Playing Hide and Seek: How Glycosylation of the Influenza Virus Hemagglutinin Can Modulate the Immune Response to Infection
Abstract
:1. Introduction
2. IAVs Show Marked Differences in Glycosylation on the Head of the Viral HA
| Subtype | Virus | Stem | Head | Stem | GenBank/GISAID Acc No | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| H3N2 | H3 numbering | 22 | 38 | 63 | 81 | 126 | 133 | 165 | 246 | 276 | 285 | ||||||
| NGT | NAT | NCT | NET | NWT | NGT | NVT | NST | NCS | NGS | ||||||||
| A/Hong Kong/1/1968 * | + | + | + | + | + | CY044261 | |||||||||||
| A/Bilthoven/1761/1976 | + | + | + | + | + | + | CY113197 | ||||||||||
| A/Netherlands/620/1989 | + | + | + | + | + | + | + | CY113421 | |||||||||
| A/Shandong/9/1993 | + | + | + | + | + | + | + | + | CY108274 | ||||||||
| A/Panama/2007/1999 | + | + | + | + | + | + | + | + | CY112917 | ||||||||
| A/Brisbane/10/2007 | + | + | + | + | + | + | + | + | EPI353304 | ||||||||
| A/Victoria/361/2011 * | + | + | + | + | + | + | + | + | EPI349103 | ||||||||
| H1N1 | H3 numbering | 21 | 33 | 63 | 65 | 95 | 123 | 129 | 130 | 158 | 162/163 | 165 | 271 | 278 | 288/289 | ||
| NST | NVT | NCS | NIT | NGT | NTS | NHT | NTT | NGS | NLS | NNS | NAS | NTT | NSS | ||||
| A/South Carolina/1/1918 * | + | + | + | + | AF117241 | ||||||||||||
| A/Wilson-Smith/1933 | + | + | + | + | DQ508905 | ||||||||||||
| A/Bellamy/1942 | + | + | + | + | + | + | HQ008263 | ||||||||||
| A/USSR/90/1977 * | + | + | + | + | + | + | + | + | + | DQ508897 | |||||||
| A/Victoria/36/1988 | + | + | + | + | + | + | JX477163 | ||||||||||
| A/New Caledonia/20/1999 | + | + | + | + | + | + | + | DQ508857 | |||||||||
| A/Brisbane/59/2007 * | + | + | + | + | + | + | CY030230 | ||||||||||
| A/California/07/2009 * | + | + | + | + | + | EPI273609 | |||||||||||
| A/Townsville/64/2010 | + | + | + | + | + | + | EPI294411 | ||||||||||
| A/Perth/500/2010 | + | + | + | + | + | + | EPI269967 | ||||||||||
| H2N2 | H3 numbering | 21 | 33 | 169 | 170 | 289 | |||||||||||
| NST | NVT | NNT | NTS | NTT | |||||||||||||
| A/Japan/305/1957 * | + | + | + | + | CY014976 | ||||||||||||
| A/Ann Arbor/7/1967 | + | + | + | + | + | CY125838 | |||||||||||
| H5N1 | H3 numbering | 21 | 33 | 158 | 169 | 219 | 289 | ||||||||||
| NST | NVT | NST | NNT | NRS | NSS | ||||||||||||
| A/Vietnam/1194/2004 * | + | + | + | + | + | GQ149237 | |||||||||||
| A/Cambodia/W0112303/2012 | + | + | + | + | + | JQ714246 | |||||||||||
| A/Indonesia/5/2005 | + | + | + | + | + | CY116646 | |||||||||||
| A/Turkey/12/2006 | + | + | + | + | EF619982 | ||||||||||||
| A/Egypt/321/2007 | + | + | + | + | EPI173707 | ||||||||||||
| A/Guizhou/1/2013 | + | + | + | + | + | EPI420386 | |||||||||||
| H7N9 | H3 numbering | 22 | 38 | 240 | |||||||||||||
| NGT | NAT | NDT | |||||||||||||||
| A/Anhui/1/2013 * | + | + | + | EPI439507 | |||||||||||||
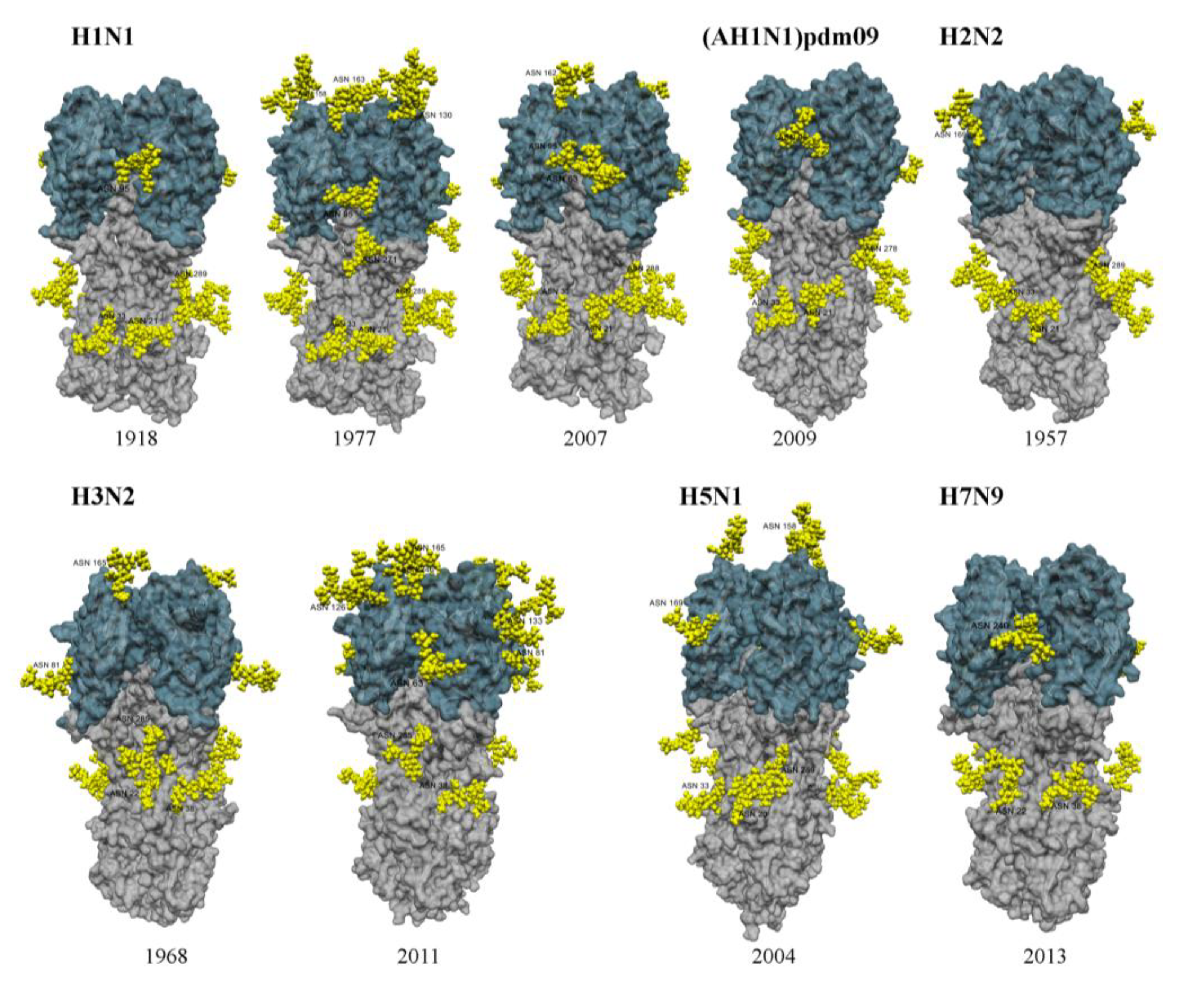
3. HA Glycosylation Determines Sensitivity of IAV to Lectins of the Innate Immune System
3.1. Soluble C-Type Lectins
3.2. Membrane-Associated C-Type Lectins
4. HA Glycosylation and Immune Evasion by IAV
4.1. HA Glycosylation Alters Ab-Mediated Recognition of Antigenic Epitopes
4.2. Effects of HA Glycosylation on T Cell-Mediated Immunity to IAV
5. Impact of HA Glycosylation on IAV Biology
5.1. Impact of HA Glycosylation on HA Receptor Avidity and Virus Fitness
5.2. Balancing the Impact of HA Glycosylation on Innate and Adaptive Immunity
6. Conclusions
Acknowledgments
Author Contributions
Conflict of Interest
References and Notes
- Kornfeld, R.; Kornfeld, S. Assembly of asparagine-linked oligosaccharides. Annu. Rev. Biochem. 1985, 54, 631–664. [Google Scholar] [CrossRef]
- Deom, C.M.; Schulze, I.T. Oligosaccharide composition of an influenza virus hemagglutinin with host-determined binding properties. J. Biol. Chem. 1985, 260, 14771–14774. [Google Scholar]
- Nakamura, K.; Compans, R.W. Host cell- and virus strain-dependent differences in oligosaccharides of hemagglutinin glycoproteins of influenza a viruses. Virology 1979, 95, 8–23. [Google Scholar] [CrossRef]
- Basak, S.; Pritchard, D.G.; Bhown, A.S.; Compans, R.W. Glycosylation sites of influenza viral glycoproteins: Characterization of tryptic glycopeptides from the a/ussr(h1n1) hemagglutinin glycoprotein. J. Virol. 1981, 37, 549–558. [Google Scholar]
- Ward, C.W.; Dopheide, T.A. Amino acid sequence and oligosaccharide distribution of the haemagglutinin from an early hong kong influenza virus variant a/aichi/2/68 (x-31). Biochem. J. 1981, 193, 953–962. [Google Scholar]
- Compans, R.W.; Pinter, A. Incorporation of sulfate into influenza virus glycoproteins. Virology 1975, 66, 151–160. [Google Scholar] [CrossRef]
- Karaivanova, V.K.; Spiro, R.G. Sulphation of n-linked oligosaccharides of vesicular stomatitis and influenza virus envelope glycoproteins: Host cell specificity, subcellular localization and identification of substituted saccharides. Biochem. J. 1998, 329, 511–518. [Google Scholar]
- Kaverin, N.V.; Klenk, H.D. Strain-specific differences in the effect of influenza a virus neuraminidase on vector-expressed hemagglutinin. Arch. Virol. 1999, 144, 781–786. [Google Scholar] [CrossRef]
- Basak, S.; Tomana, M.; Compans, R.W. Sialic acid is incorporated into influenza hemagglutinin glycoproteins in the absence of viral neuraminidase. Virus Res. 1985, 2, 61–68. [Google Scholar] [CrossRef]
- Job, E.R.; Deng, Y.M.; Barfod, K.K.; Tate, M.D.; Caldwell, N.; Reddiex, S.; Maurer-Stroh, S.; Brooks, A.G.; Reading, P.C. Addition of glycosylation to influenza a virus hemagglutinin modulates antibody-mediated recognition of h1n1 2009 pandemic viruses. J. Immunol. 2013, 190, 2169–2177. [Google Scholar] [CrossRef]
- Blake, T.A.; Williams, T.L.; Pirkle, J.L.; Barr, J.R. Targeted n-linked glycosylation analysis of h5n1 influenza hemagglutinin by selective sample preparation and liquid chromatography/tandem mass spectrometry. Anal. Chem. 2009, 81, 3109–3118. [Google Scholar] [CrossRef]
- Kasturi, L.; Eshleman, J.R.; Wunner, W.H.; Shakin-Eshleman, S.H. The hydroxy amino acid in an asn-x-ser/thr sequon can influence n-linked core glycosylation efficiency and the level of expression of a cell surface glycoprotein. J. Biol. Chem. 1995, 270, 14756–14761. [Google Scholar]
- Mellquist, J.L.; Kasturi, L.; Spitalnik, S.L.; Shakin-Eshleman, S.H. The amino acid following an asn-x-ser/thr sequon is an important determinant of n-linked core glycosylation efficiency. Biochemistry 1998, 37, 6833–6837. [Google Scholar] [CrossRef]
- Sun, S.; Wang, Q.; Zhao, F.; Chen, W.; Li, Z. Glycosylation site alteration in the evolution of influenza a (h1n1) viruses. PLoS One 2011, 6, e22844. [Google Scholar]
- Abe, Y.; Takashita, E.; Sugawara, K.; Matsuzaki, Y.; Muraki, Y.; Hongo, S. Effect of the addition of oligosaccharides on the biological activities and antigenicity of influenza a/h3n2 virus hemagglutinin. J. Virol. 2004, 78, 9605–9611. [Google Scholar] [CrossRef]
- Schulze, I.T. Effects of glycosylation on the properties and functions of influenza virus hemagglutinin. J. Infect. Dis. 1997, 176, S24–S28. [Google Scholar]
- Wagner, R.; Wolff, T.; Herwig, A.; Pleschka, S.; Klenk, H.D. Interdependence of hemagglutinin glycosylation and neuraminidase as regulators of influenza virus growth: A study by reverse genetics. J. Virol. 2000, 74, 6316–6323. [Google Scholar] [CrossRef]
- Roberts, P.C.; Garten, W.; Klenk, H.D. Role of conserved glycosylation sites in maturation and transport of influenza a virus hemagglutinin. J. Virol. 1993, 67, 3048–3060. [Google Scholar]
- Daniels, R.; Kurowski, B.; Johnson, A.E.; Hebert, D.N. N-linked glycans direct the cotranslational folding pathway of influenza hemagglutinin. Mol. Cell 2003, 11, 79–90. [Google Scholar] [CrossRef]
- Nobusawa, E.; Aoyama, T.; Kato, H.; Suzuki, Y.; Tateno, Y.; Nakajima, K. Comparison of complete amino acid sequences and receptor-binding properties among 13 serotypes of hemagglutinins of influenza a viruses. Virology 1991, 182, 475–485. [Google Scholar] [CrossRef]
- Ohuchi, R.; Ohuchi, M.; Garten, W.; Klenk, H.D. Oligosaccharides in the stem region maintain the influenza virus hemagglutinin in the metastable form required for fusion activity. J. Virol. 1997, 71, 3719–3725. [Google Scholar]
- Gallagher, P.J.; Henneberry, J.M.; Sambrook, J.F.; Gething, M.J. Glycosylation requirements for intracellular transport and function of the hemagglutinin of influenza virus. J. Virol. 1992, 66, 7136–7145. [Google Scholar]
- Blackburne, B.P.; Hay, A.J.; Goldstein, R.A. Changing selective pressure during antigenic changes in human influenza h3. PLoS Pathog. 2008, 4, e1000058. [Google Scholar] [CrossRef]
- Cherry, J.L.; Lipman, D.J.; Nikolskaya, A.; Wolf, Y.I. Evolutionary dynamics of n-glycosylation sites of influenza virus hemagglutinin. PLoS Curr. Influenza 2009, RRN1001. [Google Scholar]
- Das, S.R.; Puigbo, P.; Hensley, S.E.; Hurt, D.E.; Bennink, J.R.; Yewdell, J.W. Glycosylation focuses sequence variation in the influenza a virus h1 hemagglutinin globular domain. PLoS Pathog. 2010, 6, e1001211. [Google Scholar] [CrossRef]
- Igarashi, M.; Ito, K.; Kida, H.; Takada, A. Genetically destined potentials for n-linked glycosylation of influenza virus hemagglutinin. Virology 2008, 376, 323–329. [Google Scholar] [CrossRef]
- Zhang, M.; Gaschen, B.; Blay, W.; Foley, B.; Haigwood, N.; Kuiken, C.; Korber, B. Tracking global patterns of n-linked glycosylation site variation in highly variable viral glycoproteins: Hiv, siv, and hcv envelopes and influenza hemagglutinin. Glycobiology 2004, 14, 1229–1246. [Google Scholar] [CrossRef]
- Palese, P.; Wang, T.T. Why do influenza virus subtypes die out? A hypothesis. MBio 2011, 2, e00150-11. [Google Scholar]
- Job, E.R.; Deng, Y.M.; Tate, M.D.; Bottazzi, B.; Crouch, E.C.; Dean, M.M.; Mantovani, A.; Brooks, A.G.; Reading, P.C. Pandemic h1n1 influenza a viruses are resistant to the antiviral activities of innate immune proteins of the collectin and pentraxin superfamilies. J. Immunol. 2010, 185, 4284–4291. [Google Scholar] [CrossRef]
- Reid, A.H.; Fanning, T.G.; Hultin, J.V.; Taubenberger, J.K. Origin and evolution of the 1918 "Spanish" Influenza virus hemagglutinin gene. Proc. Natl. Acad. Sci. USA 1999, 96, 1651–1656. [Google Scholar] [CrossRef]
- Wei, C.J.; Boyington, J.C.; Dai, K.; Houser, K.V.; Pearce, M.B.; Kong, W.P.; Yang, Z.Y.; Tumpey, T.M.; Nabel, G.J. Cross-neutralization of 1918 and 2009 influenza viruses: Role of glycans in viral evolution and vaccine design. Sci. Transl. Med. 2010, 2, 24ra21. [Google Scholar]
- Medina, R.A.; Stertz, S.; Manicassamy, B.; Zimmermann, P.; Sun, X.; Albrecht, R.A.; Uusi-Kerttula, H.; Zagordi, O.; Belshe, R.B.; Frey, S.E.; et al. Glycosylations in the globular head of the hemagglutinin protein modulate the virulence and antigenic properties of the h1n1 influenza viruses. Sci. Transl. Med. 2013, 5, 187ra170. [Google Scholar]
- Tsuchiya, E.; Sugawara, K.; Hongo, S.; Matsuzaki, Y.; Muraki, Y.; Li, Z.N.; Nakamura, K. Antigenic structure of the haemagglutinin of human influenza a/h2n2 virus. J. Gen. Virol. 2001, 82, 2475–2484. [Google Scholar]
- Tsuchiya, E.; Sugawara, K.; Hongo, S.; Matsuzaki, Y.; Muraki, Y.; Li, Z.N.; Nakamura, K. Effect of addition of new oligosaccharide chains to the globular head of influenza a/h2n2 virus haemagglutinin on the intracellular transport and biological activities of the molecule. J. Gen. Virol. 2002, 83, 1137–1146. [Google Scholar]
- NetNGlyc. Available online: http://www.cbs.dtu.dk/services/NetNGlyc/ (accessed on 14 November 2013).
- Glyprot. Available online: http://ww.glycosciences.de/modeling/glyprot/ (accessed on 4 December 2013).
- Persistence of Vision Raytracer, version 3.7; Persistence of Vision Pty. Ltd: Williamstown, Victoria, Australia, 2004. Available online: http://www.povray.org/ (accessed on 4 December 2013).
- Chen, W.; Zhong, Y.; Qin, Y.; Sun, S.; Li, Z. The evolutionary pattern of glycosylation sites in influenza virus (h5n1) hemagglutinin and neuraminidase. PLoS One 2012, 7, e49224. [Google Scholar]
- Panaampon, J.; Ngaosuwankul, N.; Suptawiwat, O.; Noisumdaeng, P.; Sangsiriwut, K.; Siridechadilok, B.; Lerdsamran, H.; Auewarakul, P.; Pooruk, P.; Puthavathana, P. A novel pathogenic mechanism of highly pathogenic avian influenza h5n1 viruses involves hemagglutinin mediated resistance to serum innate inhibitors. PLoS One 2012, 7, e36318. [Google Scholar] [CrossRef]
- Kageyama, T.; Fujisaki, S.; Takashita, E.; Xu, H.; Yamada, S.; Uchida, Y.; Neumann, G.; Saito, T.; Kawaoka, Y.; Tashiro, M. Genetic analysis of novel avian a(h7n9) influenza viruses isolated from patients in china, february to april 2013. Euro. Surveill. 2013, 18, 20453. [Google Scholar]
- Gottschalk, A.; Belyavin, G.; Biddle, F. Glycoproteins as influenza virus hemagglutinin inhibitors and as cellular receptors. In Glycoproteins. Their Composition, Structure and Function; Gottschalk, A., Ed.; Elsevier Science Publishing: New York, NY, USA, 1972; pp. 1082–1096. [Google Scholar]
- Ng, W.C.; Tate, M.D.; Brooks, A.G.; Reading, P.C. Soluble host defense lectins in innate immunity to influenza virus. J. Biomed. Biotechnol. 2012, 2012, 732191. [Google Scholar]
- Anders, E.M.; Hartley, C.A.; Jackson, D.C. Bovine and mouse serum beta inhibitors of influenza a viruses are mannose-binding lectins. Proc. Natl. Acad. Sci. USA 1990, 87, 4485–4489. [Google Scholar] [CrossRef]
- Hartley, C.A.; Jackson, D.C.; Anders, E.M. Two distinct serum mannose-binding lectins function as beta inhibitors of influenza virus: Identification of bovine serum beta inhibitor as conglutinin. J. Virol. 1992, 66, 4358–4363. [Google Scholar]
- Drickamer, K. C-type lectin-like domains. Curr. Opin. Struct. Biol. 1999, 9, 585–590. [Google Scholar] [CrossRef]
- Crouch, E.C. Collectins and pulmonary host defense. Am. J. Respir. Cell Mol. Biol. 1998, 19, 177–201. [Google Scholar] [CrossRef]
- Reading, P.C.; Morey, L.S.; Crouch, E.C.; Anders, E.M. Collectin-mediated antiviral host defense of the lung: Evidence from influenza virus infection of mice. J. Virol. 1997, 71, 8204–8212. [Google Scholar]
- Tate, M.D.; Schilter, H.C.; Brooks, A.G.; Reading, P.C. Responses of mouse airway epithelial cells and alveolar macrophages to virulent and avirulent strains of influenza a virus. Viral. Immunol. 2011, 24, 77–88. [Google Scholar] [CrossRef]
- Tate, M.D.; Brooks, A.G.; Reading, P.C. Inhibition of lectin-mediated innate host defences in vivo modulates disease severity during influenza virus infection. Immunol. Cell Biol. 2011, 89, 482–491. [Google Scholar] [CrossRef]
- Hartshorn, K.L.; White, M.R.; Mogues, T.; Ligtenberg, T.; Crouch, E.; Holmskov, U. Lung and salivary scavenger receptor glycoprotein-340 contribute to the host defense against influenza a viruses. Am. J. Physiol. Lung. Cell Mol. Physiol. 2003, 285, L1066–L1076. [Google Scholar]
- White, M.R.; Tecle, T.; Crouch, E.C.; Hartshorn, K.L. Impact of neutrophils on antiviral activity of human bronchoalveolar lavage fluid. Am. J. Physiol. Lung. Cell Mol. Physiol. 2007, 293, L1293–L1299. [Google Scholar] [CrossRef]
- Fidler, K.J.; Hilliard, T.N.; Bush, A.; Johnson, M.; Geddes, D.M.; Turner, M.W.; Alton, E.W.; Klein, N.J.; Davies, J.C. Mannose-binding lectin is present in the infected airway: A possible pulmonary defence mechanism. Thorax 2009, 64, 150–155. [Google Scholar] [CrossRef]
- LeVine, A.M.; Whitsett, J.A.; Hartshorn, K.L.; Crouch, E.C.; Korfhagen, T.R. Surfactant protein d enhances clearance of influenza a virus from the lung in vivo. J. Immunol. 2001, 167, 5868–5873. [Google Scholar]
- Hawgood, S.; Brown, C.; Edmondson, J.; Stumbaugh, A.; Allen, L.; Goerke, J.; Clark, H.; Poulain, F. Pulmonary collectins modulate strain-specific influenza a virus infection and host responses. J. Virol. 2004, 78, 8565–8572. [Google Scholar] [CrossRef]
- Vigerust, D.J.; Ulett, K.B.; Boyd, K.L.; Madsen, J.; Hawgood, S.; McCullers, J.A. N-linked glycosylation attenuates h3n2 influenza viruses. J. Virol. 2007, 81, 8593–8600. [Google Scholar] [CrossRef]
- Zhang, L.; Hartshorn, K.L.; Crouch, E.C.; Ikegami, M.; Whitsett, J.A. Complementation of pulmonary abnormalities in sp-d(-/-) mice with an sp-d/conglutinin fusion protein. J. Biol. Chem. 2002, 277, 22453–22459. [Google Scholar]
- Chang, W.C.; White, M.R.; Moyo, P.; McClear, S.; Thiel, S.; Hartshorn, K.L.; Takahashi, K. Lack of the pattern recognition molecule mannose-binding lectin increases susceptibility to influenza a virus infection. BMC Immunol. 2010, 11, 64. [Google Scholar] [CrossRef]
- Hansen, S.; Selman, L.; Palaniyar, N.; Ziegler, K.; Brandt, J.; Kliem, A.; Jonasson, M.; Skjoedt, M.O.; Nielsen, O.; Hartshorn, K.; et al. Collectin 11 (cl-11, cl-k1) is a masp-1/3-associated plasma collectin with microbial-binding activity. J. Immunol. 2010, 185, 6096–6104. [Google Scholar] [CrossRef]
- Hartshorn, K.L.; Holmskov, U.; Hansen, S.; Zhang, P.; Meschi, J.; Mogues, T.; White, M.R.; Crouch, E.C. Distinctive anti-influenza properties of recombinant collectin 43. Biochem. J. 2002, 366, 87–96. [Google Scholar]
- Hartshorn, K.L.; White, M.R.; Smith, K.; Sorensen, G.; Kuroki, Y.; Holmskov, U.; Head, J.; Crouch, E.C. Increasing antiviral activity of surfactant protein d trimers by introducing residues from bovine serum collectins: Dissociation of mannan-binding and antiviral activity. Scand. J. Immunol. 2010, 72, 22–30. [Google Scholar]
- Hartshorn, K.L.; Sastry, K.; Brown, D.; White, M.R.; Okarma, T.B.; Lee, Y.M.; Tauber, A.I. Conglutinin acts as an opsonin for influenza a viruses. J. Immunol. 1993, 151, 6265–6273. [Google Scholar]
- Tate, M.D.; Brooks, A.G.; Reading, P.C. Specific sites of n-linked glycosylation on the hemagglutinin of h1n1 subtype influenza a virus determine sensitivity to inhibitors of the innate immune system and virulence in mice. J. Immunol. 2011, 187, 1884–1894. [Google Scholar] [CrossRef]
- Tate, M.D.; Job, E.R.; Brooks, A.G.; Reading, P.C. Glycosylation of the hemagglutinin modulates the sensitivity of h3n2 influenza viruses to innate proteins in airway secretions and virulence in mice. Virology 2011, 413, 84–92. [Google Scholar] [CrossRef]
- Caton, A.J.; Brownlee, G.G.; Yewdell, J.W.; Gerhard, W. The antigenic structure of the influenza virus a/pr/8/34 hemagglutinin (h1 subtype). Cell 1982, 31, 417–427. [Google Scholar] [CrossRef]
- Sun, X.; Jayaraman, A.; Maniprasad, P.; Raman, R.; Houser, K.V.; Pappas, C.; Zeng, H.; Sasisekharan, R.; Katz, J.M.; Tumpey, T.M. N-linked glycosylation of the hemagglutinin protein influences virulence and antigenicity of the 1918 pandemic and seasonal h1n1 influenza a viruses. J. Virol. 2013, 87, 8756–8766. [Google Scholar] [CrossRef]
- Hartshorn, K.L.; Webby, R.; White, M.R.; Tecle, T.; Pan, C.; Boucher, S.; Moreland, R.J.; Crouch, E.C.; Scheule, R.K. Role of viral hemagglutinin glycosylation in anti-influenza activities of recombinant surfactant protein d. Respir. Res. 2008, 9, 65. [Google Scholar] [CrossRef]
- Tokunaga, H.; Ushirogawa, H.; Ohuchi, M. The pandemic (h1n1) 2009 influenza virus is resistant to mannose-binding lectin. Virol. J. 2011, 8, 50. [Google Scholar] [CrossRef]
- Qi, L.; Kash, J.C.; Dugan, V.G.; Jagger, B.W.; Lau, Y.F.; Sheng, Z.M.; Crouch, E.C.; Hartshorn, K.L.; Taubenberger, J.K. The ability of pandemic influenza virus hemagglutinins to induce lower respiratory pathology is associated with decreased surfactant protein d binding. Virology 2011, 412, 426–434. [Google Scholar] [CrossRef]
- Reading, P.C.; University of Melbourne, Melbourne, Australia. Unpublished work. 2014.
- Ioannidis, L.J.; Verity, E.E.; Crawford, S.; Rockman, S.P.; Brown, L.E. Abortive replication of influenza virus in mouse dendritic cells. J. Virol. 2012, 86, 5922–5925. [Google Scholar]
- Hargadon, K.M.; Zhou, H.; Albrecht, R.A.; Dodd, H.A.; Garcia-Sastre, A.; Braciale, T.J. Major histocompatibility complex class ii expression and hemagglutinin subtype influence the infectivity of type a influenza virus for respiratory dendritic cells. J. Virol. 2011, 85, 11955–11963. [Google Scholar] [CrossRef]
- Hao, X.; Kim, T.S.; Braciale, T.J. Differential response of respiratory dendritic cell subsets to influenza virus infection. J. Virol. 2008, 82, 4908–4919. [Google Scholar] [CrossRef]
- Rodgers, B.; Mims, C.A. Interaction of influenza virus with mouse macrophages. Infect. Immun. 1981, 31, 751–757. [Google Scholar]
- Wells, M.A.; Albrecht, P.; Daniel, S.; Ennis, F.A. Host defense mechanisms against influenza virus: Interaction of influenza virus with murine macrophages in vitro. Infect. Immun. 1978, 22, 758–762. [Google Scholar]
- Cline, T.D.; Karlsson, E.A.; Seufzer, B.J.; Schultz-Cherry, S. The hemagglutinin protein of highly pathogenic h5n1 influenza viruses overcomes an early block in the replication cycle to promote productive replication in macrophages. J. Virol. 2013, 87, 1411–1419. [Google Scholar] [CrossRef]
- Tate, M.D.; Pickett, D.L.; van Rooijen, N.; Brooks, A.G.; Reading, P.C. Critical role of airway macrophages in modulating disease severity during influenza virus infection of mice. J. Virol. 2010, 84, 7569–7580. [Google Scholar] [CrossRef]
- GeurtsvanKessel, C.H.; Willart, M.A.; van Rijt, L.S.; Muskens, F.; Kool, M.; Baas, C.; Thielemans, K.; Bennett, C.; Clausen, B.E.; Hoogsteden, H.C.; et al. Clearance of influenza virus from the lung depends on migratory langerin+cd11b- but not plasmacytoid dendritic cells. J. Exp. Med. 2008, 205, 1621–1634. [Google Scholar] [CrossRef]
- Tumpey, T.M.; Garcia-Sastre, A.; Taubenberger, J.K.; Palese, P.; Swayne, D.E.; Pantin-Jackwood, M.J.; Schultz-Cherry, S.; Solorzano, A.; Van Rooijen, N.; Katz, J.M.; et al. Pathogenicity of influenza viruses with genes from the 1918 pandemic virus: Functional roles of alveolar macrophages and neutrophils in limiting virus replication and mortality in mice. J. Virol. 2005, 79, 14933–14944. [Google Scholar] [CrossRef]
- McGill, J.; van Rooijen, N.; Legge, K.L. Protective influenza-specific cd8 t cell responses require interactions with dendritic cells in the lungs. J. Exp. Med. 2008, 205, 1635–1646. [Google Scholar] [CrossRef]
- Cao, W.; Taylor, A.K.; Biber, R.E.; Davis, W.G.; Kim, J.H.; Reber, A.J.; Chirkova, T.; de La Cruz, J.A.; Pandey, A.; Ranjan, P.; et al. Rapid differentiation of monocytes into type i ifn-producing myeloid dendritic cells as an antiviral strategy against influenza virus infection. J. Immunol. 2012, 189, 2257–2265. [Google Scholar] [CrossRef]
- Londrigan, S.L.; Tate, M.D.; Brooks, A.G.; Reading, P.C. Cell-surface receptors on macrophages and dendritic cells for attachment and entry of influenza virus. J. Leukoc. Biol. 2012, 92, 97–106. [Google Scholar] [CrossRef]
- Reading, P.C.; Miller, J.L.; Anders, E.M. Involvement of the mannose receptor in infection of macrophages by influenza virus. J. Virol. 2000, 74, 5190–5197. [Google Scholar] [CrossRef]
- Upham, J.P.; Pickett, D.; Irimura, T.; Anders, E.M.; Reading, P.C. Macrophage receptors for influenza a virus: Role of the macrophage galactose-type lectin and mannose receptor in viral entry. J. Virol. 2010, 84, 3730–3737. [Google Scholar] [CrossRef]
- Ng, W.C.; Liong, S.; Tate, M.D.; Irimura, T.; Denda-Nagai, K.; Brooks, A.G.; Londrigan, S.L.; Reading, P.C. The macrophage galactose-type lectin can function as an attachment and entry receptor for influenza virus. J. Virol. 2013, 88, 1659–1672. [Google Scholar]
- Wang, S.F.; Huang, J.C.; Lee, Y.M.; Liu, S.J.; Chan, Y.J.; Chau, Y.P.; Chong, P.; Chen, Y.M. Dc-sign mediates avian h5n1 influenza virus infection in cis and in trans. Biochem. Biophys. Res. Commun. 2008, 373, 561–566. [Google Scholar] [CrossRef]
- Londrigan, S.L.; Turville, S.G.; Tate, M.D.; Deng, Y.M.; Brooks, A.G.; Reading, P.C. N-linked glycosylation facilitates sialic acid-independent attachment and entry of influenza a viruses into cells expressing dc-sign or l-sign. J. Virol. 2011, 85, 2990–3000. [Google Scholar]
- Hillaire, M.L.; Nieuwkoop, N.J.; Boon, A.C.; de Mutsert, G.; Vogelzang-van Trierum, S.E.; Fouchier, R.A.; Osterhaus, A.D.; Rimmelzwaan, G.F. Binding of dc-sign to the hemagglutinin of influenza a viruses supports virus replication in dc-sign expressing cells. PLoS One 2013, 8, e56164. [Google Scholar] [CrossRef] [Green Version]
- de Geus, E.D.; Tefsen, B.; van Haarlem, D.A.; van Eden, W.; van Die, I.; Vervelde, L. Glycans from avian influenza virus are recognized by chicken dendritic cells and are targets for the humoral immune response in chicken. Mol. Immunol. 2013, 56, 452–462. [Google Scholar] [CrossRef]
- Wiley, D.C.; Wilson, I.A.; Skehel, J.J. Structural identification of the antibody-binding sites of hong kong influenza haemagglutinin and their involvement in antigenic variation. Nature 1981, 289, 373–378. [Google Scholar] [CrossRef]
- Raymond, F.L.; Caton, A.J.; Cox, N.J.; Kendal, A.P.; Brownlee, G.G. Antigenicity and evolution amongst recent influenza viruses of h1n1 subtype. Nucleic Acids Res. 1983, 11, 7191–7203. [Google Scholar] [CrossRef]
- Skehel, J.J.; Stevens, D.J.; Daniels, R.S.; Douglas, A.R.; Knossow, M.; Wilson, I.A.; Wiley, D.C. A carbohydrate side chain on hemagglutinins of hong kong influenza viruses inhibits recognition by a monoclonal antibody. Proc. Natl. Acad. Sci. USA 1984, 81, 1779–1783. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhu, J.; Li, Y.; Bradley, K.C.; Cao, J.; Chen, H.; Jin, M.; Zhou, H. Glycosylation on hemagglutinin affects the virulence and pathogenicity of pandemic h1n1/2009 influenza a virus in mice. PLoS One 2013, 8, e61397. [Google Scholar]
- Throsby, M.; van den Brink, E.; Jongeneelen, M.; Poon, L.L.; Alard, P.; Cornelissen, L.; Bakker, A.; Cox, F.; van Deventer, E.; Guan, Y.; et al. Heterosubtypic neutralizing monoclonal antibodies cross-protective against h5n1 and h1n1 recovered from human igm+ memory b cells. PLoS One 2008, 3, e3942. [Google Scholar] [CrossRef]
- Han, T.; Marasco, W.A. Structural basis of influenza virus neutralization. Ann. N. Y. Acad. Sci. 2011, 1217, 178–190. [Google Scholar]
- Ekiert, D.C.; Bhabha, G.; Elsliger, M.A.; Friesen, R.H.; Jongeneelen, M.; Throsby, M.; Goudsmit, J.; Wilson, I.A. Antibody recognition of a highly conserved influenza virus epitope. Science 2009, 324, 246–251. [Google Scholar] [CrossRef]
- Bautista, E.; Chotpitayasunondh, T.; Gao, Z.; Harper, S.A.; Shaw, M.; Uyeki, T.M.; Zaki, S.R.; Hayden, F.G.; Hui, D.S.; Kettner, J.D.; et al. Clinical aspects of pandemic 2009 influenza a (h1n1) virus infection. New Engl. J. Med. 2010, 362, 1708–1719. [Google Scholar] [CrossRef]
- Hancock, K.; Veguilla, V.; Lu, X.; Zhong, W.; Butler, E.N.; Sun, H.; Liu, F.; Dong, L.; DeVos, J.R.; Gargiullo, P.M.; et al. Cross-reactive antibody responses to the 2009 pandemic h1n1 influenza virus. New Engl. J. Med. 2009, 361, 1945–1952. [Google Scholar] [CrossRef]
- Itoh, Y.; Shinya, K.; Kiso, M.; Watanabe, T.; Sakoda, Y.; Hatta, M.; Muramoto, Y.; Tamura, D.; Sakai-Tagawa, Y.; Noda, T.; et al. In vitro and in vivo characterization of new swine-origin h1n1 influenza viruses. Nature 2009, 460, 1021–1025. [Google Scholar]
- Miller, E.; Hoschler, K.; Hardelid, P.; Stanford, E.; Andrews, N.; Zambon, M. Incidence of 2009 pandemic influenza a h1n1 infection in england: A cross-sectional serological study. Lancet 2010, 375, 1100–1108. [Google Scholar] [CrossRef]
- O'Donnell, C.D.; Wright, A.; Vogel, L.N.; Wei, C.J.; Nabel, G.J.; Subbarao, K. Effect of priming with h1n1 influenza viruses of variable antigenic distances on challenge with 2009 pandemic h1n1 virus. J. Virol. 2012, 86, 8625–8633. [Google Scholar] [CrossRef]
- Kim, J.I.; Lee, I.; Park, S.; Hwang, M.W.; Bae, J.Y.; Lee, S.; Heo, J.; Park, M.S.; Garcia-Sastre, A. Genetic requirement for hemagglutinin glycosylation and its implications for influenza a h1n1 virus evolution. J. Virol. 2013, 87, 7539–7549. [Google Scholar] [CrossRef]
- Wanzeck, K.; Boyd, K.L.; McCullers, J.A. Glycan shielding of the influenza virus hemagglutinin contributes to immunopathology in mice. Am. J. Respir. Crit. Care Med. 2011, 183, 767–773. [Google Scholar] [CrossRef]
- Eggink, D.; Goff, P.H.; Palese, P. Guiding the immune response against influenza virus hemagglutinin toward the conserved stalk domain by hyper-glycosylation of the globular head domain. J. Virol. 2013. [Google Scholar] [CrossRef]
- de Vries, R.P.; Smit, C.H.; de Bruin, E.; Rigter, A.; de Vries, E.; Cornelissen, L.A.; Eggink, D.; Chung, N.P.; Moore, J.P.; Sanders, R.W.; et al. Glycan-dependent immunogenicity of recombinant soluble trimeric hemagglutinin. J. Virol. 2012, 86, 11735–11744. [Google Scholar] [CrossRef]
- Wang, C.C.; Chen, J.R.; Tseng, Y.C.; Hsu, C.H.; Hung, Y.F.; Chen, S.W.; Chen, C.M.; Khoo, K.H.; Cheng, T.J.; Cheng, Y.S.; et al. Glycans on influenza hemagglutinin affect receptor binding and immune response. Proc. Natl. Acad. Sci. USA 2009, 106, 18137–18142. [Google Scholar] [CrossRef]
- Thomas, P.G.; Keating, R.; Hulse-Post, D.J.; Doherty, P.C. Cell-mediated protection in influenza infection. Emerg. Infect. Dis. 2006, 12, 48–54. [Google Scholar] [CrossRef]
- Bui, H.H.; Peters, B.; Assarsson, E.; Mbawuike, I.; Sette, A. Ab and t cell epitopes of influenza a virus, knowledge and opportunities. Proc. Natl. Acad. Sci. USA 2007, 104, 246–251. [Google Scholar]
- Brown, L.E.; White, D.O.; Jackson, D.C. Conservation of determinants for class ii-restricted t cells within site e of influenza virus hemagglutinin and factors influencing their expression. J. Virol. 1993, 67, 2887–2893. [Google Scholar]
- Brown, L.E.; Ffrench, R.A.; Gawler, J.M.; Jackson, D.C.; Dyall-Smith, M.L.; Anders, E.M.; Tregear, G.W.; Duncan, L.; Underwood, P.A.; White, D.O. Distinct epitopes recognized by i-ad-restricted t-cell clones within antigenic site e on influenza virus hemagglutinin. J. Virol. 1988, 62, 305–312. [Google Scholar]
- Thomas, D.B.; Hodgson, J.; Riska, P.F.; Graham, C.M. The role of the endoplasmic reticulum in antigen processing. N-glycosylation of influenza hemagglutinin abrogates cd4+ cytotoxic t cell recognition of endogenously processed antigen. J. Immunol. 1990, 144, 2789–2794. [Google Scholar]
- Drummer, H.E.; Jackson, D.C.; Brown, L.E. Modulation of cd4+ t-cell recognition of influenza hemagglutinin by carbohydrate side chains located outside a t-cell determinant. Virology 1993, 192, 282–289. [Google Scholar] [CrossRef]
- Jackson, D.C.; Drummer, H.E.; Urge, L.; Otvos, L., Jr.; Brown, L.E. Glycosylation of a synthetic peptide representing a t-cell determinant of influenza virus hemagglutinin results in loss of recognition by cd4+ t-cell clones. Virology 1994, 199, 422–430. [Google Scholar] [CrossRef]
- Gambaryan, A.S.; Marinina, V.P.; Tuzikov, A.B.; Bovin, N.V.; Rudneva, I.A.; Sinitsyn, B.V.; Shilov, A.A.; Matrosovich, M.N. Effects of host-dependent glycosylation of hemagglutinin on receptor-binding properties on h1n1 human influenza a virus grown in mdck cells and in embryonated eggs. Virology 1998, 247, 170–177. [Google Scholar] [CrossRef]
- Jayaraman, A.; Koh, X.; Li, J.; Raman, R.; Viswanathan, K.; Shriver, Z.; Sasisekharan, R. Glycosylation at asn91 of h1n1 haemagglutinin affects binding to glycan receptors. Biochem. J. 2012, 444, 429–435. [Google Scholar] [CrossRef]
- Ohuchi, M.; Ohuchi, R.; Sakai, T.; Matsumoto, A. Tight binding of influenza virus hemagglutinin to its receptor interferes with fusion pore dilation. J. Virol. 2002, 76, 12405–12413. [Google Scholar] [CrossRef]
- Gallagher, P.; Henneberry, J.; Wilson, I.; Sambrook, J.; Gething, M.J. Addition of carbohydrate side chains at novel sites on influenza virus hemagglutinin can modulate the folding, transport, and activity of the molecule. J. Cell Biol. 1988, 107, 2059–2073. [Google Scholar] [CrossRef]
- Matrosovich, M.; Zhou, N.; Kawaoka, Y.; Webster, R. The surface glycoproteins of h5 influenza viruses isolated from humans, chickens, and wild aquatic birds have distinguishable properties. J. Virol. 1999, 73, 1146–1155. [Google Scholar]
- Wang, W.; Lu, B.; Zhou, H.; Suguitan, A.L., Jr.; Cheng, X.; Subbarao, K.; Kemble, G.; Jin, H. Glycosylation at 158n of the hemagglutinin protein and receptor binding specificity synergistically affect the antigenicity and immunogenicity of a live attenuated h5n1 a/vietnam/1203/2004 vaccine virus in ferrets. J. Virol. 2010, 84, 6570–6577. [Google Scholar] [CrossRef]
- Ohuchi, M.; Ohuchi, R.; Feldmann, A.; Klenk, H.D. Regulation of receptor binding affinity of influenza virus hemagglutinin by its carbohydrate moiety. J. Virol. 1997, 71, 8377–8384. [Google Scholar]
- Das, S.R.; Hensley, S.E.; David, A.; Schmidt, L.; Gibbs, J.S.; Puigbo, P.; Ince, W.L.; Bennink, J.R.; Yewdell, J.W. Fitness costs limit influenza a virus hemagglutinin glycosylation as an immune evasion strategy. Proc. Natl. Acad. Sci. USA 2011, 108, E1417–E1422. [Google Scholar] [CrossRef]
- Guo, D.; Shi, X.; Arledge, K.C.; Song, D.; Jiang, L.; Fu, L.; Gong, X.; Zhang, S.; Wang, X.; Zhang, L. A single residue within the v5 region of hiv-1 envelope facilitates viral escape from the broadly neutralizing monoclonal antibody vrc01. J. Biol. Chem. 2012, 287, 43170–43179. [Google Scholar] [CrossRef]
- van Gils, M.J.; Edo-Matas, D.; Bowles, E.J.; Burger, J.A.; Stewart-Jones, G.B.; Schuitemaker, H. Evolution of human immunodeficiency virus type 1 in a patient with cross-reactive neutralizing activity in serum. J. Virol. 2011, 85, 8443–8448. [Google Scholar] [CrossRef]
- Sun, S.; Wang, Q.; Zhao, F.; Chen, W.; Li, Z. Prediction of biological functions on glycosylation site migrations in human influenza h1n1 viruses. PLoS One 2012, 7, e32119. [Google Scholar]
- Hartley, C.A.; Reading, P.C.; Ward, A.C.; Anders, E.M. Changes in the hemagglutinin molecule of influenza type a (h3n2) virus associated with increased virulence for mice. Arch. Virol. 1997, 142, 75–88. [Google Scholar] [CrossRef]
- Reading, P.C.; Pickett, D.L.; Tate, M.D.; Whitney, P.G.; Job, E.R.; Brooks, A.G. Loss of a single n-linked glycan from the hemagglutinin of influenza virus is associated with resistance to collectins and increased virulence in mice. Respir. Res. 2009, 10, 117. [Google Scholar] [CrossRef]
- Hansen, S.; Holmskov, U. Lung surfactant protein d (sp-d) and the molecular diverted descendants: Conglutinin, cl-43 and cl-46. Immunobiology 2002, 205, 498–517. [Google Scholar] [CrossRef]
- Goh, B.C.; Rynkiewicz, M.J.; Cafarella, T.R.; White, M.R.; Hartshorn, K.L.; Allen, K.; Crouch, E.C.; Calin, O.; Seeberger, P.H.; Schulten, K.; et al. Molecular mechanisms of inhibition of influenza by surfactant protein d revealed by large-scale molecular dynamics simulation. Biochemistry 2013, 52, 8527–8538. [Google Scholar] [CrossRef]
- Krieger, E.; Joo, K.; Lee, J.; Raman, S.; Thompson, J.; Tyka, M.; Baker, D.; Karplus, K. Improving physical realism, stereochemistry, and side-chain accuracy in homology modeling: Four approaches that performed well in casp8. Proteins 2009, 77, 114–122. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Tate, M.D.; Job, E.R.; Deng, Y.-M.; Gunalan, V.; Maurer-Stroh, S.; Reading, P.C. Playing Hide and Seek: How Glycosylation of the Influenza Virus Hemagglutinin Can Modulate the Immune Response to Infection. Viruses 2014, 6, 1294-1316. https://doi.org/10.3390/v6031294
Tate MD, Job ER, Deng Y-M, Gunalan V, Maurer-Stroh S, Reading PC. Playing Hide and Seek: How Glycosylation of the Influenza Virus Hemagglutinin Can Modulate the Immune Response to Infection. Viruses. 2014; 6(3):1294-1316. https://doi.org/10.3390/v6031294
Chicago/Turabian StyleTate, Michelle D., Emma R. Job, Yi-Mo Deng, Vithiagaran Gunalan, Sebastian Maurer-Stroh, and Patrick C. Reading. 2014. "Playing Hide and Seek: How Glycosylation of the Influenza Virus Hemagglutinin Can Modulate the Immune Response to Infection" Viruses 6, no. 3: 1294-1316. https://doi.org/10.3390/v6031294
APA StyleTate, M. D., Job, E. R., Deng, Y.-M., Gunalan, V., Maurer-Stroh, S., & Reading, P. C. (2014). Playing Hide and Seek: How Glycosylation of the Influenza Virus Hemagglutinin Can Modulate the Immune Response to Infection. Viruses, 6(3), 1294-1316. https://doi.org/10.3390/v6031294





