Effects of Restoration Techniques on Soil Carbon and Nitrogen Dynamics in Florida Longleaf Pine (Pinus palustris) Sandhill Forests
Abstract
:1. Introduction
| Treatment | Period 1972 to 1995 | Period 1995 to 2009 |
|---|---|---|
| Reference | 2.2 | 6.8 |
| Burn-only | 0.6 | 4.2 |
| Delayed burn | 0.4 | 4.0 |
| Herbicide | 0.6 | 5.4 |
| Mechanical | 0.4 | 3.8 |
2. Experimental Section
2.1. Study Area
2.2. Experimental Design
2.3. Soil Analysis
2.4. Foliar Analysis
2.5. Statistical Analysis
3. Results
3.1. Multi-Response Permutation Procedure (MRPP)
| Treatment | Reference | Burn-only | Delayed burn | Herbicide | Mechanical |
|---|---|---|---|---|---|
| Pre-treatment (fall 1994) | |||||
| Reference | N.S. | N.S. | N.S. | N.S. | |
| Burn-only | N.S. | N.S. | N.S. | ||
| Delayed burn | N.S. | N.S. | |||
| Herbicide | N.S. | ||||
| Early Post-treatment (spring 1996) | |||||
| Reference | 0.003 | 0.04 | 0.001 | N.S. | |
| Burn-only | N.S. | N.S. | N.S. | ||
| Delayed burn | N.S. | N.S. | |||
| Herbicide | 0.099 | ||||
| Early Post-treatment (spring 1997) | |||||
| Reference | N.S. | 0.001 | 0.066 | 0.001 | |
| Burn-only | 0.046 | N.S. | N.S. | ||
| Delayed burn | N.S. | 0.046 | |||
| Herbicide | N.S. | ||||
| Late Post-treatment (spring 2009) | |||||
| Reference | N.S. | N.S. | 0.04 | N.S. | |
| Burn-only | N.S. | 0.035 | 0.04 | ||
| Delayed burn | N.S. | N.S. | |||
| Herbicide | N.S. |
3.2. Soil pH, Soil Bulk Density, and Soil Moisture
| Effects | df | Bulk density | Cw | Nw | C:N ratio | Cv | Nv |
|---|---|---|---|---|---|---|---|
| Treatment | 3 | N.S. | N.S. | 0.0339 | <0.0001 | N.S. | N.S. |
| Depth | 2 | <0.0001 | <0.0001 | <0.0001 | <0.0001 | <0.0001 | <0.0001 |
| T × D | 6 | N.S. | N.S. | <0.0001 | 0.0023 | N.S. | 0.0554 |
| NO3−w | NO3−v | NH4+w | NH4+v | N nitw | N nitv | ||
| Treatment | 3 | 0.0200 | 0.0145 | 0.0022 | 0.0024 | 0.0009 | 0.0070 |
| Depth | 1 | N.S. | <0.0001 | <0.0001 | <0.0001 | N.S. | N.S. |
| T × D | 3 | N.S. | N.S. | 0.0188 | N.S. | N.S. | N.S. |
| Net N minw | Net N minv | Pw | Pv | pH | Moisture | ||
| Treatment | 3 | N.S. | N.S. | 0.0152 | N.S. | N.S. | N.S. |
| Depth | 1 | 0.0017 | 0.0065 | <0.0001 | 0.0350 | <0.0001 | N.S. |
| T × D | 3 | N.S. | N.S. | 0.0283 | N.S. | N.S. | N.S. |
3.3. Carbon and Nitrogen Concentrations and Pools
| Reference | Burn-only | Delayed burn | Herbicide | Mechanical | |
|---|---|---|---|---|---|
| Bulk Density (g cm−3) | |||||
| Litter a | 0.031 (0.003) | 0.037 (0.006) a | 0.028 (0.001) a | 0.022 (0.005) a | 0.027 (0.002) a |
| Min 0–10 cm b | 0.98 (0.07) | 0.94 (0.04) a | 1.01 (0.05) a | 1.07 (0.05) a | 1.03 (0.04) a |
| Min 10–30 cm c | 1.15 (0.09) | 1.18 (0.06) a | 1.20 (0.03) a | 1.26 (0.04) a | 1.22 (0.03) a |
| pH | |||||
| Litter | NM | NM | NM | NM | NM |
| Min 0–10 cm a | 5.03 (0.05) | 4.98 (0.06) a | 5.10 (0.08) a | 5.06 (0.07) a | 5.05 (0.03) a |
| Min 10–30 cm b | 5.21 (0.05) | 5.15 (0.02) a | 5.13 (0.03) a | 5.18 (0.02) a | 5.19 (0.05) a |
| C (%) | |||||
| Litter a | 51.12 (0.90) | 50.21 (0.64) a | 49.73 (0.58) a | 50.28 (1.13) a | 50.03 (0.37) a |
| Min 0–10 cm b | 1.23 (0.17) | 1.27 (0.09) a | 1.06 (0.10) a | 0.97 (0.05) a | 0.96 (0.03) a |
| Min 10–30 cm c | 0.45 (0.03) | 0.52 (0.04) a | 0.53 (0.05) a | 0.46 (0.02) a | 0.48 (0.02) a |
| N (%) | |||||
| Litter a | 0.57 (0.03) | 0.60 (0.05) b | 0.72 (0.04) a | 0.70 (0.01) a | 0.64 (0.02) ab |
| Min 0–10 cm b | 0.046 (0.007) | 0.047 (0.003) a | 0.045 (0.003) a | 0.042 (0.002) a | 0.038 (0.002) a |
| Min 10–30 cm c | 0.019 (0.001) | 0.021 (0.002) a | 0.022 (0.001) a | 0.022 (0.002) a | 0.020 (0.001) a |
| CN ratio | |||||
| Litter a | 95.3 (3.1) | 87.3 (6.2) a | 69.9 (6.4) c | 73.6 (1.5) bc | 80.9 (1.8) ab |
| Min 0–10 cm b | 27.8 (0.7) | 26.9 (1.4) a | 23.6 (0.5) a | 23.2 (0.6) a | 25.8 (0.7) a |
| Min 10–30 cm b | 24.0 (0.7) | 24.5 (1.0) a | 23.6 (0.8) a | 21.1 (0.3) a | 23.7 (0.8) a |
| C pool (g m−2) | |||||
| Litter a | 158.7 (17.4) | 187.4 (31.6) a | 140.6 (6.4) a | 108.3 (22.9) a | 135.4 (9.5) a |
| Min 0–10 cm b | 1139.7 (71.4) | 1209.7 (63.0) a | 1055.2 (87.0) a | 1013.6 (53.0) a | 972.7 (43.2) a |
| Min 10–30 cm b | 1032.5 (103.0) | 1194.0 (69.4) a | 1276.0 (128.6) a | 1151.4 (78.5) a | 1162.8 (59.9) a |
| N pool (g m−2) | |||||
| Litter a | 1.96 (0.24) a | 2.28 (0.43) a | 2.13 (0.16) a | 1.52 (0.32) a | 1.80 (0.17) a |
| Min 0–10 cm c | 39.5 (1.96) a | 45.2 (2.82) a | 44.7 (3.31) a | 43.5 (1.67) a | 38.1 (2.40) a |
| Min 10–30 cm b | 43.3 (4.42) b | 47.6 (2.60) a | 53.7 (3.75) a | 56.3 (4.46) a | 49.4 (2.60) a |
| Moisture (%) | |||||
| Litter | NM | NM | NM | NM | NM |
| Min 0–10 cm a | 0.02 (0.01) | 0.07 (0.05) a | 0.03 (0.04) a | 0.05 (0.02) a | 0.03 (0.01) a |
| Min 10–30 cm a | 0.03 (0.01) | 0.05 (0.01) a | 0.04 (0.01) a | 0.03 (0.01) a | 0.04 (0.01) a |
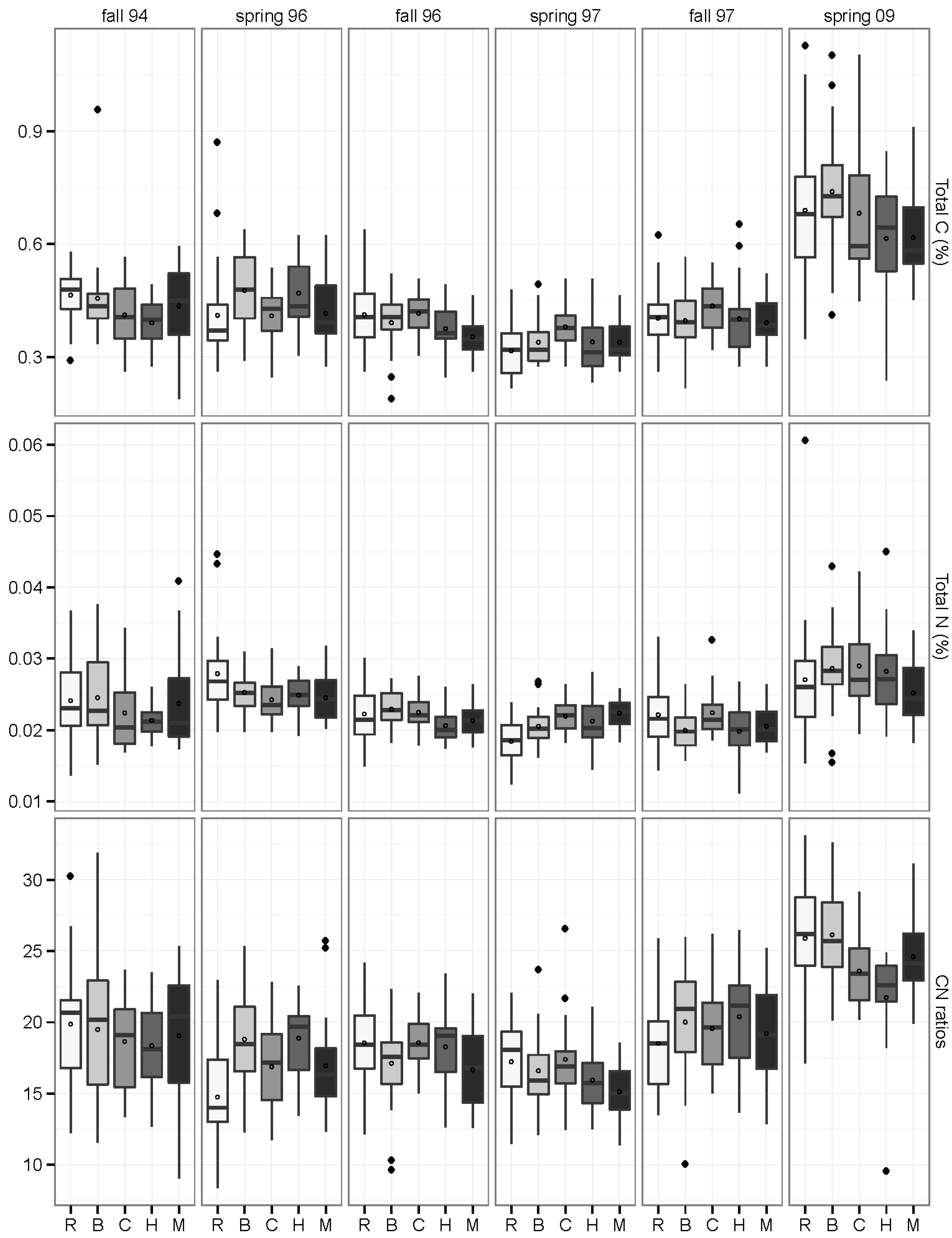
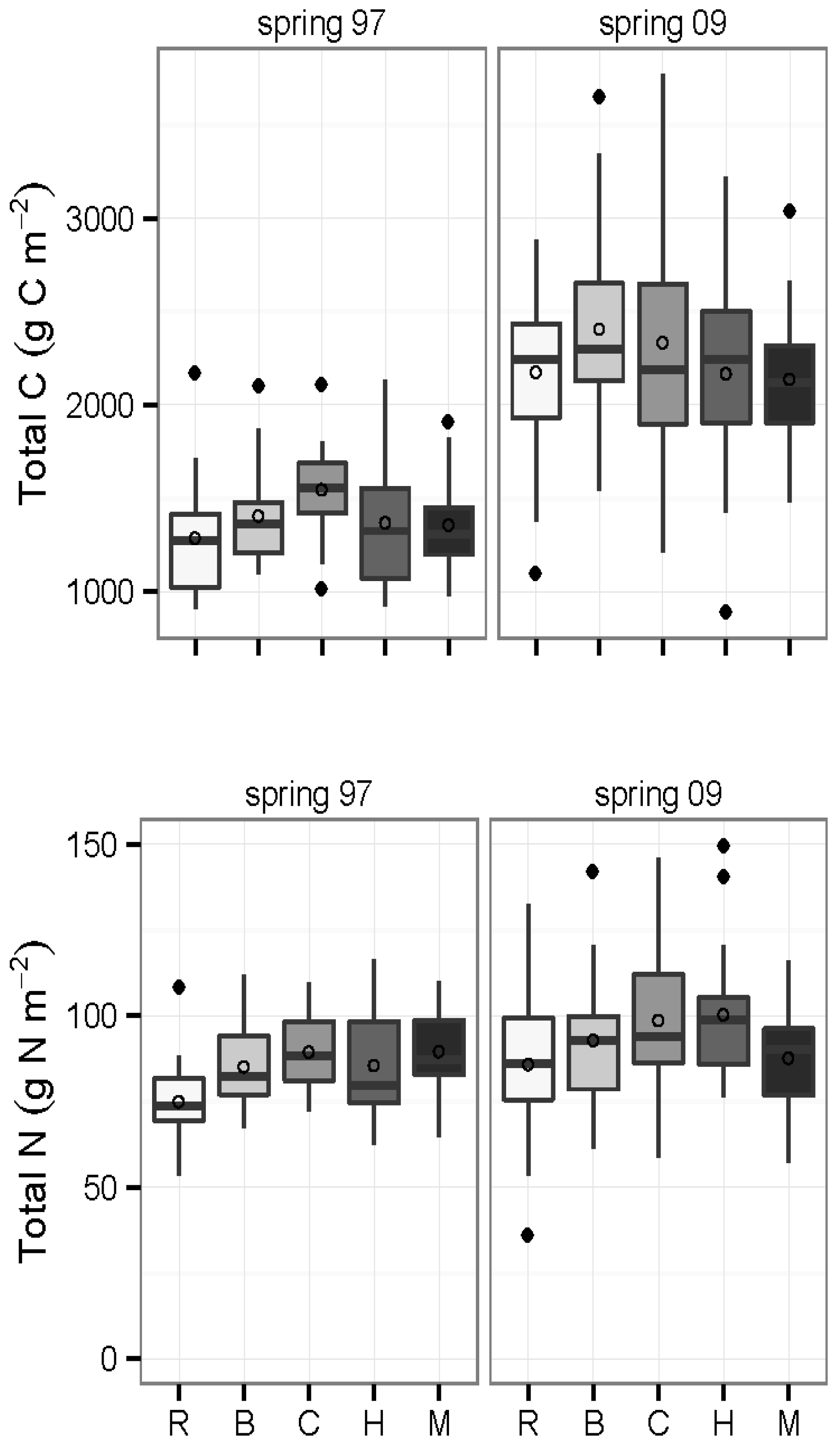
3.4. Inorganic N and Mineralization Rates: 2009
| Reference | Burn-only | Delayed burn | Herbicide | Mechanical | |
|---|---|---|---|---|---|
| NH4 (µg NH4 gdw−1) | |||||
| Min 0–10 cm a | 1.14 (0.19) | 1.64 (0.16) b | 2.60 (0.45) a | 1.81 (0.31) b | 1.48 (0.17) b |
| Min 10–30 cm b | 0.91 (0.07) | 1.08 (0.12) a | 1.29 (0.13) a | 1.27 (0.16) a | 0.97 (0.09) a |
| NO3 (µg NO3 gdw−1) | |||||
| Min 0–10 cm a | 0.12 (0.05) | 0.25 (0.06) B | 0.22 (0.06) B | 0.41 (0.10) A | 0.25 (0.08) B |
| Min 10–30 cm a | 0.15 (0.01) | 0.32 (0.08) | 0.31 (0.08) | 0.36 (0.12) | 0.24 (0.05) |
| NH4 (g NH4 m−2) | |||||
| Min 0–10 cm a | 0.12 (0.02) | 0.15 (0.02) B | 0.27 (0.05) A | 0.19 (0.04) AB | 0.15 (0.02) B |
| Min 10–30 cm b | 0.22 (0.03) | 0.24 (0.02) | 0.32 (0.04) | 0.32 (0.04) | 0.24 (0.02) |
| NO3 (g NO3 m−2) | |||||
| Min 0–10 cm a | 0.012 (0.004) | 0.022 (0.006) B | 0.022 (0.008) B | 0.045 (0.011) A | 0.026 (0.008) B |
| Min 10–30 cm b | 0.036 (0.005) | 0.075 (0.022) | 0.075 (0.022) | 0.102 (0.029) | 0.059 (0.012) |
| Nitrification (µg NO3 kg−1d−1) | |||||
| Min 0–10 cm a | 9.67 (3.64) | 8.80 (8.63) AB | 18.98 (12.22) A | −7.66 (7.96) B | 4.47 (5.10) B |
| Min 10–30 cm a | 13.92 (4.62) | 5.24 (6.65) | 18.23 (14.06) | −2.66 (5.96) | −2.03 (2.77) |
| N min (µg N min kg−1d−1) | |||||
| Min 0–10 cm a | 16.95 (6.02) | 10.22 (9.65) a | 9.97 (6.72)a | 5.59 (7.54) a | 10.84 (7.64) a |
| Min 10–30 cm b | −3.19 (7.23) | −5.42 (7.10) a | 3.47 (14.43)a | −13.87 (11.70)a | −13.57 (3.39) a |
| Nitrification (mg NO3 m−2d−1) | |||||
| Min 0–10 cm a | 0.97 (0.41) | 0.83 (0.82)AB | 1.68 (1.21) A | −0.85 (0.83) B | 0.49 (0.49) B |
| Min 10–30 cm a | 2.92 (7.18) | 0.76 (1.67) | 4.30 (3.31) | −0.73 (1.43) | −0.53 (0.70) |
| N min (mg N min m−2d−1) | |||||
| Min 0–10 cm a | 1.60 (0.64) | 0.81 (0.82) a | 0.92 (0.65) a | 0.33 (0.75) a | 1.04 (0.78) a |
| Min 10–30 cm b | −1.29 (1.43) | −1.58 (1.89) a | 0.73 (3.44) a | –3.40 (1.25) a | −3.39 (0.82) a |
| P (µg gdw−1) | |||||
| Min 0–10 cm a | 0.89 (0.12) | 0.68 (0.08) b | 1.26 (0.34) a | 0.68 (0.04) b | 0.73 (0.05) b |
| Min 10–30 cm b | 0.34 (0.03) | 0.38 (0.05)a | 0.48 (0.08) a | 0.41 (0.07) a | 0.37 (0.05) a |
| P (g m−2) | |||||
| Min 0–10 cm a | 0.088 (0.013) | 0.067 (0.008) a | 0.131 (0.043) a | 0.075 (0.009) a | 0.075 (0.062) a |
| Min 10–30 cm b | 0.084 (0.008) | 0.109 (0.021) a | 0.121 (0.018) a | 0.129 (0.039) a | 0.090 (0.011) a |
3.5. Mehlich Phosphorus
3.6. Foliar C and N, 13C and 15N
4. Discussion
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Stanturf, J.A.; Wade, D.D.; Waldrop, T.A.; Kennard, D.K.; Achtemeir, G.L. Fire in Southern Forest Landscapes. In Southern Forest Resource Assessment: Summary Report; Station General Technical Report SRS-54; Wear, D.N., Greis, J.G., Eds.; United States Department of Agriculture, Forest Service Southern Research: Asheville, NC, USA, 2002; pp. 607–630. [Google Scholar]
- Frost, C.C. History and future of the longleaf pine ecosystem. In The Longleaf Pine Ecosystem: Ecology, Silviculture, and Restoration; Springer series on Environmental Management; Jose, J., Jokela, E.J., Miller, D.L., Eds.; Springer: New York, NY, USA, 2006; pp. 9–48. [Google Scholar]
- Frost, C.C. Four centuries of changing landscape patterns in the longleaf pine ecosystem. In Proceedings of the Tall Timbers Fire Ecology Conference No. 18, the Longleaf Pine Ecosystem: Ecology, Restoration and Management; Hermann, S.M., Ed.; Tall Timbers Research Station: Tallahassee, FL, USA, 1993; pp. 17–43. [Google Scholar]
- Noss, R.F.; LaRoe, E.T.; Scott, J.M. Endangered Ecosystems of the United States: A Preliminary Assessment of Loss and Degradation; United States Department of Interior National Biological Service, Biological Report 28; United States Department of Interiors: Washington, DC, USA, 1995.
- Chapman, H.H. Is the longleaf type a climax? Ecology 1932, 45, 328–334. [Google Scholar] [CrossRef]
- Garren, K.H. Effects of fire on vegetation of the Southeastern United States. Bot. Rev. 1943, 9, 617–654. [Google Scholar] [CrossRef]
- Gilliam, F.S.; Platt, W.J. Effects of long-term fire exclusion on tree species composition and stand structure in an old-growth Pinus palustris (Longleaf pine) forest. Plant Ecol. 1999, 140, 15–26. [Google Scholar] [CrossRef]
- Van Lear, D.H.; Carroll, W.D.; Kapeluck, P.R.; Johnson, R. History and restoration of the longleaf pine-grassland ecosystem: Implications for species at risk. For. Ecol. Manag. 2005, 211, 150–165. [Google Scholar] [CrossRef]
- Hiers, J.K.; O’Brien, J.J.; Will, R.E.; Mitchell, R.J. Forest floor depth mediates understory vigor in xeric Pinus palustris ecosystems. Ecol. Appl. 2007, 17, 806–814. [Google Scholar] [CrossRef]
- Heyward, F. The relation of fire to stand composition of longleaf pine forests. Ecology 1939, 20, 287–304. [Google Scholar] [CrossRef]
- Bruce, D. Thirty-two years of annual burning in longleaf pine. J. For. 1947, 45, 809–814. [Google Scholar]
- McGuire, J.P.; Mitchell, R.J.; Moser, E.B.; Pecot, S.D.; Gjerstad, D.H.; Hedman, C.W. Gap in a gappy forest: Plant resources response to tree removal in longleaf pine savannas. Can. J. For. Res. 2011, 31, 765–778. [Google Scholar]
- Varner, J.M., III; Gordon, D.R.; Putz, F.E.; Hiers, J.K. Restoring fire to long-unburned Pinus palustris ecosystems: Novel fire effects and consequences for long-unburned ecosystems. Restor. Ecol. 2005, 13, 536–544. [Google Scholar] [CrossRef]
- Hu, H.; Wang, G.G.; Walker, J.L.; Knapp, B.O. Silvicultural treatments for converting loblolly pine to longleaf pine dominance: Effects on resource availability and their relationships with planted longleaf pine seedlings. For. Ecol. Manag. 2012, 282, 115–123. [Google Scholar] [CrossRef]
- Harrington, T.B.; Edwards, M.B. Understory vegetation, resource availability, and litterfall responses to pine thinning and woody vegetation control in longleaf pine plantations. Can. J. For. Res. 1999, 29, 1055–1064. [Google Scholar] [CrossRef]
- Kush, J.S.; Meldahl, R.S.; Boyer, W.D. Understory plant community response after 23 years of hardwood control treatments in natural longleaf pine (Pinus palustris) forests. Can. J. For. Res. 1999, 29, 1047–1054. [Google Scholar] [CrossRef]
- Brockway, D.G.; Outcalt, K.W.; Wilkins, R.N. Restoring longleaf pine wiregrass ecosystems: Plant cover, diversity and biomass following low-rate hexazinone application on Florida sandhills. For. Ecol. Manag. 1998, 103, 159–175. [Google Scholar] [CrossRef]
- Brockway, D.G.; Outcalt, K.W. Restoring longleaf pine wiregrass ecosystems: Hexazinone application enhances effects of prescribed fire. For. Ecol. Manag. 2000, 137, 121–138. [Google Scholar] [CrossRef]
- Provencher, L.; Herring, B.J.; Gordon, D.R.; Rodgers, H.L.; Tanner, G.W.; Hardesty, J.L.; Brennan, L.A.; Litt, A.R. Longleaf pine and oak responses to hardwood reduction techniques in fire-suppressed sandhills in northwest Florida. For. Ecol. Manag. 2001, 148, 63–77. [Google Scholar] [CrossRef]
- Provencher, L.; Herring, B.J.; Gordon, D.R.; Rodgers, H.L.; Galley, K.E.M.; Tanner, G.W.; Hardesty, J.L.; Brennan, L.A. Effects of hardwood reduction techniques on longleaf pine sandhill vegetation in northwest Florida. Restor. Ecol. 2001, 9, 13–27. [Google Scholar] [CrossRef]
- Archer, J.K.; Miller, D.L.; Tanner, G.W. Changes in understory vegetation and soil characteristics following silvicultural activities in a southeastern mixed pine forest. J. Torrey Bot. 2007, 134, 489–504. [Google Scholar] [CrossRef]
- Haywood, J.D. Influence of pine straw harvesting, prescribed fire, and fertilization on a Louisiana longleaf pine site. South. J. Appl. For. 2009, 33, 115–120. [Google Scholar]
- Jose, S.; Ranasinghe, S.; Ramsey, C.L. Longleaf pine (Pinus palustris P. Mill.) restoration using herbicides: Overstory and understory vegetation responses on a coastal plain flatwoods site in Florida, USA. Restor. Ecol. 2010, 18, 244–251. [Google Scholar]
- Outcalt, K.W.; Brockway, D.G. Structure and composition changes following restoration treatments of longleaf pine forests on the Gulf Coastal Plain of Alabama. For. Ecol. Manag. 2010, 259, 1615–1623. [Google Scholar] [CrossRef]
- Steen, D.A.; Smith, L.L.; Conner, L.M.; Litt, A.R.; Provencher, L.; Hiers, J.K.; Pokswinski, S.; Guyer, C. Reptile assemblage response to restoration of fire-suppressed longleaf pine sandhills. Ecol. Appl. 2013, 23, 148–158. [Google Scholar] [CrossRef]
- Steen, D.A.; Conner, L.M.; Smith, L.L.; Provencher, L.; Hiers, J.K.; Pokswinski, S.; Helms, B.S.; Guyer, C. Birds assemblage response to restoration of fire-suppressed longleaf pine sandhills. Ecol. Appl. 2013, 23, 134–147. [Google Scholar] [CrossRef]
- Hough, W.A. Impact of Prescribed Fire on Understory and Forest Floor Nutrients; Research Note SE-303; United States Department of Agriculture, Forest Service, Southeastern Forest Experiment Station: Asheville, NC, USA, 1981.
- DiStefano, J.F.; Gholz, H.L. Controls over net inorganic nitrogen transformations in an age sequence of Pinus. elliottii plantations in North Florida. For. Sci. 1989, 35, 920–934. [Google Scholar]
- Christensen, N.L. Fire and soil-plant nutrient relations in a pine-wiregrass savanna on the coastal plain of North Carolina. Oecologia 1977, 31, 27–44. [Google Scholar] [CrossRef]
- Abrahamson, W.G. Post-recovery of Florida Lake Wales Ridge vegetation. Am. J. Bot. 1984, 71, 9–21. [Google Scholar] [CrossRef]
- Binkley, D.; Richter, D.; David, M.B.; Caldwell, B. Soil chemistry in a loblolly/longleaf pine forest with interval burning. Ecol. Appl. 1992, 2, 157–164. [Google Scholar] [CrossRef]
- Boyer, W.D.; Miller, J.H. Effect of burning and brush treatments on nutrient and soil physical properties in young longleaf pine stands. For. Ecol. Manag. 1994, 70, 311–318. [Google Scholar] [CrossRef]
- Wilson, C.A.; Mitchell, R.J.; Hendricks, J.J.; Boring, L.R. Patterns and controls of ecosystem function in longleaf pine—Wiregrass savannas. II. Nitrogen dynamics. Can. J. For. Res. 1999, 29, 752–760. [Google Scholar] [CrossRef]
- Wilson, C.A.; Mitchell, R.J.; Boring, L.R.; Hendricks, J.J. Soil nitrogen dynamics in a fire-maintained forest ecosystem: Results over a 3-year burn interval. Soil Biol. Biochem. 2002, 34, 679–689. [Google Scholar] [CrossRef]
- Boring, L.R.; Hendricks, J.J.; Wilson, C.A.; Mitchell, R.J. Season of burn and nutrient losses in a longleaf pine ecosystem. Int. J. Wildland Fire 2004, 13, 443–453. [Google Scholar] [CrossRef]
- Lavoie, M.; Starr, G.; Mack, M.C.; Martin, T.A.; Gholz, H.L. Effects of a prescribed fire on understory vegetation, carbon pools, and soil nutrients in a longleaf pine-slash pine forest in Florida. Nat. Areas J. 2010, 30, 82–94. [Google Scholar] [CrossRef]
- Lavoie, M.; Mack, M.C.; Hiers, J.K.; Pokswinski, S. The effects of restoration treatments on the spatial variability of soil processes under longleaf pine trees. Forests 2012, 3, 591–604. [Google Scholar] [CrossRef]
- Palik, B.J.; Mitchell, R.J.; Houseal, G.; Pederson, N. Effects of canopy structure on resource availability and seedling responses in a longleaf pine ecosystem. Can. J. For. Res. 1997, 27, 1458–1464. [Google Scholar] [CrossRef]
- Mitchell, R.J.; Kirkman, L.K.; Pecot, S.D.; Wilson, C.A.; Palik, B.J.; Boring, L.R. Patterns and controls of ecosystems function in longleaf pine—Wiregrass savannas. I. Aboveground net primary productivity. Can. J. For. Res. 1999, 29, 743–751. [Google Scholar] [CrossRef]
- Carter, M.C.; Foster, C.D. Prescribed burning and productivity in southern pine forests: A review. For. Ecol. Manag. 2004, 191, 93–109. [Google Scholar] [CrossRef]
- Ford, C.R.; Mitchell, R.J.; Teskey, R.O. Water table depth affects productivity, water use, and the response to nitrogen addition in savanna system. Can. J. For. Res. 2008, 38, 2118–2127. [Google Scholar] [CrossRef]
- Provencher, L.; Litt, A.R.; Galley, K.E.M.; Gordon, D.R.; Tanner, G.W.; Brennan, L.A.; Gobris, N.M.; McAdoo, S.J.; McAdoo, J.P.; Herring, B.J. Restoration of Fire-Suppressed Longleaf Pine Sandhills at Eglin Air Force Base, Florida; Final Report to Natural Resources Management Division; Eglin Air Force Base: Niceville, Florida, Science Division; The Nature Conservancy: Gainesville, FL, USA, 2001.
- Provencher, L.; Gobris, N.M.; Brennan, L.A.; Gordon, D.R.; Hardesty, L. Breeding bird response to midstory hardwood reduction in Florida sandhill longleaf pine forests. J. Wildl. Manag. 2002, 66, 641–661. [Google Scholar] [CrossRef]
- Provencher, L.; Gobris, N.M.; Brennan, L.A. Effects of hardwood reduction on winter birds in northwest Florida longleaf pine sandhill forests. Auk 2002, 119, 71–87. [Google Scholar]
- Provencher, L.; Galley, K.E.M.; Litt, A.R.; Gordon, D.R.; Brennan, L.A.; Tanner, G.W.; Hardesty, J.L. Fire, herbicide, and chainsaw felling effects on arthropods in fire-suppressed longleaf pine sandhills at Eglin Air Force Base, Florida. In The Role of Fire in Nongame Wildlife Management and Community Restoration: Traditional Uses and New Directions Proceedings of a Special Workshop; General Technical Report NE-288; Ford, W.M., Russell, K.R., Moorman, C.E., Eds.; United States Department of Agriculture, Forest Service Northern Research Station: Nashville, TN, USA, 2002; pp. 24–34. [Google Scholar]
- Litt, A.R.; Provencher, L.; Tanner, G.W.; Franz, R. Herpetofaunal responses to restoration treatments of longleaf pine sandhills in Florida. Restor. Ecol. 2001, 9, 462–474. [Google Scholar] [CrossRef]
- Eglin Air Force Base, U.S. Air Force. Available online: http://www.eglin.af.mil/ (accessed on 22 December 2013).
- Overing, J.D.; Weeks, H.H.; Wilson, J.P.; Sullivan, J.; Ford, R.D. Soil Survey of Okaloosa Country, Florida. US Department of Agriculture; Natural Resource Conservation Service: Washington, DC, USA, 1995.
- Myers, R.L. Schrub and high pine. In Ecosystem of Florida; Myers, R.L., Ewel, J., Eds.; University of Florida Presses: Gainesville, FL, USA, 1990; pp. 174–193. [Google Scholar]
- Rodgers, H.L.; Provencher, L. Analysis of longleaf pine sandhill vegetation in northwest Florida. Castanea 1999, 64, 138–162. [Google Scholar]
- Mylavarapu, R.; Kennelley, E. UF/IFAS Extension Soil Testing Laboratory (ESTL) Analytical Procedures and Training Manual; University of Florida: Gainesville, FL, USA, 2002. [Google Scholar]
- Keeney, D.R.; Nelson, D.W. Nitrogen—Inorganic forms. In Methods of Soil Analysis. Part 2—Chemical and Microbial Properties, 2nd ed.; Page, A.L., Miller, R.H., Keeney, D.R., Eds.; Soil Science Society of America: Madison, WI, USA, 1982; pp. 643–698. [Google Scholar]
- Bisutti, I.; Hilke, I.; Raessler, M. Determination of total organic carbon—An overview of current methods. TRAC-Trend Anal. Chem. 2004, 23, 716–726. [Google Scholar] [CrossRef]
- Pribyl, D.W. A critical review of the conventional SOC to SOM conversion factor. Geoderma 2010, 156, 75–83. [Google Scholar] [CrossRef]
- Perié, C.; Ouimet, R. Organic carbon, organic matter and bulk density relationships in boreal forest soils. Can. J. Soil Sci. 2008, 88, 315–325. [Google Scholar] [CrossRef]
- Chatterjee, A.; Lal, R.; Wielopolski, L.; Martin, M.Z.; Ebinger, M.H. Evaluation of different soil carbon determination methods. Crit. Rev. Plant Sci. 2009, 28, 164–178. [Google Scholar] [CrossRef]
- Pereira, M.G.; Espindula, A.; Valladeres, G.S.; Cunha dos Anjos, L.H.; de Melo Benites, V.; Schultz, N. Comparison of total nitrogen methods applied for histosols and soil horizons with high organic matter content. Commun. Soil Sci. Plant 2006, 37, 939–943. [Google Scholar] [CrossRef]
- Dieckow, J.; Mielniczuk, J.; Knicker, H.; Bayer, C.; Dick, D.P.; Kogel-Knabner, I. Comparison of carbon and nitrogen determination methods for samples of a paleudult subjected to no-till cropping systems. Sci. Agric. 2007, 64, 532–540. [Google Scholar]
- McCune, B.; Grace, J.B. Analysis of Ecological Communities; MjM Software Design: Gleneden Beach, OR, 2002. [Google Scholar]
- McCune, B.; Mefford, M.J. PC-ORD, Multivariate Analysis of Ecological Data; Version 5 for Windows Edition; MjM Software Design: Gleneden Beach, OR, USA, 2006. [Google Scholar]
- Benjamini, Y.; Yekutieli, D. The control of the false discovery rate in multiple testing under dependency. Ann. Stat. 2001, 29, 1165–1188. [Google Scholar] [CrossRef]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2012. [Google Scholar]
- Bolker, B.M.; Brooks, M.E.; Clark, C.J.; Geange, S.W.; Poulsen, J.R.; Stevens, M.H.H.; White, J.-S.S. Generalized linear mixed models: A practical guide for ecology and evolution. Trends Ecol. Evol. 2009, 24, 127–135. [Google Scholar] [CrossRef]
- Bates, D.; Maechler, M.; Bolker, B. lme4.0: Linear Mixed-Effects Models Using S4 Classes. R Package Version 0.9999-1/r1692. 2012. Available online: http://R-Forge.R-project.org/projects/lme4/ (accessed on 2 August 2012).
- Crawley, M.J. The R Book; John Wiley & Sons Ltd: Chichester, UK, 2007. [Google Scholar]
- Zuur, A.; Ieno, E.N.; Walker, N.; Saveiliev, A.A.; Smith, G.M. Mixed Effects Models and Extensions in Ecology with R; Springer: New York, NY, USA, 2009. [Google Scholar]
- Hothorn, T.; Bretz, F.; Westfall, P. Simultaneous inference in general parametric models. Biom. J. 2008, 50, 346–363. [Google Scholar] [CrossRef]
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; R Package Version 2.0.9.1; Springer: Berlin, Germany, 2009. [Google Scholar]
- Anderson, R.C.; Menges, E.S. Effects of fire on sandhill herbs: Nutrients, mycorrhizae, and biomass allocation. Am. J. Bot. 1997, 84, 938–948. [Google Scholar] [CrossRef]
- Alexis, M.A.; Rasse, D.P.; Rumpel, C.; Bardoux, G.; Péchot, N.; Schmalzer, P.; Drake, B.; Mariotti, A. Fire impact on C and N losses and charcoal production in a scrub oak ecosystem. Biogeochemistry 2007, 82, 201–216. [Google Scholar] [CrossRef]
- Steen, D.A.; Smith, L.L.; Morris, G.; Conner, L.M.; Litt, A.R.; Pokswinski, S.; Guyer, C. Response of Six-Lined Racerunner (Aspidoscelis sexlineata) to habitat restoration in fire-suppressed longleaf pine (Pinus palustris) sandhills. Rest. Ecol. 2013, 21, 457–473. [Google Scholar] [CrossRef]
- Samuelson, L.J.; McLemore, P.C., III; Somers, G.L. Relationship between foliar δ13C and hydraulic pathway length in Pinus palustris. For. Sci. 2003, 49, 790–798. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lavoie, M.; Mack, M.C.; Hiers, J.K.; Pokswinski, S.; Barnett, A.; Provencher, L. Effects of Restoration Techniques on Soil Carbon and Nitrogen Dynamics in Florida Longleaf Pine (Pinus palustris) Sandhill Forests. Forests 2014, 5, 498-517. https://doi.org/10.3390/f5030498
Lavoie M, Mack MC, Hiers JK, Pokswinski S, Barnett A, Provencher L. Effects of Restoration Techniques on Soil Carbon and Nitrogen Dynamics in Florida Longleaf Pine (Pinus palustris) Sandhill Forests. Forests. 2014; 5(3):498-517. https://doi.org/10.3390/f5030498
Chicago/Turabian StyleLavoie, Martin, Michelle C. Mack, John K. Hiers, Scott Pokswinski, Analie Barnett, and Louis Provencher. 2014. "Effects of Restoration Techniques on Soil Carbon and Nitrogen Dynamics in Florida Longleaf Pine (Pinus palustris) Sandhill Forests" Forests 5, no. 3: 498-517. https://doi.org/10.3390/f5030498




