Transdermal Permeation of Drugs in Various Animal Species
Abstract
:1. Introduction
2. Skin Characteristics
3. Lipid Content
4. Hair Follicles
5. Metabolism
6. Intra- and Inter-Individual Variation
7. Effect of Skin Thickness
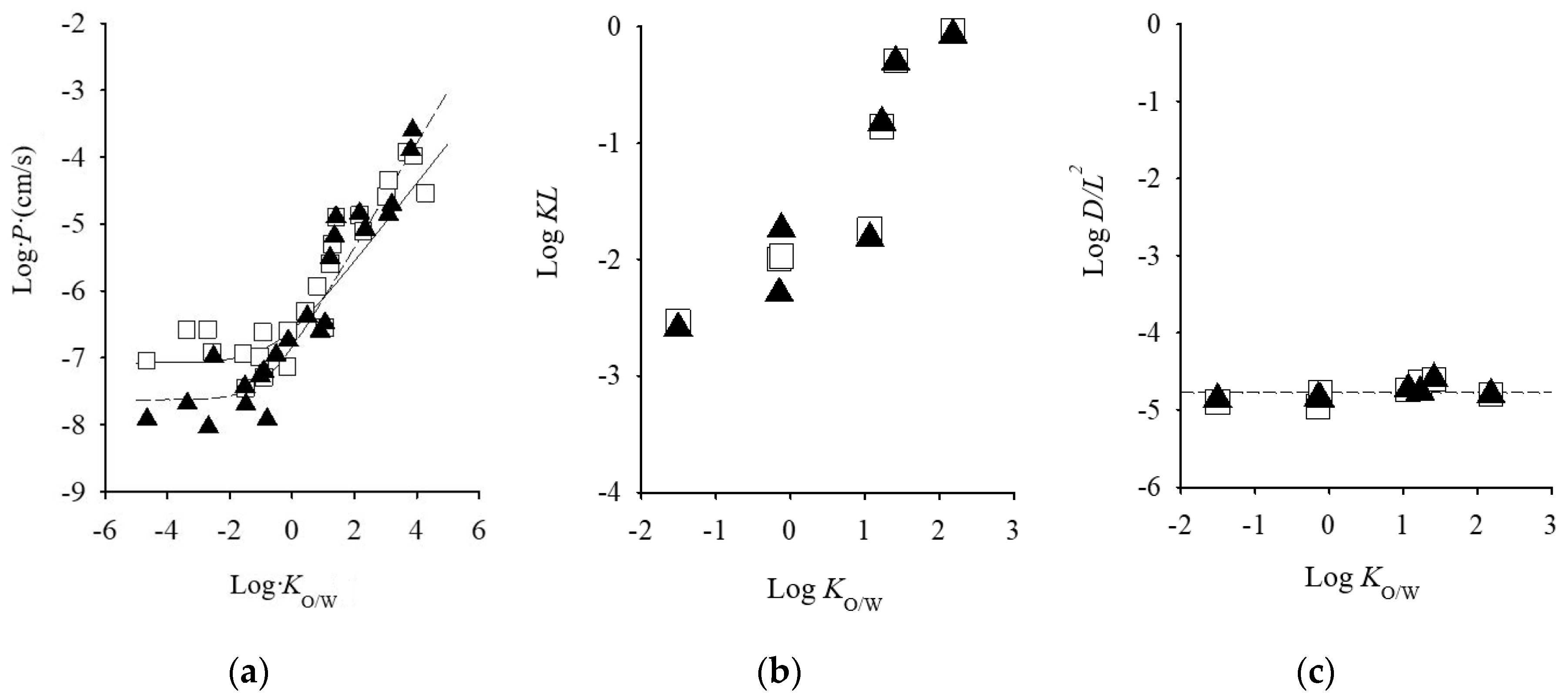
8. Permeation Parameter
9. Electrical Conductivity
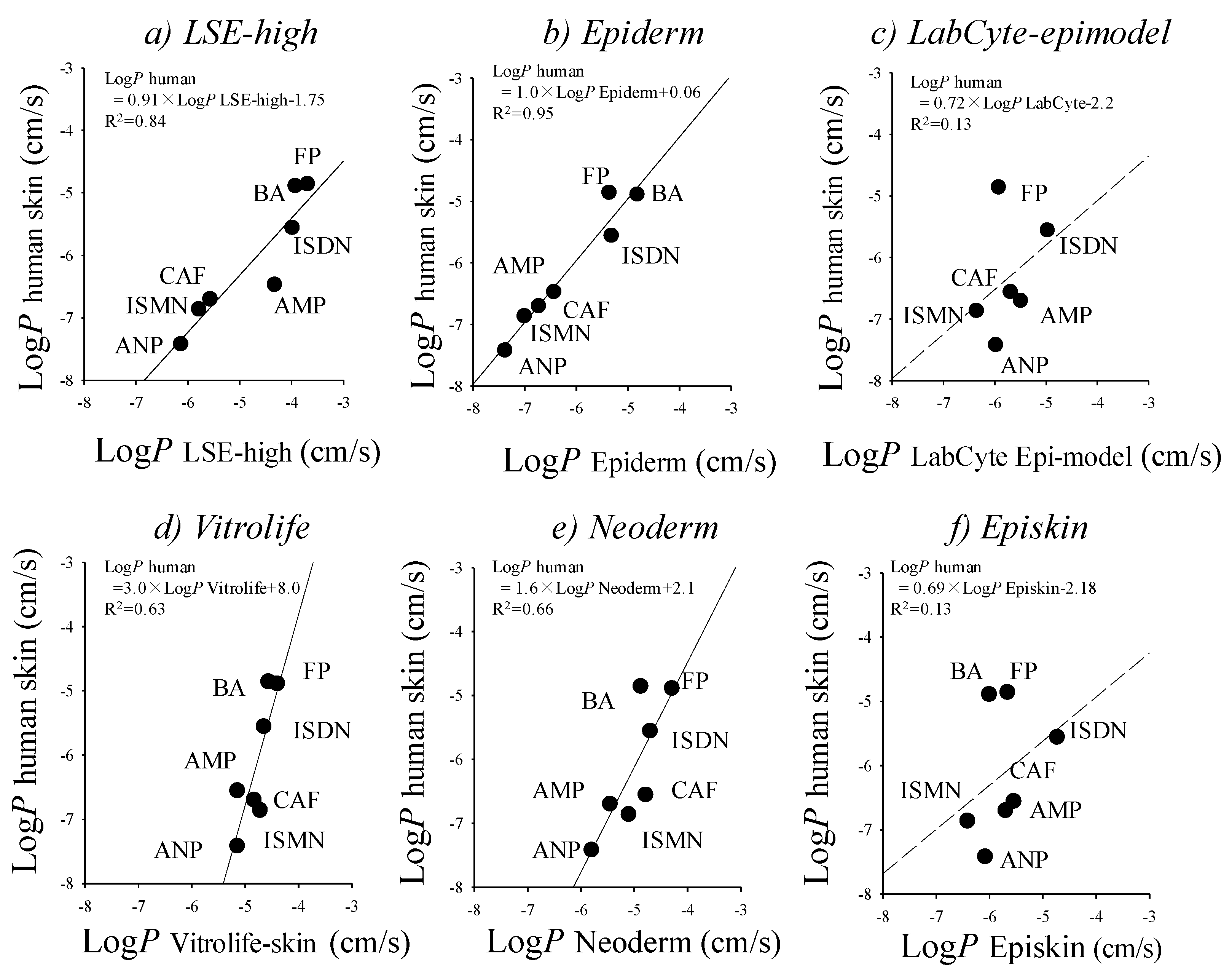
10. Cultured Human Skin Models
11. Conclusions
Conflicts of Interest
References
- Barbero, A.M.; Frasch, H.F. Pig and guinea pig skin as surrogates for human in vitro penetration studies: A quantitative review. Toxicol. Vitr. 2009, 23, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Bronaugh, R.L.; Stewart, R.F.; Congdon, E.R. Methods for in vitro percutaneous absorption studies II. Animal models for human skin. Toxicol. Appl. Pharmacol. 1982, 62, 481–488. [Google Scholar] [CrossRef]
- El Maghraby, G.M.; Barry, B.W.; Williams, A.C. Liposomes and skin: From drug delivery to model membranes. Eur. J. Pharm. Sci. 2008, 34, 203–222. [Google Scholar] [CrossRef] [PubMed]
- Godin, B.; Touitou, E. Transdermal skin delivery: Predictions for humans from in vivo, ex vivo and animal models. Adv. Drug Deliv. Rev. 2007, 59, 1152–1161. [Google Scholar] [CrossRef] [PubMed]
- Panchagnula, R.; Stemmer, K.; Ritschel, W.A. Animal models for transdermal drug delivery. Methods Find. Exp. Clin. Pharmacol. 1997, 19, 335–341. [Google Scholar] [PubMed]
- Qvist, M.H.; Hoeck, U.; Kreilgaard, B.; Madsen, F.; Frokjaer, S. Evaluation of Gottingen minipig skin for transdermal in vitro permeation studies. Eur. J. Pharm. Sci. 2000, 11, 59–68. [Google Scholar] [CrossRef]
- Sato, K.; Sugibayashi, K.; Morimoto, Y. Species differences in percutaneous absorption of nicorandil. J. Pharm. Sci. 1991, 80, 104–107. [Google Scholar] [CrossRef] [PubMed]
- Schmook, F.P.; Meingassner, J.G.; Billich, A. Comparison of human skin or epidermis models with human and animal skin in in vitro percutaneous absorption. Int. J. Pharm. 2001, 215, 51–56. [Google Scholar] [CrossRef]
- Singh, S.; Zhao, K.; Singh, J. In vitro permeability and binding of hydrocarbons in pig ear and human abdominal skin. Drug Chem. Toxicol. 2002, 25, 83–92. [Google Scholar] [CrossRef] [PubMed]
- Southwell, D.; Barry, B.W.; Woodford, R. Variations in permeability of human skin within and between specimens. Int. J. Pharm. 1984, 18, 299–309. [Google Scholar] [CrossRef]
- Nakamura, A.; Mori, D.; Tojo, K. Evaluation of the Predicted Time—Concentration Profile of Serum Tulobuterol in Human after Transdermal Application. Chem. Pharm. Bull. 2012, 60, 300–305. [Google Scholar] [CrossRef] [PubMed]
- Hatanaka, T.; Yoshida, S.; Kadhum, W.R.; Todo, H.; Sugibayashi, K. In Silico Estimation of Skin Concentration Following the Dermal Exposure to Chemicals. Pharm. Res. 2015, 32, 3965–3974. [Google Scholar] [CrossRef] [PubMed]
- Sugibayashi, K.; Todo, H.; Oshizaka, T.; Owada, Y. Mathematical model to predict skin concentration of drugs: Toward utilization of silicone membrane to predict skin concentration of drugs as an animal testing alternative. Pharm. Res. 2010, 27, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Oshizaka, T.; Kikuchi, K.; Kadhum, W.R.; Todo, H.; Hatanaka, T.; Wierzba, K.; Sugibayashi, K. Estimation of skin concentrations of topically applied lidocaine at each depth profile. Int. J. Pharm. 2014, 475, 292–297. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Polak, S.; Ghobadi, C.; Mishra, H.; Ahamadi, M.; Patel, N.; Jamei, M.; Rostami-Hodjegan, A. Prediction of concentration-time profile and its inter-individual variability following the dermal drug absorption. J. Pharm. Sci. 2012, 101, 2584–2595. [Google Scholar] [CrossRef] [PubMed]
- Sugibayashi, K.; Hayashi, T.; Matsumoto, K.; Hasegawa, T. Utility of a three-dimensional cultured human skin model as a tool to evaluate the simultaneous diffusion and metabolism of ethyl nicotinate in skin. Drug Metab. Pharmacokinet. 2004, 19, 352–362. [Google Scholar] [CrossRef] [PubMed]
- Nicoli, S.; Padula, C.; Aversa, V.; Vietti, B.; Wertz, P.W.; Millet, A.; Falson, F.; Govoni, P.; Santi, P. Characterization of rabbit ear skin as a skin model for in vitro transdermal permeation experiments: Histology, lipid composition and permeability. Skin Pharmacol. Physiol. 2008, 21, 218–226. [Google Scholar] [CrossRef] [PubMed]
- Jung, E.C.; Maibach, H.I. Animal models for percutaneous absorption. J. Appl. Toxicol. 2015, 35, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Ya-Xian, Z.; Suetake, T.; Tagami, H. Number of cell layers of the stratum corneum in normal skin—Relationship to the anatomical location on the body, age, sex and physical parameters. Arch. Dermatol. Res. 1999, 291, 555–559. [Google Scholar] [CrossRef] [PubMed]
- Gray, G.M.; Yardley, H.J. Lipid compositions of cells isolated from pig, human, and rat epidermis. J. Lipid Res. 1975, 16, 434–440. [Google Scholar] [PubMed]
- Wester, R.C.; Melendres, J.; Sedik, L.; Maibach, H.; Riviere, J.E. Percutaneous absorption of salicylic acid, theophylline, 2,4-dimethylamine, diethyl hexyl phthalic acid, and p-aminobenzoic acid in the isolated perfused porcine skin flap compared to man in vivo. Toxicol. Appl. Pharmacol. 1998, 151, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Simon, G.A.; Maibach, H.I. The pig as an experimental animal model of percutaneous permeation in man: Qualitative and quantitative observations—An overview. Skin Pharmacol. Appl. Skin Physiol. 1982, 13, 229–234. [Google Scholar] [CrossRef]
- Barber, E.D.; Teetsel, N.M.; Kolberg, K.F.; Guest, D. A comparative study of the rates of in vitro percutaneous absorption of eight chemicals using rat and human skin. Fundam. Appl. Toxicol. 1992, 19, 493–497. [Google Scholar] [CrossRef]
- Chowhan, Z.T.; Pritchard, R. Effect of surfactants on percutaneous absorption of naproxen I: Comparisons of rabbit, rat, and human excised skin. J. Pharm. Sci. 1978, 67, 1272–1274. [Google Scholar] [CrossRef] [PubMed]
- Hughes, M.F.; Edwards, B.C. In vitro dermal absorption of pyrethroid pesticides in human and rat skin. Toxicol. Appl. Pharmacol. 2010, 246, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Van Ravenzwaay, B.; Leibold, E. A comparison between in vitro rat and human and in vivo rat skin absorption studies. Hum. Exp. Toxicol. 2004, 23, 421–430. [Google Scholar] [CrossRef] [PubMed]
- Tomomi, H.; Masami, I.; Kenji, S.; Yasunori, M. Prediction of skin permeability of drugs. II. Development of composite membrane as a skin alternative. Int. J. Pharm. 1992, 79, 21–28. [Google Scholar] [CrossRef]
- Watanabe, T.; Hasegawa, T.; Takahashi, H.; Ishibashi, T.; Takayama, K.; Sugibayashi, K. Utility of the Three-dimensional Cultured Human Skin Model as a Tool to Evaluate Skin Permeation of Drugs. Altern. Anim. Test. Exp. 2001, 8, 1–14. [Google Scholar]
- Kim, M.K.; Lee, C.H.; Kim, D.D. Skin permeation of testosterone and its ester derivatives in rats. J. Pharm. Pharmacol. 2000, 52, 369–375. [Google Scholar] [CrossRef] [PubMed]
- Krishnaiah, Y.S.R.; Satyanarayana, V.; Karthikeyan, R.S. Effect of the solvent system on the in vitro permeability of nicardipine hydrochloride through excised rat epidermis. J. Pharm. Pharm. Sci. 2002, 5, 123–130. [Google Scholar] [PubMed]
- Obata, Y.; Sato, H.; Li, C.J.; Takayama, K.; Higashiyama, K.; Nagai, T.; Isowa, K. Effect of synthesized cyclohexanol derivatives using l-menthol as a lead compound on the percutaneous absorption of ketoprofen. Int. J. Pharm. 2000, 198, 191–200. [Google Scholar] [CrossRef]
- Wang, A.; Seki, T.; Yuan, D.; Saso, Y.; Hosoya, O.; Chono, S.; Morimoto, K. Effect of camellia oil on the permeation of flurbiprofen and diclofenac sodium through rat and pig skin. Biol. Pharm. Bull. 2004, 27, 1476–1479. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, H.; Mano, Y.; Terasaka, S.; Sakurai, T.; Furuya, A.; Urano, H.; Sugibayashi, K. Usefulness of rat skin as a substitute for human skin in the in vitro skin permeation study. Exp. Anim. 2011, 60, 373–384. [Google Scholar] [CrossRef] [PubMed]
- Kano, S.; Todo, H.; Sugie, K.; Fujimoto, H.; Nakada, K.; Tokudome, Y.; Hashimoto, F.; Sugibayashi, K. Utilization of Reconstructed Cultured Human Skin Models as an Alternative Skin for Permeation Studies of Chemical Compounds. Altern. Anim. Test. Exp. AATEX 2010, 15, 61–70. [Google Scholar]
- Elias, P.M.; Friend, D.S. The permeability barrier in mammalian epidermis. J. Cell Biol. 1975, 65, 180–191. [Google Scholar] [CrossRef] [PubMed]
- Lampe, M.A.; Williams, M.L.; Elias, P.M. Human epidermal lipids: Characterization and modulations during differentiation. J. Lipid Res. 1983, 24, 131–140. [Google Scholar] [PubMed]
- McIntosh, T.J.; Stewart, M.E.; Downing, D.T. X-ray diffraction analysis of isolated skin lipids: Reconstitution of intercellular lipid domains. Biochemistry 1996, 35, 3649–3653. [Google Scholar] [CrossRef] [PubMed]
- Elias, P.M. Epidermal lipids, membranes, and keratinization. Int. J. Dermatol. 1981, 20, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Williams, M.L.; Elias, P.M. The extracellular matrix of stratum corneum: Role of lipids in normal and pathological function. Crit. Rev. Ther. Drug Carr. Syst. 1987, 3, 95–122. [Google Scholar]
- Stahl, J.; Niedorf, F.; Kietzmann, M. Characterisation of epidermal lipid composition and skin morphology of animal skin ex vivo. Eur. J. Pharm. Biopharm. 2009, 72, 310–316. [Google Scholar] [CrossRef] [PubMed]
- Boncheva, M.; Damien, F.; Normand, V. Molecular organization of the lipid matrix in intact Stratum corneum using ATR-FTIR spectroscopy. Biochim. Biophys. Acta Biomembr. 2008, 1778, 1344–1355. [Google Scholar] [CrossRef] [PubMed]
- Horita, D.; Hatta, I.; Yoshimoto, M.; Kitao, Y.; Todo, H.; Sugibayashi, K. Molecular mechanisms of action of different concentrations of ethanol in water on ordered structures of intercellular lipids and soft keratin in the stratum corneum. Biochim. Biophys. Acta Biomembr. 2015, 1848, 1196–1202. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hatta, I.; Nakazawa, H.; Obata, Y.; Ohta, N.; Inoue, K.; Yagi, N. Novel method to observe subtle structural modulation of stratum corneum on applying chemical agents. Chem. Phys. Lipids 2010, 163, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Hatta, I.; Ohta, N.; Inoue, K.; Yagi, N. Coexistence of two domains in intercellular lipid matrix of stratum corneum. Biochim. Biophys. Acta Biomembr. 2006, 1758, 1830–1836. [Google Scholar] [CrossRef] [PubMed]
- Caussin, J.; Gooris, G.S.; Janssens, M.; Bouwstra, J.A. Lipid organization in human and porcine stratum corneum differs widely, while lipid mixtures with porcine ceramides model human stratum corneum lipid organization very closely. Biochim. Biophys. Acta 2008, 1778, 1472–1482. [Google Scholar] [CrossRef] [PubMed]
- Otberg, N.; Richter, H.; Schaefer, H.; Blume-Peytavi, U.; Sterry, W.; Lademann, J. Variations of Hair Follicle Size and Distribution in Different Body Sites. J. Investig. Dermatol. 2004, 122, 14–19. [Google Scholar] [CrossRef] [PubMed]
- Blume, U.; Ferracin, J.; Verschoore, M.; Czernielewski, J.M.; Schaefer, H. Physiology of the vellus hair follicle: Hair growth and sebum excretion. Br. J. Dermatol. 1991, 124, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Knorr, F.; Lademann, J.; Patzelt, A.; Sterry, W.; Blume-Peytavi, U.; Vogt, A. Follicular transport route—Research progress and future perspectives. Eur. J. Pharm. Biopharm. 2009, 71, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Mangelsdorf, S.; Vergou, T.; Sterry, W.; Lademann, J.; Patzelt, A. Comparative study of hair follicle morphology in eight mammalian species and humans. Skin Res. Technol. 2014, 20, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Kao, J.; Carver, M.P. Cutaneous metabolism of xenobiotics. Drug Metab. Rev. 1990, 22, 363–410. [Google Scholar] [CrossRef] [PubMed]
- Baron, J.M.; Höller, D.; Schiffer, R.; Frankenberg, S.; Neis, M.; Merk, H.F.; Jugert, F.K. Expression of multiple cytochrome p450 enzymes and multidrug resistance-associated transport proteins in human skin keratinocytes. J. Investig. Dermatol. 2001, 116, 541–548. [Google Scholar] [CrossRef] [PubMed]
- Swanson, H.I. Cytochrome P450 expression in human keratinocytes: An aryl hydrocarbon receptor perspective. Chem. Biol. Interact. 2004, 149, 69–79. [Google Scholar] [CrossRef] [PubMed]
- Oesch, F.; Fabian, E.; Oesch-Bartlomowicz, B.; Werner, C.; Landsiedel, R. Drug-Metabolizing Enzymes in the Skin of Man, Rat, and Pig. Drug Metab. Rev. 2007, 39, 659–698. [Google Scholar] [CrossRef] [PubMed]
- Eilstein, J.; Léreaux, G.; Budimir, N.; Hussler, G.; Wilkinson, S.; Duche, D. Comparison of xenobiotic metabolizing enzyme activities in ex vivo human skin and reconstructed human skin models from SkinEthic. Arch. Toxicol. 2014, 88, 1681–1694. [Google Scholar] [CrossRef] [PubMed]
- Eilstein, J.; Léreaux, G.; Arbey, E.; Daronnat, E.; Wilkinson, S.; Duche, D. Xenobiotic metabolizing enzymes in human skin and SkinEthic reconstructed human skin models. Exp. Dermatol. 2015, 24, 547–549. [Google Scholar] [CrossRef] [PubMed]
- Akomeah, F.K.; Martin, G.P.; Brown, M.B. Variability in human skin permeability in vitro: Comparing penetrants with different physicochemical properties. J. Pharm. Sci. 2007, 96, 824–834. [Google Scholar] [CrossRef] [PubMed]
- Schäfer-Korting, M.; Bock, U.; Diembeck, W.; Düsing, H.J.; Gamer, A.; Haltner-Ukomadu, E.; Hoffmann, C.; Kaca, M.; Kamp, H.; Kersen, S.; et al. The use of reconstructed human epidermis for skin absorption testing: Results of the validation study. ATLA Altern. Lab. Anim. 2008, 36, 161–187. [Google Scholar] [PubMed]
- Ngawhirunpat, T.; Panomsuk, S.; Opanasopit, P.; Rojanarata, T.; Hatanaka, T. Comparison of the percutaneous absorption of hydrophilic and lipophilic compounds in shed snake skin and human skin. Pharmazie 2006, 61, 331–335. [Google Scholar] [PubMed]
- Zorin, S.; Kuylenstierna, F.; Thulin, H. In vitro test of nicotine’s permeability through human skin. Risk evaluation and safety aspects. Ann. Occup. Hyg. 1999, 43, 405–413. [Google Scholar] [CrossRef]
- Fujii, M.; Yamanouchi, S.; Hori, N.; Iwanaga, N.; Naruko, K.; Matsumoto, M. Evaluation of Yucatan micropig skin for Use as an in vitro model for skin permeation study. Biol. Pharm. Bull. 1997, 20, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Príborský, J.; Mühlbachová, E. Evaluation of in vitro percutaneous absorption across human skin and in animal models. J. Pharm. Pharmacol. 1990, 42, 468–472. [Google Scholar] [CrossRef] [PubMed]
- Sato, K.; Sugibayashi, K.; Morimoto, Y.; Omiya, H.; Enomoto, N. Prediction of the In-vitro Human Skin Permeability of Nicorandil from Animal Data. J. Pharm. Pharmacol. 1989, 41, 379–383. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, K.; Mitsui, T.; Aso, Y.; Sugibayashi, K. Structure-permeability relationship analysis of the permeation barrier properties of the stratum corneum and viable epidermis/dermis of rat skin. J. Pharm. Sci. 2008, 97, 4391–4403. [Google Scholar] [CrossRef] [PubMed]
- Hatanaka, T.; Inuma, M.; Sugibayashi, K.; Morimoto, Y. Prediction of Skin Permeability of Drugs. I. Comparison with Artificial Membrane. Chem. Pharm. Bull. 1990, 38, 3452–3459. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, H.; Terasaka, S.; Sakurai, T.; Furuya, A.; Urano, H.; Sugibayashi, K. Variation assessment for in vitro permeabilities through Yucatan micropig skin. Biol. Pharm. Bull. 2011, 34, 555–561. [Google Scholar] [CrossRef] [PubMed]
- Van de Sandt, J.J.M.; Meuling, W.J.A.; Elliott, G.R.; Cnubben, N.H.P.; Hakkert, B.C. Comparative In Vitro—In Vivo Percutaneous Absorption of the Pesticide Propoxur. Toxicol. Sci. 2000, 58, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Cnubben, N.H.; Elliott, G.R.; Hakkert, B.C.; Meuling, W.J.; van de Sandt, J.J. Comparative in vitro-in vivo percutaneous penetration of the fungicide ortho-phenylphenol. Regul. Toxicol. Pharmacol. 2002, 35, 198–208. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, S.C.; Maas, W.J.M.; Nielsen, J.B.; Greaves, L.C.; van de Sandt, J.J.M.; Williams, F.M. Interactions of skin thickness and physicochemical properties of test compounds in percutaneous penetration studies. Int. Arch. Occup. Environ. Health 2006, 79, 405–413. [Google Scholar] [CrossRef] [PubMed]
- Morimoto, Y.; Hatanaka, T.; Sugibayashi, K.; Omiya, H. Prediction of skin permeability of drugs: Comparison of human and hairless rat skin. J. Pharm. Pharmacol. 1992, 44, 634–639. [Google Scholar] [CrossRef] [PubMed]
- Kano, S.; Hiroaki, T.; Furui, K.; Sugie, K.; Tokudome, Y.; Hashimoto, F.; Kojima, H.; Sugibayashi, K. Comparison of Several Reconstructed Cultured Human Skin Models by Microscopic Observation: Their Usefulness as an Alternative Membrane for Skin in Drug Permeation Experiments. Altern. Anim. Test. Exp. AATEX 2011, 16, 51–58. [Google Scholar]
- Hawkins, G.S.; Reifenrath, W.G. Influence of skin source, penetration cell fluid, and partition coefficient on in vitro skin penetration. J. Pharm. Sci. 1986, 75, 378–811. [Google Scholar] [CrossRef] [PubMed]
- Davies, D.J.; Ward, R.J.; Heylings, J.R. Multi-species assessment of electrical resistance as a skin integrity marker for in vitro percutaneous absorption studies. Toxicol. In Vitro 2004, 18, 351–358. [Google Scholar] [CrossRef] [PubMed]
- Karande, P.; Jain, A.; Mitragotri, S. Relationships between skin’s electrical impedance and permeability in the presence of chemical enhancers. J. Control. Release 2006, 110, 307–313. [Google Scholar] [CrossRef] [PubMed]
- Sekkat, N.; Kalia, Y.N.; Guy, R.H. Porcine ear skin as a model for the assessment of transdermal drug delivery to premature neonates. Pharm. Res. 2004, 21, 1390–1397. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, S.; Mahmoud, A.; Vuia, A.; Rübbelke, M.K.; Schmidt, E.; Schaller, M.; Kandárová, H.; Haberland, A.; Schäfer, U.F.; Bock, U.; et al. Reconstructed epidermis versus human and animal skin in skin absorption studies. Toxicol. In Vitro 2005, 19, 813–822. [Google Scholar] [CrossRef] [PubMed]
- Abd, E.; Yousef, S.A.; Pastore, M.N.; Telaprolu, K.; Mohammed, Y.H.; Namjoshi, S.; Grice, J.E.; Roberts, M.S. Skin models for the testing of transdermal drugs. Clin. Pharmacol. Adv. Appl. 2016, 8, 163–176. [Google Scholar] [CrossRef] [PubMed]
| Species | SC (μm) | Epidermis (μm) | Whole Skin (mm) | Reference |
|---|---|---|---|---|
| Human forearm | 17 | 36 | 1.5 | [18] |
| Human | 16.8 | 46.9 | 2.97 | [18] |
| Human | 18.2 ± 3.3 | 51.2 ± 12.2 | 2.58 ± 0.07 | [7] |
| Pig, back | 26 | 66 | 3.4 | [18] |
| Pig ear | 10 | 50 | 1.3 | [18] |
| Pig | 26.4 | 65.8 | 3.43 | [18] |
| Pig | 17.5 ± 2.4 | 50.7 ± 11.4 | 1.74 ± 0.18 | [7] |
| Guinea pig | 18.6 ± 1.2 | 20.8 ± 1.4 | 1.15 ± 0.07 | [7] |
| Mouse, back | 5 | 13 | 0.8 | [18] |
| Hairless mouse | 8.8 ± 1.0 | 18.0 ± 1.5 | 0.41 ± 0.02 | [7] |
| Rat | 18 | 32 | 2.09 | [18] |
| Hairless rat | 8.9 | 28.6 | 0.70 | [18] |
| Hairless rat | 15.4 ± 3.3 | 28.3 ± 5.3 | 0.86 ± 0.06 | [7,17,18] |
© 2017 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Todo, H. Transdermal Permeation of Drugs in Various Animal Species. Pharmaceutics 2017, 9, 33. https://doi.org/10.3390/pharmaceutics9030033
Todo H. Transdermal Permeation of Drugs in Various Animal Species. Pharmaceutics. 2017; 9(3):33. https://doi.org/10.3390/pharmaceutics9030033
Chicago/Turabian StyleTodo, Hiroaki. 2017. "Transdermal Permeation of Drugs in Various Animal Species" Pharmaceutics 9, no. 3: 33. https://doi.org/10.3390/pharmaceutics9030033





