Identification of Diaryl-Quinoline Compounds as Entry Inhibitors of Ebola Virus
Abstract
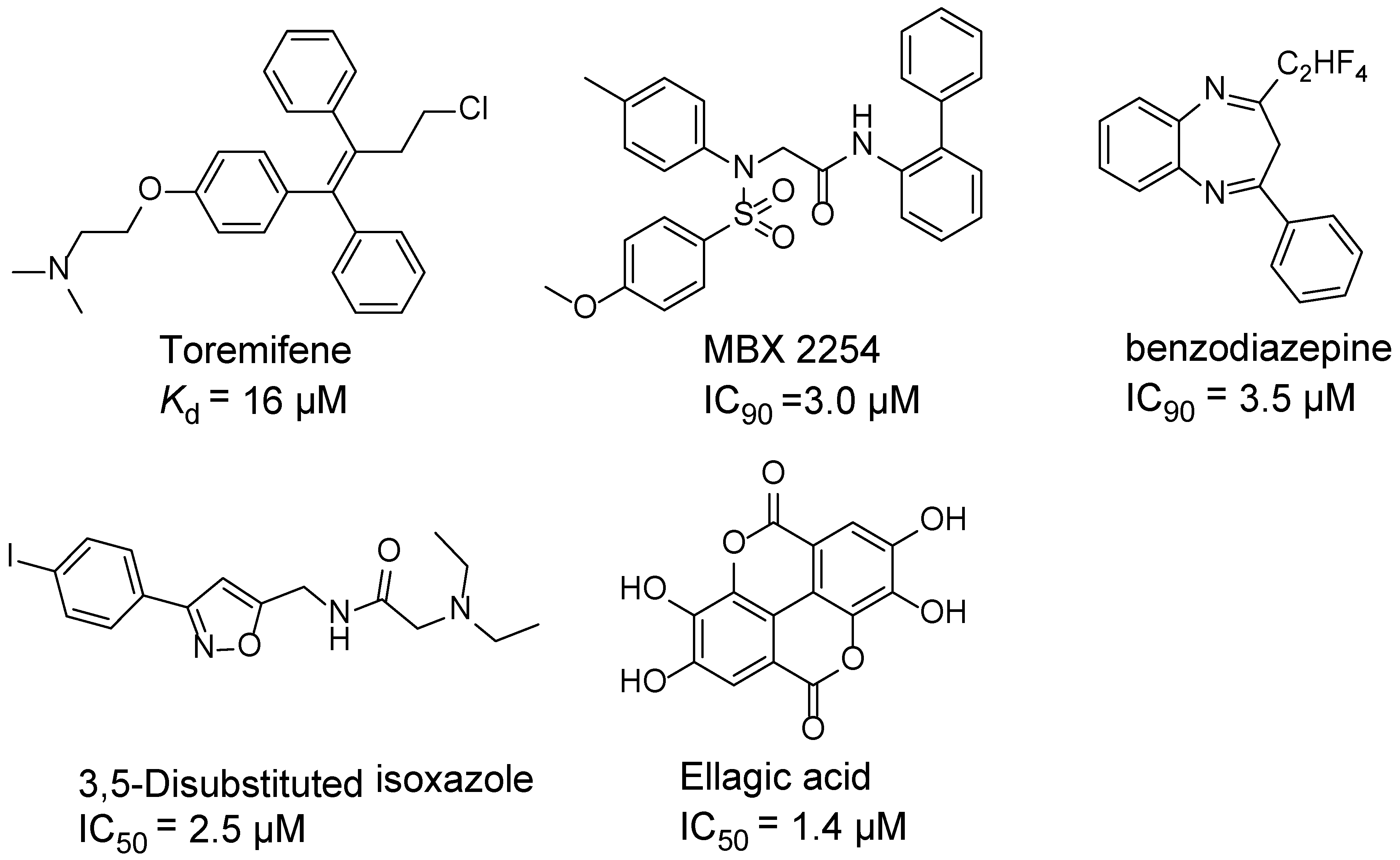
:1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. Generation of Pseudovirions
2.3. Collection of Compounds
2.4. High-Throughput Screen of Anti-EBOV Inhibitor
2.5. Computation Study
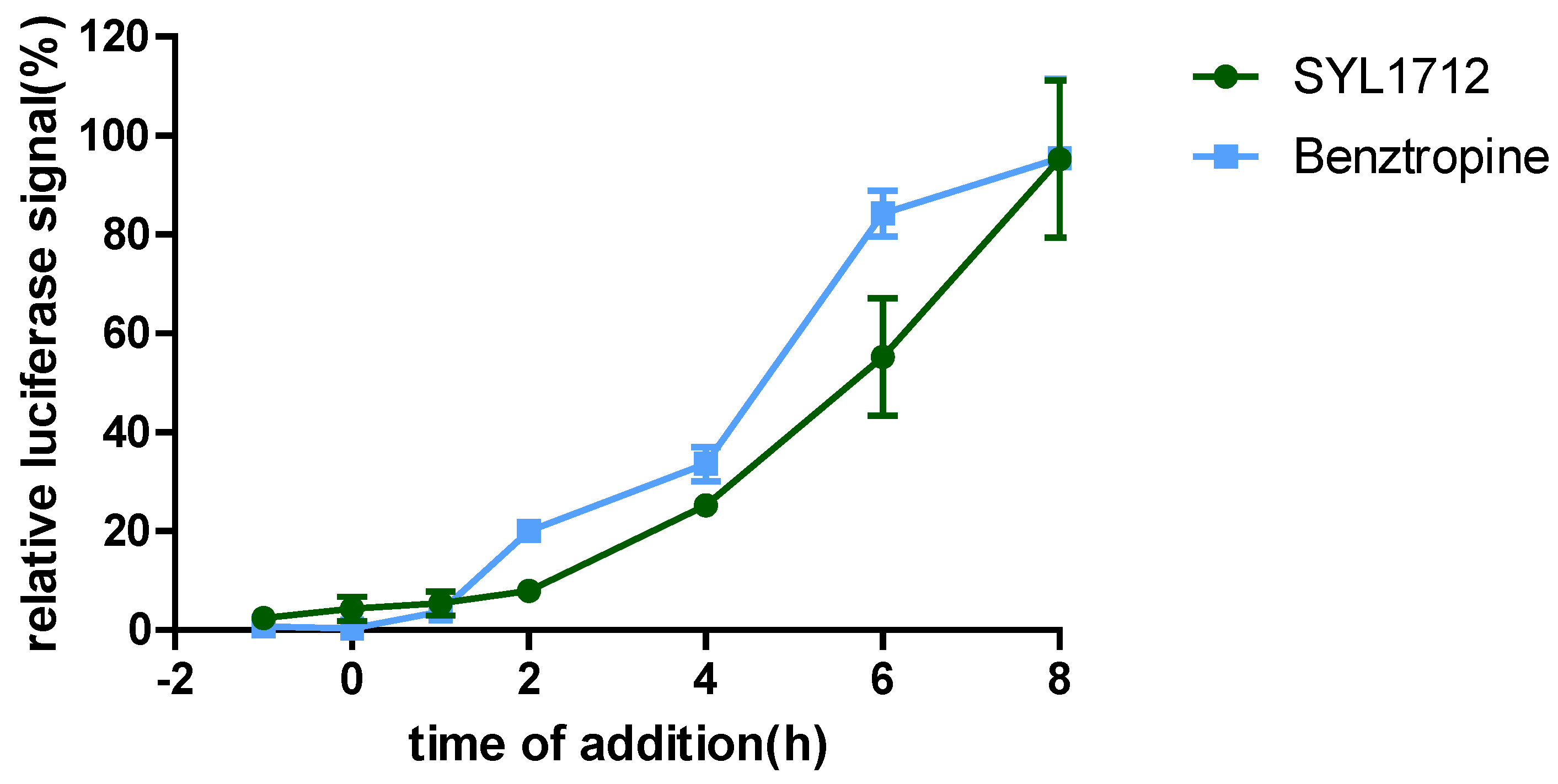
2.6. Time-of-Addition Experiment
2.7. Infectious Virus Assays
3. Results
3.1. Diaryl-Quinoline Compounds Specifically Block the Entry of Ebola Virus
3.2. Time-of-Addition (TOA) Experiments
3.3. Compound SYL1712 Inhibits Replication of Infectious EBOV
3.4. Structure-Activity Relationship and Docking Studies of the Diaryquinoline Compounds
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kuhn, J.H.; Becker, S.; Ebihara, H.; Geisbert, T.W.; Johnson, K.M.; Kawaoka, Y.; Lipkin, W.I.; Negredo, A.I.; Netesov, S.V.; Nichol, S.T.; et al. Proposal for a revised taxonomy of the family filoviridae: Classification, names of taxa and viruses, and virus abbreviations. Arch. Virol. 2010, 155, 2083–2103. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Ebola Virus Disease—Democratic Republic of the Congo. Available online: http://www.Who.Int/csr/don/23-may-2018-ebola-drc/en/ (accessed on 10 May 2018).
- Qiu, X.; Wong, G.; Audet, J.; Bello, A.; Fernando, L.; Alimonti, J.B.; Fausther-Bovendo, H.; Wei, H.; Aviles, J.; Hiatt, E.; et al. Reversion of advanced Ebola virus disease in nonhuman primates with ZMAPP. Nature 2014, 514, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Walldorf, J.A.; Cloessner, E.A.; Hyde, T.B.; MacNeil, A.; CDC Emergency Ebola Vaccine Taskforce. Considerations for use of Ebola vaccine during an emergency response. Vaccine 2017. [Google Scholar] [CrossRef] [PubMed]
- Oestereich, L.; Ludtke, A.; Wurr, S.; Rieger, T.; Munoz-Fontela, C.; Gunther, S. Successful treatment of advanced Ebola virus infection with T-705 (favipiravir) in a small animal model. Antivir. Res. 2014, 105, 17–21. [Google Scholar] [CrossRef] [PubMed]
- Warren, T.K.; Jordan, R.; Lo, M.K.; Ray, A.S.; Mackman, R.L.; Soloveva, V.; Siegel, D.; Perron, M.; Bannister, R.; Hui, H.C.; et al. Therapeutic efficacy of the small molecule gs-5734 against Ebola virus in rhesus monkeys. Nature 2016, 531, 381–385. [Google Scholar] [CrossRef] [PubMed]
- Brown, C.S.; Lee, M.S.; Leung, D.W.; Wang, T.; Xu, W.; Luthra, P.; Anantpadma, M.; Shabman, R.S.; Melito, L.M.; MacMillan, K.S.; et al. In silico derived small molecules bind the filovirus vp35 protein and inhibit its polymerase cofactor activity. J. Mol. Biol. 2014, 426, 2045–2058. [Google Scholar] [CrossRef] [PubMed]
- Schafer, A.M.; Cheng, H.; Lee, C.; Du, R.; Han, J.; Perez, J.; Peet, N.; Manicassamy, B.; Rong, L. Development of potential small molecule therapeutics for treatment of Ebola virus. Curr. Med. Chem. 2018, 25, 5177–5190. [Google Scholar] [CrossRef] [PubMed]
- Cui, Q.; Du, R.; Anantpadma, M.; Schafer, A.; Hou, L.; Tian, J.; Davey, R.A.; Cheng, H.; Rong, L. Identification of ellagic acid from plant Rhodiola rosea l. As an anti-Ebola virus entry inhibitor. Viruses 2018, 10, 152. [Google Scholar] [CrossRef] [PubMed]
- Sakurai, Y.; Kolokoltsov, A.A.; Chen, C.C.; Tidwell, M.W.; Bauta, W.E.; Klugbauer, N.; Grimm, C.; Wahl-Schott, C.; Biel, M.; Davey, R.A. Ebola virus. Two-pore channels control Ebola virus host cell entry and are drug targets for disease treatment. Science 2015, 347, 995–998. [Google Scholar] [CrossRef] [PubMed]
- Cote, M.; Misasi, J.; Ren, T.; Bruchez, A.; Lee, K.; Filone, C.M.; Hensley, L.; Li, Q.; Ory, D.; Chandran, K.; et al. Small molecule inhibitors reveal niemann-pick c1 is essential for Ebola virus infection. Nature 2011, 477, 344–348. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.H.; Guo, J.T.; Du, Y.M.; Block, T. Imino sugar glucosidase inhibitors as broadly active anti-filovirus agents. Emerg. Microbes Infect. 2013, 2, 77. [Google Scholar] [CrossRef] [PubMed]
- Fabozzi, G.; Nabel, C.S.; Dolan, M.A.; Sullivan, N.J. Ebolavirus proteins suppress the effects of small interfering RNA by direct interaction with the mammalian RNA interference pathway. J. Virol. 2011, 85, 2512–2523. [Google Scholar] [CrossRef] [PubMed]
- Warren, T.K.; Warfield, K.L.; Wells, J.; Swenson, D.L.; Donner, K.S.; Van Tongeren, S.A.; Garza, N.L.; Dong, L.; Mourich, D.V.; Crumley, S.; et al. Advanced antisense therapies for postexposure protection against lethal filovirus infections. Nat. Med. 2010, 16, 991–994. [Google Scholar] [CrossRef] [PubMed]
- Manicassamy, B.; Wang, J.; Jiang, H.; Rong, L. Comprehensive analysis of Ebola virus gp1 in viral entry. J. Virol. 2005, 79, 4793–4805. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, A. Analysis of filovirus entry into vero E6 cells, using inhibitors of endocytosis, endosomal acidification, structural integrity, and cathepsin (B and L) activity. J. Infect. Dis. 2007, 196, S251–S258. [Google Scholar] [CrossRef] [PubMed]
- Davidson, E.; Bryan, C.; Fong, R.H.; Barnes, T.; Pfaff, J.M.; Mabila, M.; Rucker, J.B.; Doranz, B.J. Mechanism of binding to Ebola virus glycoprotein by the zmapp, zmab, and mb-003 cocktail antibodies. J. Virol. 2015, 89, 10982–10992. [Google Scholar] [CrossRef] [PubMed]
- Olinger, G.G.; Pettitt, J.; Kim, D.; Working, C.; Bohorov, O.; Bratcher, B.; Hiatt, E.; Hume, S.D.; Johnson, A.K.; Morton, J.; et al. Delayed treatment of Ebola virus infection with plant-derived monoclonal antibodies provides protection in rhesus macaques. Proc. Natl. Acad. Sci. USA 2012, 109, 18030–18035. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Basu, A.; Mills, D.M.; Mitchell, D.; Ndungo, E.; Williams, J.D.; Herbert, A.S.; Dye, J.M.; Moir, D.T.; Chandran, K.; Patterson, J.L.; et al. Novel small molecule entry inhibitors of Ebola virus. J. Infect. Dis. 2015, 212, S425–S434. [Google Scholar] [CrossRef] [PubMed]
- Basu, A.; Li, B.; Mills, D.M.; Panchal, R.G.; Cardinale, S.C.; Butler, M.M.; Peet, N.P.; Majgier-Baranowska, H.; Williams, J.D.; Patel, I.; et al. Identification of a small-molecule entry inhibitor for filoviruses. J. Virol. 2011, 85, 3106–3119. [Google Scholar] [CrossRef] [PubMed]
- Yermolina, M.V.; Wang, J.; Caffrey, M.; Rong, L.L.; Wardrop, D.J. Discovery, synthesis, and biological evaluation of a novel group of selective inhibitors of filoviral entry. J. Med. Chem. 2011, 54, 765–781. [Google Scholar] [CrossRef] [PubMed]
- He, C.X.; Preiss, L.; Wang, B.; Fu, L.; Wen, H.; Zhang, X.; Cui, H.Q.; Meier, T.; Yin, D.L. Structural simplification of bedaquiline: The discovery of 3-(4-(N,N-dimethylaminomethyl)phenyl)quinoline-derived antitubercular lead compounds. ChemMedChem 2017, 12, 106–119. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Rumschlag-Booms, E.; Wang, J.Z.; Xiao, H.X.; Yu, J.; Wang, J.W.; Guo, L.; Gao, G.F.; Cao, Y.J.; Caffrey, M.; et al. Analysis of hemagglutinin-mediated entry tropism of H5N1 avian influenza. Virol. J. 2009, 6, 39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Radoshitzky, S.R.; Abraham, J.; Spiropoulou, C.F.; Kuhn, J.H.; Nguyen, D.; Li, W.H.; Nagel, J.; Schmidt, P.J.; Nunberg, J.H.; Andrews, N.C.; et al. Transferrin receptor 1 is a cellular receptor for new world haemorrhagic fever arenaviruses. Nature 2007, 446, 92–96. [Google Scholar] [CrossRef] [PubMed]
- He, J.L.; Choe, S.; Walker, R.; Dimarzio, P.; Morgan, D.O.; Landau, N.R. Human-immunodeficiency-virus type-1 viral-protein-r (VPR) arrests cells in the G(2) phase of the cell-cycle by inhibiting p34(CDC2) activity. J. Virol. 1995, 69, 6705–6711. [Google Scholar] [PubMed]
- Schafer, A.; Cheng, H.; Xiong, R.; Soloveva, V.; Retterer, C.; Mo, F.; Bavari, S.; Thatcher, G.; Rong, L. Repurposing potential of 1st generation h1-specific antihistamines as anti-filovirus therapeutics. Antivir. Res. 2018, 157, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Anantpadma, M.; Kouznetsova, J.; Wang, H.; Huang, R.; Kolokoltsov, A.; Guha, R.; Lindstrom, A.R.; Shtanko, O.; Simeonov, A.; Maloney, D.J.; et al. Large-scale screening and identification of novel Ebola virus and marburg virus entry inhibitors. Antimicrob. Agents Chemother. 2016, 60, 4471–4481. [Google Scholar] [CrossRef] [PubMed]
- Ebihara, H.; Theriault, S.; Neumann, G.; Alimonti, J.B.; Geisbert, J.B.; Hensley, L.E.; Groseth, A.; Jones, S.M.; Geisbert, T.W.; Kawaoka, Y.; et al. In vitro and in vivo characterization of recombinant Ebola viruses expressing enhanced green fluorescent protein. J. Infect. Dis. 2007, 196, S313–S322. [Google Scholar] [CrossRef] [PubMed]
- Barrientos, L.G.; Rollin, P.E. Release of cellular proteases into the acidic extracellular milieu exacerbates Ebola virus-induced cell damage. Virology 2007, 358, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Shah, P.P.; Wang, T.H.; Kaletsky, R.L.; Myers, M.C.; Purvis, J.E.; Jing, H.Y.; Huryn, D.M.; Greenbaum, D.C.; Smith, A.B.; Bates, P.; et al. A small-molecule oxocarbazate inhibitor of human cathepsin l blocks severe acute respiratory syndrome and Ebola pseudotype virus infection into human embryonic kidney 293T cells. Mol. Pharmacol. 2010, 78, 319–324. [Google Scholar] [CrossRef] [PubMed]
- Shang, X.F.; Morris-Natschke, S.L.; Liu, Y.Q.; Guo, X.; Xu, X.S.; Goto, M.; Li, J.C.; Yang, G.Z.; Lee, K.H. Biologically active quinoline and quinazoline alkaloids part I. Med. Res. Rev. 2018, 38, 775–828. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Cheng, H.; Ratia, K.; Varhegyi, E.; Hendrickson, W.G.; Li, J.; Rong, L.J. A comparative high-throughput screening protocol to identify entry inhibitors of enveloped viruses. J. Biomol. Screen. 2014, 19, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Ren, J.; Zhao, Y.; Fry, E.E.; Stuart, D.I. Target identification and mode of action of four chemically divergent drugs against Ebola virus infection. J. Med. Chem. 2018, 61, 724–733. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.; Lear-Rooney, C.M.; Johansen, L.; Varhegyi, E.; Chen, Z.W.; Olinger, G.G.; Rong, L.J. Inhibition of Ebola and Marburg virus entry by G protein-coupled receptor antagonists. J. Virol. 2015, 89, 9932–9938. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Ren, J.; Harlos, K.; Jones, D.M.; Zeltina, A.; Bowden, T.A.; Padilla-Parra, S.; Fry, E.E.; Stuart, D.I. Toremifene interacts with and destabilizes the Ebola virus glycoprotein. Nature 2016, 535, 169–172. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Ren, J.; Fry, E.E.; Xiao, J.; Townsend, A.R.; Stuart, D.I. Structures of ebola virus glycoprotein complexes with tricyclic antidepressant and antipsychotic drugs. J. Med. Chem. 2018, 61, 4938–4945. [Google Scholar] [CrossRef] [PubMed]
- Kaletsky, R.L.; Simmons, G.; Bates, P. Proteolysis of the Ebola virus glycoproteins enhances virus binding and infectivity. J. Virol. 2007, 81, 13378–13384. [Google Scholar] [CrossRef] [PubMed]
- Gnirss, K.; Kuhl, A.; Karsten, C.; Glowacka, I.; Bertram, S.; Kaup, F.; Hofmann, H.; Pohlmann, S. Cathepsins B and L activate Ebola but not Marburg virus glycoproteins for efficient entry into cell lines and macrophages independent of tmprss2 expression. Virology 2012, 424, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Van der Linden, W.A.; Schulze, C.J.; Herbert, A.S.; Krause, T.B.; Wirchnianski, A.A.; Dye, J.M.; Chandran, K.; Bogyo, M. Cysteine cathepsin inhibitors as anti-Ebola agents. ACS Infect. Dis. 2016, 2, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Nyakatura, E.K.; Frei, J.C.; Lai, J.R. Chemical and structural aspects of Ebola virus entry inhibitors. ACS Infect. Dis. 2015, 1, 42–52. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Vedantham, P.; Lu, K.; Agudelo, J.; Carrion, R.; Nunneley, J.W.; Barnard, D.; Pohlmann, S.; McKerrow, J.H.; Renslo, A.R.; et al. Protease inhibitors targeting coronavirus and filovirus entry. Antivir. Res. 2015, 116, 76–84. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, F.R.; Liang, Q.R.; Abi-Mosleh, L.; Das, A.; De Brabander, J.K.; Goldstein, J.L.; Brown, M.S. Identification of npc1 as the target of u18666a, an inhibitor of lysosomal cholesterol export and ebola infection. eLife 2015, 4, e12177. [Google Scholar] [CrossRef] [PubMed]
- Johansen, L.M.; Brannan, J.M.; Delos, S.E.; Shoemaker, C.J.; Stossel, A.; Lear, C.; Hoffstrom, B.G.; DeWald, L.E.; Schornberg, K.L.; Scully, C.; et al. FDA-approved selective estrogen receptor modulators inhibit Ebola virus infection. Sci. Transl. Med. 2013, 5, 190ra79. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.; Schafer, A.; Soloveva, V.; Gharaibeh, D.; Kenny, T.; Retterer, C.; Zamani, R.; Bavari, S.; Peet, N.P.; Rong, L.J. Identification of a coumarin-based antihistamine-like small molecule as an anti-filoviral entry inhibitor. Antivir. Res. 2017, 145, 24–32. [Google Scholar] [CrossRef] [PubMed]
| ID | Structure | Inhibition | Cell Viability | ||
|---|---|---|---|---|---|
| HIV-1/H5N1 | HIV-1/LASV | HIV-1/EBOV | |||
| SYL1640 |  | −1.97% | −15.08% | 79.06% | 97.64% |
| SYL1642 |  | 14.11% | 34.99% | 92.15% | 72.30% |
| SYL1654 |  | 8.04% | 18.79% | 84.84% | 94.74% |
| SYL1655 |  | −17.17% | 28.87% | 90.29% | 95.31% |
| SYL1660 |  | 3.34% | −26.55% | 97.48% | 106.02% |
| SYL1657 |  | 0.16% | 25.76% | 89.23% | 93.92% |
| SYL1658 |  | 36.88% | 19.23% | 77.41% | 72.67% |
| SYL1683 |  | 21.73% | 26.27% | 95.73% | 104.43% |
| SYL1711 |  | 38.33% | 25.47% | 98.65% | 90.69% |
| SYL1712 |  | 5.75% | 55.35% | 99.39% | 96.93% |
| ID | IC50 (μM) | CC50 (μM) | SI |
|---|---|---|---|
| SYL1640 | 2.96 | 190.4 | 64.3 |
| SYL1642 | 5.21 | 152.2 | 29.2 |
| SYL1654 | 4.98 | 222.5 | 44.7 |
| SYL1655 | 2.65 | 132.3 | 49.9 |
| SYL1657 | 3.56 | 214.6 | 60.3 |
| SYL1658 | 8.65 | 109.5 | 12.7 |
| SYL1660 | 2.58 | 184.7 | 71.6 |
| SYL1683 | 2.93 | 235.4 | 80.3 |
| SYL1711 | 4.11 | 241.9 | 58.9 |
| SYL1712 | 0.95 | 214.6 | 225.9 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cui, Q.; Cheng, H.; Xiong, R.; Zhang, G.; Du, R.; Anantpadma, M.; Davey, R.A.; Rong, L. Identification of Diaryl-Quinoline Compounds as Entry Inhibitors of Ebola Virus. Viruses 2018, 10, 678. https://doi.org/10.3390/v10120678
Cui Q, Cheng H, Xiong R, Zhang G, Du R, Anantpadma M, Davey RA, Rong L. Identification of Diaryl-Quinoline Compounds as Entry Inhibitors of Ebola Virus. Viruses. 2018; 10(12):678. https://doi.org/10.3390/v10120678
Chicago/Turabian StyleCui, Qinghua, Han Cheng, Rui Xiong, Gang Zhang, Ruikun Du, Manu Anantpadma, Robert A. Davey, and Lijun Rong. 2018. "Identification of Diaryl-Quinoline Compounds as Entry Inhibitors of Ebola Virus" Viruses 10, no. 12: 678. https://doi.org/10.3390/v10120678






