1. Introduction
The rubber tree,
Hevea brasiliensis, is the only cultivated species for latex [
1,
2], and has often been described as an out-breeding species that is pollinated by insects such as thrips and midges [
3,
4]. Natural rubber is one of the most important raw materials in industry, agriculture, defense, transportation, and daily life [
5,
6]. Demand for rubber is increasing with economic development; however, the regions where rubber trees are planted are limited due to the stringent environmental requirements for their growth [
7]. In addition, alternatives to natural rubber are still limited because synthetic rubber produced from petroleum does not match its resilience, elasticity, and abrasion resistance [
8,
9].
Substantial efforts have been expended to solve the problem of the imbalance between rubber supply and demand. Efforts by breeders led to many rubber tree cultivar clones being selected and planted in non-traditional planting areas, such as Chinese rubber plantations. These were established in Hainan and Yunnan Provinces, in areas as far north as 22° N, while rubber plantations are typically located in latitudes that range from 10° N to 10° S [
6,
10]. Two new rubber tree cultivars, Yunyan 77-2 and Yunyan 77-4, were selected and confirmed as triploids which were largely planted in Yunnan, China [
11]. Some reports showed that these triploids had a relatively high rubber yield and good cold resistance [
12,
13]. These two triploid cultivars both were derived from GT1 × PR 107 [
13,
14]. In many studies, this parental cross was considered a special case regarding triploid formation, particularly because GT1 is male-sterile and PR 107 has very low fruit production, often considered to be female-sterile [
11,
13]. To date, no efforts have been made to explore these special rubber tree materials, and the mechanism of triploid formation in rubber trees has been forever obscure.
Generally, triploid plants can be created by crossing tetraploids with diploids or by combining unreduced (2
n) gametes with reduced (
n) gametes [
15,
16,
17,
18]. No rubber tree tetraploids have been reported in the wild; therefore, triploid rubber trees most likely arise from the combination of 2
n gametes with reduced gametes. Unreduced gametes are crucial for triploid formation and can be formed via several mechanisms, namely, premeiotic doubling, first-division restitution (FDR), chromosome doubling during the meiotic interphase, postmeiotic restitution (PMR), and indeterminate meiotic restitution (IMR) [
19,
20,
21]. Many studies have reported that FDR and SDR are the predominant mechanisms of 2
n gamete formation based on their genetic consequences [
22]. It has been theorized that 2
n gametes from FDR transmit roughly 80% of parental heterozygosity to the progeny, while 2
n gametes from SDR transmit about 40% [
23,
24,
25].
In this study, the rubber tree clone GT1 was chosen as the mother tree. Its open pollinated half sib progeny were collected to screen the triploids. Then, the origin of 2n gametes and the male parents of these triploids were determined using SSR markers. The mechanism of 2n gamete formation was also studied. This study will be helpful for the study of rubber trees especially in polyploid breeding.
2. Materials and Methods
2.1. Plant Materials
The mother tree, GT1 (Original clone, 2
n = 2
x = 36), was located in the flower park of the XiShuangBanNa, Yunnan Institute of Tropical Crops, China. Seventeen rubber trees were selected as candidate male parents which were nearby GT1 (
Table 1). These materials derived from Wickham germplasm (PB 310, Yunyan 277-5, RRIC 103), and IRRDB’ 81 (International Rubber Research and Development Board) germplasm (A
1-A
12). All rubber trees were identified by clonal inspectors.
2.2. Collection of Seeds and Sowing
The fruits of GT1 were bagged before seed dispersal from the fruit shell, and then seeds were collected and sown in sandy soil that had been disinfected using carbendazim.
2.3. Ploidy Analysis by Flow Cytometry
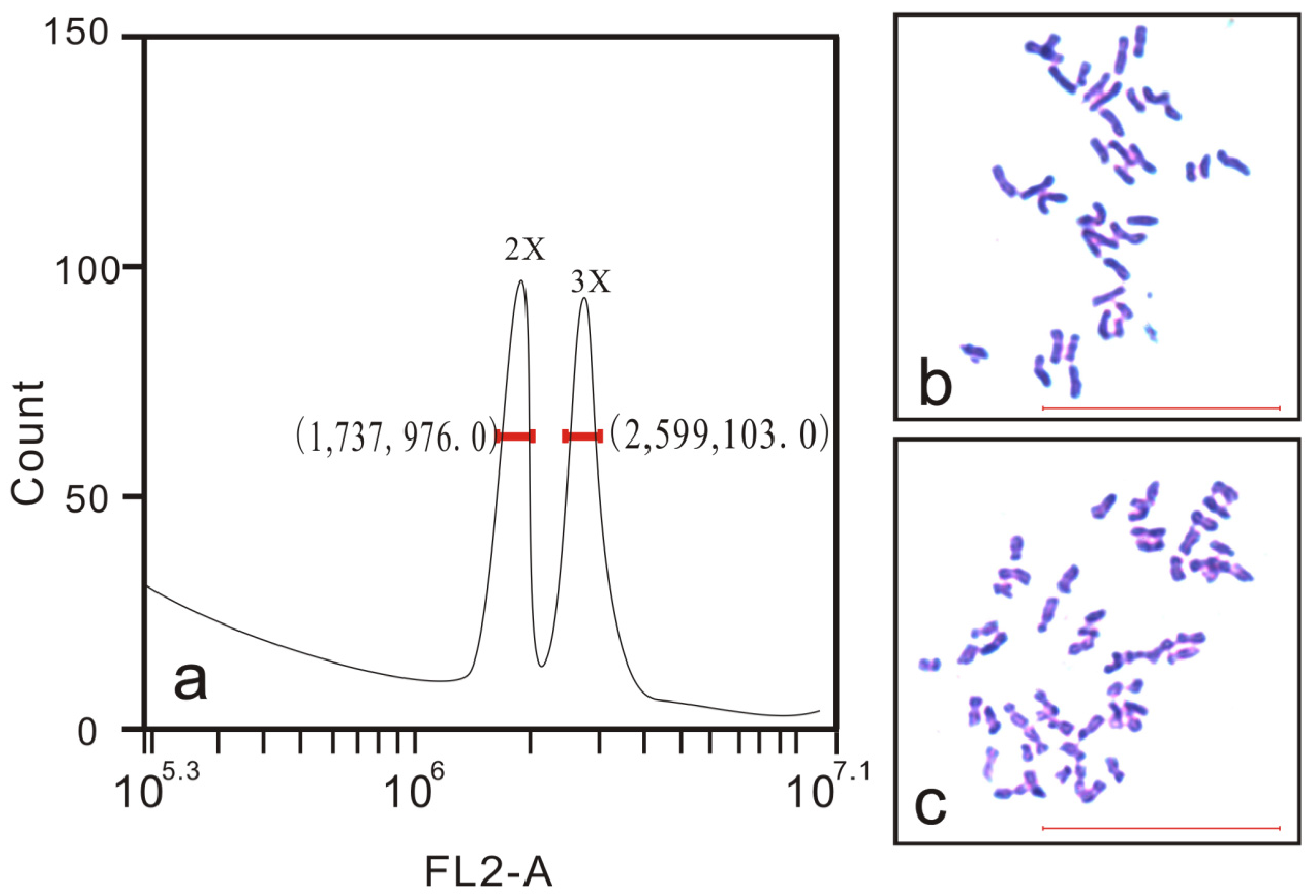
Flow cytometry was performed using an Accuri™ C6 Flow Cytometer (BD Biosciences, Franklin Lakes, NJ, USA). About 20 mg of fresh young leaves were cut into pieces with a sharp blade in 0.5 mL of nuclear extraction solution (45 mM MgCl2, 30 mM sodium citrate, 20 mM 4-morpholinepropane sulfonate, 0.1% (v/v) Triton X-100, pH 7.0), and then filtered through 40-µm nylon mesh after being left to stand for 3 min. The suspension of released nuclei was stained with 50 µL of propidium iodide (PI) for 5 min. The leaf sample from a known diploid rubber tree was used as a control, which was balanced by mixing with samples. A seedling was recorded as triploid when there were two peaks and the ratio of their peak values was 3:2. Samples were assessed independently three times for each putative triploid.
2.4. Chromosome Counting
The ploidy level of plantlets can be further confirmed by chromosome counting. Very young leaves which were reddish in color were collected from the seedlings and pretreated in a saturated solution of para-dichlorobenzene for at least 3 h, washed once using distilled water, and then fixed in Carnoy’s fluid (ethanol/acetic acid, 3:1) for at least 24 h at 4 °C. Next, the materials were transferred to 1 mol/L HCl for 10–15 min at 60 °C, washed with water, and then immersed in distilled water for 10 min. These hydrolyzed materials were stained with carbolfuchsin solution. Chromosome counts of at least 10 cells with a well-spread metaphase per seedling were observed using a microscope (Olympus CX41; Olympus, Tokyo, Japan).
2.5. DNA Extraction and SSR Analysis
DNA was extracted from approximately 300 mg of each leaf sample using a DNeasy
® Plant Mini Kit (Tiangen Biotech Co. Ltd., Beijing, China) following the manufacturer’s instructions. The SSR primers used in this study were derived from many previous studies [
26,
27,
28]. The fluorescently labeled TP-M13-SSR method [
29] was adopted in this work. Three primers were included for the polymerase chain reaction (PCR), including a forward primer with the tail of a universal primer, M13 (5′-TGTAAAACGACGGCCAGT-3′), at the 5′ end, a reverse primer, and a universal primer, M13, fluorescently labeled with 6-carboxyfluorescein (FAM), 6-carboxy-x-rhodamine (ROX), hexachloro-6-carboxyfluorescein (HEX), or tetrachloro-6-carboxyrhodamine (TAMARA). PCR was carried out in a total volume of 20 µL containing 2 µL of DNA template, 10 µL of PCR Master Mixes (Bo Maide Biotech Company, Beijing, China), 0.08 µL of forward primer, 0.32 µL of reverse primer, and 0.4 µL of fluorescence primer. Amplification was performed using the following conditions: 94 °C for 5 min; followed by 11 cycles of 94 °C for 30 s, 30 s at the optimal annealing temperature for each SSR marker, and 72 °C for 30 s; 20 cycles of 94 °C for 30 s, 55 °C for 30 s, and 72 °C for 30 s; 8 cycles of 94 °C for 30 s, 53 °C for 30 s, and 72 °C for 30 s; and a final extension step at 72 °C for 10 min. Finally, capillary electrophoresis fluorescence-based SSR analyses were conducted on an ABI 3730xl DNA Analyzer (Genewiz Inc., Beijing, China). Fragment sizes and peak areas were determined automatically using GeneMarker software v2.2 (Soft Genetics, LLC. College Station, PA, USA).
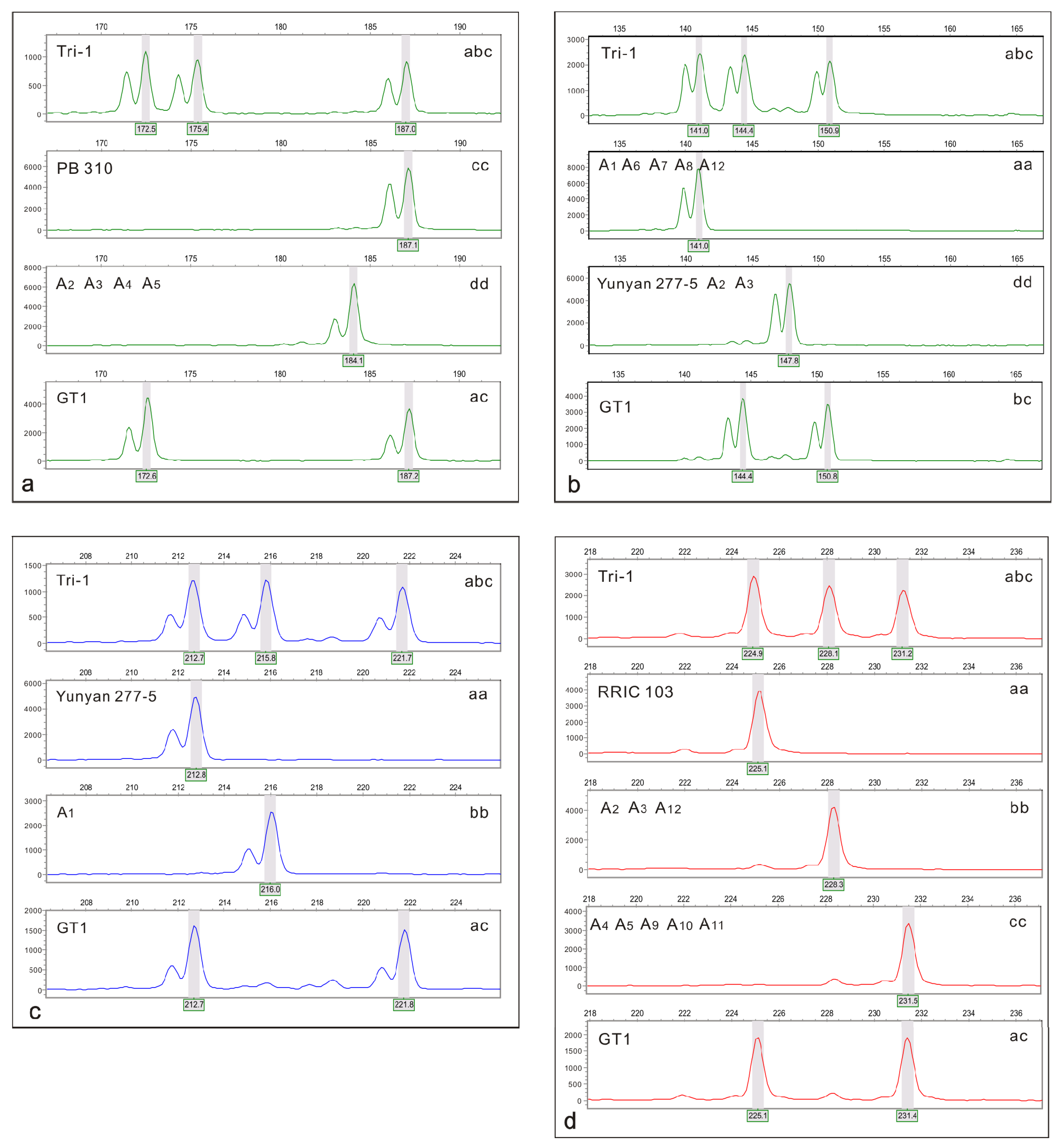
Selected SSR markers should be heterozygous for triploids and be polymorphic between maternal parent GT1 and candidate male parents. When one allele was shared between the two parents, the MAC-PR [
30,
31] was used to determine the allelic configuration. The origin of 2
n gametes is determined by comparing SSR marker results among GT1, triploids, and candidate male parents. Theoretically, a triploid and its parent which provided 2
n gamete should share two alleles for a locus when the null alleles and dropout alleles are neglected. So 2
n gametes origin can be determined if one parent always can share possible two alleles with the triploid. Based on the knowledge of 2
n gametes origin, the triploid’s male parent is determined by comparing SSR marker results among GT1, triploids, and candidate male parents. A triploid and its male parent share at least one allele for a locus when the null alleles and dropout alleles are neglected. So the male parent can be determined only, if all other candidate males are excluded by mismatches.
Based on the results of 2
n gametes origin and determined parents, the mechanism of 2
n gamete formation was identified by comparing allelic configurations among the triploid, its female parent, GT1, and its male parent. The genotypes of 2
n gametes were directly read when parental alleles displayed as completely different based on the allele number and type. The rate of maternal HR (HR%) was estimated as described by Xie et al. [
32], calculated as:
where n
ab is the number of heterozygous 2
n gametes, and n
aa and n
bb are two kinds of homozygous 2
n gametes.
4. Discussion
In this study using co-dominant SSR markers, the clone GT1 was unambiguously determined to be a 2
n gamete donor for triploid rubber tree formation. We demonstrated the advantages of the selected SSR markers and the capabilities of determining the origin and ploidy levels of gametes. Ideal markers should be different and heterozygous in both parents with three different alleles in triploids, such as the RUB 16, RUB 19, RUB 95, RUB 98, RUB 103, RUB 156, RUB 183, RUB 184, and RUB 199 markers for Tri-1 used in this study. We can directly determine the allelic configuration via differences in alleles at a locus, and if the same alleles are present in both parents and offspring, MAC-PR should be used to determine the donor genome in triploids, such as demonstrated with RUB 178 for Tri-1. More crosses among
Hevea brasiliensis clones are required to confirm whether other clones are related to 2
n gamete formation. In other plants the 2
n gametes probably originated from certain species or clones. Raboin et al. [
33] reported that unreduced gametes were produced by partially sterile diploid cultivars and reduced gametes by fertile diploid cultivars in the banana. Chen et al. [
34] reported that 2
n eggs originated only from the maternal parent in
Citrus sinensis ×
Poncirus trifoliata. The presence of 2
n gametes in many other clones would have significant potential for rubber tree breeding to select desired triploids. If only 2
n gametes are produced from the clone GT1, further efforts should focus on male parent selection. Crosses between high-quality rubber tree cultivars to produce triploids can likely yield high-quality triploids.
SSR markers have been applied in many studies for parent identification. For example, this method was used to identify 220 open-pollination progeny of
Liriodendron spp., of which 49 male parents were identified for 138 progeny [
35]. SSR was also used to identify the male parent of 41 elite clones derived from sugarcane polycross families, showing the importance of using molecular marker technology in the identification and confirmation of male parents of high-performance clones in sugarcane breeding programs [
36]. In our study, parents of all triploid clones were successfully identified. A large set of SSR markers should be adopted when using the exclusion method, as in this study. Additionally, if specific alleles can be amplified in one or two clones, such as the band ‘b’ was only amplified in A
1 and A
12 in all candidate male parents by the marker RUB 178. These markers can improve the efficiency of male parent identification. The results highlight the usefulness of SSR markers in the identification of male parents of triploid rubber trees, and will provide guidance for parent identification and early selection of rubber trees, providing references for improving the efficiency of rubber tree breeding.
Without previous knowledge of the positions of markers relative to the centromeres, Park et al. [
37] suggested that the rate of HR varies between 0% and 100% for SDR 2
n gametes, and between 50% and 100% for FDR 2
n gametes, under the hypothesis that only one crossover occurred between the locus and centromere. Xie et al. [
32] reported that SDR was the mechanism of 2
n megagametophyte formation in ‘Nadorcott’ tangor, because the rate of maternal HR varied from 0.00% to 87.80% for 22 SSR markers, with 13 exhibiting a heterozygosity rate <50%. In the present study, the rate of maternal HR of all 25 markers varied from 0% to 100%, with a heterozygosity rate <50% for 10 markers, suggesting that SDR may be the mechanism underlying 2
n megagametophyte formation. Cuenca et al. [
38] reported that maternal heterozygosity transmitted to each SDR 2
n megagametophyte varied from 15.38% to 100%, with a mean value of 54.98%. In this study, based on all loci analyzed, the rate of maternal HR transmitted to each triploid hybrid ranged from 27.78% to 75.00%, with a mean of 51.46%. Therefore, compared with FDR, SDR is more likely the mechanism of 2
n megagametophyte formation in the rubber tree clone GT1. Although one mechanism may be predominantly observed in certain clones, it was by no means ruled out that other mechanisms did not operate in these clones at the same time because substantial influences from the environment likely disturb the processes of meiosis and meiotic nuclear restitution. For example, high temperature can induce 2
n female gametes in
Populus [
39,
40]. However, gamete formation is controlled mainly by genes [
32,
41]. Some genes, such as
AtPS1 [
42] and
JASON [
43], were reported to be involved in 2
n gamete formation in
Arabidopsis. However, the molecular mechanism in rubber trees remains unclear.
For practical application, the level of heterozygosity in the triploid progeny is of empirical importance regardless of the mechanism of gamete production. Therefore, use of SSR analysis is important as a determinant of maternal HR and as an indicator in early breeding projects involving rubber trees. In future studies, we will examine the selection of male parents based on allelic differentiation from GT1 to determine whether this will result in triploid progeny that are also highly heterozygous at loci controlling production traits.