Abstract
The frequency and magnitude of recognized and declared filovirus-disease outbreaks have increased in recent years, while pathogenic filoviruses are potentially ubiquitous throughout sub-Saharan Africa. Meanwhile, the efficiency and effectiveness of filovirus-disease outbreak preparedness and response efforts are currently limited by inherent challenges and persistent shortcomings. This paper delineates some of these challenges and shortcomings and provides a proposal for enhancing future filovirus-disease outbreak preparedness and response. The proposal serves as a call for prompt action by the organizations that comprise filovirus-disease outbreak response teams, namely, Ministries of Health of outbreak-prone countries, the World Health Organization, Médecins Sans Frontières, the Centers for Disease Control and Prevention—Atlanta, and others.
Keywords:
Ebola; ebolavirus; Marburg virus; marburgvirus; Filoviridae; filovirus; outbreak; preparedness; response; data collection; treatment; guidelines; surveillance 1. Introduction
Ebola virus disease (EVD) and Marburg virus disease (MVD) in human and non-human primates (NHPs) are caused by seven distinct viruses that produce filamentous, enveloped particles with negative-sense, single-stranded ribonucleic acid genomes. These viruses belong to the Filoviridae family and its Ebolavirus and Marburgvirus genera, respectively []. An eighth filovirus, Lloviu virus (LLOV), assigned to the third filovirus genus, Cuevavirus has thus far not been associated with human disease [,]. To date, no human or NHP infections of LLOV have been known to occur [].
1.1. Outbreak Response Teams, Objectives, and Components
For a human filovirus-disease outbreak to be declared, a single laboratory-confirmed case must be identified. If filovirus infection is suspected but not laboratory-confirmed, the possible outbreak remains speculative and is not recognized and declared by the World Health Organization (WHO), the acknowledged authority for international filovirus reporting. As described elsewhere [,,,], filovirus-disease outbreak response teams (ORTs) typically comprise first-line medical professionals reinforced by the relevant Ministry of Health, the WHO, Médecins Sans Frontières (MSF), the Centers for Disease Control and Prevention—Atlanta (CDC), and others.
Overall filovirus-disease outbreak response objectives are to (1) prevent and control the spread of the disease and (2) provide infected patients with optimal monitoring and medical treatment. To achieve these objectives, the following outbreak-response components are realized. Within an affected community: (1) epidemiological surveillance for case detection, (2) burial and disinfection, (3) home-based risk reduction, (4) peripheral health-facility support, (5) psychosocial support (6) information and education campaigns, and (7) ecological studies. Within a filovirus ward and health facility: (1) design and construction of the filovirus ward, (2) case diagnosis, (3) case detection in the health facility, (4) case management, (5) psychological care, and (6) infection control in the health facility.
1.2. Response-Component Protocol Modifications
Due to numerous past impediments to efficiency and effectiveness, protocols corresponding to each filovirus-disease outbreak-response component have purportedly been modified for improvement [,,,,,,,,,,,]. These modifications aimed to promote cultural sensitivity, community collaboration, transparency of activities, improved data collection initiatives, and the active involvement of all stakeholders during all phases of the response [].
Further, it is now understood that the acceptability of a filovirus ward in a host community requires that psychological and cultural factors be considered during all stages of filovirus ward planning and implementation, including the provision of optimal medical care, which increases the acceptability of response components within the affected community and may improve survival rates for some patients [,,]. Additionally, as filovirus clinicians often triage patients based on presenting signs and symptoms and contact history, ORTs should now be cognizant of the crucial importance of collecting and analysing high-quality epidemiological and clinical data, which contribute to case definition refinement, and thereby facilitate outbreak control and treatment strategies [,,,,].
2. Delineation of the Problem
Despite the purported protocol modifications, limitations to efficient and effective filovirus-disease outbreak preparedness and response remain [,,,]. Thus, ensuing the acknowledgement of challenges inherent to and identification of shortcomings in current outbreak preparedness and response, a proposal for future enhancement is herein provided. A brief overview of human filovirus-disease outbreak frequency, magnitude, and geographic distribution evinces the pertinence of the proposal, while the proposal itself serves as a call for prompt action by Ministries of Health of outbreak-prone countries, the WHO, MSF, CDC, and others.
2.1. Outbreak Frequency and Magnitude
Since the initial 1967 filovirus discovery [,], a total of 41 human filovirus-disease outbreaks have been recognized and declared; 29 of these were EVD and 12, MVD; each outbreak occurred in or was thought to have originated from widely distributed areas of sub-Saharan Africa. As of 18 September 2014, these outbreaks have resulted in 8883 laboratory-confirmed or putative filovirus-disease cases and 4921 deaths, yielding a mean case fatality ratio (CFR) of 55.4% [,,,,,,,,,,,,] (Table 1, Figure 1).
An increase in frequency and magnitude of recognized and declared human filovirus-disease outbreaks have occurred in the recent 1994 to 2014 time period (Table 1, Figure 1). The only two recognized major MVD outbreaks to occur in their natural setting (sub-Saharan Africa) transpired within this period: Durba and Watsa, DRC (1998–2000) and Uige, Angola (2005) []. Remarkably, the current outbreaks of 2014 have thus far yielded nearly sixty-seven percent of all recognized and declared filovirus infections known to have occurred since 1967 (Table 1).
Filovirus-disease outbreaks are currently unpredictable in their timing and, within sub-Saharan Africa, their location [,]. The extent to which the recent increase in outbreak frequency can be attributed to improved surveillance and/or laboratory diagnostic capacity rather than an actual increase in number of outbreaks is uncertain. Seroprevalence studies [,,,,,,,] suggest that symptomatic and asymptomatic endemic filovirus infections occur, but transmission is typically recognized only when amplified [,,,]. There is also a suggested high likelihood of unrecognised outbreaks or isolated cases in unmonitored areas [,,,]. Further research regarding filovirus-disease outbreak frequency and magnitude is warranted.
2.2. Outbreak Geographic Distribution
Although recent research has implicated fruit bats of multiple species as natural reservoirs [,,,,,,,], detailed ecology of ebolaviruses and marburgviruses and their complete maintenance cycle are, to date, uncertain and are the subject of ongoing study [,,,,]. Nonetheless, high seroprevalence of Ebola virus-specific immunoglobulin G (IgG) in chimpanzees residing in Republic of the Congo, Gabon, and Cameroon [], and bats from Republic of the Congo and Gabon [] suggest that Ebola virus circulates continuously and with long-term persistence in tropical forest regions of sub-Saharan Africa, causing lethal and non-lethal infections in human and NHPs []. A serological survey in Gabon found an Ebola virus-specific IgG seroprevalence of 15.3% among rural human populations; the highest reported to date, suggesting a common source of human exposure, such as fruit contaminated by bat saliva [,]. Furthermore, excluding accidental exposures in biosafety level-4 laboratories, all recognized human filovirus-disease outbreaks to date can be traced back to tropical forest regions and other widely distributed areas of sub-Saharan Africa [,,,,]. As of 18 September 2014, human filovirus-disease outbreaks have been laboratory-confirmed and declared in the following sub-Saharan African countries: Republic of the Congo, Gabon, Zaire (and present-day Democratic Republic of the Congo), Rhodesia (present-day Zimbabwe), South Africa, Kenya, Angola, Côte d’Ivoire, Uganda, Sudan (present-day South Sudan), Guinea, Liberia, Sierra Leone, Nigeria, and Senegal (Table 1) [,].

Table 1.
Recognised and declared filovirus-disease outbreaks in humans (1967–18 September 2014). Note: Biosafety level-4 laboratory accidental exposures are categorized as filovirus-disease outbreaks as they involve human cases.
| Number | Year | Filovirus | Outbreak Location | Laboratory Confirmed Cases | Putative Cases | Total cases (Laboratory Confirmed Plus Putative) | Deaths | CFR* (%) |
|---|---|---|---|---|---|---|---|---|
| 1 | 1967 | Marburg virus | Marburg and Frankfurt, West Germany and Belgrade, Yugoslavia | 23 | 9 | 31 | 7 | 22.6 |
| 2 | 1975 | Marburg virus | Johannesburg, South Africa (Imported from Rhodesia) | 3 | 0 | 3 | 1 | 33.3 |
| 3 | 1976 | Sudan virus | Maridi and Nzara, Sudan | § | § | 284 | 151 | 53.2 |
| 4 | 1976 | Ebola virus | Yambuku, Zaire | § | § | 318 | 280 | 88.1 |
| 5 | 1976 | Sudan virus | Porton, United Kingdom laboratory accident at the Microbiological Research Establishment | 1 | 0 | 1 | 0 | 0.0 |
| 6 | 1977 | Ebola virus | Tandala, Zaire | 1 | 0 | 1 | 1 | 100.0 |
| 7 | 1979 | Sudan virus | Nzara, Sudan | 2 | 32 | 34 | 22 | 64.7 |
| 8 | 1980 | Marburg virus | Kisumu and Nairobi, Kenya | 2 | 0 | 2 | 1 | 50.0 |
| 9 | 1987 | Ravn virus | Mombasa, Kenya | 1 | 0 | 1 | 1 | 100.0 |
| 10 | 1988 | Marburg virus | USSR laboratory accident | 1 | 0 | 1 | 1 | 100.0 |
| 11 | 1990 | Marburg virus | USSR laboratory accident | 1 | 0 | 1 | 0 | 0.0 |
| 12 | 1994 | Ebola virus | Mékouka, Ogooué-Ivindo Province, Gabon | 7 | 45 | 52 | 32 | 61.5 |
| 13 | 1994 | Taï Forest virus | Taï Forest, Côte d’Ivoire (Treated in Switzerland) | 1 | 0 | 1 | 0 | 0.0 |
| 14 | 1995 | Ebola virus | Kikwit, Zaire | § | § | 315 | 254 | 80.6 |
| 15 | 1996 | Ebola virus | Mayibout, Ogooué-Ivindo Province, Gabon | 3 | 28 | 31 | 21 | 67.7 |
| 16 | 1996–1997 | Ebola virus | Booué, Ogooué-Ivindo Province, Gabon | 6 | 54 | 60 | 45 | 75.0 |
| 17 | 1996 | Ebola virus | Johannesburg, South Africa (Imported from Gabon) [linked to 1996-97 Booue, Gabon outbreak] | 1 | 1 | 2 | 1 | 50.0 |
| 18 | 1998–2000 | Marburg virus. Ravn virus | Durba and Watsa, Democratic Republic of the Congo | 51 | 103 | 154 | 128 | 83.1 |
| 19 | 2000–2001 | Sudan virus | Gulu, Uganda | 218 | 207 | 425 | 224 | 52.7 |
| 20 | 2001–2002 | Ebola virus | Ogooué-Ivindo Province, Gabon and Cuvette Ouest Region, Republic of the Congo | § | § | 124 | 97 | 78.2 |
| 21 | 2002 | Ebola virus | Ogooué-Ivindo Province, Gabon and Cuvette Ouest Region, Republic of the Congo | § | § | 11 | 10 | 90.9 |
| 22 | 2002–2003 | Ebola virus | Kellé, Cuvette Ouest Region, Republic of the Congo | 2 | 141 | 143 | 128 | 89.5 |
| 23 | 2003 | Ebola virus | Mbandza Mbomo, Cuvette Ouest Region, RC | § | § | 35 | 29 | 82.9 |
| 24 | 2004 | Ebola virus | Koltsovo, Russian Federation laboratory accident at the State Research Center of Virology and Biotechnology (Vector) | 1 | 0 | 1 | 1 | 100.0 |
| 25 | 2004 | Sudan virus | Yambio, Sudan (currently South Sudan) | § | § | 17 | 7 | 41.2 |
| 26 | 2005 | Ebola virus | Etoumbi, Republic of the Congo | 1 | 11 | 12 | 9 | 75.0 |
| 27 | 2004–2005 | Marburg virus | Uíge, Angola | 158 | 216 | 374 | 329 | 88.0 |
| 28 | 2007 | Ebola virus | Kasai Occidental Province, Democratic Republic of the Congo | 21 | 202 | 223 | 179 | 80.3 |
| 29 | 2007 | Marburg virus , Ravn virus | Kamwenge, Uganda | 4 | 0 | 4 | 1 | 25.0 |
| 30 | 2007–2008 | Bundibugyo virus | Kikyo and Bundibugyo, Uganda | 30 | 119 | 149 | 37 | 24.8 |
| 31 | 2008 | Marburg virus | Holland (Imported from Uganda) | 1 | 0 | 1 | 1 | 100.0 |
| 32 | 2008–2009 | Ebola virus | Mweka and Luebo health zones in the Province of Kasai Occidental Province, Democratic Republic of the Congo | § | § | 32 | 15 | 46.9 |
| 33 | 2008–2009 | Marburg virus | USA (Imported from Uganda) | 1 | 0 | 1 | 0 | 0.0 |
| 35 | 2011 | Sudan virus | Luwero District, Uganda | 1 | 0 | 1 | 1 | 100.0 |
| 36 | 2012 | Marburg virus | Kabale, Ibanda, Mbarara, and Kampala, Uganda | 15 | 8 | 23 | 9 | 39.1 |
| 37 | 2012 | Sudan virus | Kagadi, Kibaale District, Uganda | 11 | 13 | 24 | 17 | 70.8 |
| 38 | 2012 | Bundibugyo virus | Isiro and Viadana, Haut-Uélé District, Province Orientale, Democratic Republic of the Congo | 36 | 16 | 52 | 25 | 48.1 |
| 39 | 2012 | Sudan virus | Luweero District, Uganda | 6 | 1 | 7 | 4 | 57.1 |
| 40** | 2014 (as of 18 Sept. 2014) | Ebola virus | West Africa (To date: Guinea, Liberia, Sierra Leone, Nigeria, and Senegal) Cases exported to USA, Spain, and others were recorded in the country where the transmission occurred. | 3341 | 2523 | 5864 | 2811 | 47.9 |
| 41** | 2014 (as of 18 Sept. 2014) | Ebola virus | Boende District, Equateur Province, Democratic Republic of the Congo | 28 | 40 | 68 | 41 | 60.3 |
| Total | 3979 | 3769 | 8883 | 4921 | 55.4 | |||
* CFR = case fatality ratio = deaths divided by total cases. § Although an undetermined number of cases were stated to be laboratory-confirmed by the World Health Organization and/or peer-reviewed and published scientific articles, the precise number of confirmed versus putative cases was not mentioned. ** Outbreak is ongoing at time of publication of this paper.
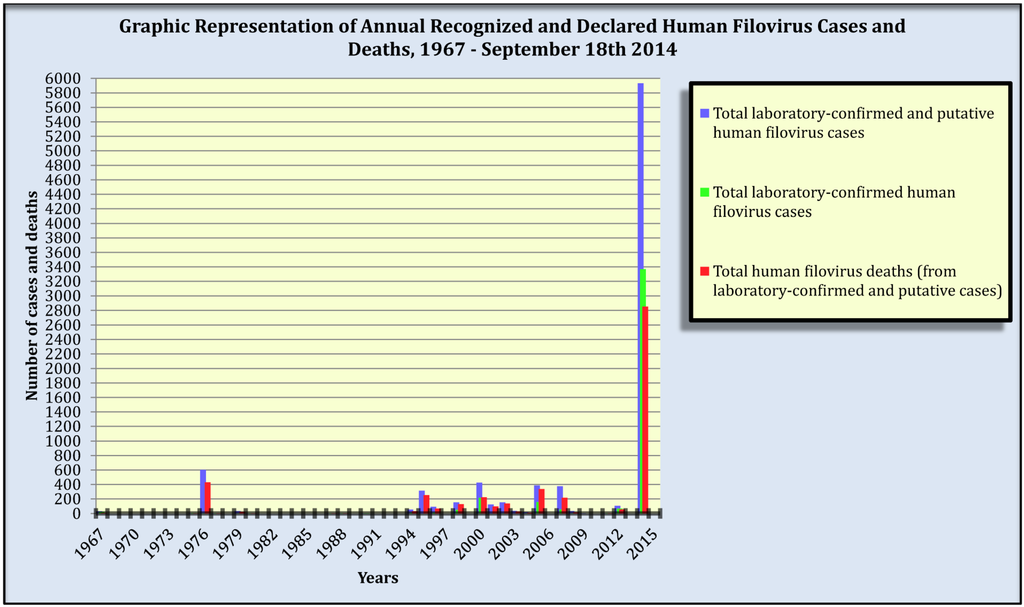
Figure 1.
Annual recognized and declared filovirus-disease cases and deaths in humans, 1967–18 September 2014. Note: Biosafety level-4 laboratory accidental filovirus exposures are included as they involve human cases. Filovirus-disease cases and deaths from outbreaks that occurred over multiple years are assigned to their first year of occurrence. Data source: Table 1.
2.3. Challenges and Shortcomings in Outbreak Preparedness and Response
2.3.1. Challenges
The 2014 EVD outbreak in West Africa (To date: Guinea, Liberia, Sierra Leone, Nigeria, and Senegal) [,,,,,] prompts recollection of some of the inherent, formidable, and reoccurring challenges filovirus ORTs experience when implementing disease control and treatment strategies in geographically dispersed communities served by antiquated health systems [,]. For example, to effectively manage and implement response components, teams must continuously replenish their numerous multidisciplinary and multisectoral human resources, who routinely operate in remote locations [,,,,,]. Response efforts are further complicated when components are suspended or diminished ensuing community resistance to the intervention due to fear of the disease and misconception of outbreak response objectives and components, as transpired in Gabon in 2002 [], the Republic of the Congo in 2002 and 2003 [,,], Angola in 2005 [], and West Africa in 2014 [,,,,]. Finally, as seen in previous outbreaks [] and most recently in 2014 in Guinea [] and the Democratic Republic of the Congo [], another challenge to filovirus-disease outbreak response includes the weeks or months of habitually unrecognized secondary transmission occurring in a community prior to the recognition and declaration of the outbreak, which contribute to high filovirus-disease case numbers and wide geographic spread [,].
ORTs diligently work to overcome these and other challenges, in part by sensitising affected communities about filovirus disease, transmission routes, and outbreak response objectives and components. Notwithstanding, below is a non-exhaustive list of identified and current filovirus-disease outbreak preparedness and response shortcomings, followed by a proposal aimed at Ministries of Health of outbreak-prone countries, the WHO, MSF, CDC, and others to consider for enhancing future efforts.
2.3.2. Shortcoming #1—Data Collection Initiatives
2.3.2.1. Epidemiological Data
Filovirus-disease outbreaks continue to be plagued by poor epidemiological and clinical data collection initiatives. Surveillance teams typically use epidemiological data to identify and follow-up primary and/or secondary transmission contact links [,,,,], an essential outbreak control activity []. Contact tracing databases such as the WHO Field Information Management System (FIMS) [], schematic secondary transmission-chain representations of epidemiological contact-tracing investigations [], and/or an Epi Info™ application recently designed by the CDC [,] have been created to facilitate these efforts.
Regrettably, these databases currently lack involved inter-organizational ownership, regularly scheduled user training, and—particularly when data comprise patient demographic, epidemiological, and clinical variables—data-sharing agreements approved by the ethical review mechanisms of each ORT organization, including the relevant Ministries of Health. These lacunae have likely contributed to the intermittent employment of FIMS in filovirus-disease outbreak settings since its 2005 inception and to the inter-outbreak methodological variance in schematic secondary transmission-chain representations. Also, despite its stated potential for inter-agency communication and data management efficiency, as well as its epidemiology, laboratory, clinical, and mapping module design input received from individual members of the WHO, MSF, and the Uganda Ministry of Health, and subsequent pilot testing [], the effectiveness of the proposed Epi Info™ application would likely be impeded by the non-proficiency in Epi Info™ among an ORT’s high human-resource numbers and turn-over rate [,,]. Onsite Epi Info™ training conducted near the end of an outbreak, when incidence rates have abated, would largely be ineffective for facilitating control efforts for that particular outbreak, while training sessions conducted during the height of an outbreak would be quixotic and inadvisable [,] as ORT members are responsible for and immersed in a multitude of intervention activities, leaving insufficient time to attend software training sessions. Ideally, relevant inter-organizational ORT members from relevant Ministries of Health, the WHO, MSF, CDC, others would receive regularly scheduled database training between outbreak occurrences and deploy to outbreak settings with the required software proficiency.
Despite the current lacunae, these databases facilitate outbreak control, and their future use is encouraged. However, outbreak control efficiency and effectiveness can be strengthened through inter-organizational preparedness, which would remove a multidisciplinary and multisectoral ORT’s dependence on a single organization to manage and analyze epidemiological and clinical data for real-time, intra-outbreak decision making. Ministries of Health of outbreak-prone countries and international ORT organizations must foster involved ownership, commit to regularly scheduled human-resource training, particularly between outbreak occurrences, and ensure the ethical use of patient data.
2.3.2.2. Clinical Data
Filovirus-disease clinical data-collection initiatives in human outbreak settings have consistently yielded low-quality data and few peer-reviewed published analyses to contribute knowledge of these poorly understood diseases. Moreover, to date, despite the same organizations responding to all 24 recognized human filovirus-disease outbreaks that have occurred in sub-Saharan Africa since 1995 (Table 1), clinical data have not been systematically collected; habitually fail to record patients’ symptom onset, frequency, and duration; are often obtained without written and informed patient or caregiver consent [,]; and lamentably, for many outbreaks, not collected at all. Stated previously [,,,], and with continued relevance today, concise yet thorough data collection guidelines, templates, training, and armamentarium, similar to those used for intensive care patients in industrialized countries, must be prioritized through inter-organizational preparedness initiatives prior to the next outbreak occurrence and beyond.
2.3.3. Shortcoming #2—Evidence-based Case Management
Coupled with the feasibility of provision in an outbreak setting and an affected community’s values and preferences, optimal filovirus-disease medical care should be defined by methodologically sound, patient-centered clinical research [,,,]. However, to date, best practice for filovirus-disease case management is primarily based on anecdotal evidence, while the impact of supportive and/or innovative treatment on clinical outcome is unknown []. Moreover, few scientific studies have been designed and implemented to critically evaluate treatment effectiveness. Beyond the current primary focus on filovirus-disease containment [], ORTs must aim to apply an appropriate and Ethical Review Board-approved study design for the collection and analyses of high-quality epidemiological and clinical data to generate improved evidence for supportive and/or innovative treatment effectiveness in human outbreak settings [].
2.3.4. Shortcoming #3—Outbreak Preparedness and Response Guidelines
In 1998, together with contributions from over 20 relevant organizations, the CDC and the WHO jointly produced an infection control manual for viral haemorrhagic fevers in the African health care setting []. Later, in 2008, the WHO produced an interim summary of infection control recommendations when providing care to filovirus patients []. That same year, MSF developed an internal filovirus-disease outbreak-response guideline to provide relevant MSF staff with a practical summary of filovirus-disease intervention objectives, activities, and lessons learned from previous outbreaks []. The MSF internal guideline summarized a draft of what was meant to be the official MSF Filovirus Outbreak Control Guidance Manual; to date this manual has not yet been completed. Most recently, during the 2014 EVD outbreak in West Africa, the WHO released an interim guideline for filovirus-disease outbreak preparedness, alert, control, and evaluation [], an interim infection-prevention and control guideline for the care of patients with filovirus disease [], and a guideline for the clinical management of patients with viral haemorrhagic fever [].
Collectively, the above-mentioned filovirus guidelines are informative and advise ORT control and treatment strategies. Notwithstanding, the technical content provided in these guidelines, particularly with respect to filovirus epidemiology, ecology, data collection templates and procedures, information and education campaigns, case definitions, laboratory diagnoses, treatment, and lessons learned, all require further elaboration, improvement, harmonization, and updating, ideally prior to the next outbreak occurrence. Ministries of Health of outbreak-prone countries, the WHO, MSF, CDC, and others would greatly enhance the efficiency and effectiveness of their filovirus-disease outbreak preparedness and response if they collaboratively developed and implemented a technically sound, comprehensive, and updated inter-organizational guideline that incorporates scientific and technical advances since 2008 and responds to the increasing expectation on their ORTs to improve their data collection and case management strategies [,,].
2.3.5. Shortcoming #4—Surveillance in Outbreak-prone Countries
In sub-Saharan Africa, antiquated health systems and the non-ubiquity of filovirus-disease surveillance mechanisms, trained human resources, and diagnostic capacity all contribute to the paucity of functioning filovirus-disease surveillance systems. The 2014 EVD outbreak in West Africa has demonstrated, once again, that filovirus-disease outbreaks are often unpredictable in their timing and, within sub-Saharan Africa, their location [,]; unrecognized—particularly in unmonitored areas [,,,]; and undiagnosed until disease amplification occurs in a health facility and/or community, often weeks or months after secondary transmission commenced [,,,,,,,,,,,,,,].
Yet, outbreak control and treatment efforts are facilitated when an intervention follows early recognition of disease transmission [,]. ORTs must initiate discussion on how to best create and sustain a functional local, national, regional, and/or international filovirus-disease surveillance network in outbreak-prone countries, a formidable yet plausible endeavor. Established high-containment laboratories in filovirus-disease outbreak-prone countries that strengthen local and national surveillance and perform filovirus and differential diagnostics, pathogenesis, ecological research, and more, such as the International Center for Medical Research in Franceville, Gabon (CIRMF) [], the Uganda Virus Research Institute (UVRI) and CDC in Entebbe, Uganda [], mobile laboratory units [,], and others may provide guidance for creating, implementing, and sustaining functional filovirus-disease surveillance systems. Additionally, relevant Ministries of Health have recently adopted regulations [], strategies [], and frameworks [,], and created national and regional surveillance initiatives [,], which should inform these efforts.
3. Proposal for Enhancing Filovirus-Disease Outbreak Preparedness and Response
To date, within ORTs no single individual or group has been able to devote the time and resources required to adequately advance filovirus-disease data-collection initiatives, evidence-based case management, comprehensive outbreak-response guidelines, international surveillance networks, and more. Between outbreaks, when thorough preparedness and research endeavours are ideally conducted, ORT members are occupied with other non-filovirus-disease-related components of their employment. As such, much of the preparedness and research for the benefit of the efficiency and effectiveness of future outbreak response is forsaken.
It is proposed that, in accordance with the principles of the ethical practice of public health [] and research [], and previously suggested frameworks [,], the Ministries of Health of outbreak-prone countries, the WHO, MSF, CDC, and others promptly create and sustain a functional, multisectoral, and multidisciplinary filovirus-disease working group to advance outbreak preparedness and response efforts. The pertinence of such a working group is underscored by the recent increase in frequency and magnitude of recognized and declared human-filovirus disease outbreaks and the potential ubiquity of pathogenic filoviruses throughout sub-Saharan Africa. Critical components of this proposal include the following.
- As the WHO is responsible for providing leadership on global health matters, shaping health research agendas, setting norms and standards, articulating evidence-based policy options, providing technical support to countries, and monitoring and assessing health trends [,] it should convene, coordinate, and ensure maintained funding for a functional filovirus-disease working group.
- The filovirus-disease working group would promptly convene to define its objectives, activities, timelines, and expected outputs and deliverables.
- The proposed overall objective of the filovirus-disease working group would be to develop and implement frameworks, guidelines, protocols, templates, and trainings for efficient and effective filovirus-disease outbreak preparedness and response.
- The participating members of the filovirus-disease working group would collaboratively address the preparedness and response shortcomings identified in this paper and elsewhere.
- All patient data collection and analysis initiatives would prioritize and seek the guidance and approval of relevant Ethical Review Boards.
- Core members of the filovirus-disease working group should be agile and resilient to be deployed immediately on outbreak recognition [].
- Proposed outputs and deliverables of the filovirus-disease working group include, but are not limited to, the development and implementation in outbreak settings of the following.
- Protocols, templates, and trainings that ensure the collection and analyses of high-quality epidemiological and clinical data;
- Study protocol(s) with an appropriate evaluation scheme for evidence-based case management;
- Inter-organizational, technically-sound, comprehensive, and updated, outbreak-response guidelines;
- Framework(s) for a filovirus-disease surveillance network in outbreak-prone countries, including corresponding guidelines, inter-organizational agreements, protocols, trainings, and templates.
4. Conclusion
With uncertain timing and location in sub-Saharan Africa, frequent and high magnitude filovirus-disease outbreaks will likely occur again. The idea of improving filovirus-disease outbreak preparedness and response must therefore be embraced. This paper stands as a call for prompt action by Ministries of Health of outbreak-prone countries, the WHO, MSF, CDC, and others to create and sustain a functional, multisectoral, and multidisciplinary filovirus-disease working group to address the inherent challenges and current shortcomings in filovirus-disease outbreak preparedness and response, which would greatly enhance future efforts.
Conflicts of Interest
The author declares no conflicts of interest.
References and Notes
- Kuhn, J.H.; Becker, S.; Ebihara, H.; Geisbert, T.W.; Jahrling, P.B.; Kawaoka, Y.; Netesov, S.V.; Nichol, S.T.; Peters, C.J.; Volchkov, V.E.; et al. Family Filoviridae. In Virus Taxonomy: Ninth Report of the International Committee on Taxonomy of Viruses; King, A.M.Q., Adams, M.J., Carstens, E.B., Lefkowitz, E.J., Eds.; Elsevier/Academic Press: London, UK, 2011; pp. 665–671. [Google Scholar]
- Adams, M.J.; Lefkowitz, E.J.; King, A.M.Q.; Carstens, E.B. Ratification vote on taxonomic proposals to the International Committee on Taxonomy of Viruses (2014). Arch. Virol. 2014. [Google Scholar] [CrossRef]
- Kuhn, J.H.; Becker, S.; Ebihara, H.; Geisbert, T.W.; Johnson, K.M.; Kawaoka, Y.; Lipkin, W.I.; Negredo, A.I.; Netesov, S.V.; Nichol, S.T.; et al. Proposal for a revised taxonomy of the family Filoviridae: classification, names of taxa and viruses, and virus abbreviations. Arch. Viro. 2010, 155, 2083–2103. [Google Scholar] [CrossRef]
- Negredo, A.; Palacios, G.; Vazquez-Moron, S.; González, F.; Dopazo, H.; Molero, F.; Juste, J.; Quetglas, J.; Savji, N.; de la Martínez, M; et al. Discovery of an Ebolavirus-Like Filovirus in Europe. PLoS Pathogens 2011, 7, e1002304. [Google Scholar] [CrossRef]
- Jeffs, B.; Roddy, P.; Weatherill, D.; de la Rosa, O.; Dorion, C.; Iscla, M.; Grovas, I.; Palma, P.P.; Villa, L.; Bernal, O.; et al. The Médecins Sans Frontières intervention in the Marburg hemorrhagic fever epidemic, Uige/Angola, 2005. I. Lessons learned in the hospital. J. Infect. Dis. 2007, 196, S154–S161. [Google Scholar]
- Roddy, P.; Weatherill, D.; Jeffs, B.; Abaakouk, Z.; Dorion, C.; Rodríguez-Martínez, J.; Palma, P.P.; de la Rosa, O.; Villa, L.; Grovas, I.; Borchert, M. The Médecins Sans Frontières intervention in the Marburg hemorrhagic fever epidemic, Uige, Angola, 2005. II. Lessons learned in the community. J. Infect. Dis. 2007, 196, S162–S167. [Google Scholar]
- The WHO. Public Health Events of Initially Unknown Etiology: A Framework for Preparedness and Response in the African Region; WHO Regional Office for Africa: Geneva, Switzerland, 2014. ISBN: 978-929-023-2476 (NLM Classification: WA 105). Available online: http://www.afro.who.int/en/media-centre/pressreleases/item/6551-who-afro-issues-guidance-on-preparing-for-responding-to-public-health-events.html (accessed on 31 August 2014).
- Roddy, P. Ebola: Anticipating challenges and improving patient health and safety measures on makeshift filovirus wards. Humanitarian Practice Network Online Exchange. 25 May 2014. Available online: http://www.odihpn.org/the-humanitarian-space/news/announcements/blog-articles/ebola-anticipating-challenges-and-imJzproving-patient-health-and-safety-measures-on-makeshift-filovirus-wards (accessed on 31 August 2014).
- WHO; CDC. Infection Control for Viral Haemorrhagic Fevers in the African Health Care Setting; WHO: Geneva, Switzerland, 1998. Available online: http://www.who.int/csr/resources/publications/ebola/whoemcesr982sec1-4.pdf (accessed on 31 August 2014).
- Hewlett, B.S.; Amola, R.P. Cultural contexts of Ebola in Northern Uganda. Emerg. Infect. Dis. 2003, 9, 1242–1248. [Google Scholar] [PubMed]
- WHO. Outbreak(s) of Ebola haemorrhagic fever, Congo and Gabon, October 2001–July 2002. Wkly. Epidemiol. Rec 2003, 78, 223–228. [Google Scholar]
- Larkin, M. Ebola outbreak in the news. Lancet Infect. Dis. 2003, 3, 255. [Google Scholar]
- Formenty, P.; Libama, F.; Epelboin, A.; Allarangar, Y.; Leroy, E.; Moudzeo, H.; Tarangonia, P.; Molamou, A.; Lenzi, M.; Ait-Ikhlef, K.; et al. L’épidémie de fièvre hémorragique a virus Ebola en République du Congo, 2003: une nouvelle stratégie? Méd. Trop. 2003, 63, 291–295. [Google Scholar]
- Hewlett, B.S.; Epelboin, A.; Hewlett, B.L.; Formenty, P. Medical anthropology and Ebola in Congo: cultural models and humanistic care. Bull. Exot. Pathol. Soc. 2005, 98, 230–236. [Google Scholar]
- Bausch, D.G.; Feldmann, H.; Geisbert, T.W.; Bray, M.; Sprecher, A.G.; Boumandouki, P.; Rollin, P.E.; Roth, C. Outbreaks of filovirus hemorrhagic fever: Time to refocus on the patient. J. Infect. Dis. 2007, 196, S136–S141. [Google Scholar] [PubMed]
- Raabe, V.; Mutyaba, I.; Roddy, P.; Lutwama, J.J.; Geissler, W.; Borchert, M. Infection control during filoviral hemorrhagic fever outbreaks: Preferences of community members and health workers in Masindi, Uganda. Trans. Royal Soc. Trop. Med. Hyg. 2010, 104, 48–50. [Google Scholar]
- Roddy, P.; Colebunders, R.; Jeffs, B.; Palma, P.P.; Van Herp, M.; Brochert, M. Filovirus hemorrhagic fever outbreak case management: A review of current and future treatment options. J. Infect. Dis. 2011, 204, S791–S795. [Google Scholar]
- Roddy, P.; Howard, N.; Van Kerkhove, M.D; Lutwama, J.; Wamala, J.; Yoti, Z.; Colebunders, R.; Palma, P.P.; Sterk, E.; Jeffs, B.; et al. Clinical Manifestations and Case Management of Ebola Haemorrhagic Fever Caused by a Newly Identified Virus Strain, Bundibugyo, Uganda, 2007–2008. PLoS One 2012, 7, e52986. [Google Scholar] [CrossRef]
- Roddy, P.; Thomas, S.L.; Jeffs, B.; Folo, P.N.; Palma, P.P.; Henrique, B.M.; Villa, L.; Machado, F.P.D.; Bernal, O.; Jones, S.M.; et al. Factors associated with Marburg hemorrhagic fever: Analysis of patient data from Uige, Angola. J. Infect. Dis. 2010, 201, 1909–1918. [Google Scholar]
- Baize, S.; Pannetier, D.; Oestereich, L.; Rieger, T.; Koivogui, L.; Magassouba, N.; Soropogui, B.; Sow, M.S.; Keita, S.; De Clerk, H.; et al. Emergence of Zaire Ebola Virus Disease in Guinea—Preliminary Report. New Engl. J. Med. 2014. [Google Scholar] [CrossRef]
- Feldman, H. Ebola—A Growing Threat? New Engl. J. Med. 2014. [Google Scholar] [CrossRef]
- Siegert, R.; Shu, H.L.; Slenczka, W.; Peters, D.; Müller, G. On the etiology of an unknown human infection originating from monkeys. Deutsche Medizinische Wochenschrif 1967, 92, 2341–2343. [Google Scholar]
- Martini, G.A.; Knauff, H.G.; Schmidt, H.A.; Mayer, G.; Baltzer, G. Über eine bisher unbekannte, von Affen eingeschleppte Infektionskrankheit: Marburg-Virus-Krankheit. “Marburgvirus” disease. Dtsch. Med. Wochenschr. 1968, 93, 559–571. [Google Scholar] [PubMed]
- Kuhn, J.H. Filoviruses: A Compendium of 40 Years of Epidemiological, Clinical, and Laboratory Studies; Springer: Wien, Austria, 2008. [Google Scholar]
- Bausch, D.G.; Sprecher, A.G.; Jeffs, B.; Boumandouki, P. Treatment of Marburg and Ebola hemorrhagic fevers: A strategy for testing new drugs and vaccines under outbreak conditions. Antivir. Res. 2008, 78, 150–161. [Google Scholar] [PubMed]
- WHO. Case of Marburg Haemorrhagic Fever Imported into the Netherlands from Uganda; WHO: Geneva, Switzerland, 2008. Available online: http://www.who.int/csr/don/2008_07_10/en/index.html (accessed on 31 August 2014).
- WHO. Ebola Haemorrhagic Fever in the Democratic Republic of the Congo; WHO: Geneva, Switzerland, 2008. Available online: http://www.who.int/csr/don/2008_12_26a/en/index.html (accessed on 31 August 2014).
- CDC. Imported case of Marburg hemorrhagic fever—Colorado, 2008. Morb. Mortal. Wkly. Rep. 2009, 58, 1377–1381. [Google Scholar]
- Tuffs, A. Trial vaccine may have saved Hamburg scientist from Ebola fever. Br. Med. J. 2009, 338, b1223. [Google Scholar]
- WHO. Ebola in Uganda; WHO: Geneva, Switzerland, 2011. Available online: http://www.who.int/csr/don/2011_05_18/en/index.html (accessed on 31 August 2014).
- WHO. End of Ebola Outbreak in Uganda; WHO: Geneva, Switzerland, 2012. Available online: http://www.who.int/csr/don/2012_10_04/en/index.html (accessed on 31 August 2014).
- WHO. End Outbreak in Democratic Republic of Congo; Update. WHO: Geneva, Switzerland, 2012. Available online: http://www.who.int/csr/don/2012_10_26/en/index.html (accessed on 31 August 2014).
- WHO. Marburg Haemorrhagic Fever in Uganda; Update. WHO: Geneva, Switzerland, 22 October 2012. Available online: http://www.who.int/csr/don/2012_10_22/en/index.html (accessed on 31 August 2014).
- WHO. Ebola in Uganda—Update; WHO: Geneva, Switzerland, 2012. Available online: http://www.who.int/csr/don/2012_11_30_ebola/en/index.html (accessed on 31 August 2014).
- Albariño, C.G.; Shoemaker, T.; Khristova, M.L.; Wamala, J.F.; Muyembe, J.J.; Balinandi, S.; Tumusime, A.; Campbell, S.; Cannon, D.; Gibbons, A.; et al. Genomic analysis of filoviruses associated with four viral hemorrhagic fever outbreaks in Uganda and the Democratic Republic of the Congo in 2012. Virology 2013, 442, 97–100. [Google Scholar]
- WHO. WHO Ebola Response Roadmap; Updated on 22 September 2014. WHO: Geneva, Switzerland, 2014. Available online: http://apps.who.int/iris/bitstream/10665/134449/1/roadmapupdate22sept14_eng.pdf?ua=1 (accessed on 23 September 2014).
- Bray, M.; Murphy, F.A. Filovirus research: Knowledge expands to meet a growing threat. J. Infect. Dis. 2007, 196, S438–S443. [Google Scholar] [PubMed]
- Brown, H.; Kelly, A.H. Material Proximities and Hotspots: Toward an Anthropology of Viral Hemorrhagic Fevers. Med. Anthropol. Q. 2014. [Google Scholar] [CrossRef]
- WHO. Ebola haemorrhagic fever in Zaire, 1976: Report of an international commission. Bull. WHO 1978, 56, 271–293. [Google Scholar]
- Baron, R.C.; McCormick, J.B.; Zubeir, O.A. Ebola virus disease in southern Sudan: Hospital dissemination and intrafamilial spread. Bull. WHO 1983, 61, 997–1003. [Google Scholar] [PubMed]
- Ignatyev, G.M.; Streltsova, M.A.; Kashentseva, E.A.; Patrushev, N.A.; Ginko, S.I.; Agafanov, A.A. Immunity Indices in the Personnel Involved in Hemorrhagic Virus Investigation. In ERDEC Scientific Conference on Chemical and Biological Defense Research—Final Report; Berg Dorothy, A., Ed.; Edgewood Research Development & Engineering Center, U.S. Army Chemical and Biological Defense Command, Aberdeen Proving Ground: Washington, DC, USA, 19–22 November 1997; pp. 323–330. [Google Scholar]
- Leroy, E.M.; Baize, S.; Debre, P.; Lansoud-Soukate, J.; Mavoungou, E. Early immune responses accompanying human asymptomatic Ebola infections. Clin. Exp. Immunol. 2001, 124, 453–460. [Google Scholar] [PubMed]
- Borchert, M.; Muyembe-Tamfum, J.J.; Colebunders, R.; Libande, M.; Sabue, M.; Van der Stuyft, P. A cluster of Marburg virus disease involving an infant. Trop. Med. Int. Health 2002, 10, 902–906. [Google Scholar]
- Leroy, E.M.; Telfer, P.; Kumulungui, B.; Yaba, P.; Rouquet, P.; Roques, P.; Gonzalez, J.P.; Ksiazek, T.G; Rollin, P.; Nerrienet, E. A serological survey of Ebola virus infection in central African nonhuman primates. J. Infect. Dis. 2004, 190, 1895–1899. [Google Scholar] [PubMed]
- Gonzalez, J.P.; Herbreteau, V.; Morvan, J.; Leroy, E.M. Ebola virus circulation in Africa: A balance between clinical expression and epidemiological silence. Bull. Soc. Pathol. Exot. 2005, 98, pp. 210–217. Available online: http://www.pathexo.fr/bull_bulletin.php?id_bull=98&id_volume=250 (accessed on 31 August 2014). [PubMed]
- Pourrut, X.; Delicat, A.; Rollin, P.E.; Ksiazek, T.G.; Gonzalez, J.P.; Leroy, E.M. Spatial and temporal patterns of Zaire ebolavirus antibody prevalence in the possible reservoir bat species. J. Infect. Dis. 2007, 196, S176–S183. [Google Scholar] [PubMed]
- Monath, T.P. Ecology of Marburg and Ebola viruses: Speculations and directions for future research. J. Infect. Dis. 1999, 179, S127–S138. [Google Scholar] [PubMed]
- Bausch, D.G.; Borchert, M.; Grein, T.; Roth, C.; Swanepoel, R.; Libande, M.L.; Talamine, A.; Bertherat, E.; Muyembe-Tamfum, J.J.; Tugume, B.; et al. Risk Factors for Marburg hemorrhagic fever, DRC. Emerg. Infect. Dis. 2003, 9, 1531–1537. [Google Scholar] [PubMed]
- Gonzalez, J.P.; Nakoune, E.; Slenczka, W.; Vidal, P.; Morvan, J.M. Ebola and Marburg virus antibody prevalence in selected populations of the Central African Republic. Microb. Infect. 2000, 2, 39–44. [Google Scholar]
- Allela, L.; Boury, O.; Pouillot, R.; Delicat, A.; Yaba, P.; Kumulungui, B.; Rouquet, P.; Gonzalez, J.P.; Leroy, E.M. Ebola virus antibody prevalence in dogs and human risk. Emerg. Infect. Dis. 2005, 11, 385–390. [Google Scholar] [PubMed]
- Johnson, E.D.; Johnson, B.K.; Silverstein, D.; Tukei, P.; Geisbert, T.W.; Sanchez, A.N.; Jahrling, P.B. Characterization of a new Marburg virus isolated from a 1987 fatal case in Kenya. Arch. Virol. 1996, 11, 101–114. [Google Scholar]
- Leroy, E.M.; Kumulungui, B.; Pourrut, X.; Rouquet, P.; Hassanin, A.; Yaba, P.; Delicat, A.; Paweska, J.T.; Gonzales, J.-P.; Swanepoel, R. Fruit bats as reservoirs of Ebola virus. Nature 2005, 438, 575–576. [Google Scholar] [PubMed]
- Towner, J.S.; Pourrut, X.; Albarino, C.G.; Nkogue, C.Z.; Bird, B.H.; Ksiazek, T.G.; Gonzales, J.-P.; Nichol, S.T.; Leroy, E.M. Marburg virus infection detected in a common African bat. PLoS One 2007, 2, e764. [Google Scholar] [PubMed]
- Swanepoel, R.; Smit, S.B.; Rollin, P.E.; Formenty, P.; Leman, P.A.; Kemp, A.; Burt, F.J.; Grobbelaar, A.A.; Croft, J.; Bausch, D.G.; et al. Studies of reservoir hosts for Marburg virus. Emerg.Infect.Dis. 2007, 13, 1847–1851. [Google Scholar] [PubMed]
- Towner, J.S.; Amman, B.R.; Sealy, T.K.; Reeder Carroll, S.A.; Comer, J.A.; Kemp, A.; Swanepoel, R.; Paddock, C.D.; Balinandi, S.; Khristova, M.L.; et al. Isolation of Genetically Diverse Marburg Viruses from Egyptian Fruit Bats. LoS Pathogens 2009, 5, e1000536. [Google Scholar] [PubMed]
- Leroy, E.M.; Epelboin, A.; Mondonge, V.; Pourrut, X.; Gonzales, J-P.; Muyembe-Tamfum, J.J.; Formenty, P. Human ebola outbreak resulting from direct exposure to fruit bats in Luebo, Democratic Republic of Congo, 2007. Vector-Borne Zoonot. Dis. 2009, 9, 723–728. [Google Scholar] [CrossRef]
- Taniguchi, S.; Watanabe, S.; Masangkay, J.S.; Omatsu, T.; Ikegami, T.; Alviola, P.; Ueda, N.; Iha, K.; Fujii, H.; Ishii, Y.; et al. Reston Ebolavirus antibodies in bats, the Philippines. Emerg. Infect. Dis. 2011, 17, 1559–1560. [Google Scholar] [PubMed]
- Pourrut, X.; Souris, M.; Towner, J.S.; Rollin, P.E.; Nichol, S.T.; Gonzales, J-P.; Leroy, E. Large serological survey showing cocirculation of Ebola and Marburg viruses in Gabonese bat populations, and a high seroprevalence of both viruses in Rousettus aegyptiacus. BMC Infect. Dis. 2009, 9. [Google Scholar] [CrossRef]
- Becquart, P.; Wauquier, N.; Mahlakoiv, T.; Nkoghe, D.; Padilla, C.; Souris, M.; Ollomo, B.; Gonzales, J-P.; De Lamballerie, X.; Kazanji, M.; Leroy, E.M. High Prevalence of Both Humoral and Cellular Immunity to Zaire ebolavirus among Rural Populations in Gabon. PLoS One 2010, 5, e9126. [Google Scholar] [PubMed]
- Kuzmin, I.V.; Bozick, B.; Guagliardo, S.A.; Kunkel, R.; Shak, J.R.; Tong, S.; Rupprecht, C.E. Bats, emerging infectious diseases, and the rabies paradigm revisited. Emerg. Health Threat. J. 2011, 4. [Google Scholar] [CrossRef]
- Weingartl, H.M.; Ebury-Hyatt, C.; Nfon, C.; Leung, A.; Smith, G.; Kobinger, G. Transmission of Ebola virus from pigs to non-human primates. Sci. Rep. 2012, 2. [Google Scholar] [CrossRef] [PubMed]
- WHO. Ebola Virus Disease, West Africa; Updated on 3 July 2014. WHO AFRO: Brazzaville, Republic of Congo, 2014. Available online: http://www.afro.who.int/en/clusters-a-programmes/dpc/epidemic-a-pandemic-alert-and-response/outbreak-news/4216-ebola-virus-disease-west-africa-3-july-2014.html (accessed on 31 August 2014).
- WHO. Ebola Virus Disease in Guinea; WHO AFRO: Brazzaville, Republic of Congo, 2014. Available online: http://www.afro.who.int/en/clusters-a-programmes/dpc/epidemic-a-pandemic-alert-and-response/outbreak-news/4063-ebola-hemorrhagic-fever-in-guinea.html (accessed on 31 August 2014).
- WHO. Ebola Virus Disease, Liberia; Situation as of 30 March 2014. WHO AFRO: Brazzaville, Republic of Congo, 2014. Available online: http://www.afro.who.int/en/clusters-a-programmes/dpc/epidemic-a-pandemic-alert-and-response/outbreak-news/4072-ebola-virus-disease-liberia.html (accessed on 31 August 2014).
- WHO. Ebola Virus Disease, West Africa; Update of 26 May 2014. WHO AFRO: Brazzaville, Republic of Congo, 2014. Available online: http://www.afro.who.int/en/clusters-a-programmes/dpc/epidemic-a-pandemic-alert-and-response/outbreak-news/4143-ebola-virus-disease-west-africa-26-may-2014.html (accessed on 31 August 2014).
- The Lancet. Ebola in west Africa: Gaining community trust and confidence. Lancet 2014, 383. [Google Scholar] [CrossRef]
- Green, A. West Africa struggles to contain Ebola outbreak. Lancet 2014, 383. [Google Scholar] [CrossRef] [PubMed]
- Médecins Sans Frontières. Response to West Africa Ebola Epidemic Remains Dangerously Inadequate. 15 August 2014. Available online: http://www.doctorswithoutborders.org/news-stories/field-news/response-west-africa-ebola-epidemic-remains-dangerously-inadequate (accessed on 31 August 2014).
- Borchert, M.; Mutyaba, I.; Van Kerkhove, M.D.; Lutwama, J.; Luwaga, H.; Bisoborwa, G.; Turyagaruka, J.; Pirard, P.; Ndayimirije, N.; Roddy, P. Ebola haemorrhagic fever outbreak in Masindi District, Uganda: outbreak description and lessons learned. BMC Infect. Dis. 2011, 11, 357. [Google Scholar] [PubMed]
- Médecins Sans Frontières. Successes and Challenges in Response to Guinea Ebola Epidemic. 7 April 2014. Available online: http://www.doctorswithoutborders.org/article/successes-and-challenges-response-guinea-ebola-epidemic (accessed on 31 August 2014).
- Tweh, JD. Liberia: Protest in Kru Town Over Ebola Virus. 7 April 2014. Available online: http://allafrica.com/stories/201404072519.html (accessed on 31 August 2014).
- WHO. Ebola Virus Disease, West Africa; Situation as of 27 May 2014. WHO AFRO: Brazzaville, Republic of Congo, 2014. Available online: http://www.afro.who.int/en/clusters-a-programmes/dpc/epidemic-a-pandemic-alert-and-response/outbreak-news/4150-ebola-virus-disease-west-africa-27-may-2014.html (accessed on 31 August 2014).
- WHO. Ebola Virus Disease, West Africa; Situation as of 5 June 2014. WHO AFRO: Brazzaville, Republic of Congo, 2014. Available online: http://www.afro.who.int/en/clusters-a-programmes/dpc/epidemic-a-pandemic-alert-and-response/outbreak-news/4158-ebola-virus-disease-west-africa-5-june-2014.html (accessed on 31 August 2014).
- Peters, C.J. Marburg and Ebola: Arming ourselves against deadly Filoviruses. New Engl. J. Med. 2005, 352, 2571–2573. [Google Scholar] [PubMed]
- WHO. Ebola Virus Disease—Democratic Republic of Congo; WHO: Geneva, Switzerland, 27 August 2014. Available online: http://who.int/csr/don/2014_08_27_ebola/en/ (accessed on 31 August 2014).
- Georges, A.J.; Leroy, E.M.; Renaut, A.A.; Benissan, C.T.; Nabias, RJ; Ngoc, M.T.; Obiang, P.I.; Lepage, J.P.M.; Bertherat, E.T.; et al. Ebola hemorrhagic fever outbreaks in Gabon, 1994–1997: Epidemiologic and health control issues. J. Infect. Dis. 1999, 179, S65–S75. [Google Scholar] [PubMed]
- Leroy, E.M.; Rouquet, P.; Formenty, P.; Souquière, S.; Kilbourne, A.; Froment, J. M.; Bermejo, M.; Smit, S.; Karesh, W.; Swanepoel, R.; et al. Multiple Ebola virus transmission events and rapid decline of central African wildlife. Science 2004, 303, 387–390. [Google Scholar] [PubMed]
- Rouquet, P.; Froment, J.M.; Bermejo, M.; Kilbourn, A.; Karesh, W.; Reed, P.; Kumulungui, B.; Yaba, P.; Delicat, A.; Rollin, P.E.; et al. Wild animal mortality monitoring and human Ebola outbreaks, Gabon and Republic of Congo, 2001–2003. Emerg. Infect. Dis. 2005, 11, 283–290. [Google Scholar] [PubMed]
- Towner, J.S.; Khristova, M.L.; Sealy, T.K.; Vincent, M.J.; Erickson, B.R.; Bawiec, D.A.; Hartman, A.L.; Comer, J.A.; Zaki, S.R.; Stroher, U.; et al. Marburg virus genomics and association with a large hemorrhagic fever outbreak in Angola. J. Virol. 2006, 80, 6497–6516. [Google Scholar] [PubMed]
- Bejtullahu, A.; Hughes, C.; Anderson, S. Field Information Management System; WHO: Geneva, Switzerland, 2005. Available online: http://www.inf.ed.ac.uk/publications/report/1048.html (accessed on 31 August 2014).
- CDC. CDC Disease Detectives Using New Software Tool in Ebola Hemorrhagic Fever Outbreak; Press Release 29 April 2014. Available online: http://www.cdc.gov/media/releases/2014/p0429-new-software.html (accessed on 31 August 2014).
- Schafer, I.J.; E. Knudsen, E.; Aponte, J.; Knust, B.; Ströher, U.; Rollin, P.E.; Nichol, S.T.; Islam, A. Creating an Epi Info Application for Viral Hemorrhagic Fever Outbreak Data Management. In Proceedings of the 6th International Symposium of Filoviruses, Galveston, TX, USA, 30 April–2 May 2014.
- Colebunders, R.; Tshomba, A.; Van Kerkhove, M.; Bausch, D.G.; Campbell, P.; Libande, M.; Pirard, P.; Tshioko, F.; Mardel, S.; Mulangu, S.; et al. Marburg hemorrhagic fever in Durba and Watsa, Democratic Republic of the Congo: Clinical documentation, features of illness, and treatment. The J. Infect. Dis. 2007, 196, S148–S153. [Google Scholar]
- Sackett, D.L.; Rosenberg, W.M.; Gray, J.A.; Haynes, R.B.; Richarson, W.S. cEvidence based medicine: what it is and what it isn’t. BMJ 1996, 312, 71–72. [Google Scholar] [PubMed]
- Blanchet, K.; Sistenich, V.; Ramesh, A.; Frison, S.; Warren, E.; Hossain, M.; Knight, A.; Lewis, C.; Smith, J.; Woodward, A.; et al. An Evidence Review of Research on Health Interventions in Humanitarian Crises; London School of Hygiene and Tropical Medicine/Harvard School of Public Health: London, UK, 2013. [Google Scholar]
- Ratnayake, R.; Degomme, O.; Roberts, B.; Spiegel, P. Conflict and Health: seven years of advancing science in humanitarian crises. Conf. Health. 2014, 7, 1–6. [Google Scholar]
- Gerdin, M.; Clarke, M.; Allen, C.; Kayabu, B.; Summerskill, W.; Devane, D.; MacLachlan, M.; Spiegel, P.; Ghosh, A.; Zachariah, R.; et al. Optimal Evidence in Difficult Settings: Improving Health Interventions and Decision Making in Disasters. PLoS Medicine 2014, 11, e1001632. [Google Scholar] [CrossRef] [PubMed]
- WHO. Interim Infection Control Recommendations for Care of Patients with Suspected or Confirmed Filovirus (Ebola, Marburg) Haemorrhagic Fever; WHO: Geneva, Switzerland, March 2008. Available online: http://www.who.int/csr/bioriskreduction/interim_recommendations_filovirus.pdf?ua=1 (accessed on 31 August 2014).
- Sterk, E. Filovirus Haemorrhagic Fever Guidelines; Médecins Sans Frontières Operational Center Barcelona: Barcelona, Spain, 2008. [Google Scholar]
- WHO. Ebola and Marburg Virus Disease Epidemics: Preparedness, Alert, Control, and Evaluation, Interim Version 1.1; WHO: Geneva, Switzerland, June 2014. Available online: http://who.int/csr/disease/ebola/PACE_outbreaks_ebola_marburg_en.pdf?ua=1 (accessed on 31 August 2014).
- WHO. Interim Infection Prevention and Control Guidance for Care of Patients with Suspected or Confirmed Filovirus Haemorrhagic Fever in Health-Care Settings, with Focus on Ebola; WHO: Geneva, Switzerland, August 2014. Available online: http://www.who.int/csr/resources/who-ipc-guidance-ebolafinal-09082014.pdf (accessed on 31 August 2014).
- WHO. Clinical Management of Patients with Viral Haemorrhagic Fever: A Pocket Guide for the Front-line Health Worker; WHO: Geneva, Switzerland, 13 April 2014. Available online: http://apps.who.int/iris/bitstream/10665/130883/2/WHO_HSE_PED_AIP_14.05.pdf?ua=1 (accessed on 31 August 2014).
- CDC. Outbreak of Ebola Hemorrhagic Fever Uganda, August 2000–January 2001. Morb. Mortal. Wkly. Rep. 2001, 50, 73–77. [Google Scholar]
- Wamala, J.F.; Lukwago, L.; Malimbo, M.; Nguku, P.; Yoti, Z.; Musenero, M.; Amone, J.; Mbabazi, W.; Nanyunja, M.; Zaramba, S.; et al. Ebola hemorrhagic fever associated with novel virus strain, Uganda, 2007–2008. Emerg. Infect. Dis. 2010, 6, 1087–1092. [Google Scholar] [CrossRef]
- Leroy, E.; Gonzalez, J.P. Filovirus Research in Gabon and Equatorial Africa: The Experience of a Research Center in the Heart of Africa. Viruses 2012, 4, 1592–1604. [Google Scholar] [CrossRef] [PubMed]
- Shoemaker, T.; MacNeil, A.; Balinandi, S.; Campbell, S.; Wamala, J.F.; McMullan, L.K.; Downing, R.; Lutwama, J.; Mbiddle, E.; Ströher, U.; et al. Reemerging Sudan Ebola Virus Disease in Uganda, 2011. Emerg. Infect. Dis. 2012, 18, 1480–1483. [Google Scholar] [CrossRef] [PubMed]
- Grolla, A.; Jones, S.; Kobinger, G.; Sprecher, A.; Girard, G.; Yao, M.; Roth, C.; Artsob, H.; Feldmann, H.; Strong, J.E. Flexibility of Mobile Laboratory Unit in Support of Patien Management During the 2007 Ebola-Zaire Outbreak in the Democratic Republic of Congo. Zoon. Public Health. 2012, 59, 151–157. [Google Scholar] [CrossRef]
- European Mobile Laboratory Project. Available online: http://www.emlab.eu/ (accessed on 31 August 2014).
- WHO. International Health Regulations (2005); WHO: Geneva, Switzerland, 2005, 2nd ed. Available online: http://www.who.int/ihr/publications/9789241596664/en/index.html (accessed on 31 August 2014).
- WHO. Technical Guidelines for Integrated Disease Surveillance and Response in the Africa Region; WHO: Geneva, Switzerland, 2010, 2nd ed. Available online: http://www.afro.who.int/en/clusters-a-programmes/dpc/integrated-disease-surveillance (accessed on 31 August 2014).
- United Nations. Contributing to One World, One Health: A Strategic Framework for Reducing Risk of Infectious Diseases at the Animal-Human-Ecosystem Interface; FAO/OIE/WHO/ UNICEF/UNSIC/World Bank, 2008. Available online: http://www.fao.org/docrep/011/aj137e/aj137e00.HTM (accessed on 31 August 2014).
- ProMED-mail. Ebola Virus Disease—West Africa (38): Sierra Leone Preparedness. 12 May 2014. Available online: http://www.promedmail.org (accessed on 31 August 2014).
- WHO. Ebola Virus Disease, West Africa; Update of 25 April 2014. WHO AFRO: Brazzaville, Republic of Congo, 2014. Available online: http://www.afro.who.int/en/clusters-a-programmes/dpc/epidemic-a-pandemic-alert-and-response/outbreak-news/4121-ebola-virus-disease-west-africa-25-april-2014.html (accessed on 31 August 2014).
- Public Health Leadership Society. Principles of the Ethical Practice of Public Health. 2002. Available online: http://www.apha.org/NR/rdonlyres/1CED3CEA-287E-4185–9CBD-BD405FC60856/0/ethicsbrochure.pdf (accessed on 31 August 2014).
- Calain, P.; Fiore, N.; Poncin, M.; Hurst, S.A. Research Ethics and International Epidemic Response: The Case of Ebola and Marburg Hemorrhagic Fevers. Public Health Eth. 2009, 1, 7–29. [Google Scholar] [CrossRef]
- WHO. About WHO; WHO: Geneva, Switzerland, 2014. Available online: http://www.who.int/about/en/ (accessed on 31 August 2014).
- Clift, C. What’s the World Health Organization For? Final Report from the Centre on Global Health Security Working Group on Health Governance; Chatham House Report. May 2014. Available online: http://www.chathamhouse.org/sites/files/chathamhouse/field/field_document/20140521WHOHealthGovernanceClift.pdf (accessed on 31 August 2014).
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).