Soil Quality Indices for Evaluating Smallholder Agricultural Land Uses in Northern Ethiopia
Abstract
:1. Introduction
- (1)
- To evaluate effects of F. albida based agroforestry (AF), irrigation based Psidium guajava fruit production (IR) and a tree-less row-crop management (RF) (Figure 1) on selected physical, chemical and biological soil quality indicators and,
- (2)
- To compute an overall integrated soil quality index (SQI) for each land use system and compare among the indices.

2. Materials and Methods
2.1. Descriptions of the Study Site
2.2. Soil Sampling and Analysis
Microbial Biomass Carbon (MBC)
2.3. Soil Quality Assessment
| Function | Weight | Indicator Level 1 | Weight | Indicator Level 2 | Weight | Source for Indicators/Weights |
|---|---|---|---|---|---|---|
| Accommodate Water Entry | 0.20 | WSA | 0.40 | [17,31] | ||
| BD | 0.20 | [17] | ||||
| POR | 0.20 | [12] | ||||
| SOC | 0.20 | [12] | ||||
| Facilitate Water Movement and Availability | 0.20 | POR | 0.60 | [12,17,31] | ||
| SOC | 0.40 | [17,31] | ||||
| Resist Surface Degradation | 0.20 | WSA | 0.60 | [17,31] | ||
| Microbial Processes | 0.40 | MBC | 0.60 | [12,17,31] | ||
| SOC | 0.20 | [12,17,31] | ||||
| TN | 0.20 | [12;31] | ||||
| Supply Plant Nutrient | 0.40 | Nutrient Storage | 0.40 | CEC | 0.40 | [12] |
| SOC | 0.40 | [12] | ||||
| TN | 0.20 | [12] | ||||
| Nutrient Cycling | 0.20 | SOC | 0.40 | [12,31] | ||
| MBC | 0.20 | [12,31] | ||||
| TN | 0.40 | [31] | ||||
| Nutrient Availability | 0.40 | SOC | 0.20 | [12] | ||
| pH | 0.20 | [31] | ||||
| TN | 0.20 | [12] | ||||
| AVP | 0.20 | [12] | ||||
| AVK | 0.20 | [12] |
| Soil Quality Indicator | Weight | Soil Function |
|---|---|---|
| Soil organic carbon | 0.264 | Accommodate water entry |
| Facilitate Water movement and availability | ||
| Resist Surface structure degradation | ||
| Supply plant nutrients | ||
| Aggregate Stability | 0.200 | Accommodate water entry |
| Facilitate Water movement and availability | ||
| Resist surface structure degradation | ||
| Bulk density | 0.040 | Accommodate water entry |
| Porosity | 0.160 | Accommodate water entry |
| Facilitate water movement and availability | ||
| Microbial biomass carbon | 0.064 | Resist surface structure degradation |
| Supply plant nutrients | ||
| Cation exchange capacity | 0.064 | Supply plant nutrients |
| Total Nitrogen | 0.112 | Supply plant nutrients |
| Resist surface structure degradation | ||
| Available phosphorus | 0.032 | Supply plant nutrients |
| Available Potassium | 0.032 | Supply plant nutrients |
| pH | 0.032 | Supply plant nutrients |
| Total | 1.00 |
| Indicator | Scoring Curve | Depth (cm) | LT | UT | LB | UB | OPT | Slope | Source of Threshold/Baseline Values |
|---|---|---|---|---|---|---|---|---|---|
| Physical properties | |||||||||
| BD (Mgm−3) | Less is better | 0–15 cm | 1.0 | 2.0 | 1.5 | - | - | −2.0832 | [31]; Adjacent grass pasture |
| WSA (>0.5 mm) | More is better | 0–10 cm | 0.0 | 40.0 | 20.0 | - | - | 0.0339 | Adjacent grass pasture |
| TP (V%) | Optimum | 0–15 cm | 20.0 | 80.0 | 40.0 | 60.0 | 50.0 | 0.0644 | [12,31]; Adjacent grass pasture |
| Chemical Properties | |||||||||
| CEC (cmol (+) kg−1) | More is better | 0–15 cm | 0.0 | 18.0 | 9.0 | - | - | 0.0757 | [12]; Adjacent grass pasture |
| pH (1:2.5) | Optimum | 0–15 cm | 3.0 | 9.0 | 5.0 | 8.0 | 7.0 | 0.5332; −0.496 | [18] |
| TN (kgha−1) | More is better | 0–15 cm | 0.0 | 2000.0 | 1000.0 | - | - | 0.0007 | [12]; Adjacent grass pasture |
| AVP (kgha−1) | More is better | 0–15 cm | 0.0 | 50.0 | 25.0 | - | - | 0.0226 | [12] |
| AVK (kg·ha−1) | More is better | 0–15 cm | 0.0 | 400.0 | 200.0 | - | - | 0.0036 | [12] |
| Biological Properties | |||||||||
| SOC (gkg−1) | More is better | 0–15 cm | 0.0 | 10.0 | 5.0 | - | - | 0.1341 | [12]; Adjacent grass pasture |
| MBC (mgkg−1) | More is better | 0–15 cm | 0.0 | 300.0 | 150.0 | - | - | 0.0042 | [12]; Adjacent grass pasture |
2.4. Statistical Analyses
3. Results and Discussion
3.1. Soil Physical Quality Indicators
| Soil Quality Indicator | Land Use | |||
|---|---|---|---|---|
| RF | AF | IR | ||
| Physical | ||||
| BD (Mg·m−3) | 1.57 (0.03) | 1.48 (0.05) | 1.57 (0.02) | NS |
| WSA (>0.5 mm) | 11.3 (1.8) | 17.3 (2.5) | 13.6 (3.6) | NS |
| TP (V%) | 35.4 (3.6) | 43.5 (2.0) | 44.9 (2.7) | NS |
| Chemical | ||||
| CEC (cmol (p+) kg−1) | 5.4 (1.0) b | 11.5 (0.8) a | 4.8 (1.8) b | ** |
| pH | 6.6 (0.3) b | 6.4 (0.2) b | 8.0 (0.03) a | *** |
| TN (kg·ha−1) | 809.7 (134.6) b | 1568.6 (85.4) a | 1042.7 (244.6) a,b | * |
| AVP (kg·ha−1) | 24.4 (10.7) | 39.1 (4.3) | 39.8 (4.7) | NS |
| AVK (kg·ha−1) | 216.5 (56.9) b | 1019.1 (161.0) a | 297.7 (71.8) b | *** |
| Biological | ||||
| SOC (g·kg−1) | 3.2 (0.7) b | 6.4 (0.3) a | 5.9 (1.1) a,b | * |
| MBC (mg·kg−1) | 75.5 (24.1) | 95.9 (10.3) | 100.1 (31.3) | NS |
3.2. Soil Chemical Quality Indicators
3.3. Soil Biological Quality Indicators
3.4. Soil Quality Indicators Integration and Assessment
| Soil Function | Land Use | |||
|---|---|---|---|---|
| RF | AF | IR | ||
| Accommodate Water Entry (0.20) | 0.09 (0.00) b | 0.11 (0.002) a | 0.10 (0.004) a,b | * |
| Facilitate Water Entry and Availability (0.20) | 0.10 (0.004) b | 0.12 (0.004) a | 0.11 (0.004) a,b | * |
| Resist Surface Degradation (0.20) | 0.09 (0.003) | 0.11 (0.002) | 0.09 (0.005) | NS |
| Source of Plant Nutrients (0.40) | 0.19 (0.01) b | 0.24 (0.004) a | 0.21 (0.015) a,b | * |
| Integrated Soil Quality Index (1.00) | 0.47 (0.01) b | 0.58 (0.01) a | 0.51 (0.02) a,b | ** |
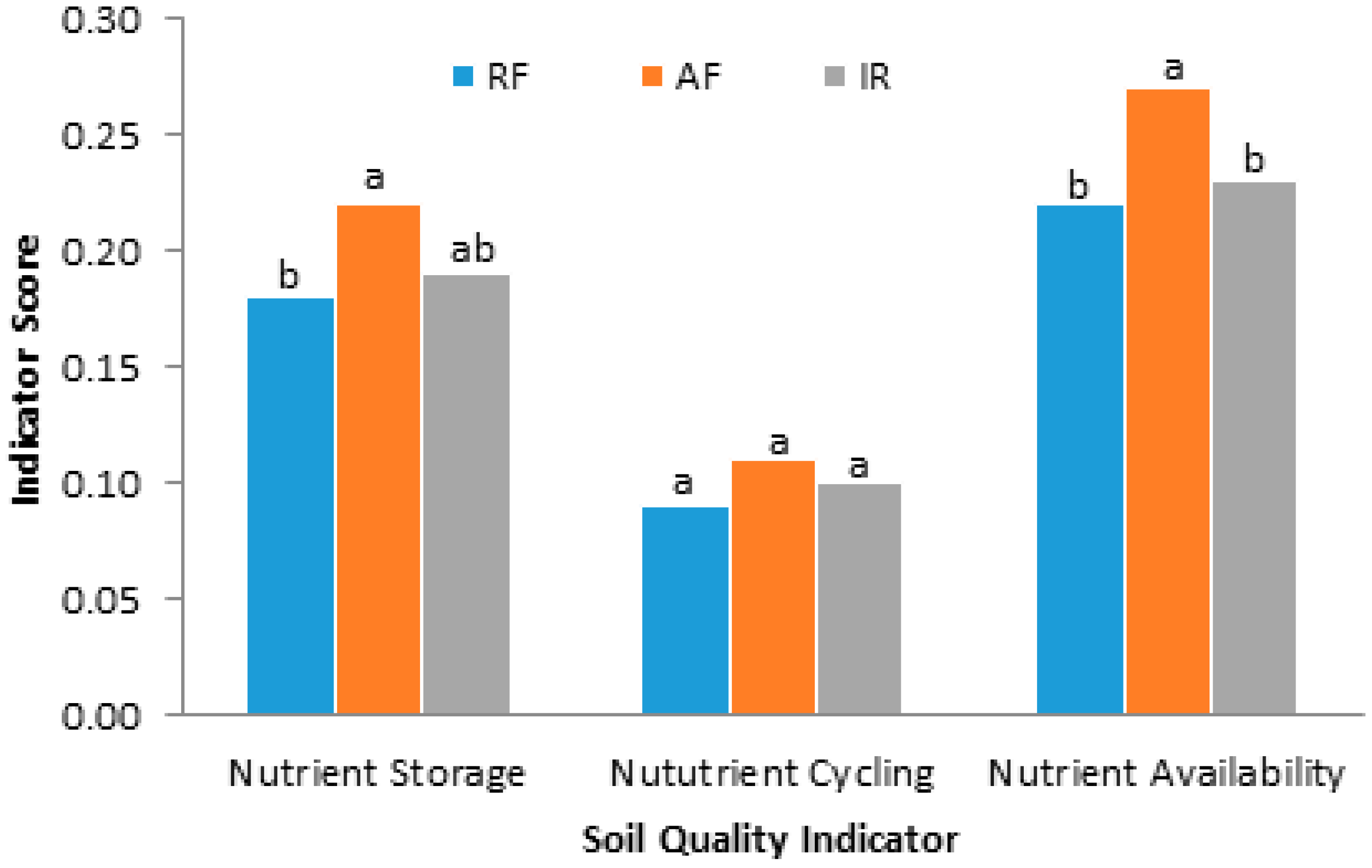
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sanchez, P.A.; Shepherd, K.D.; Soule, M.J.; Place, F.M.; Buresh, R.J.; Izac, A.M.N.; Mokwunye, A.U.; Kwesiga, F.R.; Ndiritu, C.G.; Woomer, P.L.; et al. Soil Fertility Replenishment in Africa: An Investment in Natural Resource Capital. In Replenishing Soil Fertility in Africa; Buresh, R.J., Sanchez, P.A., Calhoun, F., Eds.; SSSA and ICRAF: Madison, WI, USA, 1997; pp. 1–46. [Google Scholar]
- Smaling, E. An Agroecological Framework for Integrating Nutrient Management, with Special Reference to Kenya. Ph.D. Thesis, Agricultural University of Wageningen, Wageningen, The Netherlands, 1993; p. 250. [Google Scholar]
- Agricultural Transformation Agency (ATA). Status of soil resources in Ethiopia and priorities for sustainable management. In Proceedings of the Global Soil Partnership (GSP) for Eastern and Southern Africa Launching Workshop, Nairobi, Kenya, 25–27 March 2013.
- Eweg, H.P.A.; van Lammeren, R.; Deurloo, H.; Woldu, Z. Analysing degradation and rehabilitation for sustainable land management in the highlands of Ethiopia. Land Degrad. Dev. 1998, 9, 529–542. [Google Scholar] [CrossRef]
- Hengsdijk, H.; Meijerink, G.W.; Mosugu, M.E. Modeling the effect of three soil and water conservation practices in Tigray, Ethiopia. Agric. Ecosyst. Environ. 2005, 105, 29–40. [Google Scholar] [CrossRef]
- Belete, T. Efforts for Sustainable Land Management in Tigray: The role of Extension. In Policies for Sustainable Land Management in the Highlands of Tigray, Northern Ethiopia; Gebremedhin, B., Pender, J., Ehui, S., Haile, M., Eds.; International Food Policy Research Institute (IFPRI): Mekele, Ethiopia, 2002. [Google Scholar]
- Girmay, G. Land Use Change Effects in Northern Ethiopia: Runoff, Soil and Nutrient Losses, Soil Quality, and Sediment as Nutrient Sources. PhD Thesis, Norwegian University of Life Sciences, Ås, Norway, 2009; p. 140. [Google Scholar]
- Moges, G.; Hengsdijk, H.; Jansen, H.C. Review and quantitative assessment of ex situ household rainwater harvesting systems in Ethiopia. Agric. Water Manag. 2011, 98, 1215–1227. [Google Scholar] [CrossRef]
- Feng, Z.; Wang, X.; Feng, Z. Soil N and Salinity leaching after the autumn irrigation and its impact on ground water in Hetao irrigation district, China. Agric. Water Manag. 2005, 71, 131–143. [Google Scholar] [CrossRef]
- Hadgu, K.M.; Kooistra, L.; Rossing, W.A.H.; van Bruggen, A.H.C. Assessing the effect of Faidherbia albida based land use systems on barley yield at field and regional scale in the highlands of Tigray, Northern Ethiopia. Food Secur. 2008, 1, 337–350. [Google Scholar] [CrossRef]
- Karlen, D.L.; Mausbach, J.W.; Doran, J.W.; Cline, R.G.; Harris, R.F.; Schuman, G.E. Soil quality: A concept, definition and framework for evaluation. Soil Sci. Soc. Am. J. 1997, 61, 4–10. [Google Scholar] [CrossRef]
- Masto, R.E.; Chhonkar, P.K.; Singh, D.; Patra, A.K. Soil quality response to long-term nutrient and crop management on a semi-arid Inceptisol. Agric. Ecosyst. Environ. 2007, 118, 130–142. [Google Scholar] [CrossRef]
- Brejda, J.J.; Moorman, T.B.; Karlen, D.L.; Dao, T.H. Identification of regional soil quality factors and indicators: I. Central and southern high plains. Soil Sci. Soc. Am. J. 2000, 64, 2115–2124. [Google Scholar] [CrossRef]
- Islam, K.R.; Weil, R.R. Land use effects on soil quality in a tropical forest ecosystem of Bangladesh. Agric. Ecosyst. Environ. 2000, 79, 9–16. [Google Scholar] [CrossRef]
- Sanchez-Maranon, M.; Soriano, M.; Delgado, G.; Delgado, R. Soil quality in Mediterranean mountain environments: Effects of land use change. Soil Sci. Soc. Am. J. 2002, 66, 948–958. [Google Scholar] [CrossRef]
- Karlen, D.L.; Stott, D.E. A framework for evaluating physical and chemical indicators of soil quality. In Defining Soil Quality for a Sustainable Environment; Doran, J.W., Coleman, D.C., Bezdicek, D.F., Stewart, B.A., Eds.; ASA and SSSA: Madison, WI, USA, 1994; pp. 53–72. [Google Scholar]
- Glover, J.D.; Reganold, J.P.; Andrews, P.K. Systematic method for rating soil quality of conventional, organic, and integrated apple orchards in Washington State. Agric. Ecosyst. Environ. 2000, 80, 29–45. [Google Scholar] [CrossRef]
- Fernandes, J.C.; Gamero, C.A.; Rodrigues, J.G.L.; Mirás-Avalos, J.M. Determination of the quality index of a Paleudult under sunflower culture and different management systems. Soil Tillage Res. 2011, 112, 167–174. [Google Scholar] [CrossRef]
- WRB; International Union of Soil Science Working Group. World Soil Resources Reports No. 103; FAO: Rome, Italy, 2006. [Google Scholar]
- Rabia, A.H.; Afifi, R.R.; Gelaw, A.M.; Bianchi, S.; Figueredo, H.; Huong, T.L.; Lopez, A.A.; Mandala, S.D.; Matta, E.; Ronchi, M.; et al. Soil mapping and classification: A case study in the Tigray Region, Ethiopia. JAEID 2013, 107, 73–99. [Google Scholar]
- Blake, G.R.; Hartge, K.H. Bulk density. In Methods of Soil Analysis. Part 1, 2nd ed.; Klute, A., Ed.; American Society of Agronomy—Soil Science Society of America: Madison, WI, USA, 1986; pp. 363–375. [Google Scholar]
- Nelson, D.W.; Sommers, L.E. Total carbon, Organic carbon and Organic matter. In Methods of Soil Analysis. Part 3; Book Series 5; Sparks, D.L., Page, A.L., Helmke, P.A., Loeppert, R.H., Soltanpour, P.N., Tabatabai, M.A., Johnston, C.T., Sumner, M.E., Eds.; SSSA: Madison, WI, USA, 1996; pp. 961–1010. [Google Scholar]
- Yoder, R.E. A direct method of aggregate analysis of soils and a study of the physical nature of erosion losses. J. Am. Soc. Agron. 1936, 28, 337–351. [Google Scholar] [CrossRef]
- Olsen, S.R.; Cole, C.V.; Watanabe, F.S.; Dean, L.A. Estimation of Available Phosphorous in Soils by Extraction with Sodium Bicarbonate; USDA: Washington, DC, USA, 1954. [Google Scholar]
- Chapman, H.D. Cation exchange capacity. In Methods of Soil Analysis; Black, C.A., Evans, D.D., Ensminger, L.E., White, J.L., Clark, F.E., Eds.; American Society of Agronomy: Madison, WI, USA, 1965; Volume 9, pp. 891–901. [Google Scholar]
- Brookes, P.C.; Landman, A.; Pruden, G. Chloroform fumigation and release of soil N: A rapid direct extraction method to measure microbial biomass N in soil. Soil Biol. Biochem. 1985, 17, 837–842. [Google Scholar] [CrossRef]
- Vance, E.D.; Brookes, P.C.; Jenkinson, D.S. An extraction method for measuring soil microbial biomass C. Soil Biol. Biochem. 1987, 19, 703–707. [Google Scholar] [CrossRef]
- Wu, J.; Joergensen, R.G.; Pommerening, B.; Chaussod, R.; Brookes, P.C. Measurement of soil microbial biomass Carbon by fumigation-extraction an automated procedure. Soil Biol. Biochem. 1990, 22, 1167–1169. [Google Scholar] [CrossRef]
- Wienhold, B.J.; Karlen, D.L.; Andrews, S.S.; Stott, D.E. Protocol for Soil Management Assessment Framework (SMAF) soil indicator scoring curve development. Renew. Agric. Food Syst. 2009, 24, 260–266. [Google Scholar] [CrossRef]
- Andrews, S.S.; Karlen, D.L.; Cambardella, C.A. The soil management assessment framework: A quantitative soil quality evaluation method. Soil Sci. Soc. Am. J. 2004, 68, 1945–1962. [Google Scholar] [CrossRef]
- Karlen, D.L.; Wollenhaupt, N.C.; Erbach, D.C.; Berry, E.C.; Swan, J.B.; Eash, N.S.; Jordahl, J.L. Residue effects on soil quality following 10-years of no-till corn. Soil Tillage Res. 1994, 31, 149–167. [Google Scholar] [CrossRef]
- Karlen, D.L.; Wollenhaupt, N.C.; Erbach, D.C.; Berry, E.C.; Swan, J.B.; Eash, N.S.; Jordahl, J.L. Long-term tillage effects on soil quality. Soil Tillage Res. 1994, 32, 313–327. [Google Scholar] [CrossRef]
- Wymore, A.W. Model-Based Systems Engineering. In An Introduction to the Mathematical Theory of Discrete Systems and to the Tricotyledon Theory of System Design; CRC: Boca Raton, FL, USA, 1993. [Google Scholar]
- Hussain, I.; Olson, K.R.; Wander, M.M.; Karlen, D.L. Adaptation of soil quality indices and application to three tillage systems in southern Illinois. Soil Tillage Res. 1999, 50, 237–249. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing: Vienna, Austria, 2012. Available online: http://www.R-project.org. (Accessed on 9 March 2014).
- Tisdale, J.M.; Oades, J.M. Organic matter and water stable aggregates in soils. J. Soil Sci. 1982, 33, 141–161. [Google Scholar] [CrossRef]
- Gelaw, A.M.; Singh, B.R.; Lal, R. Organic carbon and nitrogen associated with soil aggregates and particle sizes under different land uses in Tigray, Northern Ethiopia. Land Degrad. Dev. 2013. [Google Scholar] [CrossRef]
- Hartemink, A.E.; Huting, J. Land Cover, Extent, and Properties of Arenosols in Southern Africa. Arid Land Res. Manag. 2008, 22, 134–147. [Google Scholar] [CrossRef]
- Arevalo, L.A.; Alegre, J.C.; Bandy, D.E.; Szott, L.T. The effect of cattle grazing on soil physical and chemical properties in a silvopastoral system in the Peruvian Amazon. Agrofor. Syst. 1998, 40, 109–124. [Google Scholar] [CrossRef]
- Sanchez, P.A. Science in agroforestry. Agrofor. Syst. 1995, 30, 5–55. [Google Scholar] [CrossRef]
- Nair, P.K.R. An Introduction to Agroforestry; Kluwer Academic Publishers: London, UK, 1993; p. 489. [Google Scholar]
- Franchini, J.C.; Crispino, C.C.; Souza, R.A.; Torres, E.; Hungria, M. Microbiological parameters as indicators of soil quality under various soil management and crop rotation systems in southern Brazil. Soil Tillage Res. 2007, 92, 18–29. [Google Scholar] [CrossRef]
- Stott, D.E.; Cambardella, C.A.; Karlen, D.L.; Harmel, R.D. A Soil Quality and Metabolic Activity Assessment after Fifty-Seven Years of Agricultural Management. Soil Sci. Soc. Am. J. 2013, 77, 903–913. [Google Scholar] [CrossRef]
- Girmay, G.; Singh, B.R.; Mitiku, H.; Borresen, T.; Lal, R. Carbon Stocks in Ethiopian Soils in Relation to Land Use and Soil Management. Land Degrad. Dev. 2008, 19, 351–367. [Google Scholar] [CrossRef]
- Sanchez, P.A. Tropical soil fertility research, towards the second paradigm. In Transactions 15th World Congress of Soil Science; International Society of Soil Science and Mexican Society of Soil Science: Acapulco, Mexico, 1994; Volume 1, pp. 65–88. [Google Scholar]
- Palm, C.A. Contribution of agroforestry trees to nutrient requirements of intercropped plants. Agrofor. Syst. 1995, 30, 105–124. [Google Scholar] [CrossRef]
- Poschen, P. An evaluation of the Acacia albida-based agroforestry practices in the Hararghe highlands of Eastern Ethiopia. Agrofor. Syst. 1986, 4, 129–143. [Google Scholar] [CrossRef]
- Kamara, C.S.; Haque, I. Faidherbia albida and its effects on Ethiopian highland Vertisols. Agrofor. Syst. 1992, 18, 17–29. [Google Scholar] [CrossRef]
- Asfaw, Z.; Ågren, G.I. Farmers’ local knowledge and topsoil properties of agroforestry practices in Sidama, Southern Ethiopia. Agrofor. Syst. 2007, 71, 35–48. [Google Scholar] [CrossRef]
- Kwesiga, F.; Coe, R. The effect of short-rotation Sesbania sesban planted fallows on maize yields. For. Ecol. Manag. 1994, 64, 199–208. [Google Scholar] [CrossRef]
- Sanchez, P.A.; Palm, C.A. Nutrient cycling and agroforestry in Africa. Unasylva 1996, 47, 24–28. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gelaw, A.M.; Singh, B.R.; Lal, R. Soil Quality Indices for Evaluating Smallholder Agricultural Land Uses in Northern Ethiopia. Sustainability 2015, 7, 2322-2337. https://doi.org/10.3390/su7032322
Gelaw AM, Singh BR, Lal R. Soil Quality Indices for Evaluating Smallholder Agricultural Land Uses in Northern Ethiopia. Sustainability. 2015; 7(3):2322-2337. https://doi.org/10.3390/su7032322
Chicago/Turabian StyleGelaw, Aweke M., B. R. Singh, and R. Lal. 2015. "Soil Quality Indices for Evaluating Smallholder Agricultural Land Uses in Northern Ethiopia" Sustainability 7, no. 3: 2322-2337. https://doi.org/10.3390/su7032322
APA StyleGelaw, A. M., Singh, B. R., & Lal, R. (2015). Soil Quality Indices for Evaluating Smallholder Agricultural Land Uses in Northern Ethiopia. Sustainability, 7(3), 2322-2337. https://doi.org/10.3390/su7032322







