Resiliency of an Interior Ponderosa Pine Forest to Bark Beetle Infestations Following Fuel-Reduction and Forest-Restoration Treatments
Abstract
:1. Introduction
2. Experimental Section
| Plot | Stand Structure 1 | Prescribed Burn | Sample Period 1 2 | Sample Period 2 | Sample Period 3 3 | |
|---|---|---|---|---|---|---|
| Unburned | Burned | All | All | |||
| September 1998 | August 1999 | September 2002 | June 2008 | |||
| 39 | LoD | October 1997 | September 1998 | September 1999 | September 2002 | June 2008 |
| 40 | LoD | October 2000 | August 2002 | July 2002 | August 2005 | May 2012 |
| 41 | HiD | October 1997 | August 1998 | August 1999 | September 2002 | June 2008 |
| 42 | HiD | November 1999 | September 2001 | August 2001 | September 2004 | October 2009 |
| 43 | LoD | October 1997 | August 1998 | August 1999 | October 2002 | June 2008 |
| 44 | LoD | November 1999 | July 2001 | August 2001 | August 2004 | August 2009 |
| 45 | LoD | November 1999 | October 2001 | October 2001 | October 2004 | October 2009 |
| 46 | LoD | October 2000 | June 2002 | June 2002 | 4 | 4 |
| 47 | HiD | November 1999 | July 2002 | July 2002 | August 2004 | August 2009 |
| 48 | HiD | October 2000 | July 2002 | July 2002 | August 2005 | May 2012 |
| 49 | HiD | October 2000 | August 2002 | August 2002 | October 2005 | May 2012 |
3. Results and Discussion
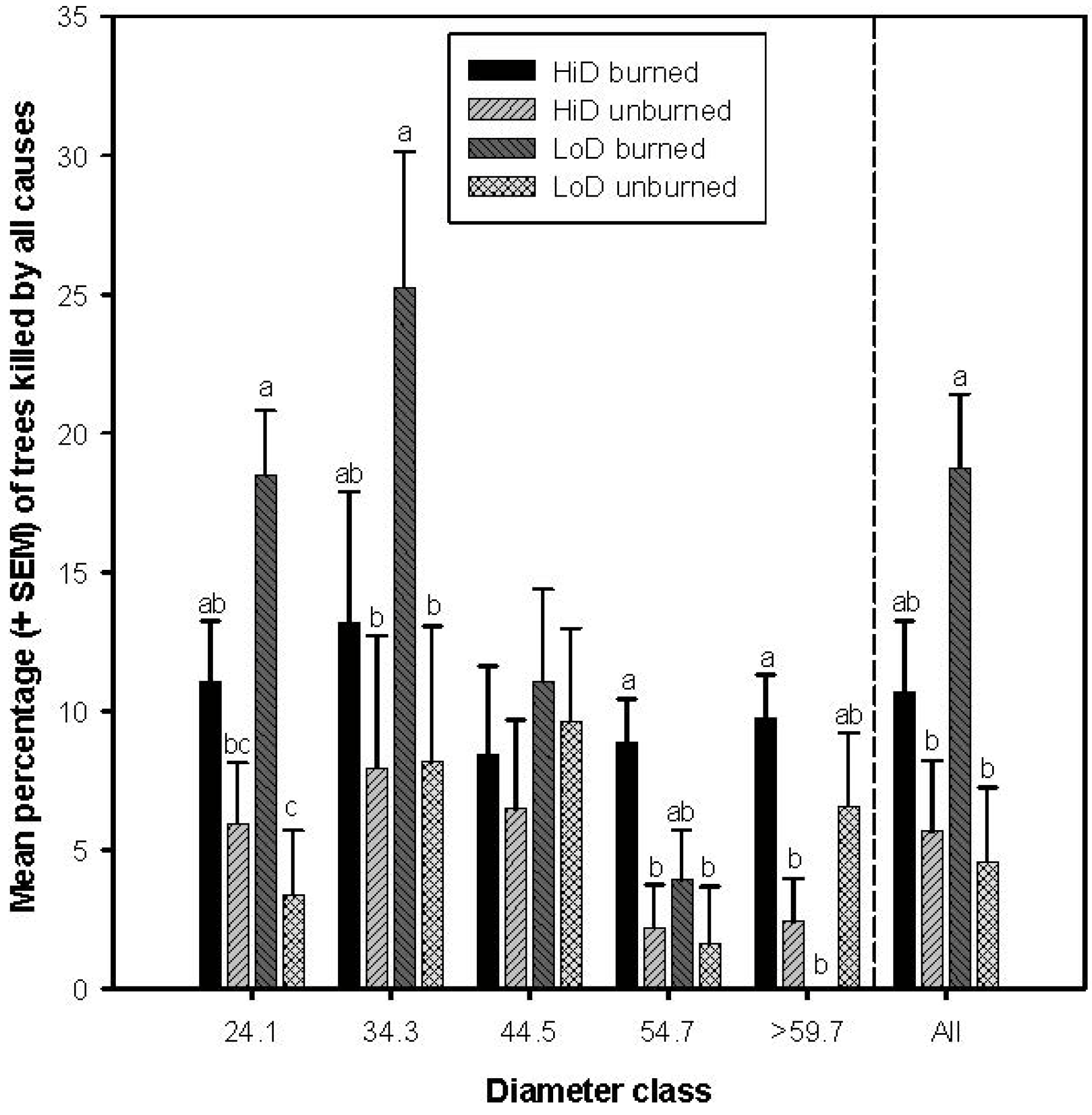
3.1. Tree Mortality
| Treatment | Dbh Class | Dendroctonus brevicomis 1 | Dendroctonus ponderosae | Dendroctonus jeffreyi | Ips spp. | Scolytus ventralis | Total |
|---|---|---|---|---|---|---|---|
| HiD + B | 24.1 | 190 | 448 | 7 | 129 | 1354 | 2,128 |
| 34.3 | 198 | 227 | 11 | 28 | 537 | 1,001 | |
| 44.5 | 102 | 59 | 2 | 1 | 157 | 321 | |
| 54.7 | 66 | 10 | 2 | 2 | 27 | 107 | |
| >59.7 | 250 | 28 | 2 | 1 | 14 | 295 | |
| All | 806 | 772 | 24 | 161 | 2089 | 3,852 | |
| HiD | 24.1 | 119 | 450 | 1 | 2 | 1071 | 1,643 |
| 34.3 | 89 | 175 | 1 | 0 | 626 | 891 | |
| 44.5 | 33 | 37 | 2 | 0 | 200 | 272 | |
| 54.7 | 27 | 10 | 0 | 0 | 29 | 66 | |
| >59.7 | 100 | 13 | 0 | 0 | 17 | 130 | |
| All | 368 | 685 | 4 | 2 | 1943 | 3,002 | |
| LoD + B | 24.1 | 65 | 221 | 5 | 252 | 1151 | 1,694 |
| 34.3 | 86 | 104 | 3 | 60 | 549 | 802 | |
| 44.5 | 18 | 13 | 1 | 2 | 66 | 100 | |
| 54.7 | 0 | 4 | 0 | 0 | * | 4 | |
| >59.7 | * | * | 0 | * | 0 | 0 | |
| All | 169 | 342 | 9 | 314 | 1766 | 2,600 | |
| LoD | 24.1 | 12 | 68 | 4 | 6 | 586 | 676 |
| 34.3 | 27 | 30 | 1 | 1 | 381 | 440 | |
| 44.5 | 11 | 3 | 0 | 0 | 67 | 81 | |
| 54.7 | 1 | 0 | 0 | 0 | * | 1 | |
| >59.7 | 0 | 2 | 0 | 0 | 1 | 3 | |
| All | 51 | 103 | 5 | 7 | 1035 | 1,201 | |
| Total | 1394 | 1902 | 42 | 484 | 6833 | 10,655 |
3.2. Tree Mortality Attributed to Bark Beetles
| Sample Period | Interval 1 | Dendroctonus brevicomis | Dendroctonus ponderosae | Dendroctonus jeffreyi | Ips spp. | Scolytus ventralis | Total |
|---|---|---|---|---|---|---|---|
| 1 | 1–2 | 442 | 468 | 18 | 456 2 | 1300 | 2,684 |
| 2 | 3–5 | 747 | 947 | 17 | 15 | 2467 | 4,193 |
| 3 | 6–10 | 205 | 487 | 7 | 13 | 3066 | 3,778 |
| Total | 1394 | 1902 | 42 | 484 | 6833 | 10,655 |
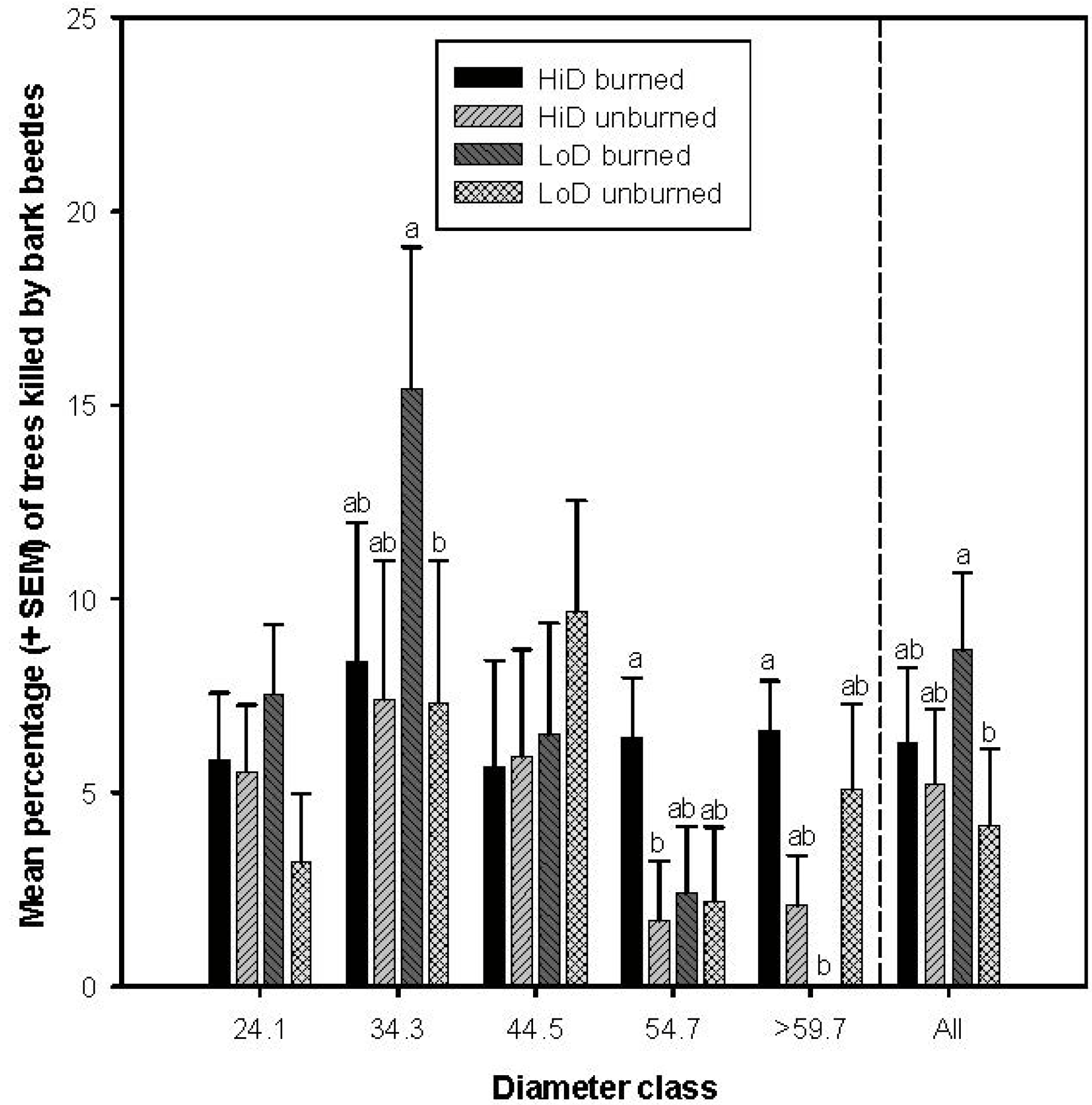
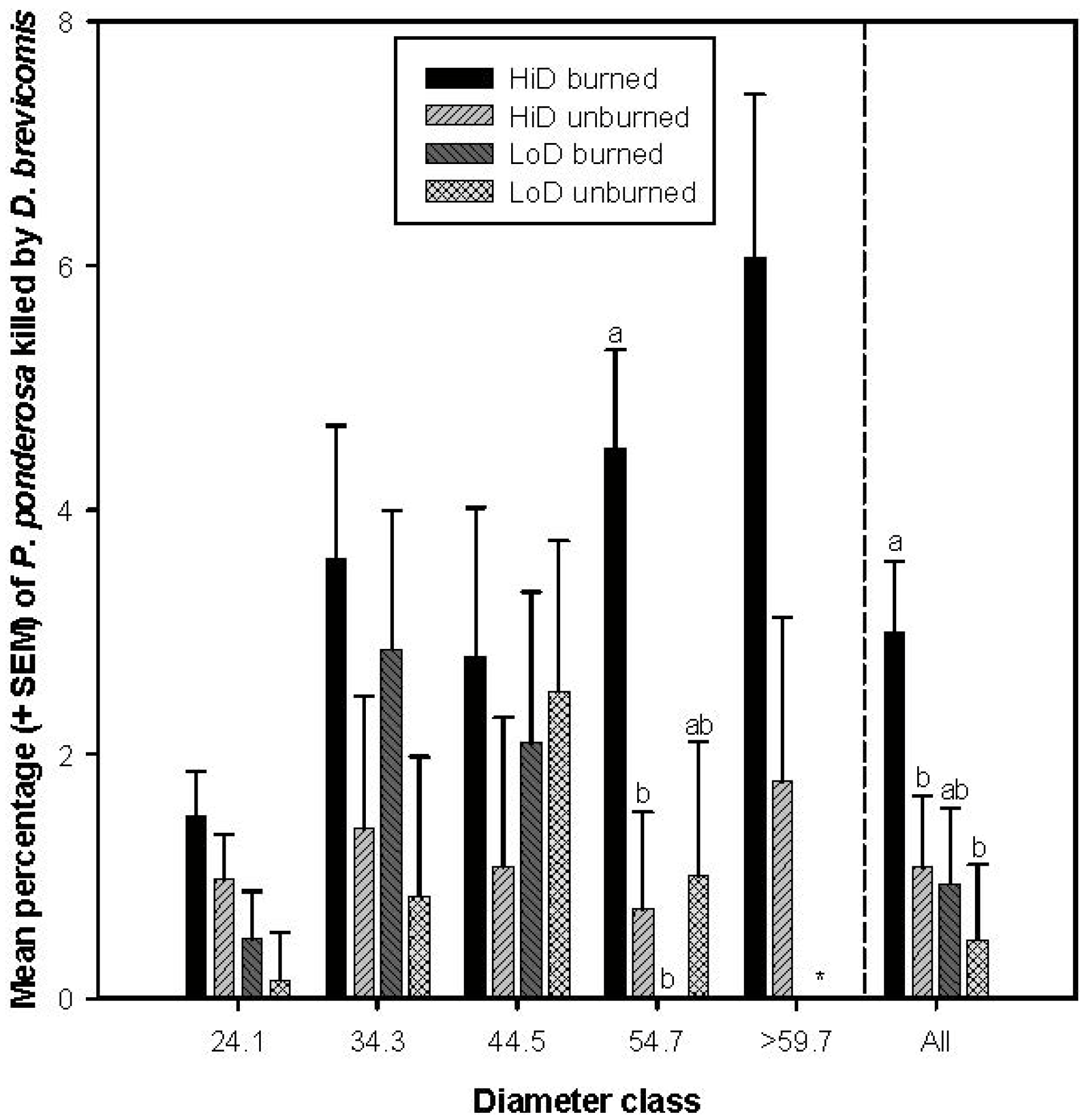
3.2.1. Tree Mortality Attributed to Dendroctonus brevicomis
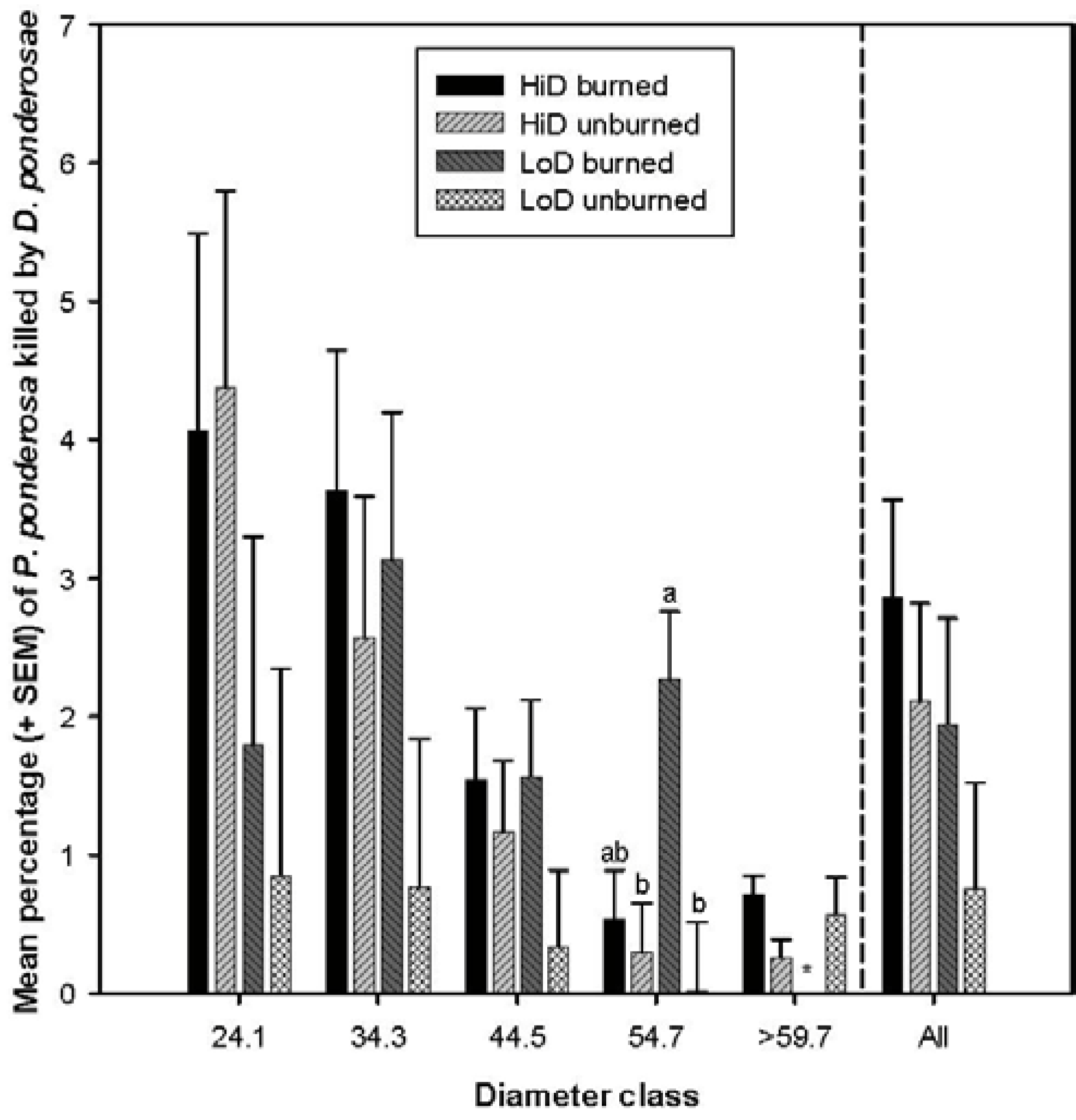
3.2.2. Tree Mortality Attributed to Dendroctonus ponderosae
3.2.3. Tree Mortality Attributed to Dendroctonus jeffreyi
3.2.4. Tree Mortality Attributed to Ips spp.
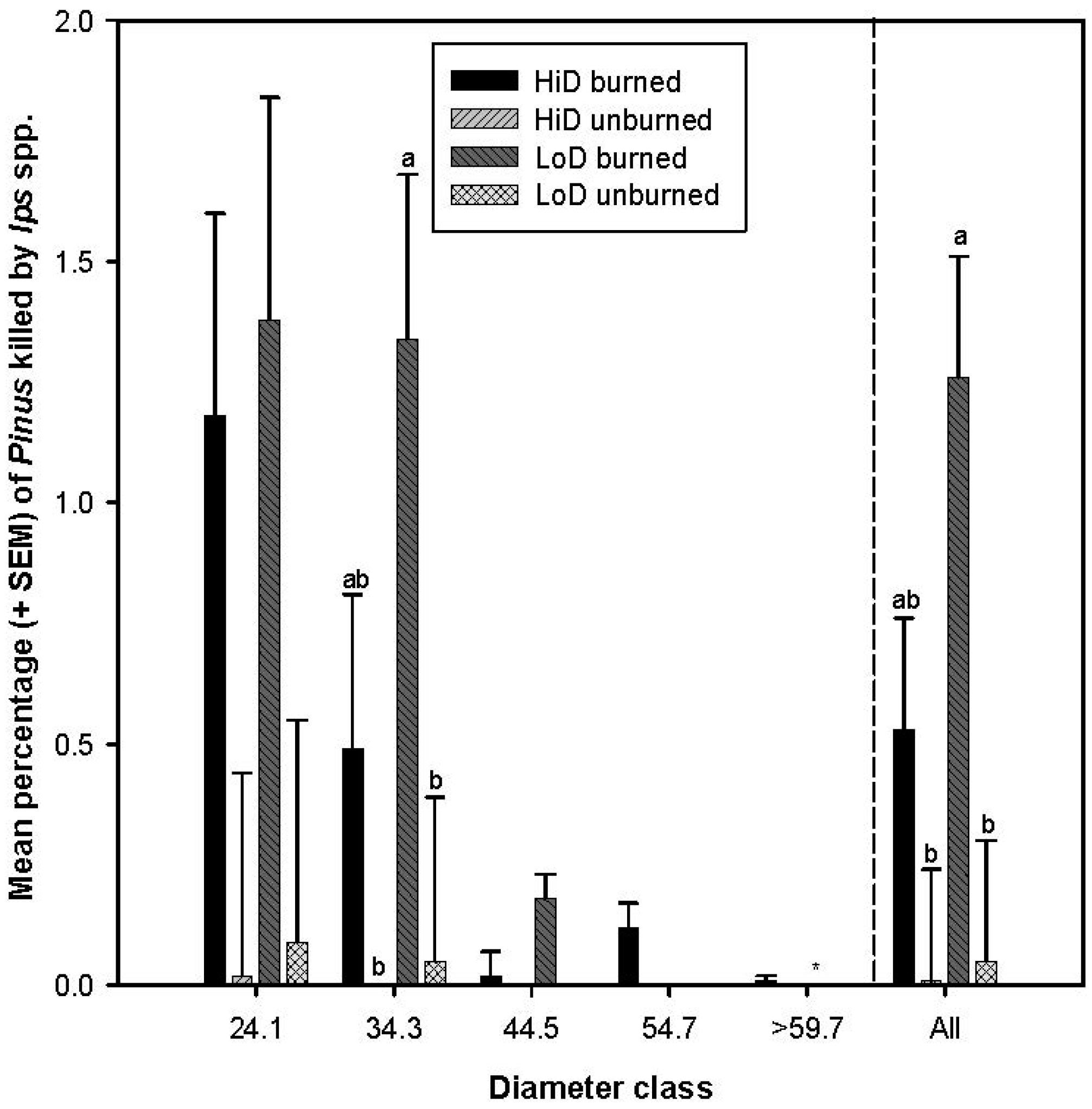
3.2.5. Tree Mortality Attributed to Scolytus ventralis
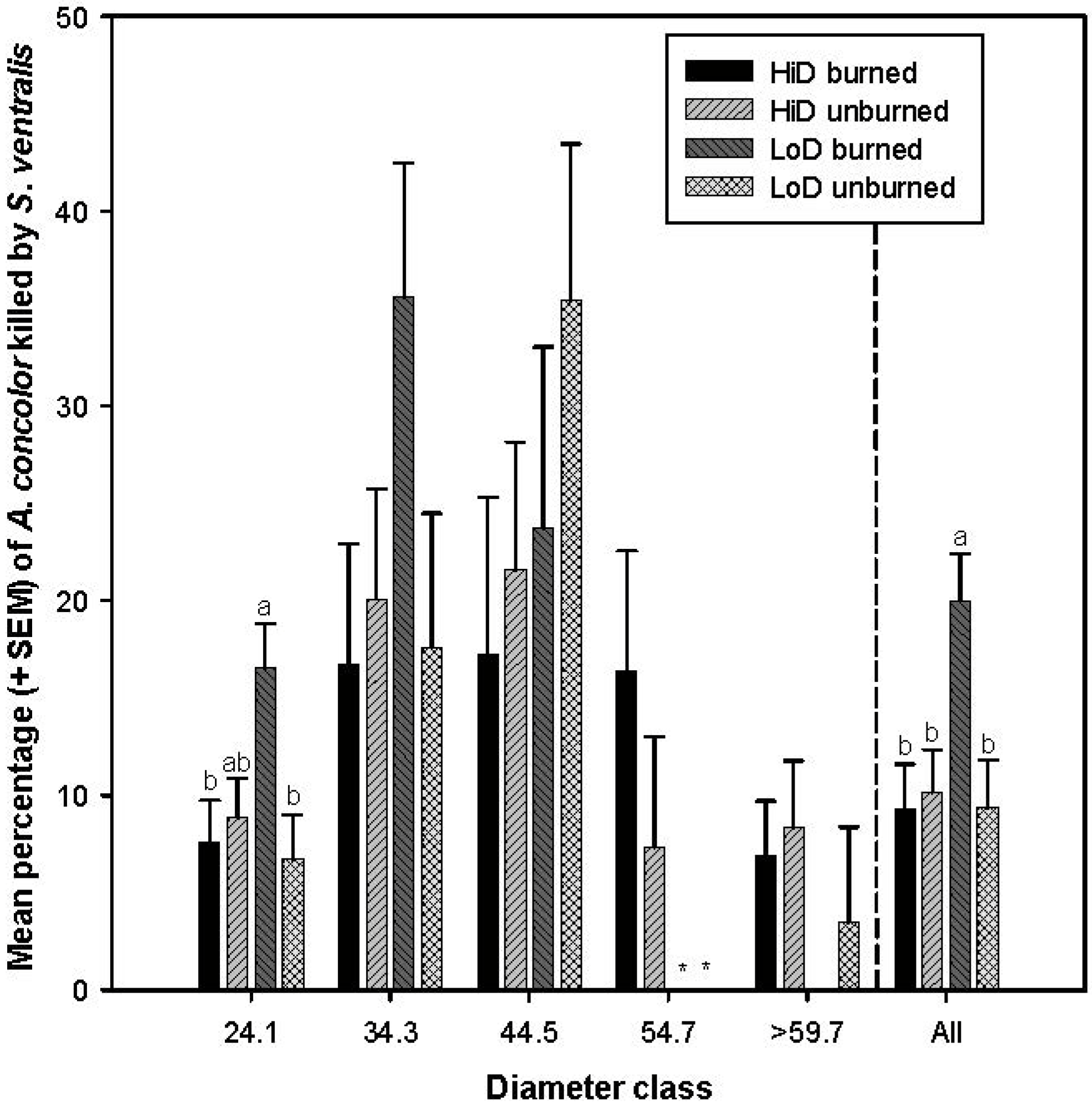
3.3. Tree Mortality Attributed to Wood Borers
3.4. Tree Mortality and Stand Density
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Stephens, S.L.; Ruth, L.W. Federal forest-fire policy in the United States. Ecol. Appl. 2005, 15, 532–542. [Google Scholar] [CrossRef]
- Arno, S.F. Forest fire history of the northern Rockies. J. For. 1980, 78, 460–465. [Google Scholar]
- Swetnam, T.W. Fire History and Climate in the Southwestern United States. In Effects of Fire Management on Southwestern Natural Resources. Proceedings of the Symposium; Krammes, J.S., Ed.; US Department of Agriculture, Forest Service, Rocky Mountain Research Station: Fort Collins, CO, USA, 1990; pp. 6–17. [Google Scholar]
- Agee, J.K. Fire history along an elevational gradient in the Siskiyou Mountains, Oregon. Northwest Sci. 1993, 65, 188–199. [Google Scholar]
- Covington, W.W.; Moore, M. Southwestern ponderosa pine forest structure: Changes since Euro-American settlement. J. For. 1994, 92, 39–47. [Google Scholar]
- Skinner, C.N.; Chang, C. Fire Regimes, Past and Present. In Sierra Nevada Ecosystem Project: Final Report to Congress. Volume II: Assessments and Scientific Basis for Management Options; Water Resources Center Report No. 37; University of California: Davis, CA, USA, 1996; pp. 1041–1069. [Google Scholar]
- Taylor, A.H. Fire regimes and forest changes along a montane forest gradient, Lassen Volcanic National Park, southern Cascade Mountains, USA. J. Biogeogr. 2000, 27, 87–104. [Google Scholar] [CrossRef]
- Taylor, A.H.; Skinner, C.N. Fire history and landscape dynamics in a late-successional reserve, Klamath Mountains, California, USA. For. Ecol. Manag. 1998, 111, 285–301. [Google Scholar] [CrossRef]
- Swetnam, T.W.; Baisan, C.H. Tree-Ring Reconstructions of Fire and Climate History in the Sierra Nevada and Southwestern United States. In Fire and Climatic Change in Temperate Ecosystems of the Western Americas; Veblen, T.T., Baker, W., Montenegro, G., Swetnam, T.W., Eds.; Springer-Verlag: New York, NY, USA, 2002; pp. 154–191. [Google Scholar]
- Stephens, S.L.; Skinner, C.N.; Gill, S.J. Dendrochronology-based fire history of Jeffrey pine-mixed conifer forests in the Sierra San Pedro Martir, Mexico. Can. J. For. Res. 2003, 33, 1090–1101. [Google Scholar] [CrossRef]
- Burns, R.M.; Honkala, B.H. Silvics of North. America. Vol. I. Conifers; US Department of Agriculture, Forest Service: Washington, DC, USA, 1990; p. 675.
- Oliver, W.W. Ecological Research at the Blacks Mountain Experimental Forest in Northeastern California; US Department of Agriculture, Forest Service, Pacific Southwest Research Station: Albany, CA, USA, 2000; p. 66.
- Stephens, S.L.; McIver, J.D.; Boerner, R.E.J.; Fettig, C.J.; Fontaine, J.B.; Hartsough, B.R.; Kennedy, P.L.; Schwilk, D.W. The effects of forest fuel-reduction treatments in the United States. Bioscience 2012, 62, 549–560. [Google Scholar] [CrossRef]
- McIver, J.D.; Stephens, S.L.; Agee, J.K.; Barbour, J.; Boerner, R.E.J.; Edminster, C.B.; Erickson, K.L.; Farris, K.L.; Fettig, C.J.; Fiedler, C.E.; et al. Ecological effects of alternative fuel reduction treatments:Highlights of the national Fire and Fire Surrogate study (FFS). Int. J. Wildland Fire 2013, 22, 63–82. [Google Scholar] [CrossRef]
- Stephens, S.L.; Moghaddas, J.J.; Edminster, C.; Fiedler, C.E.; Haase, S.; Harrington, M.; Keeley, J.E.; Knapp, E.E.; McIver, J.D.; Metlen, K.; et al. Fire treatment effects on vegetation structure, fuels, and potential fire severity in western U.S. forests. Ecol. Appl. 2009, 19, 305–320. [Google Scholar] [CrossRef]
- Ritchie, M.W.; Skinner, C.N.; Hamilton, T.A. Probability of tree survival after wildfire in an interior pine forest of northern California: Effects of thinning and prescribed fire. For. Ecol. Manag. 2007, 247, 200–208. [Google Scholar] [CrossRef]
- Hood, S.M.; Smith, S.L.; Cluck, D.R. Delayed Conifer Tree Mortality Following Fire in California. In Restoring Fire-Adapted Ecosystems: Proceedings of the 2005 National Silviculture Workshop; U.S. Department of Agriculture, Forest Service, Pacific Southwest Research Station: Albany, CA, USA, 2007; pp. 261–283. [Google Scholar]
- Parker, T.J.; Clancy, K.M.; Mathiasen, R.L. Interactions among fire, insects and pathogens in coniferous forests of the interior western United States and Canada. Agric. For. Entomol. 2006, 8, 167–189. [Google Scholar] [CrossRef]
- Rasmussen, L.A.; Amman, G.D.; Vandygriff, J.C.; Oakes, R.D.; Munson, A.S.; Gibson, K.E. Bark Beetle and Wood Borer Infestations in the Greater Yellowstone Area During Four Postfire Years; US Department of Agriculture, Forest Service, Intermountain Research Station: Ogden, UT, USA, 2000; p. 66.
- Aronson, G.; Kulakowski, D. Bark beetle outbreaks, wildfires and defensible space: How much area do we need to treat to protect homes and communities? Int. J. Wildland Fire 2012, 22, 256–265. [Google Scholar] [CrossRef]
- Fettig, C.J.; Klepzig, K.D.; Billings, R.F.; Munson, A.S.; Nebeker, T.E.; Negrón, J.F.; Nowak, J.T. The effectiveness of vegetation management practices for prevention and control of bark beetle outbreaks in coniferous forests of the western and southern United States. For. Ecol. Manag. 2007, 238, 24–53. [Google Scholar] [CrossRef]
- Schwilk, D.W.; Keeley, J.E.; Knapp, E.E.; McIver, J.; Bailey, J.D.; Fettig, C.J.; Fielder, C.E.; Harrod, R.J.; Moghaddas, J.J.; Outcalt, K.W.; et al. The National Fire and Fire Surrogate Study: Effects of alternative fuel reduction methods on forest structure. Ecol. Appl. 2009, 19, 285–304. [Google Scholar] [CrossRef]
- Seybold, S.J.; Huber, D.P.W.; Lee, J.C.; Graves, A.D.; Bohlmann, J. Pine monoterpenes and pine bark beetles: A marriage of convenience for defense and chemical communication. Phytochem. Rev. 2006, 5, 143–178. [Google Scholar] [CrossRef]
- Fettig, C.J.; McMillin, J.D.; Anhold, J.A.; Hamud, S.M.; Borys, R.R.; Dabney, C.P.; Seybold, S.J. The effects of mechanical fuel reduction treatments on the activity of bark beetles (Coleoptera: Scolytidae) infesting ponderosa pine. For. Ecol. Manag. 2006, 230, 55–68. [Google Scholar] [CrossRef]
- Davis, R.S.; Hood, S.; Bentz, B.J. Fire-injured ponderosa pine provide a pulsed resource for bark beetles. Can. J. For. Res. 2012, 42, 2022–2036. [Google Scholar] [CrossRef]
- DeGomez, T.; Fettig, C.J.; McMillin, J.D.; Anhold, J.A.; Hayes, C.J. Managing Slash to Minimize Colonization of Residual Leave Trees by Ips and Other Bark Beetle Species Following Thinning in Southwestern Ponderosa Pine; University of Arizona: Tucson, AZ, USA, 2008; p. 12. [Google Scholar]
- Bartos, D.L.; Amman, G.D. Microclimate: An Alternative to Tree Vigor as a Basis for Mountain Pine Beetle Infestations; U.S. Department of Agriculture, Forest Service, Intermountain Research Station: Ogden, UT, USA, 1989; p. 10.
- Thistle, H.W.; Peterson, H.; Allwine, G.; Lamb, B.K.; Strand, T.; Holsten, E.H.; Shea, P.J. Surrogate pheromone plumes in three forest trunk spaces: Composite statistics and case studies. For. Sci. 2004, 50, 610–625. [Google Scholar]
- Hayes, C.J.; Fettig, C.J.; Merrill, L.D. Evaluation of multiple funnel traps and stand characteristics for estimating western pine beetle-caused tree mortality. J. Econ. Entomol. 2009, 102, 2170–2182. [Google Scholar] [CrossRef]
- Bradley, T.; Tueller, P. Effects of fire on bark beetle presence on Jeffrey pine in the Lake Tahoe Basin. For. Ecol. Manag. 2001, 142, 205–214. [Google Scholar] [CrossRef]
- Ganz, D.J.; Dahlsten, D.L.; Shea, P.J. The Post-Burning Responses of Bark Beetles to Prescribed Burning Treatments. In Fire, Fuel Treatments, and Ecological Restoration: Conference. Proceedings; Omi, P.N., Joyce, L.A., Eds.; US Department of Agriculture, Forest Service, Rocky Mountain Research Station: Fort Collins, CO, USA, 2003; pp. 143–158. [Google Scholar]
- McHugh, C.W.; Kolb, T.E. Ponderosa pine mortality following fire in northern Arizona. Int. J. Wildland Fire 2003, 12, 7–22. [Google Scholar] [CrossRef]
- McHugh, C.W.; Kolb, T.E.; Wilson, J.L. Bark beetle attacks on ponderosa pine following fire in northern Arizona. Environ. Entomol. 2003, 32, 510–522. [Google Scholar] [CrossRef]
- Schwilk, D.W.; Knapp, E.E.; Ferrenberg, S.M.; Keeley, J.E.; Caprio, A.C. Tree mortality from fire and bark beetles following early and late season prescribed fires in a Sierra Nevada mixed-conifer forest. For. Ecol. Manag. 2006, 232, 36–45. [Google Scholar] [CrossRef]
- Breece, C.R.; Kolb, T.E.; Dickson, B.G.; McMillin, J.D.; Clancy, K.M. Prescribed fire effects on bark beetle activity and tree mortality in southwestern ponderosa pine forests. For. Ecol. Manag. 2008, 255, 119–128. [Google Scholar] [CrossRef]
- Fettig, C.J.; Borys, R.R.; McKelvey, S.R.; Dabney, C.P. Blacks Mountain Experimental Forest: Bark beetle responses to differences in forest structure and the application of prescribed fire in interior ponderosa pine. Can. J. For. Res. 2008, 38, 924–935. [Google Scholar] [CrossRef]
- Fettig, C.J.; McKelvey, S.R. Bark beetle responses to stand structure and prescribed fire at Blacks Mountain Experimental Forest, California, USA: 5-year data. Fire Ecol. 2010, 6, 26–42. [Google Scholar] [CrossRef]
- Fettig, C.J.; Borys, R.R.; Dabney, C.P. Effects of fire and fire surrogate treatments on bark beetle-caused tree mortality in the Southern Cascades, California. For. Sci. 2010, 56, 60–73. [Google Scholar]
- Fettig, C.J.; McKelvey, S.R.; Cluck, D.R.; Smith, S.L.; Otrosina, W.J. Effects of prescribed fire and season of burn on direct and indirect levels of tree mortality in ponderosa and Jeffrey pine forests in California, USA. For. Ecol. Manag. 2010, 260, 207–218. [Google Scholar] [CrossRef]
- Maloney, P.E.; Smith, T.F.; Jensen, C.E.; Innes, J.; Rizzo, D.M.; North, M.P. Initial tree mortality and insect and pathogen response to fire and thinning restoration treatments in an old-growth mixed-conifer forest of the Sierra Nevada, California. Can. J. For. Res. 2008, 38, 3011–3020. [Google Scholar] [CrossRef]
- Stark, D.T.; Wood, D.L.; Storer, A.J.; Stephens, S.L. Prescribed fire and mechanical thinning effects on bark beetle caused tree mortality in a mid-elevation Sierran mixed-conifer forest. For. Ecol. Manag. 2013, 306, 61–67. [Google Scholar] [CrossRef]
- Salman, K.A.; Bongberg, J.W. Logging high-risk trees to control insects in the pines stands of northeastern California. J. For. 1942, 40, 533–539. [Google Scholar]
- Zhang, J.; Ritchie, M.W.; Oliver, W.W. Vegetation responses to stand structure and prescribed fire in an interior ponderosa pine ecosystem. Can. J. For. Res. 2008, 38, 909–918. [Google Scholar] [CrossRef]
- Furniss, R.L.; Carolin, V.M. Western Forest Insects; US Department of Agriculture, Forest Service: Washington, DC, USA; p. 654.
- Fischer, W.C. Prescribed fire and bark beetle attack in ponderosa pine forests. Fire Manag. Notes 1980, 41, 10–12. [Google Scholar]
- Kegley, S.J.; Livingston, R.L.; Gibson, K.E. Pine Engraver, Ips pini (Say), in the United States; US Department of Agriculture, Forest Service: Portland, OR, USA, 1997; p. 8.
- Agee, J.K.; Skinner, C.N. Basic principles of forest fuel reduction treatments. For. Ecol. Manag. 2005, 211, 83–96. [Google Scholar] [CrossRef]
- Regelbrugge, J.C.; Conard, S.G. Modeling tree mortality following wildfire in Pinus ponderosa forests in the central Sierra Nevada of California. Int. J. Wildland Fire 1993, 3, 139–148. [Google Scholar] [CrossRef]
- Fulé, P.Z.; Covington, W.W.; Smith, H.B.; Springer, J.D.; Heinlein, T.A.; Huisinga, K.D.; Moore, M.M. Comparing ecological restoration alternatives: Grand Canyon, Arizona. For. Ecol. Manag. 2002, 170, 19–41. [Google Scholar] [CrossRef]
- Kolb, T.E.; Agee, J.K.; Fulé, P.Z.; McDowell, N.G.; Pearson, K.; Sala, A.; Waring, R.H. Perpetuating old growth ponderosa pine. For. Ecol. Manag. 2007, 249, 141–157. [Google Scholar] [CrossRef]
- Ritchie, M.W.; Wing, B.M.; Hamilton, T.A. Stability of the large tree component in treated and untreated late-seral interior ponderosa pine stands. Can. J. For. Res. 2008, 38, 919–923. [Google Scholar] [CrossRef]
- DeMars, C.J.; Roettgering, B.H. Western Pine Beetle; US Department of Agriculture, Forest Service: Portland, OR, USA, 1982; p. 8.
- Walker, R.; Rosenberg, M.; Warbington, R.; Schwind, B.; Beardsley, D.; Ramirez, C.; Fischer, L.; Frerichs, B. Inventory of Tree Mortality in Southern California Mountains (2001–2004) due to Bark Beetle Impacts. Available online: http://www.frap.fire.ca.gov/projects/mast/reports.html (accessed on 11 November 2013).
- Sánchez-Martínez, G.; Wagner, M.R. Bark beetle community structure under four ponderosa pine forest stand conditions in northern Arizona. For. Ecol. Manag. 2002, 170, 145–160. [Google Scholar] [CrossRef]
- Fettig, C.J.; McKelvey, S.R.; Dabney, C.P.; Huber, D.P.W.; Lait, C.G.; Fowler, D.L.; Borden, J.H. Efficacy of “Verbenone Plus” for protecting ponderosa pine trees and stands from Dendroctonus brevicomis (Coleoptera: Curculionidae) attack in British Columbia and California. J. Econ. Entomol. 2012, 105, 1668–1680. [Google Scholar] [CrossRef]
- Fettig, C.J.; Grosman, D.M.; Munson, A.S. Advances in Insecticide Tools and Tactics for Protecting Conifers from Bark Beetle Attack in the Western United States. In Insecticides—Development of Safer and More Effective Technologies; Trdan, S., Ed.; InTech: Rijeka, Croatia, 2013; pp. 472–492. [Google Scholar]
- Gibson, K.; Kegley, S.; Bentz, B. Mountain Pine Beetle; US Department of Agriculture, Forest Service: Portland, OR, USA, 2009; p. 12.
- Jenkins, M.J.; Runyon, J.B.; Fettig, C.J.; Page, W.G.; Bentz, B.J. Interactions among the mountain pine beetle, fires, and fuels. For. Sci. 2014. [Google Scholar] [CrossRef]
- Smith, S.L.; Borys, R.R.; Shea, P.J. Jeffrey Pine Beetle; US Department of Agriculture, Forest Service: Portland, OR, USA, 2009; p. 8.
- Kolb, T.E.; Guerard, N.; Hofstetter, R.W.; Wagner, M.R. Attack preference of Ips pini on Pinus ponderosa in northern Arizona: Tree size and bole position. Agric. For. Entomol. 2006, 8, 295–303. [Google Scholar] [CrossRef]
- Fiddler, G.O.; Hart, D.R.; Fiddler, T.A.; McDonald, P.M. Thinning Decreases Mortality and Increases Growth of Ponderosa Pine in Northeastern California; US Department of Agriculture, Forest Service, Pacific Southwest Research Station: Albany, CA, USA, 1989; p. 12.
- Oliver, W.W. Fifteen-Year Growth Patterns after Thinning a Ponderosa Pine-Jeffrey Pine Plantation in Northeastern California; US Department of Agriculture, Forest Service, Pacific Southwest Research Station: Albany, CA, USA, 1979; p. 10.
- Oliver, W.W. Is Self-Thinning in Ponderosa Pine Ruled by Dendroctonus Bark Beetles? In Forest Health Through Silviculture: Proceedings of the 1995 National Silviculture Workshop; Eskew, L.G., Ed.; US Department of Agriculture, Forest Service, Rocky Mountain Research Station: Fort Collins, CO, USA, 1995; pp. 213–218. [Google Scholar]
- Holling, C.S. Resilience and stability of ecosystems. Annu. Rev. Ecol. Syst. 1973, 4, 1–23. [Google Scholar]
- Progar, R.A.; Gillette, N.; Fettig, C.J.; Hrinkevich, K. Applied chemical ecology of the mountain pine beetle. For. Sci. 2014. [Google Scholar] [CrossRef]
- Perrakis, D.B.; Agee, J.K.; Eglitis, A. Effects of prescribed burning on mortality and resin defenses in old growth ponderosa pine (Crater Lake, Oregon): Four years of post-fire monitoring. Nat. Areas J. 2011, 31, 14–25. [Google Scholar] [CrossRef]
- Fowler, J.F.; Sieg, C.H.; Wadleigh, L.L. Effectiveness of litter removal to prevent cambial kill-caused mortality in northern Arizona ponderosa pine. For. Sci. 2010, 56, 166–171. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Fettig, C.J.; McKelvey, S.R. Resiliency of an Interior Ponderosa Pine Forest to Bark Beetle Infestations Following Fuel-Reduction and Forest-Restoration Treatments. Forests 2014, 5, 153-176. https://doi.org/10.3390/f5010153
Fettig CJ, McKelvey SR. Resiliency of an Interior Ponderosa Pine Forest to Bark Beetle Infestations Following Fuel-Reduction and Forest-Restoration Treatments. Forests. 2014; 5(1):153-176. https://doi.org/10.3390/f5010153
Chicago/Turabian StyleFettig, Christopher J., and Stephen R. McKelvey. 2014. "Resiliency of an Interior Ponderosa Pine Forest to Bark Beetle Infestations Following Fuel-Reduction and Forest-Restoration Treatments" Forests 5, no. 1: 153-176. https://doi.org/10.3390/f5010153




