Murine Leukemia Virus Uses TREX Components for Efficient Nuclear Export of Unspliced Viral Transcripts
Abstract
:1. Introduction
2. Results and Discussion
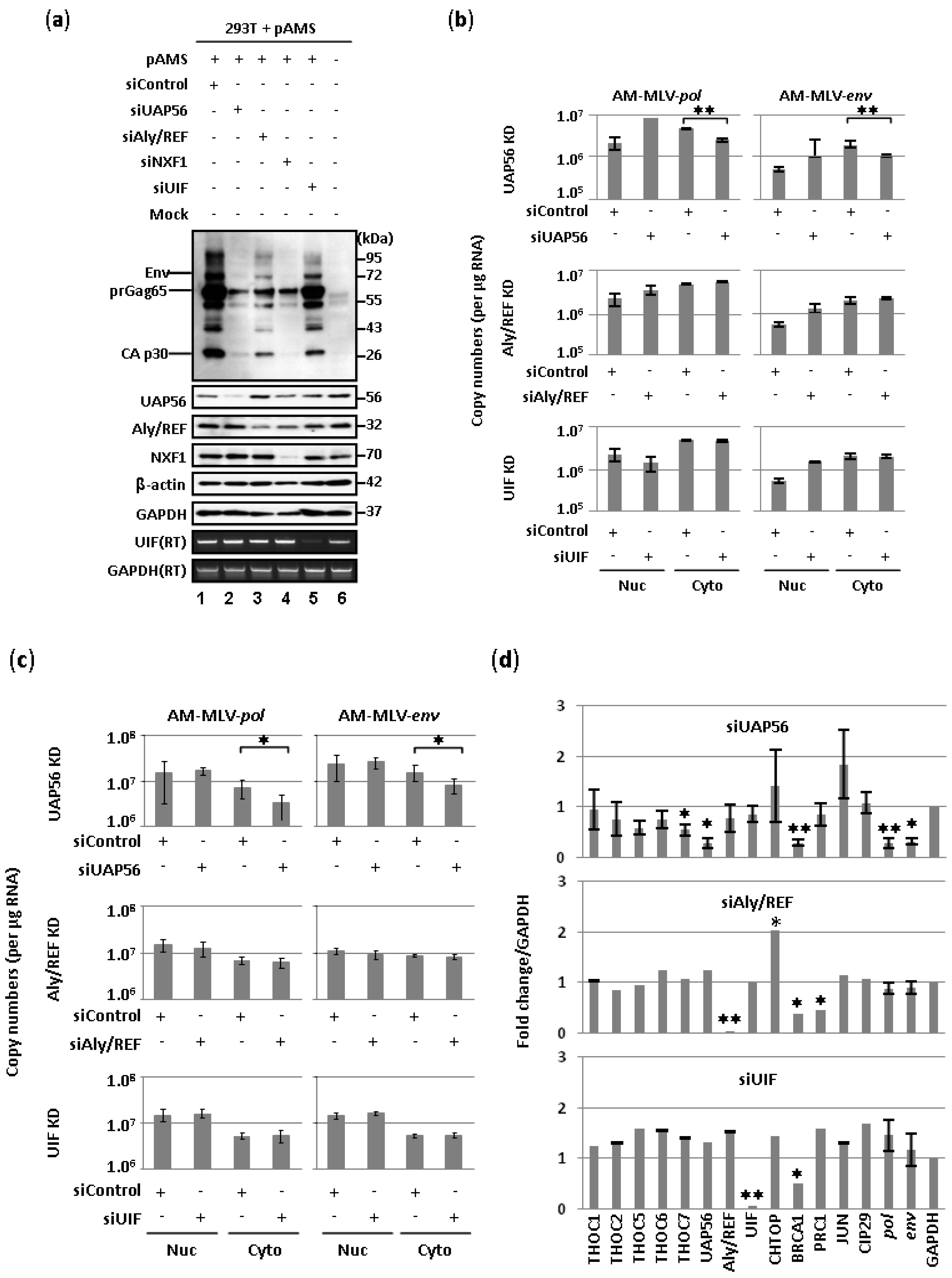
2.1. Involvement of UAP56 in the Nuclear Export of MLV Transcripts
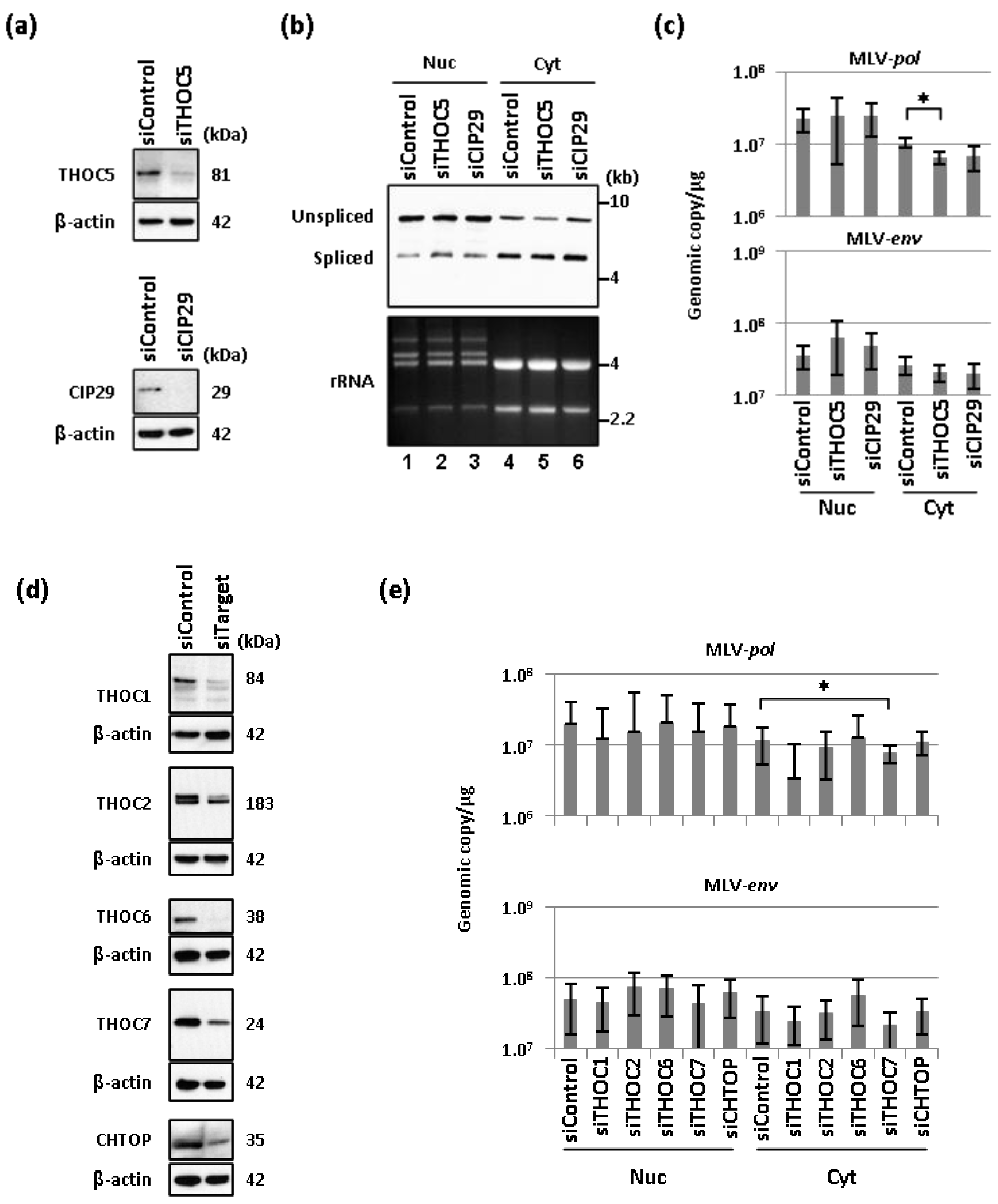
2.2. Depletion of THO Complex Prevents the Cytoplasmic Accumulation of Unspliced MLV Transcripts


3. Experimental Section
3.1. Cell Lines and Transfection
3.2. Northern Blotting
3.3. RT-PCR/RT-qPCR
3.4. Western Blot
3.5. RIP (RNA Immunoprecipitation) Assay
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References and Notes
- Sandri-Goldin, R.M. Viral regulation of mRNA export. J. Virol. 2004, 78, 4389–4396. [Google Scholar] [CrossRef]
- Gruter, P.; Tabernero, C.; von Kobbe, C.; Schmitt, C.; Saavedra, C.; Bachi, A.; Wilm, M.; Felber, B.K.; Izaurralde, E. TAP, the human homolog of Mex67p, mediates CTE-dependent RNA export from the nucleus. Mol. Cell 1998, 1, 649–659. [Google Scholar] [CrossRef]
- Zapp, M.L.; Green, M.R. Sequence-specific RNA binding by the HIV-1 Rev protein. Nature 1989, 342, 714–716. [Google Scholar] [CrossRef]
- Daly, T.J.; Cook, K.S.; Gray, G.S.; Maione, T.E.; Rusche, J.R. Specific binding of HIV-1 recombinant Rev protein to the Rev-responsive element in vitro. Nature 1989, 342, 816–819. [Google Scholar] [CrossRef]
- Popa, I.; Harris, M.E.; Donello, J.E.; Hope, T.J. CRM1-dependent function of a cis-acting RNA export element. Mol. Cell. Biol. 2002, 22, 2057–2067. [Google Scholar] [CrossRef]
- Ahmed, Y.F.; Hanly, S.M.; Malim, M.H.; Cullen, B.R.; Greene, W.C. Structure-function analyses of the HTLV-I Rex and HIV-1 Rev RNA response elements: Insights into the mechanism of Rex and Rev action. Genes Dev. 1990, 4, 1014–1022. [Google Scholar] [CrossRef]
- Hanly, S.M.; Rimsky, L.T.; Malim, M.H.; Kim, J.H.; Hauber, J.; Duc Dodon, M.; Le, S.Y.; Maizel, J.V.; Cullen, B.R.; Greene, W.C. Comparative analysis of the HTLV-I Rex and HIV-1 Rev trans-regulatory proteins and their RNA response elements. Genes Dev. 1989, 3, 1534–1544. [Google Scholar]
- Indik, S.; Gunzburg, W.H.; Salmons, B.; Rouault, F. A novel, mouse mammary tumor virus encoded protein with Rev-like properties. Virology 2005, 337, 1–6. [Google Scholar] [CrossRef]
- Mertz, J.A.; Simper, M.S.; Lozano, M.M.; Payne, S.M.; Dudley, J.P. Mouse mammary tumor virus encodes a self-regulatory RNA export protein and is a complex retrovirus. J. Virol. 2005, 79, 14737–1447. [Google Scholar] [CrossRef]
- Nitta, T.; Hofacre, A.; Hull, S.; Fan, H. Identification and mutational analysis of a Rej response element in Jaagsiekte sheep retrovirus RNA. J. Virol. 2009, 83, 12499–12511. [Google Scholar] [CrossRef]
- Bodem, J.; Schied, T.; Gabriel, R.; Rammling, M.; Rethwilm, A. Foamy virus nuclear RNA export is distinct from that of other retroviruses. J. Virol. 2011, 85, 2333–2341. [Google Scholar] [CrossRef]
- Liker, E.; Fernandez, E.; Izaurralde, E.; Conti, E. The structure of the mRNA export factor TAP reveals a cis arrangement of a non-canonical RNP domain and an LRR domain. EMBO J. 2000, 19, 5587–5598. [Google Scholar] [CrossRef]
- Bachi, A.; Braun, I.C.; Rodrigues, J.P.; Pante, N.; Ribbeck, K.; von Kobbe, C.; Kutay, U.; Wilm, M.; Gorlich, D.; Carmo-Fonseca, M.; et al. The C-terminal domain of TAP interacts with the nuclear pore complex and promotes export of specific CTE-bearing RNA substrates. RNA 2000, 6, 136–158. [Google Scholar] [CrossRef]
- Sakuma, T.; Davila, J.I.; Malcolm, J.A.; Kocher, J.P.; Tonne, J.M.; Ikeda, Y. Murine leukemia virus uses NXF1 for nuclear export of spliced and unspliced viral transcripts. J. Virol. 2014. [Google Scholar] [CrossRef]
- Houseley, J.; Tollervey, D. The many pathways of RNA degradation. Cell 2009, 136, 763–776. [Google Scholar] [CrossRef]
- Kohler, A.; Hurt, E. Exporting RNA from the nucleus to the cytoplasm. Nat. Rev. Mol. Cell Biol. 2007, 8, 761–773. [Google Scholar] [CrossRef]
- Culjkovic-Kraljacic, B.; Borden, K.L. Aiding and abetting cancer: mRNA Export and the nuclear pore. Trends Cell Biol. 2013, 23, 328–335. [Google Scholar] [CrossRef]
- Strasser, K.; Masuda, S.; Mason, P.; Pfannstiel, J.; Oppizzi, M.; Rodriguez-Navarro, S.; Rondon, A.G.; Aguilera, A.; Struhl, K.; Reed, R.; et al. TREX is a conserved complex coupling transcription with messenger RNA export. Nature 2002, 417, 304–308. [Google Scholar] [CrossRef]
- Reed, R. Coupling transcription, splicing and mRNA export. Curr. Opin. Cell Biol. 2003, 15, 326–331. [Google Scholar] [CrossRef]
- Taniguchi, I.; Ohno, M. ATP-dependent recruitment of export factor Aly/REF onto intronless mRNAs by RNA helicase UAP56. Mol. Cell. Biol. 2008, 28, 601–608. [Google Scholar] [CrossRef]
- Lei, H.; Dias, A.P.; Reed, R. Export and stability of naturally intronless mRNAs require specific coding region sequences and the TREX mRNA export complex. Proc. Natl. Acad. Sci. USA 2011, 108, 17985–17990. [Google Scholar] [CrossRef]
- Lei, H.; Zhai, B.; Yin, S.; Gygi, S.; Reed, R. Evidence that a consensus element found in naturally intronless mRNAs promotes mRNA export. Nucleic Acids Res. 2013, 41, 2517–2525. [Google Scholar] [CrossRef]
- Fleckner, J.; Zhang, M.; Valcarcel, J.; Green, M.R. U2AF65 recruits a novel human DEAD box protein required for the U2 snRNP-branchpoint interaction. Genes Dev. 1997, 11, 1864–1872. [Google Scholar] [CrossRef]
- Luo, M.L.; Zhou, Z.; Magni, K.; Christoforides, C.; Rappsilber, J.; Mann, M.; Reed, R. Pre-mRNA splicing and mRNA export linked by direct interactions between UAP56 and Aly. Nature 2001, 413, 644–647. [Google Scholar] [CrossRef]
- Gatfield, D.; Izaurralde, E. REF1/Aly and the additional exon junction complex proteins are dispensable for nuclear mRNA export. J. Cell Biol. 2002, 159, 579–588. [Google Scholar] [CrossRef]
- Hautbergue, G.M.; Hung, M.L.; Walsh, M.J.; Snijders, A.P.; Chang, C.T.; Jones, R.; Ponting, C.P.; Dickman, M.J.; Wilson, S.A. UIF, a new mRNA export adaptor that works together with REF/ALY, requires FACT for recruitment to mRNA. Curr. Biol. 2009, 19, 1918–1924. [Google Scholar] [CrossRef]
- Wickramasinghe, V.O.; McMurtrie, P.I.; Mills, A.D.; Takei, Y.; Penrhyn-Lowe, S.; Amagase, Y.; Main, S.; Marr, J.; Stewart, M.; Laskey, R.A. mRNA export from mammalian cell nuclei is dependent on GANP. Curr. Biol. 2010, 20, 25–31. [Google Scholar] [CrossRef]
- Chang, C.T.; Hautbergue, G.M.; Walsh, M.J.; Viphakone, N.; van Dijk, T.B.; Philipsen, S.; Wilson, S.A. Chtop is a component of the dynamic TREX mRNA export complex. EMBO J. 2013, 32, 473–486. [Google Scholar] [CrossRef]
- Dufu, K.; Livingstone, M.J.; Seebacher, J.; Gygi, S.P.; Wilson, S.A.; Reed, R. ATP is required for interactions between UAP56 and two conserved mRNA export proteins, Aly and CIP29, to assemble the TREX complex. Genes Dev. 2010, 24, 2043–2053. [Google Scholar] [CrossRef]
- Viphakone, N.; Hautbergue, G.M.; Walsh, M.; Chang, C.T.; Holland, A.; Folco, E.G.; Reed, R.; Wilson, S.A. TREX exposes the RNA-binding domain of Nxf1 to enable mRNA export. Nat. Commun. 2012, 3. [Google Scholar] [CrossRef]
- Masuda, S.; Das, R.; Cheng, H.; Hurt, E.; Dorman, N.; Reed, R. Recruitment of the human TREX complex to mRNA during splicing. Genes Dev. 2005, 19, 1512–1517. [Google Scholar] [CrossRef]
- Cheng, H.; Dufu, K.; Lee, C.S.; Hsu, J.L.; Dias, A.; Reed, R. Human mRNA export machinery recruited to the 5' end of mRNA. Cell 2006, 127, 1389–1400. [Google Scholar] [CrossRef]
- Stutz, F.; Bachi, A.; Doerks, T.; Braun, I.C.; Seraphin, B.; Wilm, M.; Bork, P.; Izaurralde, E. REF, an evolutionary conserved family of hnRNP-like proteins, interacts with TAP/Mex67p and participates in mRNA nuclear export. RNA 2000, 6, 638–650. [Google Scholar] [CrossRef]
- Cullen, B.R. Connections between the processing and nuclear export of mRNA: Evidence for an export license? Proc. Natl. Acad. Sci. USA 2000, 97, 4–6. [Google Scholar] [CrossRef]
- Boyne, J.R.; Colgan, K.J.; Whitehouse, A. Recruitment of the complete hTREX complex is required for Kaposi’s sarcoma-associated herpesvirus intronless mRNA nuclear export and virus replication. PLoS Pathog. 2008, 4, e1000194. [Google Scholar] [CrossRef]
- Read, E.K.; Digard, P. Individual influenza A virus mRNAs show differential dependence on cellular NXF1/TAP for their nuclear export. J. Gen. Virol. 2010, 91, 1290–1301. [Google Scholar] [CrossRef]
- Colgan, K.J.; Boyne, J.R.; Whitehouse, A. Uncoupling of hTREX demonstrates that UAP56 and hTHO-complex recruitment onto herpesvirus saimiri intronless transcripts is required for replication. J. Gen. Virol. 2009, 90, 1455–1460. [Google Scholar] [CrossRef]
- Rehwinkel, J.; Herold, A.; Gari, K.; Kocher, T.; Rode, M.; Ciccarelli, F.L.; Wilm, M.; Izaurralde, E. Genome-wide analysis of mRNAs regulated by the THO complex in Drosophila melanogaster. Nat. Struct. Mol. Biol. 2004, 11, 558–566. [Google Scholar] [CrossRef]
- Jackson, B.R.; Boyne, J.R.; Noerenberg, M.; Taylor, A.; Hautbergue, G.M.; Walsh, M.J.; Wheat, R.; Blackbourn, D.J.; Wilson, S.A.; Whitehouse, A. An interaction between KSHV ORF57 and UIF provides mRNA-adaptor redundancy in herpesvirus intronless mRNA export. PLoS Pathog. 2011, 7, e1002138. [Google Scholar] [CrossRef]
- Yamazaki, T.; Fujiwara, N.; Yukinaga, H.; Ebisuya, M.; Shiki, T.; Kurihara, T.; Kioka, N.; Kambe, T.; Nagao, M.; Nishida, E.; et al. The closely related RNA helicases, UAP56 and URH49, preferentially form distinct mRNA export machineries and coordinately regulate mitotic progression. Mol. Biol. Cell 2010, 21, 2953–2965. [Google Scholar] [CrossRef]
- Herold, A.; Teixeira, L.; Izaurralde, E. Genome-wide analysis of nuclear mRNA export pathways in Drosophila. EMBO J. 2003, 22, 2472–2483. [Google Scholar] [CrossRef]
- Pena, A.; Gewartowski, K.; Mroczek, S.; Cuellar, J.; Szykowska, A.; Prokop, A.; Czarnocki-Cieciura, M.; Piwowarski, J.; Tous, C.; Aguilera, A.; et al. Architecture and nucleic acids recognition mechanism of the THO complex, an mRNP assembly factor. EMBO J. 2012, 31, 1605–1616. [Google Scholar] [CrossRef] [Green Version]
- Chi, B.; Wang, Q.; Wu, G.; Tan, M.; Wang, L.; Shi, M.; Chang, X.; Cheng, H. Aly and THO are required for assembly of the human TREX complex and association of TREX components with the spliced mRNA. Nucleic Acids Res. 2013, 41, 1294–1306. [Google Scholar] [CrossRef]
- Reed, R.; Cheng, H. TREX, SR proteins and export of mRNA. Curr. Opin. Cell Biol. 2005, 17, 269–273. [Google Scholar] [CrossRef]
- Cenik, C.; Chua, H.N.; Zhang, H.; Tarnawsky, S.P.; Akef, A.; Derti, A.; Tasan, M.; Moore, M.J.; Palazzo, A.F.; Roth, F.P. Genome analysis reveals interplay between 5'UTR introns and nuclear mRNA export for secretory and mitochondrial genes. PLoS Genet. 2011, 7, e1001366. [Google Scholar] [CrossRef]
- Muller-McNicoll, M.; Neugebauer, K.M. How cells get the message: Dynamic assembly and function of mRNA-protein complexes. Nat. Rev. Genet. 2013, 14, 275–287. [Google Scholar] [CrossRef]
- Katahira, J.; Inoue, H.; Hurt, E.; Yoneda, Y. Adaptor Aly and co-adaptor Thoc5 function in the Tap-p15-mediated nuclear export of HSP70 mRNA. EMBO J. 2009, 28, 556–567. [Google Scholar] [CrossRef]
- Sakuma, R.; Sakuma, T.; Ohmine, S.; Silverman, R.H.; Ikeda, Y. Xenotropic murine leukemia virus-related virus is susceptible to AZT. Virology 2010, 397, 1–6. [Google Scholar] [CrossRef]
- Sakuma, T.; Hue, S.; Squillace, K.A.; Tonne, J.M.; Blackburn, P.R.; Ohmine, S.; Thatava, T.; Towers, G.J.; Ikeda, Y. No Evidence of XMRV in prostate cancer cohorts in the Midwestern United States. Retrovirology 2011, 8. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Sakuma, T.; Tonne, J.M.; Ikeda, Y. Murine Leukemia Virus Uses TREX Components for Efficient Nuclear Export of Unspliced Viral Transcripts. Viruses 2014, 6, 1135-1148. https://doi.org/10.3390/v6031135
Sakuma T, Tonne JM, Ikeda Y. Murine Leukemia Virus Uses TREX Components for Efficient Nuclear Export of Unspliced Viral Transcripts. Viruses. 2014; 6(3):1135-1148. https://doi.org/10.3390/v6031135
Chicago/Turabian StyleSakuma, Toshie, Jason M. Tonne, and Yasuhiro Ikeda. 2014. "Murine Leukemia Virus Uses TREX Components for Efficient Nuclear Export of Unspliced Viral Transcripts" Viruses 6, no. 3: 1135-1148. https://doi.org/10.3390/v6031135



