Process-Based Modeling to Assess the Effects of Recent Climatic Variation on Site Productivity and Forest Function across Western North America
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area
2.2. Forest Inventory and Analysis (FIA) Data
2.3. Process-Based Growth Model
2.4. Climatic Data
2.5. Model Simulations
3. Results
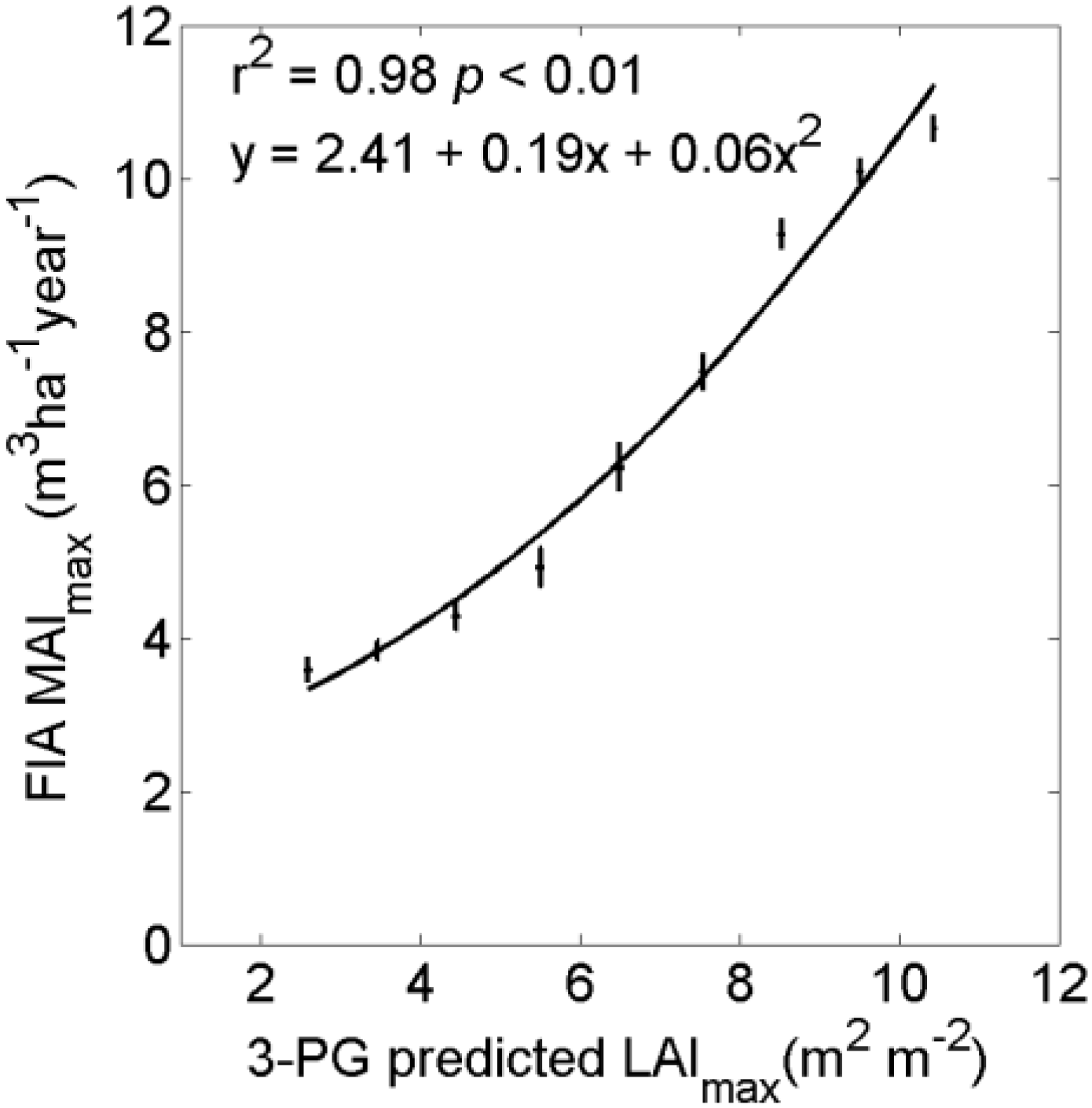
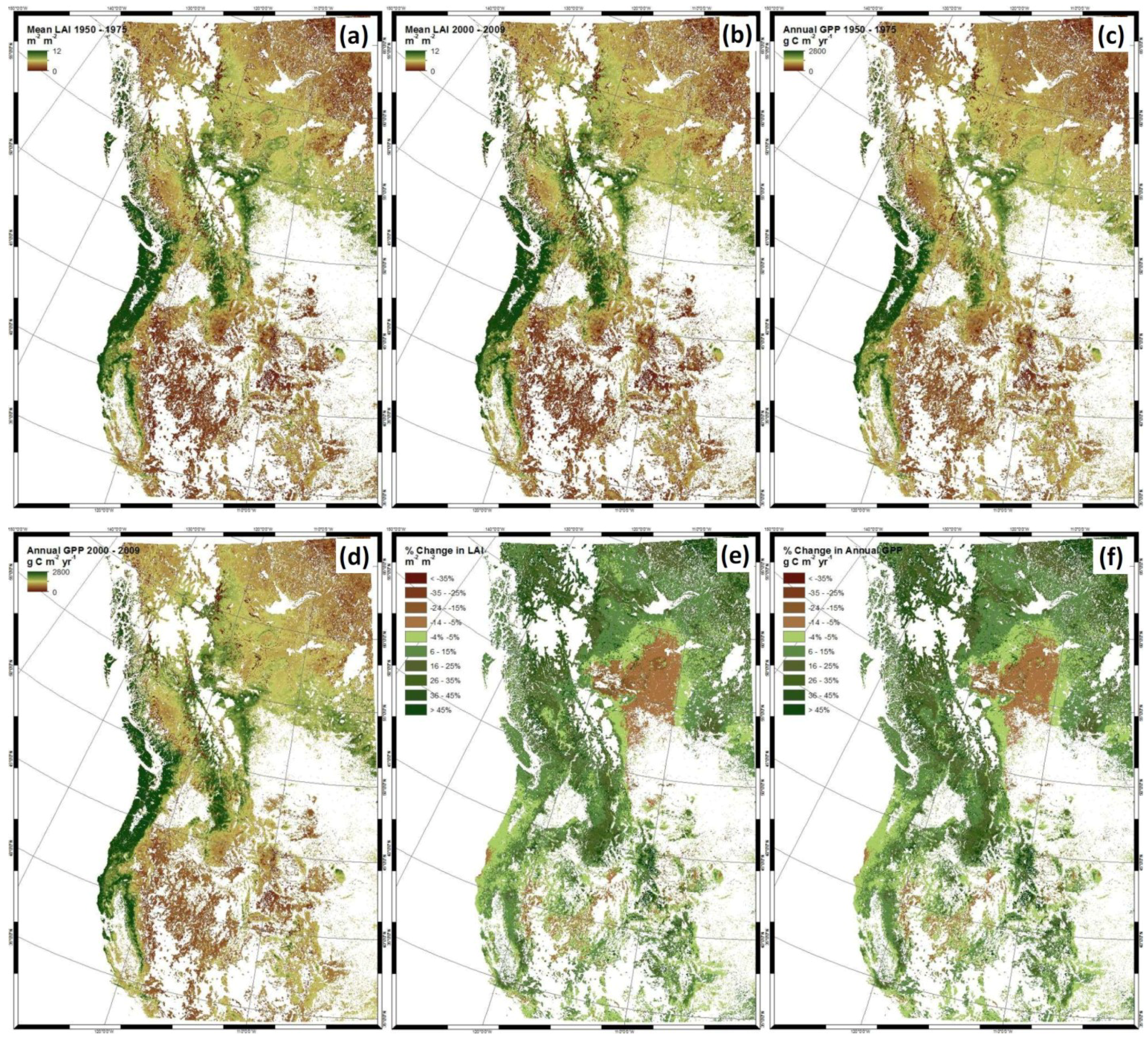

4. Discussion
4.1. General Points
4.2. Future Improvements in the Approach
4.3. Implications on Forest Health
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Woods, A.; Coates, K.D.; Hamann, A. Is an unprecedented Dothistroma needle blight epidemic related to climate change? BioScience 2005, 55, 761–769. [Google Scholar] [CrossRef]
- Westerling, A.L.; Hidalgo, H.G.; Cayan, D.R.; Swetnam, T.W. Warming and earlier spring increase western U.S. forest wildfire activity. Science 2006, 313, 940–943. [Google Scholar] [CrossRef]
- Raffa, K.; Aukema, B.; Bentz, B.; Carroll, A. Cross-scale drivers of natural disturbances prone to anthropogenic amplification: The dynamics of bark beetle eruptions. Bioscience 2008, 58, 501–517. [Google Scholar] [CrossRef]
- Wulder, M.A.; White, J.C.; Fournier, R.A.; Luther, J.E.; Magnussen, S. Spatially explicit large area biomass estimation: Three approaches using forest inventory and remotely sensedimagery in a GIS. Sensors 2008, 8, 529–560. [Google Scholar] [CrossRef]
- Mildrexler, D.J.; Zhao, M.; Running, S.W. Testing a MODIS Global Disturbance Index across North America. Remote Sens. Environ. 2009, 113, 2103–2117. [Google Scholar] [CrossRef]
- Parker, T.J.; Clancy, K.M.; Mathiasen, R.L. Interactions among fire, insects and pathogens in coniferous forests of the interior western United States and Canada. Agric. For. Entomol. 2006, 8, 167–189. [Google Scholar] [CrossRef]
- Jessie, E.; Fettig, C.J.; Billings, R.F.; Munson, A.S.; Nebeker, T.E.; Klepzig, K.D.; Nowak, J.T. The effectiveness of vegetation management practices for prevention and control of bark beetle infestations in coniferous forests of the western and southern United States. For. Ecol. Manag. 2007, 238, 24–53. [Google Scholar] [CrossRef]
- Cattelino, P.J.; Noble, I.R.; Slatyer, R.O.; Kessell, S.R. Predicting the multiple pathways of plant succession. Environ. Manag. 1979, 3, 41–50. [Google Scholar] [CrossRef]
- Anderson-Teixeira, K.J.; Miller, A.D.; Mohan, J.E.; Hudiburg, T.W.; Duval, B.D.; Delucia, E.H. Altered dynamics of forest recovery under a changing climate. Glob. Chang. Biol. 2013, 19, 2001–2021. [Google Scholar] [CrossRef]
- Waring, R.H.; Coops, N.C.; Running, S.W. Predicting satellite-derived patterns of large-scale disturbances in forests of the Pacific Northwest Region in response to recent climatic variation. Remote Sens. Environ. 2011, 115, 3554–3566. [Google Scholar] [CrossRef]
- Running, S. Estimating terrestrial primary productivity by combining remote sensing and ecosystem simulation. In Remote Sensing of Biosphere Functioning; Springer New York: New York, NY, USA, 1990; pp. 65–86. [Google Scholar]
- Coops, N.C.; Waring, R.H. The use of multiscale remote sensing imagery to derive regional estimates of forest growth capacity using 3-PGS. Remote Sens. Environ. 2001, 75, 324–334. [Google Scholar] [CrossRef]
- Runyon, J.; Waring, R.H.; Goward, S.N.; Welles, J.M. Environmental limits on net primary production and light-use efficiency across the Oregon transect. Ecol. Appl. 1994, 4, 226–237. [Google Scholar] [CrossRef]
- Law, B.E.; Turner, D.; Campbell, J.; Lefsky, M. Carbon fluxes across regions. In Scaling and Uncertainy Analysis in Ecology: Methods and Applications; Wu, J., Jones, K.B., Loucks, O.L., Eds.; Springer: Dordrecht, The Netherlands, 2006; pp. 167–190. [Google Scholar]
- Latta, G.; Temesgen, H.; Barrett, T.M. Mapping and imputing potential productivity of Pacific Northwest forests using climate variables. Can. J. For. Res. 2009, 39, 1197–1207. [Google Scholar] [CrossRef]
- Landsberg, J. Modelling forest ecosystems: State of the art, challenges, and future direction. Can. J. For. Res. 2003, 33, 385–397. [Google Scholar] [CrossRef]
- Mäkelä, A.; Landsberg, J.; Ek, A. Process-based models for forest ecosystem management: Current state of the art and challenges for practical implementation. Tree Physiol. 2000, 20, 289–298. [Google Scholar] [CrossRef]
- Coops, N.C.; Waring, R.H.; Brown, S.R.; Running, S.W. Comparison of predictions of Net Primary Production and seasonal patterns in water use derived with two forest growth models in southwestern Oregon. Ecol. Model. 2001, 142, 61–81. [Google Scholar] [CrossRef]
- Peterman, W.; Waring, R.H.; Seager, T.; Pollock, W.L. Soil properties affect pinyon pine–juniper response to drought. Ecohydrology 2013, 6, 455–463. [Google Scholar] [CrossRef]
- Piao, S.; Sitch, S.; Ciais, P.; Friedlingstein, P.; Peylin, P.; Wang, X.; Zeng, N. Evaluation of terrestrial carbon cycle models for their response to climate variability and to CO2 trends. Glob. Chang. Biol. 2013, 19, 2117–2132. [Google Scholar] [CrossRef]
- Oren, R.; Ellsworth, D.S.; Johnsen, K.H.; Phillips, N.; Ewers, B.E.; Maier, C.; Katul, G.G. Soil fertility limits carbon sequestration by forest ecosystems in a CO2-enriched atmosphere. Nature 2001, 411, 469–472. [Google Scholar] [CrossRef]
- Gómez-Guerrero, A.; Silva, L.C.R.; Barrera-Reyes, M.; Kishchuk, B.; Velázquez-Martínez, A.; Martínez-Trinidad, T.; Horwath, W.R. Growth decline and divergent tree ring isotopic composition δ13C and δ18O contradict predictions of CO2 stimulation in high altitudinal forests. Glob. Chang. Biol. 2013, 19, 1748–1758. [Google Scholar] [CrossRef]
- Nightingale, J.M.; Coops, N.C.; Waring, R.H.; Hargrove, W.W. Comparison of MODIS gross primary production estimates for forests across the U.S.A. with those generated by a simple process model, 3-PGS. Remote Sens. Environ. 2007, 109, 500–509. [Google Scholar]
- Coops, N.C.; Waring, R.H.; Hilker, T. Prediction of soil properties using a process-based forest growth model to match satellite-derived estimates of leaf area index. Remote Sens. Environ. 2012, 126, 160–173. [Google Scholar] [CrossRef]
- Mathys, A.; Coops, N.C.; Waring, R.H. Soil water availability effects on the distribution of 20 tree species in western North America. For. Ecol. Manag. 2014, 313, 144–152. [Google Scholar] [CrossRef]
- Waring, R.H.; Franklin, J.F. Evergreen coniferous forests of the Pacific Northwest. Science 1979, 204, 1380–1386. [Google Scholar]
- Landsberg, J.J.; Waring, R.H. A generalised model of forest productivity using simplified concepts of radiation-use efficiency, carbon balance and partitioning. For. Ecol. Manag. 1997, 95, 209–228. [Google Scholar] [CrossRef]
- Digital Representation of Tree Species Range Maps. Available online: http://esp.cr.usgs .gov/data/little/ (accessed on 18 March 2014).
- Friedl, M.; McIver, D.; Hodges, J.C.; Zhang, X.; Muchoney, D.; Strahler, A.; Schaaf, C. Global land cover mapping from MODIS: Algorithms and early results. Remote Sens. Environ. 2002, 83, 287–302. [Google Scholar] [CrossRef]
- U.S. Environmental Protection Agency. Available online: http://www.epa.gov/wed/pages/ecoregions/level_iii_iv.htm (accessed on 18 March 2014).
- Franklin, J.; Dyrness, C. Natural vegetation of Oregon and Washington; United States Forest Service General Techical Report PNW-8; USDA Forest Service: Portland, OR, USA, 1973. [Google Scholar]
- Whittaker, R.H. Vegetation of the Siskiyou Mountains, Oregon and California. Ecol. Monogr. 1960, 30, 279–338. [Google Scholar] [CrossRef]
- McLaughlin, S. Floristic analysis of the southwestern United States. Gt. Basin Nat. 1986, 46, 46–65. [Google Scholar]
- Roesch, F.A.; Reams, G.A. Analytical alternatives for an annual inventory system. J. For. 1999, 97, 44–48. [Google Scholar]
- Czaplewski, R.L. Forest survey sampling designs: A history. J. For. 1999, 97, 4–10. [Google Scholar]
- Hanson, E.J.; Azuma, D.L.; Hiserote, B.A. Site Index Equations and Mean Annual Increment Equations for Pacific Northwest Research Station Forest Inventory and Analysis Inventories, 1985–2001. USDA Forest Service Research Note PNW-RN-533. U.S. Department of Agriculture, Forest Service, Pacific Northwest Research Station: Portland, OR, USA, 2002; 24. [Google Scholar]
- McArdle, R.E.; Meyers, W.H.; Bruce, D. The Yield of Douglas Fir in the Pacific Northwest; Technical Bulletin No. 201; US Department of Agriculture: Washington, DC, USA, 1961; p. 64.
- Meyers, W.H. Yield of Even-Aged Stands of Ponderosa Pine; Techical Bulletin No. 630; US Department of Agriculture, US Forest Service: Washington, DC, USA, 1961; p. 59.
- Barnes, G.H. Yield of Even-Aged Stands of Western Hemlock; Technical Bulletin No. 1273; US Department of Agriculture: Washington, DC, USA, 1962; p. 52.
- Cochran, P.H. Gross Yields for Even-Aged Stands of Douglas-fir and White or Grand Fir East of the Cascades in Oregon and Washington; Research Paper PNW-263; US Department of Agriculture, Forest Service, Pacific Northwest Forest and Range Experiment Station: Portland, OR, USA, 1979; p. 17. [Google Scholar]
- Hember, R.A.; Coops, N.C.; Black, T.A.; Guy, R.D. Simulating gross primary production across a chronosequence of coastal Douglas-fir forest stands with a production efficiency model. Agric. For. Meteorol. 2010, 150, 238–253. [Google Scholar] [CrossRef]
- Hadley, J. Effect of daily minimum temperature on photosynthesis in eastern hemlock (Tsuga canadensis L.) in autumn and winter. Arct. Antarct. Alp. Res. 2000, 32, 368–374. [Google Scholar] [CrossRef]
- Landsberg, J.J.; Sands, P. Physiological Ecology of Forest Production: Principles, Processes and Models; Academic Press (Elsevier): New York, NY, USA, 2010. [Google Scholar]
- Climate BC/WNA. Available online: http://www.genetics.forestry.ubc.ca/cfcg/ClimateWNA/ClimateWNA.html (accessed on 18 March 2014).
- Wang, T.; Hamann, A. Development of scale free climate data for western Canada for use in resource management. Int. J. Climatol. 2006, 26, 383–397. [Google Scholar] [CrossRef]
- Kimball, J.; Running, S.; Nemani, R. An improved method for estimating surface humidity from daily minimum temperature. Agric. For. Meteorol. 1997, 85, 87–98. [Google Scholar] [CrossRef]
- Fu, P.; Rich, P.M. A geometric solar radiation model with applications in agriculture and forestry. Comput. Electron. Agric. 2002, 37, 25–35. [Google Scholar]
- Schroeder, T.; Hember, R.; Coops, N.C.; Liang, S. Validation of solar radiation surfaces from MODIS and reanalysis data over topographically complex terrain. J. Appl. Meteorol. Climatol. 2009, 48, 2241–2458. [Google Scholar]
- Magnani, F.; Mencuccini, M.; Grace, J. Age-related decline in stand productivity: the role of structural acclimation under hydraulic constraints. Plant Cell Environ. 2000, 23, 251–263. [Google Scholar] [CrossRef]
- Data Basin. Available online: http://consbio.org/products/categories/data-basin (assessed on 18 March 2014).
- Weiss, J.L.; Castro, C.L.; Overpeck, H.T. Distinguishing pronounced droughts in the Southwestern United States: Seasonality and effects of warmer temperature. J. Clim. 2008, 22, 5918–5932. [Google Scholar]
- USGCP United States Global Change Research Program. Available online: http:www.globalchange.gov (accessed on 18 March 2014).
- Williams, A.P.; Allen, C.D.; Millar, C.I.; Swetnam, T.W.; Michaelsen, J.; Still, C.J.; Leavitt, S.W. Forest responses to increasing aridity and warmth in the southwestern United States. Proc. Natl. Acad. Sci. USA 2010, 107, 21289–21294. [Google Scholar]
- Grant, G.; Tague, C.; Allen, C. Watering the forest for the trees: An emerging priority for managing water in forest landscapes. Front. Ecol. Environ. 2013, 11, 314–321. [Google Scholar] [CrossRef]
- Mueller, R.C.; Scudder, C.M.; Porter, M.E.; Talbot Trotter, R.; Gehring, C.A.; Whitham, T.G. Differential tree mortality in response to severe drought: Evidence for long-term vegetation shifts. J. Ecol. 2005, 93, 1085–1093. [Google Scholar] [CrossRef]
- Anthoni, P.M.; Unsworth, M.H.; Law, B.E.; Irvine, J.; Baldocchi, D.D.; van Tuyl, S. Seasonal differences in carbon and water vapor exchange in young and old-growth ponderosa pine ecosystems. Agric. For. Meteorol. 2002, 111, 203–222. [Google Scholar] [CrossRef]
- Chen, J.; Ustin, S.; Suchanek, T. Net ecosystem exchanges of carbon, water, and energy in young and old-growth Douglas-fir forests. Ecosystems 2004, 7, 534–544. [Google Scholar]
- Morgenstern, K.; Black, T.A.; Humphreys, E.R.; Griffis, T.J.; Drewitt, G.B.; Cai, T.; Livingston, N.J. Sensitivity and uncertainty of the carbon balance of a Pacific Northwest Douglas-fir forest during an El Niño/La Niña cycle. Agric. For. Meteorol. 2004, 123, 201–219. [Google Scholar] [CrossRef]
- Lefsky, M.A.; Cohen, W.B.; Parker, G.G.; Harding, D.J. Lidar remote sensing for ecosystem studies. BioScience 2002, 52, 19–30. [Google Scholar] [CrossRef]
- Wei, L.; Marshall, J.; Timothy, E.L.; Kathleen, L.K.; Enhao, D.; Robert, E.P.; Peter, J.G.; Nerea, U. Constraining 3-PG with a new δ13C submodel: A test using the δ13C of tree rings. Plant Cell Environ. 2013, 37, 82–100. [Google Scholar]
- Waring, R.H.; Oregon State University, Corvallis, OR, USA. Personal Correspondence with Dr. Liang Wei. University of Idaho: Moscow, ID, USA, 2013. [Google Scholar]
- Almeida, A.C.; Soares, J.V.; Landsberg, J.J.; Rezende, G.D. Growth and water balance of Eucalyptus grandis hybrid plantations in Brazil during a rotation for pulp production. For. Ecol. Manag. 2007, 251, 10–21. [Google Scholar] [CrossRef]
- Dye, P.; Jacobs, S.; Drew, D. Verification of 3-PG growth and water-use predictions in twelve Eucalyptus plantation stands in Zululand, South Africa. For. Ecol. Manag. 2004, 193, 197–218. [Google Scholar] [CrossRef]
- Landsberg, J.J.; Waring, R.; Coops, N. Performance of the forest productivity model 3-PG applied to a wide range of forest types. For. Ecol. Manag. 2003, 172, 199–214. [Google Scholar] [CrossRef]
- Stape, J.L.; Ryan, M.G.; Binkley, D. Testing the utility of the 3-PG model for growth of with natural and manipulated supplies of water and nutrients. For. Ecol. Manag. 2004, 193, 219–234. [Google Scholar] [CrossRef]
- Van Mantgem, P.J.; Stephenson, N.L.; Byrne, J.C.; Daniels, L.D.; Franklin, J.F.; Fulé, P.Z.; Veblen, T.T. Widespread increase of tree mortality rates in the western United States. Science 2009, 323, 521–524. [Google Scholar] [CrossRef]
- Van Mantgem, P.J.; Nesmith, J.C.B.; Keifer, M.; Knapp, E.E.; Flint, A.; Flint, L. Climatic stress increases forest fire severity across the western United States. Ecol. Lett. 2013, 16, 1151–1156. [Google Scholar] [CrossRef]
- Woods, A.J.; Heppner, D.; Kope, H.H.; Burleigh, J.; Maclauchian, L. Forest health and climate change: A British Columbia perspective. For. Chron. 2010, 86, 412–422. [Google Scholar]
- Bentz, B.; Régnière, J.; Fettig, C.; Hansen, E. Climate change and bark beetles of the western United States and Canada: Direct and indirect effects. BioScience 2010, 60, 603–613. [Google Scholar]
- Adams, H.D.; Kolb, T.E. Drought responses of conifers in ecotone forests of northern Arizona: Tree ring growth and leaf δ13C. Oecologia 2004, 140, 217–225. [Google Scholar]
- Lapointe-Garant, M.-P.; Huang, J.-G.; Gea-Izquierodo, G.; Raulier, F. Use of tree rings to study the effect of climate change on tremling aspen in Quebec. Glob. Chang. Biol. 2010, 16, 2039–2051. [Google Scholar] [CrossRef]
- Anyomi, K.A.; Raulier, F.; Mailly, D.; Girardin, M.P.; Bergeron, Y. Using height growth to model local and regional responses of trembling aspen (Populus tremuloides Michaux) to climate within the boreal forests of western Quebec. Ecol. Model. 2012, 243, 123–132. [Google Scholar] [CrossRef]
- Bontemps, J.-D.; Herve, J.-C.; Dhote, J.F. Long-term changes in forest productivity: A consistent assessment in even-aged stands. For. Sci. 2009, 55, 549–564. [Google Scholar]
- Coops, N.C.; Waring, R.H.; Beier, C.; Roy-Jauvin, R.; Wang, T. Modeling the occurrence of fifteen coniferous tree species throughout the Pacific Northwest of North America using a hybrid approach of a generic process-based growth model and decision tree analysis. Appl. Vegetation Sci. 2011, 14, 401–414. [Google Scholar]
- Jackson, S.T.; Betancourt, J.L.; Booth, R.K.; Gray, S.T. Ecology and the ratchet of events: Climate variability, niche dimensions, and species distributions. Proc. Natl. Acad. Sci. USA 2009, 106 (Suppl. 20), 19685–19692. [Google Scholar]
- Rehfeldt, G.E.; Crookston, N.L.; Warwell, M.V.; Evans, J.S. Empirical Analyses of Plant-Climate Relationships for the Western United States. Int. J. Plant Sci. 2006, 167, 1123–1150. [Google Scholar] [CrossRef]
- Bonan, G.B. Forests and climate change: Forcing, feedbacks, and the climate benefits of forests. Science 2008, 320, 1444–1449. [Google Scholar] [CrossRef]
- Richardson, A.D.; Anderson, R.S.; Arain, M.A.; Barr, A.G.; Bohrer, G.; Chen, G.; Chen, J.M.; Ciais, P.; Davis, K.J.; Desai, A.R.; et al. Terrestrial biosphere models need better representation of vegetation phenology: Results from the North American carbon program site synthesis. Glob. Chang. Biol. 2012, 18, 566–584. [Google Scholar] [CrossRef]
- Treuhaft, R.N.; Law, B.E.; Asner, G.P. Forest attributes from radar interferometric structure and its function with iptical remote sensing. BioScience 2004, 54, 561–571. [Google Scholar] [CrossRef]
- Swenson, J.; Waring, R. Predicting site index with a physiologically based growth model across Oregon, USA. Can. J. Res. 2005, 35, 1697–1707. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Waring, R.H.; Coops, N.C.; Mathys, A.; Hilker, T.; Latta, G. Process-Based Modeling to Assess the Effects of Recent Climatic Variation on Site Productivity and Forest Function across Western North America. Forests 2014, 5, 518-534. https://doi.org/10.3390/f5030518
Waring RH, Coops NC, Mathys A, Hilker T, Latta G. Process-Based Modeling to Assess the Effects of Recent Climatic Variation on Site Productivity and Forest Function across Western North America. Forests. 2014; 5(3):518-534. https://doi.org/10.3390/f5030518
Chicago/Turabian StyleWaring, Richard H., Nicholas C. Coops, Amanda Mathys, Thomas Hilker, and Greg Latta. 2014. "Process-Based Modeling to Assess the Effects of Recent Climatic Variation on Site Productivity and Forest Function across Western North America" Forests 5, no. 3: 518-534. https://doi.org/10.3390/f5030518
APA StyleWaring, R. H., Coops, N. C., Mathys, A., Hilker, T., & Latta, G. (2014). Process-Based Modeling to Assess the Effects of Recent Climatic Variation on Site Productivity and Forest Function across Western North America. Forests, 5(3), 518-534. https://doi.org/10.3390/f5030518





