, M.; Mortimer, M.; Gichuki, F. More People, Less Erosion; John Wiley & Sons, Ltd.: Chichester, UK, 1994; pp. 1–326. [Google Scholar]
- Barbier, E.B. The economic linkages between rural poverty and land degradation: Some evidence from Africa. Agr. Ecosyst. Environ. 2000, 82, 355–370. [Google Scholar] [CrossRef]
- McCall, M. Environmental and agricultural impacts of Tanzania’s villagization programme. In Population and Development Projects in Africa; Clark, J.I., Khogali, M., Kosinski, L.A., Eds.; Cambridge University Press: Cambridge, UK, 1985; pp. 123–140. [Google Scholar]
- Thomas, D.S.G.; Sporton, D.; Perkins, J. The environmental impact of livestock ranches in the Kalahari, Botswana: Natural resource use, ecological change and human response in a dynamic dryland system. Land Degrad. Dev. 2000, 11, 327–341. [Google Scholar] [CrossRef]
- Rohde, R.F.; Moleele, N.M.; Mphale, M.; Allsopp, N.; Chanda, R.; Hoffman, M.T.; Magole, L.; Young, E. Dynamics of grazing policy and practice: Environmental and social impacts in three communal areas of southern Africa. Environ. Sci. Policy 2006, 9, 302–316. [Google Scholar] [CrossRef]
- Mafongoya, P.L.; Bationo, A.; Kihara, J.; Waswa, B.S. Appropriate technologies to replenish soil fertility in southern Africa. Nutr. Cycl. Agroecosyst. 2006, 76, 137–151. [Google Scholar] [CrossRef]
- Boserup, E. The Conditions of Agricultural Growth; Adine Publishing Company: Chicago, IL, USA, 1965. [Google Scholar]
- Hoddinott, J.; Haddad, L. Does female income share influence household expenditures? Evidence from Côte d'Ivoire. Oxford Bull. Econ. Stat. 1995, 57, 77–96. [Google Scholar] [CrossRef]
- Nkonya, E.; Moore, K. Smallholder Adoption of Integrated Soil Fertility Management; USAID&Agrilinks: Washington, DC, USA, 2015; p. 51. [Google Scholar]
- Nkonya, E. Soil Conservation Practices and Non-Agricultural Land Use in the South-Western Highlands of Uganda; The International Food Policy Research Institute (IFPRI): Washington, DC, USA, 2002; pp. 1–31. [Google Scholar]
- Tamene, L.; Vlek, P.L.G. Soil Erosion Studies in Northern Ethiopia. In Land Use and Soil Resources; Braimoh, A.K., Vlek, P.L.G., Eds.; Springer Netherlands: Dordrecht, The Netherlands, 2008; pp. 73–100. [Google Scholar]
- Tamene, L.; Park, S.J.; Dikau, R.; Vlek, P.L.G. Reservoir siltation in the semi-arid highlands of northern Ethiopia: Sediment yield–catchment area relationship and a semi-quantitative approach for predicting sediment yield. Earth Surf. Process. Landforms 2006, 31, 1364–1383. [Google Scholar] [CrossRef]
- Kimaro, D.N.; Deckers, J.A.; Poesen, J.; Kilasara, M.; Msanya, B.M. Short and medium term assessment of tillage erosion in the Uluguru Mountains, Tanzania. Soil Tillage Res. 2005, 81, 97–108. [Google Scholar] [CrossRef]
- Collins, A.L.; Walling, D.E.; Sichingabula, H.M.; Leeks, G.J.L.G. Suspended sediment source fingerprinting in a small tropical catchment and some management implications. Appl. Geogr. 2001, 21, 387–412. [Google Scholar] [CrossRef]
- Taddese, G.; Saleem, M.A.M.; Abyie, A.; Wagnew, A. Impact of grazing on plant species richness, plant biomass, plant attribute, and soil physical and hydrological properties of vertisol in East African highlands. Environ. Manag. 2002, 29, 279–289. [Google Scholar] [CrossRef]
- Van N du Toit, G.; Snyman, H.A.; Malan, P.J. Physical impact of grazing by sheep on soil parameters in the Nama Karoo subshrub/grass rangeland of South Africa. J. Arid Environ. 2009, 73, 804–810. [Google Scholar] [CrossRef]
- Weil, R.R.; Brady, N. Nature and Properties of Soils, 15 ed.; 2015; unpublished work. [Google Scholar]
- Nye, P.H.; Greenland, D.J. The Soil under Shifting Cultivation, 51st ed.; Commonwealth Agricultural Bureau, Farnham Royal: Berks, Great Britain, 1960; p. 156. [Google Scholar]
- Vitousek, P.M.; Naylor, R.; Crews, T.; David, M.B.; Drinkwater, L.E.; Holland, E.; Johnes, P.J.; Katzenberger, J.; Martinelli, L.A.; Matson, P.A.; et al. Nutrient Imbalances in Agricultural Development. Science 2009, 324, 1519–1520. [Google Scholar] [CrossRef] [PubMed]
- Oenema, O.; de Vries, W. Approaches and uncertainties in nutrient budgets: Implications for nutrient management and environmental policies. Eur. J. Agron. 2003, 20, 3–16. [Google Scholar] [CrossRef]
- Henao, J.; Baanante, C.A. Estimating Rates of Nutrient Depletion in Soils of Agricultural Lands of Africa; Intl Fertilizer Development Center: Muscle Shoals, AL, USA, 1999. [Google Scholar]
- Cobo, J.G.; Dercon, G.; Cadisch, G. Nutrient balances in African land use systems across different spatial scales: A review of approaches, challenges and progress. Agric. Ecosyst. Environ. 2010, 136, 1–15. [Google Scholar] [CrossRef]
- Amede, T.; Belachew, T.; Geta, E. Reversing the Degradation of Arable Land in the Ethiopian Highlands; IIED: London, UK, 2001; p. 23. [Google Scholar]
- Juo, A.S.R.; Dabiri, A.; Franzluebbers, K. Acidification of a kaolinitic Alfisol under continuous cropping with nitrogen fertilization in West Africa. Plant Soil 1995, 171, 245–253. [Google Scholar] [CrossRef]
- Barbiéro, L.; Mohamedou, A.O.; Roger, L.; Furian, S.; Aventurier, A.; Rémy, J.C.; Marlet, S. The origin of Vertisols and their relationship to Acid Sulfate Soils in the Senegal valley. CATENA 2005, 59, 93–116. [Google Scholar] [CrossRef]
- Van Asten, P.J.A.; Barbiéro, L.; Wopereis, M.C.S.; Maeght, J.L.; van der Zee, S.E.A.T.M. Actual and potential salt-related soil degradation in an irrigated rice scheme in the Sahelian zone of Mauritania. Agric. Water Manag. 2003, 60, 13–32. [Google Scholar] [CrossRef]
- Soil Atlas of Africa; Jones, A. (Ed.) Publications Office of the European Union: Luxembourg, 2013.
- HarvestChoice Updating Soil Functional Capacity Classification System. Available online: http://harvestchoice.org/node/1435 (accessed on 24 January 2015).
- Vlek, P.L.G. The role of fertilizers in sustaining agriculture in sub-Saharan Africa. Fert. Res. 1990, 26, 327–339. [Google Scholar] [CrossRef]
- Bationo, A.; Lompo, F.; Koala, S. Research on nutrient flows and balances in West Africa: State-of-the-art. Agr. Ecosyst. Environ. 1998, 71, 19–35. [Google Scholar] [CrossRef]
- Gray, L.C. Is land being degraded? A multi-scale investigation of landscape change in southwestern Burkina Faso. Land Degrad. Dev. 1999, 10, 329–343. [Google Scholar] [CrossRef]
- Lemenih, M.; Karltun, E.; Olsson, M. Assessing soil chemical and physical property responses to deforestation and subsequent cultivation in smallholders farming system in Ethiopia. Agric. Ecosyst. Environ. 2005, 105, 373–386. [Google Scholar] [CrossRef]
- Bossio, D.A.; Girvan, M.S.; Verchot, L.; Bullimore, J.; Borelli, T.; Albrecht, A.; Scow, K.M.; Ball, A.S.; Pretty, J.N.; Osborn, A.M. Soil Microbial Community Response to Land Use Change in an Agricultural Landscape of Western Kenya. Microb. Ecol. 2005, 49, 50–62. [Google Scholar] [CrossRef] [PubMed]
- Brussaard, L.; de Ruiter, P.C.; Brown, G.G. Soil biodiversity for agricultural sustainability. Agr. Ecosyst. Environ. 2007, 121, 233–244. [Google Scholar] [CrossRef]
- Wood, S.A.; Almaraz, M.; Bradford, M.A.; McGuire, K.L.; Naeem, S.; Palm, C.A.; Tully, K.L.; Zhou, J. Farm management, not soil microbial diversity, controls nutrient loss from smallholder tropical agriculture. Front. Micr. 2015, 6, 1–10. [Google Scholar]
- Swift, M.J.; Anderson, J.M. Biodiversity and ecosystem function in agricultural systems. In Biodiversity and Ecosystem Function; Schulze, E.D., Mooney, H.A., Eds.; Springer: Berlin, Germany, 1994; pp. 15–41. [Google Scholar]
- Beare, M.H.; Reddy, M.V.; Tian, G.; Srivastava, S.C. Agricultural intensification, soil biodiversity and agroecosystem function in the tropics: The role of decomposer biota. Appl. Soil Ecol. 1997, 6, 87–108. [Google Scholar] [CrossRef]
- Palm, C.; Sanchez, P.; Ahamed, S.; Awiti, A. Soils: A Contemporary Perspective. Annu. Rev. Environ. Resourc. 2007, 32, 99–129. [Google Scholar] [CrossRef]
- Manlay, R.J.; Feller, C.; Swift, M.J. Historical evolution of soil organic matter concepts and their relationships with the fertility and sustainability of cropping systems. Agric. Ecosyst. Environ. 2007, 119, 217–233. [Google Scholar] [CrossRef]
- Mathieu, J.; Rossi, J.P.; Mora, P.; Lavelle, P.; Martins, P.F.D.S.; Rouland, C.; Grimaldi, M. Recovery of soil macrofauna communities after forest clearance in eastern Amazonia, Brazil. Conserv. Biol. 2005, 19, 1598–1605. [Google Scholar] [CrossRef]
- Mutema, M.; Mafongoya, P.L.; Nyagumbo, I.; Chikukura, L. Effects of crop residues and reduced tillage on macrofauna abundance. J. Org. Syst. 2013, 8, 5–16. [Google Scholar]
- Wood, S.A.; Bradford, M.A.; Gilbert, J.A.; McGuire, K.L.; Palm, C.A.; Tully, K.; Zhou, J.; Naeem, S. Agricultural intensification and the functional capacity of soil microbes on smallholder African farms. J. Appl. Ecol. 2015. [Google Scholar] [CrossRef]
- Bradford, M.A.; Wood, S.A.; Bardgett, R.D.; Black, H.I.J.; Bonkowski, M.; Eggers, T.; Grayston, S.J.; Kandeler, E.; Manning, P.; Setala, H.; et al. Discontinuity in the responses of ecosystem processes and multifunctionality to altered soil community composition. Proc. Natl. Acad. Sci. USA 2014, 111, 14478–14483. [Google Scholar] [CrossRef] [PubMed]
- De Vries, F.T.; Bardgett, R.D. Plant–microbial linkages and ecosystem nitrogen retention: lessons for sustainable agriculture. Front. Ecol. Environ. 2012, 10, 425–432. [Google Scholar] [CrossRef]
- R Development Core Team 2008. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2008. [Google Scholar]
- Adejuwon, J.O.; Ekanade, O. A comparison of soil properties under different landuse types in a part of the Nigerian Cocoa Belt. CATENA 1988, 15, 319–331. [Google Scholar] [CrossRef]
- Awiti, A.O.; Walsh, M.G.; Shepherd, K.D.; Kinyamario, J. Soil condition classification using infrared spectroscopy: A proposition for assessment of soil condition along a tropical forest-cropland chronosequence. Geoderma 2008, 143, 73–84. [Google Scholar] [CrossRef]
- Demessie, A.; Singh, B.R.; Lal, R. Soil carbon and nitrogen stocks under chronosequence of farm and traditional agroforestry land uses in Gambo District, Southern Ethiopia. Nutr. Cycl. Agroecosyst. 2013, 95, 365–375. [Google Scholar] [CrossRef]
- Juo, A.S.R.; Franzluebbers, K.; Dabiri, A.; Ikhile, B. Changes in soil properties during long-term fallow and continuous cultivation after forest clearing in Nigeria. Agr. Ecosyst. Environ. 1995, 56, 9–18. [Google Scholar] [CrossRef]
- Karltun, E.; Lemenih, M.; Tolera, M. Comparing farmers’ perception of soil fertility change with soil properties and crop performance in Beseku, Ethiopia. Land Degrad. Dev. 2011, 24, 228–235. [Google Scholar] [CrossRef]
- Kimetu, J.M.; Lehmann, J.; Ngoze, S.O.; Mugendi, D.N.; Kinyangi, J.M.; Riha, S.; Verchot, L.; Recha, J.W.; Pell, A.N. Reversibility of Soil Productivity Decline with Organic Matter of Differing Quality Along a Degradation Gradient. Ecosystems 2008, 11, 726–739. [Google Scholar] [CrossRef]
- Kiunsi, R.B.; Meadows, M.E. Assessing land degradation in the Monduli District, northern Tanzania. Land Degrad. Dev. 2006, 17, 509–525. [Google Scholar] [CrossRef]
- Lal, R. Deforestation and land-use effects on soil degradation and rehabilitation in western Nigeria. II. Soil chemical properties. Land Degrad. Dev. 1998, 7, 87–98. [Google Scholar] [CrossRef]
- Lemenih, M.; Karltun, E.; Olsson, M. Soil organic matter dynamics after deforestation along a farm field chronosequence in southern highlands of Ethiopia. Agric. Ecosyst. Environ. 2005, 109, 9–19. [Google Scholar] [CrossRef]
- Moebius-Clune, B.N.; van Es, H.M.; Idowu, O.J.; Schindelbeck, R.R.; Kimetu, J.M.; Ngoze, S.; Lehmann, J.; Kinyangi, J.M. Long-term soil quality degradation along a cultivation chronosequence in western Kenya. Agr. Ecosyst. Environ. 2011, 141, 86–99. [Google Scholar] [CrossRef]
- Ngoze, S.; Riha, S.; Lehmann, J.; Verchot, L.; Kinyagi, J.; Mbuga, D.; Pell, A. Nutrient constraints to tropical agroecosystem productivity in long-term degrading soils. Global Change Biol. 2008, 14, 2810–2822. [Google Scholar] [CrossRef]
- Nyberg, G.; Bargués Tobella, A.; Kinyangi, J.; Ilstedt, U. Soil property changes over a 120-yr chronosequence from forest to agriculture in western Kenya. Hydrol. Earth Syst. Sci. 2012, 16, 2085–2094. [Google Scholar] [CrossRef]
- Peters, J.B. Gambian soil fertility trends, 1991–1998. Commun. Soil Sci. Plan. 2000, 31, 2201–2210. [Google Scholar] [CrossRef]
- Solomon, D.; Lehmann, J.; Kinyagi, J.; Amelung, W.; Lobe, I.; Pell, A.; Riha, S.; Ngoze, S.; Verchot, L.; Mbuga, D.; et al. Long-term impacts of anthropogenic perturbations on dynamics and speciation of organic carbon in tropical forest and subtropical grassland ecosystems. Global Change Biol. 2007, 13, 511–530. [Google Scholar] [CrossRef]
- Vågen, T.-G.; Shepherd, K.D.; Walsh, M.G. Sensing landscape level change in soil fertility following deforestation and conversion in the highlands of Madagascar using Vis-NIR spectroscopy. Geoderma 2006, 133, 281–294. [Google Scholar] [CrossRef]
- Awiti, A.O.; Walsh, M.G.; Kinyamario, J. Dynamics of topsoil carbon and nitrogen along a tropical forest–cropland chronosequence: Evidence from stable isotope analysis and spectroscopy. Agr. Ecosyst. Environ. 2008, 127, 265–272. [Google Scholar] [CrossRef]
- Sanchez, P.A.; Palm, C.A.; Davey, C.B.; Szott, L.T.; Russell, C.E. Tree Crops as Soil Improvers in the Humid Tropics? In Attributes of Trees as Crop Plants; Cannell, M.G.R., Jackson, J.E., Eds.; Institute of Terrestrial Ecology: Huntingdon, UK, 1985; pp. 327–358. [Google Scholar]
- Russell, C.E. Nutrient Cycling and Productivity of Native and Plantation Forests at Jari Florestal, Pará, Brazil; Institute of Ecology, University of Georgia: Athens, GA, USA, 1983. [Google Scholar]
- Tesfahunegn, G.B.; Tamene, L.; Vlek, P.L.G. A participatory soil quality assessment in Northern Ethiopia’s Mai-Negus catchment. CATENA 2011, 86, 1–13. [Google Scholar] [CrossRef]
- Sanchez, P.A. Properties and Management of Soils in the Tropics; John Wiley & Sons Inc.: Hoboken, NJ, USA, 1976. [Google Scholar]
- Hudson, B. Soil organic matter and available water capacity. J. Soli Water Conserv. 1994, 49, 189–194. [Google Scholar]
- Weil, R.R.; Magdoff, F. Significance of soil organic matter to soil quality and health. In Soil Organic Matter in Sustainable Agriculture; Magdoff, F., Weil, R.R., Eds.; CRC Press: Boca Raton, FL, USA, 2004; pp. 1–42. [Google Scholar]
- Lotter, D.W.; Seidel, R.; Liebhardt, W. The performance of organic and conventional cropping systems in an extreme climate year. Am. J. Alt. Agric. 2003, 18, 146–154. [Google Scholar] [CrossRef]
- Vigiak, O.; Okoba, B.O.; Sterk, G.; Groenenberg, S. Modelling catchment-scale erosion patterns in the East African Highlands. Earth Surf. Process. Landforms 2005, 30, 183–196. [Google Scholar] [CrossRef]
- Tisdall, J.M. Formation of soil aggregates and accumulation of soil organic matter. In Structure and Organic Matter Storage in Agricultural Soils; Carter, M.R., Stewart, B.A., Eds.; CRC Press: Boca Raton, FL, USA, 1996; pp. 57–96. [Google Scholar]
- Wander, M.M.; Traina, S.J.; Stinner, B.R.; Peters, S.E. Organic and Conventional Management Effects on Biologically Active Soil Organic Matter Pools. Soil Sci. Soc. Am. J. 1994, 58, 1130–1139. [Google Scholar] [CrossRef]
- Pierson, F.B.; Blackburn, W.H.; Vactor, S.S.; Wood, J.C. Partitioning small scale spatial variability of runoff and erosion on sagebrush rangeland. J. Am. Water Resour. As. 1994, 30, 1081–1089. [Google Scholar] [CrossRef]
- Blackburn, W.H.; Pierson, F.B. Sources of Variation in Interrill Erosion on Rangelands. In Variability in Rangeland Water Erosion Processes; Blackburn, W.H., Pierson, F.B., Schulman, G.E., Zartman, R., Eds.; SSSA Special Publication; Soil Science Society of America: Madison, WI, USA, 1994; pp. 1–9. [Google Scholar]
- Herrick, J.E.; Whitford, W.G.; de Soyza, A.G.; van Zee, J.W. Field soil aggregate stability kit for soil quality and rangeland health evaluations. CATENA 2001, 44, 27–35. [Google Scholar] [CrossRef]
- Lucas, S.T.; Weil, R.R. Can a Labile Carbon Test be Used to Predict Crop Responses to Improve Soil Organic Matter Management? Agron. J. 2012, 104, 1160–1170. [Google Scholar] [CrossRef]
- Khan, S.A.; Mulvaney, R.L.; Ellsworth, T.R. The potassium paradox: Implications for soil fertility, crop production and human health. Renew. Agric. Food Syst. 2013, 29, 3–27. [Google Scholar] [CrossRef]
- Marín-Spiotta, E.; Silver, W.L.; Ostertag, R. Long-term patterns in tropical reforestation: Plant community composition and aboveground biomass accumulation. Ecol. Appl. 2007, 17, 828–839. [Google Scholar] [CrossRef] [PubMed]
- Murage, E.W.; Karanja, N.K.; Smithson, P.C.; Woomer, P.L. Diagnostic indicators of soil quality in productive and non-productive smallholders’ fields of Kenya’s Central Highlands. Agr Ecosyst. Environ. 2000, 79, 1–8. [Google Scholar] [CrossRef]
- Verdoodt, A.; Mureithi, S.M.; Ye, L.; van Ranst, E. Chronosequence analysis of two enclosure management strategies in degraded rangeland of semi-arid Kenya. Agric. Ecosyst. Environ. 2009, 129, 332–339. [Google Scholar] [CrossRef]
- Fterich, A.; Mahdhi, M.; Mars, M. Impact of grazing on soil microbial communities along a chronosequence of Acacia tortilis subsp. raddiana in arid soils in Tunisia. Eur. J. Soil Biol. 2012, 50, 56–63. [Google Scholar] [CrossRef]
- Dawoe, E.K.; Quashie-Sam, J.S.; Oppong, S.K. Effect of land-use conversion from forest to cocoa agroforest on soil characteristics and quality of a Ferric Lixisol in lowland humid Ghana. Agroforest. Syst. 2013, 88, 87–99. [Google Scholar] [CrossRef]
- Mando, A.; Ouattara, B.; Somado, A.E.; Wopereis, M.C.S.; Stroosnijder, L.; Breman, H. Long-term effects of fallow, tillage and manure application on soil organic matter and nitrogen fractions and on sorghum yield under Sudano-Sahelian conditions. Soil Use Manag. 2005, 21, 25–31. [Google Scholar] [CrossRef]
- Vanlauwe, B.; Diels, J.; Sanginga, N.; Merckx, R. Long-term integrated soil fertility management in South-western Nigeria: Crop performance and impact on the soil fertility status. Plant Soil 2005, 273, 337–354. [Google Scholar] [CrossRef]
- Zingore, S.; Murwira, H.K.; Delve, R.J.; Giller, K.E. Soil type, management history and current resource allocation: Three dimensions regulating variability in crop productivity on African smallholder farms. Field Crops Res. 2007, 101, 296–305. [Google Scholar] [CrossRef]
- Vanlauwe, B.; Bationo, A.; Chianu, J.; Giller, K.E.; Merckx, R.; Mokwunye, U.; Ohiokpehai, O.; Pypers, P.; Tabo, R.; Shepherd, K.D.; et al. Integrated soil fertility management. Outlook Agric. 2010, 39, 17–24. [Google Scholar] [CrossRef]
- Vanlauwe, B.; Giller, K. Popular myths around soil fertility management in sub-Saharan Africa. Agric. Ecosyst. Environ. 2006, 116, 34–46. [Google Scholar] [CrossRef]
- Defoer, T. Learning about methodology development for integrated soil fertility management. Agric. Syst. 2002, 73, 57–81. [Google Scholar] [CrossRef]
- Zingore, S.; Tittonell, P.; Corbeels, M.; van Wijk, M.T.; Giller, K.E. Managing soil fertility diversity to enhance resource use efficiencies in smallholder farming systems: a case from Murewa District, Zimbabwe. Nutr. Cycl. Agroecosyst. 2011, 90, 87–103. [Google Scholar] [CrossRef]
- Chivenge, P.; Vanlauwe, B.; Six, J. Does the combined application of organic and mineral nutrient sources influence maize productivity? A meta-analysis. Plant Soil 2011, 342, 1–30. [Google Scholar] [CrossRef]
- Bationo, A.; Waswa, B.; Kihara, J.; Six, J. Advances in integrated soil fertility management in sub Saharan Africa: Challenges and opportunities. Nutr. Cycl. Agroecosyst. 2006. [Google Scholar] [CrossRef]
- Bot, A.; Benites, J. Conservation agriculture: Case studies in Latin American and Africa; Food & Agriculture Organization: Rome, Italy, 2001. [Google Scholar]
- Erenstein, O.; Sayre, K.; Wall, P.; Dixon, J.; Hellin, J. Adapting No-Tillage Agriculture to the Conditions of Smallholder Maize and Wheat Farmers in the Tropics and Sub-Tropics. Goddard, T., Zoebisch, M., Gan, Y., Ellis, W., Watson, A., Sombatpanit, S., Eds.; In No-till Farming Systems; Special Publication 3 World Association of Soil and Water Conservation (WASWC): Bangkok, Thailand, 2007; pp. 253–274. [Google Scholar]
- Chivenge, P.; Murwira, H.; Giller, K.; Mapfumo, P.; Six, J. Long-term impact of reduced tillage and residue management on soil carbon stabilization: Implications for conservation agriculture on contrasting soils. Soil Till. Res. 2007, 94, 328–337. [Google Scholar] [CrossRef]
- Palm, C.; Blanco-Canqui, H.; DeClerck, F.; Gatere, L.; Grace, P. Conservation agriculture andecosystem services: An overview. Agric. Ecosyst. Environ. 2014, 187, 87–105. [Google Scholar] [CrossRef]
- Verhulst, N.; Govaerts, B.; Verachtert, E.; Castellanos-Navarrete, A.; Mezzalama, M.; Wall, P.; Deckers, J.; Sayre, K. Conservation agriculture, improving soil quality for sustainable production systems? In Food Security and Soil Quality; Lal, R., Stewart, B.A., Eds.; CRC Press: Boca Raton, FL, USA, 2010; pp. 137–208. [Google Scholar]
- Thierfelder, C.; Cheesman, S.; Rusinamhodzi, L. Benefits and challenges of crop rotations in maize-based conservation agriculture (CA) cropping systems of southern Africa. Int. J. Agric. Sustain. 2013, 11, 108–124. [Google Scholar] [CrossRef]
- Nyssen, J.; Poesen, J.; Haile, M.; Moeyersons, J.; Deckers, J. Tillage erosion on slopes with soil conservation structures in the Ethiopian highlands. Soil Till. Res. 2000, 57, 115–127. [Google Scholar] [CrossRef]
- Valentin, C.; Poesen, J.; Li, Y. Gully erosion: Impacts, factors and control. CATENA 2005, 63, 132–153. [Google Scholar] [CrossRef]
- Gebrernichael, D.; Nyssen, J.; Poesen, J.; Deckers, J.; Haile, M.; Govers, G.; Moeyersons, J. Effectiveness of stone bunds in controlling soil erosion on cropland in the Tigray Highlands, northern Ethiopia. Soil Use Manag. 2005, 21, 287–297. [Google Scholar] [CrossRef]
- Astatke, A.; Jabbar, M.; Tanner, D. Participatory conservation tillage research: an experience with minimum tillage on an Ethiopian highland Vertisol. Agric. Ecosyst. Environ. 2003, 95, 401–415. [Google Scholar] [CrossRef]
- Bryan, R.B. Soil Erosion, Land Degradation and Social Transition: Geoecological Analysis of A Semi-Arid Tropical Region, Kenya; Catena-Verlag: Cremlingen Destedt, Germay, 1994. [Google Scholar]
- Beukes, P.C.; Cowling, R.M. Non-Selective Grazing Impacts on Soil-Properties of the Nama Karoo. J. Range Manag. 2003, 56, 547–552. [Google Scholar] [CrossRef]
- Gebremeskel, K.; Pieterse, P.J. Impact of grazing around a watering point on soil status of a semi-arid rangeland in Ethiopia. Afr. J. Ecol. 2007, 45, 72–79. [Google Scholar] [CrossRef]
- Mekuria, W.; Veldkamp, E.; Tilahun, M.; Olschewski, R. Economic valuation of land restoration: The case of exclosures established on communal grazing lands in Tigray, Ethiopia. Land Degrad. Dev. 2011, 22, 334–344. [Google Scholar] [CrossRef]
- Bünemann, E.K.; Smithson, P.C.; Jama, B.; Frossard, E.; Oberson, A.; Oberson, A. Maize productivity and nutrient dynamics in maize-fallow rotations in western Kenya. Plant Soil 2004, 264, 195–208. [Google Scholar] [CrossRef]
- Ojiem, J.O.; Vanlauwe, B.; de Ridder, N.; Giller, K.E. Niche-based assessment of contributions of legumes to the nitrogen economy of Western Kenya smallholder farms. Plant Soil 2007, 292, 119–135. [Google Scholar] [CrossRef]
- Oikeh, S.O.; Carsky, R.J.; Kling, J.G.; Chude, V.O.; Horst, W.J. Differential N uptake by maize cultivars and soil nitrate dynamics under N fertilization in West Africa. Agric. Ecosyst. Environ. 2003, 100, 181–191. [Google Scholar] [CrossRef]
- Adiku, S.G.K.; Jones, J.W.; Kumaga, F.K.; Tonyigah, A. Effects of crop rotation and fallow residue management on maize growth, yield and soil carbon in a savannah-forest transition zone of Ghana. J. Agric. Sci. 2009, 147, 313–322. [Google Scholar] [CrossRef]
- Chikowo, R.; Mapfumo, P.; Nyamugafata, P.; Giller, K.E. Mineral N dynamics, leaching and nitrous oxide losses under maize following two-year improved fallows on a sandy loam soil in Zimbabwe. Plant Soil 2004, 259, 315–330. [Google Scholar] [CrossRef]
- Ndufa, J.K.; Gathumbi, S.M.; Kamiri, H.W.; Giller, K.E.; Cadisch, G. Do mixed-species Legume fallows provide long-term maize yield benefit compared with monoculture legume fallows? Agron. J. 2009, 101, 1352–1362. [Google Scholar] [CrossRef]
- Bado, B.V.; Bationo, A.; Cescas, M.P. Assessment of cowpea and groundnut contributions tosoil fertility and succeeding sorghum yields in the Guinean savannah zone of Burkina Faso (West Africa). Biol. Fert. Soils 2006, 43, 171–176. [Google Scholar] [CrossRef]
- Shepherd, K.D.; Ohlsson, E.; Okalebo, J.R.; Ndufa, J.K. Potential impact of agroforestry on soil nutrient balances at the farm scale in the East African Highlands. Fert. Res. 1996, 44, 87–99. [Google Scholar] [CrossRef]
- Buresh, R.J. Soil improvement by trees in sub-Saharan Africa. Agroforest. Syst. 1998, 38, 51–76. [Google Scholar] [CrossRef]
- Odhiambo, H.O.; Ong, C.K.; Deans, J.D.; Wilson, J.; Khan, A.; Sprent, J.I. Roots, soil water and crop yield: Tree crop interactions in a semi-arid agroforestry system in Kenya. Plant Soil 2001, 235, 221–233. [Google Scholar] [CrossRef] [Green Version]
- Akinnifesi, F.K.; Makumba, W.; Sileshi, G.; Ajayi, O.C.; Mweta, D. Synergistic effect ofinorganic N and P fertilizers and organic inputs from Gliricidia sepium on productivity of intercropped maize in Southern Malawi. Plant Soil 2007, 294, 203–217. [Google Scholar] [CrossRef]
- Van Noordwijk, M.; Lusiana, B. WaNuLCAS, a model of water, nutrient and light capture in agroforestry systems. In Agroforestry for Sustainable Land-Use Fundamental Research and Modelling with Emphasis on Temperate and Mediterranean Applications; Springer Netherlands: Dordrecht, The Netherlands, 1999; Volume 60, pp. 217–242. [Google Scholar]
- Sileshi, G.; Akinnifesi, F.K.; Ajayi, O.C.; Place, F. Meta-analysis of maize yield response to woody and herbaceous legumes in sub-Saharan Africa. Plant Soil 2008, 307, 1–19. [Google Scholar] [CrossRef]
- Nyamadzawo, G.; Nyamugafata, P.; Wuta, M.; Nyamangara, J.; Chikowo, R. Infiltration and runoff losses under fallowing and conservation agriculture practices on contrasting soils, Zimbabwe. Water South Africa 2012, 38, 233–240. [Google Scholar]
- Munodawafa, A. Assessing nutrient losses with soil erosion under different tillage systems and their implications on water quality. Phys. Chem. Earth Pt. A/B/C 2007, 32, 1135–1140. [Google Scholar] [CrossRef]
- Agbede, T. Nutrient availability and cocoyam yield under different tillage practices. Soil Till. Res. 2008, 99, 49–57. [Google Scholar] [CrossRef]
- Enfors, E.; Barron, J.; Makurira, H.; Rockström, J.; Tumbo, S. Yield and soil system changes from conservation tillage in dryland farming: A case study from North Eastern Tanzania. Agric. Water Manag. 2011, 98, 1687–1695. [Google Scholar] [CrossRef]
- Mchunu, C.N.; Lorentz, S.; Jewitt, G.; Manson, A.; Chaplot, V. No-Till Impact on Soil and SoilOrganic Carbon Erosion under Crop Residue Scarcity in Africa. Soil Sci. Soc. Am. J. 2011, 75, 1503–1512. [Google Scholar] [CrossRef]
- Ouédraogo, E.; Mando, A.; Stroosnijder, L. Effects of tillage, organic resources and nitrogen fertiliser on soil carbon dynamics and crop nitrogen uptake in semi-arid West Africa. Soil Till. Res. 2006, 91, 57–67. [Google Scholar] [CrossRef]
- Sanchez, P.A.; Denning, G.L.; Nziguheba, G. The African Green Revolution moves forward. Food Sec. 2009, 1, 37–44. [Google Scholar] [CrossRef]
- Sanchez, P.A. En route to plentiful food production in Africa. Nature Plants 2015, 1, 1–2. [Google Scholar] [CrossRef]
- Alliance for a Green Revolution in Africa (AGRA). Building on the New Momentum in African Agriculture: AGRA in 2008; Alliance for a Green Revolution in Africa: Nairobi, Kenya, 2009. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tully, K.; Sullivan, C.; Weil, R.; Sanchez, P. The State of Soil Degradation in Sub-Saharan Africa: Baselines, Trajectories, and Solutions. Sustainability 2015, 7, 6523-6552. https://doi.org/10.3390/su7066523
Tully K, Sullivan C, Weil R, Sanchez P. The State of Soil Degradation in Sub-Saharan Africa: Baselines, Trajectories, and Solutions. Sustainability. 2015; 7(6):6523-6552. https://doi.org/10.3390/su7066523
Chicago/Turabian StyleTully, Katherine, Clare Sullivan, Ray Weil, and Pedro Sanchez. 2015. "The State of Soil Degradation in Sub-Saharan Africa: Baselines, Trajectories, and Solutions" Sustainability 7, no. 6: 6523-6552. https://doi.org/10.3390/su7066523






