Ecosystem Responses to Partial Harvesting in Eastern Boreal Mixedwood Stands
Abstract
:1. Introduction
2. Experimental Section
2.1. Study Area
2.2. Experimental Design and Treatment Description

2.3. Field Methods
2.4. Statistical Analyses
3. Results and Discussion
3.1. Initial Stand Composition and Regeneration
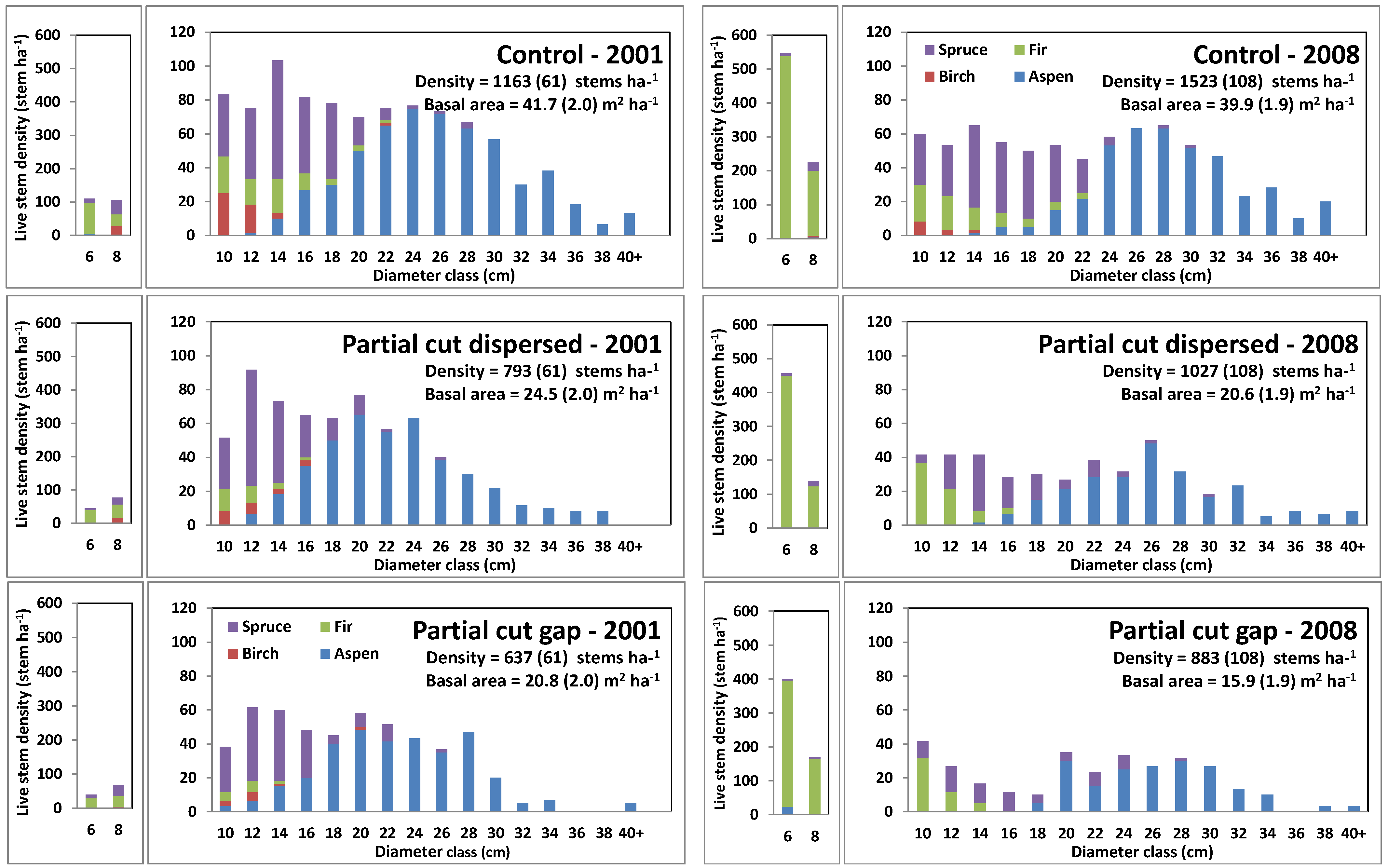
3.2. Harvesting Effects on Stand Characteristics
| Fixed Factors | Time | C vs. PC | CC vs. PC | PCD vs. PCG | Time × Treatment |
|---|---|---|---|---|---|
| Live stems (trees >5 cm dbh) | |||||
| Density | *** | *** | NA | NS | NS |
| Basal area | *** | *** | NA | NS | NS |
| Snag density (snags >5 cm dbh) | |||||
| Aspen snags | * | NS | *** | * | *** |
| Softwood snags | *** | NS | * | NS | * |
| Snags >20 cm | *** | NS | *** | NS | * |
| Downed log volume | |||||
| Fresh logs | NS | NS | NS | NS | *** |
| Well decayed logs | * | NS | * | NS | NS |
| Logs >20 cm | NS | NS | NS | NS | NS |
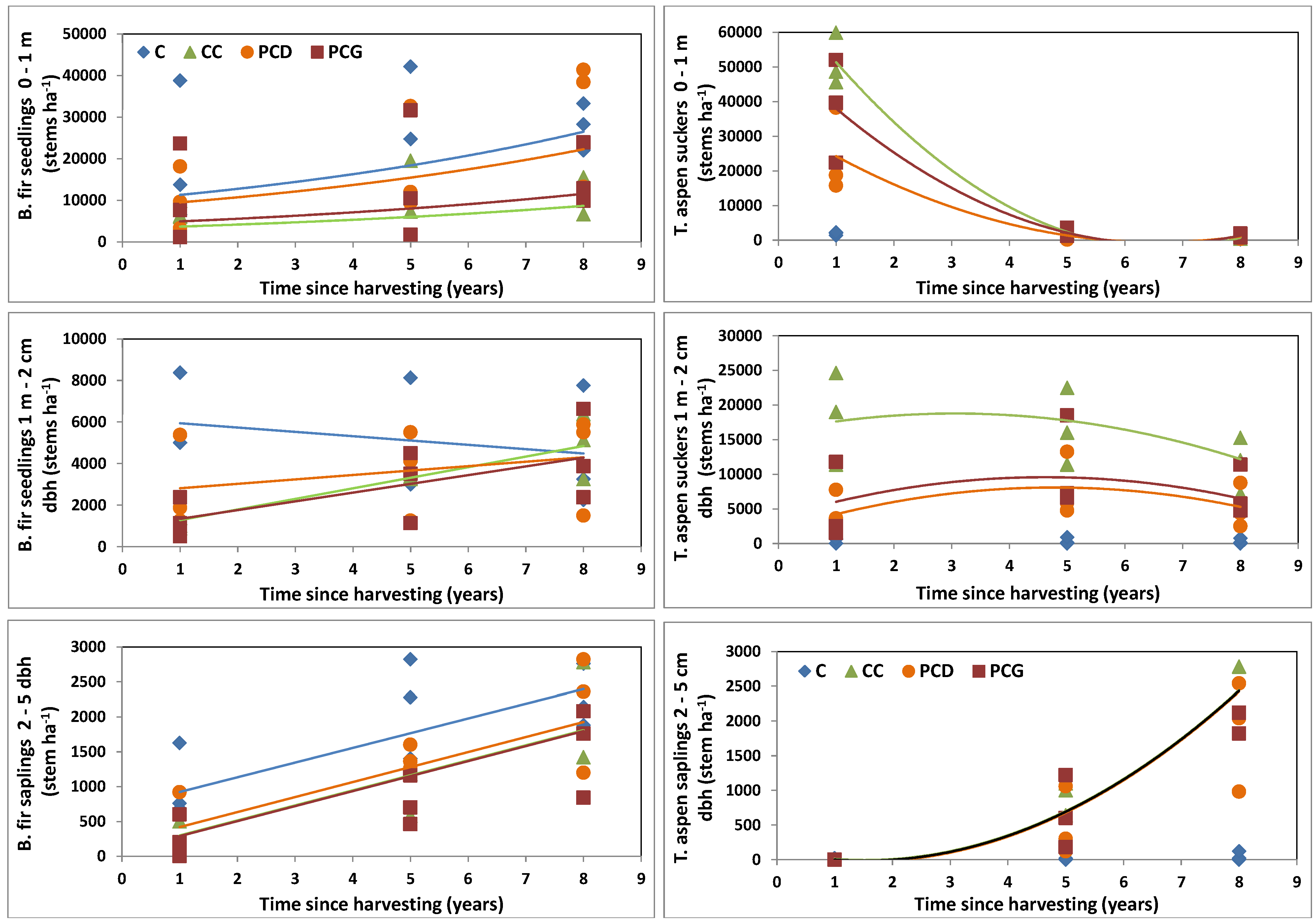
3.3. Balsam Fir Regeneration Response to Harvesting
| Factors | Time | Time2 | C vs. PC | CC vs. PC | PCD vs. PCG | Time × Treatment | Time2 × Treatment |
|---|---|---|---|---|---|---|---|
| Balsam fir regeneration density | |||||||
| Seedlings <1 m (log) | ** | NS | * | ** | * | NS | NS |
| Seedlings 1 m–2 cm dbh | *** | NS | *** | NS | NS | *** | NS |
| Saplings 2–5 cm dbh | *** | NS | ** | NS | NS | NS | NS |
| Balsam fir small sapling growth | |||||||
| Annual basal radial growth | *** | *** | NS | *** | NS | *** | *** |
| Annual height growth | *** | ** | NS | NS | NS | *** | * |
| Total height | *** | *** | * | NS | NS | ** | *** |
| Trembling aspen regeneration density | |||||||
| Seedlings <1 m | *** | *** | NA | ** | * | *** | ** |
| Seedlings 1 m–2 cm dbh | NS | *** | NA | *** | NS | * | NS |
| Saplings 2–5 cm dbh | *** | *** | NA | NS | NS | NS | NS |
| Trembling aspen small sapling growth | |||||||
| Annual basal radial growth | *** | *** | NA | NS | NS | NS | NS |

3.4. Trembling Aspen Regeneration Response to Harvesting
3.5. Changes in Stand Diameter Distribution in Response to Harvesting
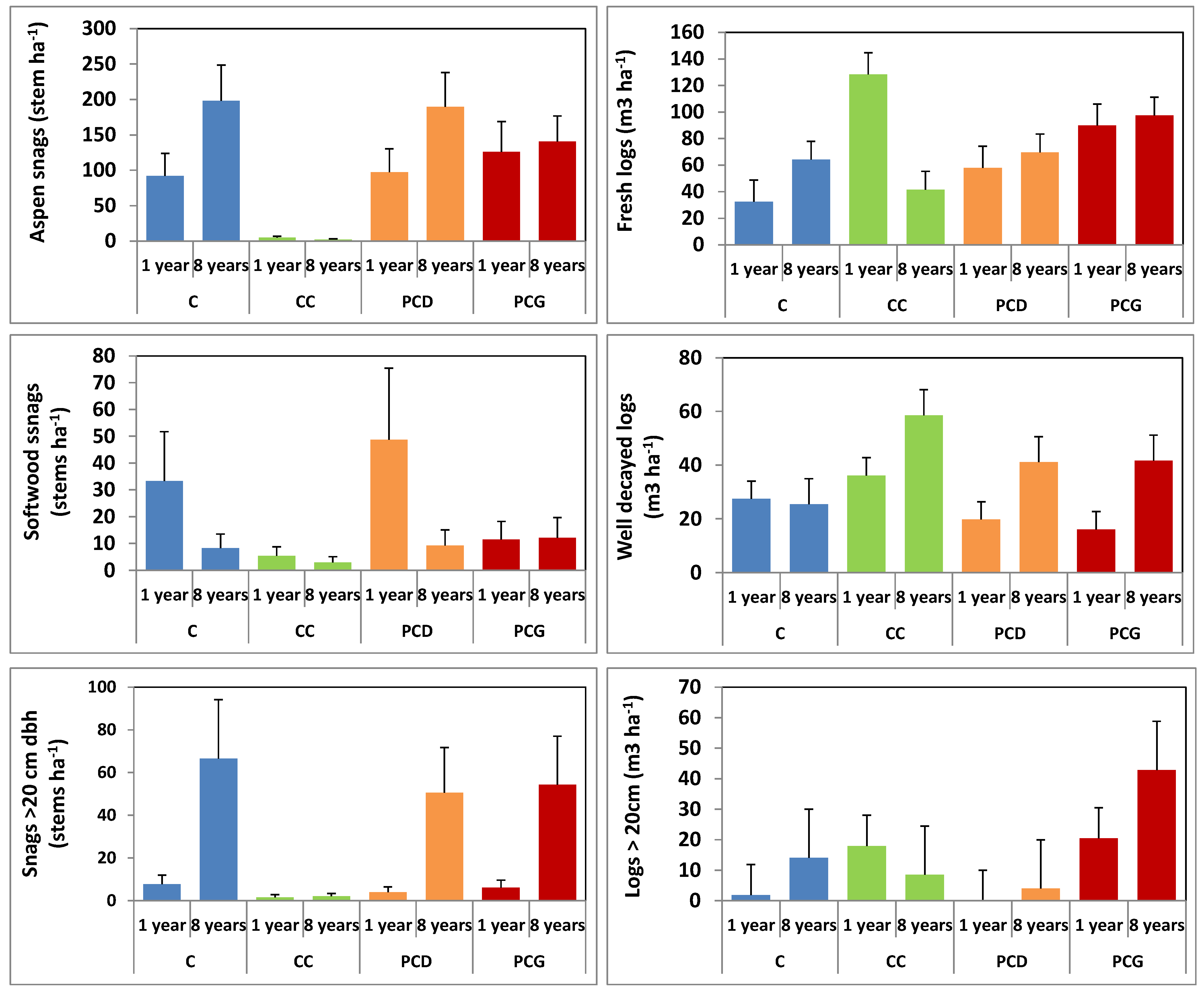
3.6. Recruitment and Volume of Deadwood Following Partial and Clear Cut Harvesting
3.7. Response of Ground Beetles to Harvesting
| Split-1 | Split-2 | Split-3 | Split-4 | Split-5 | Tree Total | Species Total | |
|---|---|---|---|---|---|---|---|
| Agonum affine Kirby | 0.020 | 0 | 0 | 0.030 | 0.030 | 0.080 | 0.684 |
| Agonum cupripenne (Say) | 0.005 | 0 | 0.015 | 0.030 | 0 | 0.050 | 0.352 |
| Agonum retractum LeConte | 0.100 | 0.001 | 0.050 | 0.005 | 0.063 | 0.218 | 1.342 |
| Agonum tenue (LeConte) | 0.005 | 0 | 0.015 | 0 | 0.030 | 0.050 | 0.352 |
| Amara lunicollis Schiodte | 0.028 | 0.043 | 0 | 0 | 0 | 0.071 | 0.499 |
| Amara patruelis Dejean | 0.035 | 0.052 | 0 | 0 | 0 | 0.088 | 0.613 |
| Badister obtustus LeConte | 0.514 | 0 | 1.543 | 0.008 | 0 | 2.066 | 3.661 |
| Bembidion praticola Lindroth | 0.001 | 0.043 | 0.060 | 0.121 | 0 | 0.225 | 1.135 |
| Bembidion wingatei Bland | 0.254 | 0 | 0.054 | 0.205 | 0.188 | 0.700 | 2.627 |
| Bradycellus lugubris (LeConte) | 0.168 | 0.994 | 0.490 | 0.087 | 0 | 1.738 | 3.507 |
| Calathus ingratus Dejean | 0.645 | 0.201 | 0.716 | 0.069 | 0.026 | 1.657 | 6.374 |
| Carabus maeander Fischer von Waldheim | 0.005 | 0 | 0.015 | 0.030 | 0 | 0.050 | 0.352 |
| Cymindis cribricollis Dejean | 0.247 | 0 | 0.015 | 0.030 | 0.483 | 0.775 | 2.043 |
| Harpalus egregius Casey | 0.046 | 0.121 | 0.015 | 0 | 0.030 | 0.212 | 0.999 |
| Harpalus fulvilabris Mannerheim | 0.020 | 0.030 | 0 | 0 | 0 | 0.050 | 0.354 |
| Harpalus herbivagus Say | 0.001 | 0.354 | 0.371 | 0.134 | 0.948 | 1.808 | 4.152 |
| Notiophilus aeneus (Herbst) | 0.170 | 0 | 0.021 | 0.16 | 0.068 | 0.422 | 2.151 |
| Olisthopus parmatus (Say) | 0.001 | 0.121 | 0.185 | 0.37 | 0 | 0.679 | 1.924 |
| Patrobus foveocollis (Escherholtz) | 0.012 | 0.014 | 0.145 | 0.030 | 0.037 | 0.238 | 3.049 |
| Platynus decentis (Say) | 0.301 | 1.402 | 2.197 | 0.268 | 0.511 | 4.679 | 9.257 |
| Pseudoamara arenaria (LeConte) | 0.623 | 0.935 | 0 | 0 | 0 | 1.558 | 2.605 |
| Pterostichus adstrictus Eschscholtz | 1.909 | 0.906 | 4.57 | 0.35 | 0.690 | 8.425 | 10.462 |
| Pterostichus coracinus (Newman) | 0.686 | 0.256 | 0.001 | 0.18 | 0.150 | 1.27 | 2.827 |
| Pterostichus luctuosus (Dejean) | 0.005 | 0 | 0.015 | 0.030 | 0 | 0.050 | 0.352 |
| Pterostichus melanarius (Illiger) | 0.051 | 0 | 0.154 | 0.020 | 0 | 0.225 | 1.135 |
| Pterostichus pensylvanicus LeConte | 1.564 | 0.319 | 0.045 | 0.051 | 0.195 | 2.174 | 4.184 |
| Pterostichus punctatissimus (Randall) | 0.010 | 0.218 | 1.155 | 0.030 | 0.082 | 1.494 | 5.078 |
| Scaphinotus bilobus (Say) | 0.869 | 0.629 | 0 | 0 | 0 | 1.498 | 2.191 |
| Sphaeroderus nitidicollis (Guérin-Méneville) | 0.031 | 0.053 | 0.033 | 0.068 | 0.211 | 0.396 | 1.524 |
| Sphaeroderus stenostomus (Dejean) | 11.586 | 0 | 0.277 | 0.480 | 0.104 | 12.448 | 13.158 |
| Syntomus americanus (Dejean) | 0.107 | 0 | 0.102 | 0.078 | 0.030 | 0.317 | 1.749 |
| Synuchus impunctatus (Say) | 0.718 | 0.005 | 0.006 | 0.584 | 0.841 | 2.154 | 4.092 |
| Trechus crassiscapus Lindroth | 0.221 | 0.062 | 0.009 | 0.875 | 1.141 | 2.308 | 5.215 |
| Total | 20.958 | 6.760 | 12.278 | 4.325 | 5.856 | 50.176 | 100 |

4. Conclusions
Acknowledgments
Conflict of Interest
References
- Bergeron, Y. Species and stand dynamics in the mixed woods of Quebec’s southern boreal forest. Ecology 2000, 81, 1500–1516. [Google Scholar] [CrossRef]
- Chen, H.Y.H.; Popadiouk, R.V. Dynamics of North American boreal mixedwoods. Environ. Rev. 2002, 10, 137–166. [Google Scholar] [CrossRef]
- Hunter, M.L., Jr. Wildlife, Forests, and Forestry. Principles of Managing Forests for Biological Diversity; Englewood Cliffs: Prentice Hall, NJ, USA, 1990; p. 370. [Google Scholar]
- Bergeron, Y.; Harvey, B.D. Basing silviculture on natural ecosystem dynamics: An approach applied to the southern boreal mixedwood forest of Quebec. For. Ecol. Manag. 1997, 92, 235–242. [Google Scholar] [CrossRef]
- Franklin, J.F.; Spies, T.A.; van Pelt, R.; Carey, A.B.; Thornburgh, D.A.; Berg, D.R.; Lindenmayer, D.B.; Harmon, M.E.; Keeton, W.S.; Chaw, D.C.; et al. Disturbances and structural development of natural forest ecosystems with silvicultural implications, using Douglas-fir forests as an example. For. Ecol. Manag. 2002, 155, 399–423. [Google Scholar] [CrossRef]
- Lieffers, V.J.; Beck, J.A., Jr. A semi-natural approach to mixedwood management in the prairie provinces. For. Chron. 1994, 70, 260–264. [Google Scholar]
- Harvey, B.D.; Leduc, A.; Gauthier, S.; Bergeron, Y. Stand-landscape integration in natural disturbance-based management of the southern boreal forest. For. Ecol. Manag. 2002, 155, 369–385. [Google Scholar] [CrossRef]
- Work, T.T.; Shorthouse, D.P.; Spence, J.R.; Volney, W.J.A.; Langor, D. Stand composition and structure of the boreal mixedwood and epigaeic arthropods of the Ecosystem Management Emulating Natural Disturbance (EMEND) landbase in northwestern Alberta. Can. J. For. Res. 2004, 34, 417–430. [Google Scholar] [CrossRef]
- Harvey, B.D.; Brais, S. Partial cutting as an analogue to stem exclusion and dieback in trembling aspen (Populus tremuloides) dominated boreal mixedwoods: Implications for deadwood dynamics. Can. J. For. Res. 2007, 37, 1525–1533. [Google Scholar] [CrossRef]
- Nyland, R.D. Even- to uneven-aged: The challenges of conversion. For. Ecol. Manag. 2003, 172, 291–300. [Google Scholar] [CrossRef]
- Drever, C.R.; Peterson, G.; Messier, C.; Bergeron, Y.; Flannigan, M. Can forest management based on natural disturbances maintain ecological resilience? Can. J. For. Res. 2009, 36, 2285–2299. [Google Scholar]
- Rainio, J.; Niemelä, J. Ground beetles (Coleoptera:Carabidae) as bioindicators. Biol. Conserv. 2003, 12, 487–506. [Google Scholar]
- Work, T.T.; Koivula, M.; Klimaszewski, J.; Langor, D.; Spence, J.R.; Sweeney, J.; Hébert, C. Evaluation of carabid beetles as indicators of forest change in Canada. Can. Entomol. 2008, 393, 393–414. [Google Scholar]
- Niemelä, J.; Langor, D.; Spence, J.R. Effects of clear-cut harvesting on boreal ground-beetle assemblages (Coleoptera:Carabidae) in Western Canada. Conserv. Biol. 1993, 7, 551–561. [Google Scholar]
- Koivula, M.; Kukkonen, J.; Niemelä, J. Boreal carabid-beetle (Coleoptera:Carabidae) assemblages along the clear-cut originated succession gradient. Biodivers. Conserv. 2002, 11, 1269–1288. [Google Scholar] [CrossRef]
- Buddle, C.M.; Langor, D.W.; Pohl, G.R.; Spence, J.R. Arthropod responses to harvesting and wildfire: Implications for emulation of natural disturbance in forest management. Biol. Conserv. 2006, 128, 346–357. [Google Scholar] [CrossRef]
- Koivula, M.; Spence, J.R. Effects of post-fire salvage logging on boreal mixed-wood ground beetle assemblages (Coleoptera:Carabidae). For. Ecol. Manag. 2006, 236, 102–112. [Google Scholar] [CrossRef]
- Martikainen, P.; Kouki, J.; Heikkala, O. The effects of green tree retention and subsequent prescribed burning on ground beetles (Coleoptera:Carabidae) in boreal pine-dominated forests. Ecography 2006, 29, 659–670. [Google Scholar] [CrossRef]
- Halaj, J.; Halpern, C.B.; Yi, H. Responses of litter-dwelling spiders and carabid beetles to varying levels and patterns of green-tree retention. For. Ecol. Manag. 2008, 255, 887–900. [Google Scholar] [CrossRef]
- Work, T.T.; Jacobs, J.M.; Spence, J.R.; Volney, W.J.A. High levels of green retention are required to preserve ground beetle diversity in boreal mixedwood forests. Ecol. Appl. 2010, 20, 741–751. [Google Scholar] [CrossRef]
- Brais, S.; Harvey, B.D.; Bergeron, Y.; Messier, C.; Greene, D.; Belleau, A.; Paré, D. Testing forest ecosystem management in boreal mixedwoods of northwestern Quebec: Initial response of aspen stands to different levels of harvesting. Can. J. For. Res. 2004, 34, 431–446. [Google Scholar] [CrossRef]
- Haeussler, S.; Bergeron, Y.; Brais, S.; Harvey, B.D. Natural dynamics-based silviculture for maintaining plant biodiversity in Populus tremuloides-dominated boreal forests of eastern Canada. Can. J. Bot. 2007, 85, 1158–1170. [Google Scholar] [CrossRef]
- Kebli, H.; Brais, S.; Kernaghan, G.; Drouin, P. Impact of harvesting intensity on wood-inhabiting fungi in boreal aspen forests of eastern Canada. For. Ecol. Manag. 2012, 279, 45–54. [Google Scholar] [CrossRef]
- Harvey, B. The Lake Duparquet research and teaching forest: Building a foundation for ecosystem management. For. Chron. 1999, 75, 389–393. [Google Scholar]
- Saucier, J.-P.; Bergeron, J.-F.; Grondin, P.; Robitaille, A. Les régions écologiques du Québec méridional (3e version): Un des éléments du système hiérarchique de classification écologique du territoire mis au point par le ministère des Ressources naturelles du Québec. Supplément de l'Aubelle 1998, 124:1–124:12. [Google Scholar]
- Veillette, J.J. Evolution and paleohydrology of glacial Lakes Barlow and Ojibway. Quat. Sci. Rev. 1994, 13, 945–971. [Google Scholar] [CrossRef]
- Environment Canada, Canadian Climate Normals or Averages 1971–2000. In National Climate Data and Information Archive; Available online: www.climate.weatheroffice.gc.ca. (accessed on 16 May 2013).
- Imbeau, L.; Desrochers, A. Foraging ecology and use of drumming trees by three-toed woodpeckers. J. Wildl. Manag. 2002, 66, 222–231. [Google Scholar] [CrossRef]
- Van Wagner, C.E. Practical Aspects of the Line Intercept Method; Information Report PI-X-12; ISSN 0706–1854; Petawawa National Forestry Institute, Canadian Forestry Service: Chalk River, Canada, 1982. [Google Scholar]
- Robert, E.; Brais, S.; Harvey, B.D.; Greene, D. Seedling establishment and survival on decaying logs in boreal mixedwood stands following a mast year. Can. J. For. Res. 2012, 42, 1446–1455. [Google Scholar] [CrossRef]
- Morin, H.; Laprise, D; Bergeron, Y. Chronology of spruce budworm outbreaks near Lake Duparquet, Abitibi region, Quebec. Can. J. For. Res. 1993, 23, 1497–1506. [Google Scholar] [CrossRef]
- SAS Institute Inc, SAS/STAT® 9.2 User’s Guide, 2nd Edition, SAS Institute Inc.: Cary, NC, USA, 2009.
- De’ath, G. Multivariate regression trees: A new technique for modeling species-environment relationships. Ecology 2002, 83, 1105–1117. [Google Scholar]
- Brassard, W.B.; Chen, H.Y.H. Stand structural dynamics of North American boreal forests. Crit. Rev. Plant Sci. 2006, 25, 115–137. [Google Scholar] [CrossRef]
- Bergeron, Y.; Leduc, A.; Joyal, C.; Morin, H. Balsam fir mortality following the last spruce budworm outbreak in northwestern Quebec. Can. J. For. Res. 1995, 25, 1375–1384. [Google Scholar] [CrossRef]
- Messier, C.; Doucet, R.; Ruel, J.-C.; Claveau, Y.; Kelly, C.; Lechowicz, M.J. Functional ecology of advance regeneration in relation to light in boreal forests. Can. J. For. Res. 1999, 29, 812–823. [Google Scholar] [CrossRef]
- Parent, S.; Ruel, J.C. Chronologie de la croissance chez des semis de sapin baumier (Abies balsamea (L.) Mill.) après une coupe à blanc avec protection de la régénération. For. Chron. 2002, 8, 876–885. [Google Scholar]
- Beaudet, M.; Harvey, B.D.; Messier, C.; Coates, K.D.; Poulin, J.; Kneeshaw, D.D.; Brais, S.; Bergeron, Y. Managing understory light conditions in boreal mixedwoods through variation in the intensity and spatial pattern of harvest: A modelling approach. For. Ecol. Manag. 2011, 261, 84–94. [Google Scholar] [CrossRef]
- Claveau, Y.; Messier, C.; Comeau, P.G.; Coates, K.D. Growth and crown morphological responses of boreal conifer seedlings and saplings with contrasting shade tolerance to a gradient of light and height. Can. J. For. Res. 2002, 32, 458–468. [Google Scholar] [CrossRef]
- Parent, S.; Messier, C. Effets d'un gradient de lumière sur la croissance en hauteur et la morphologie de la cime du sapin baumier régénéré naturellement. Can. J. For. Res. 1995, 25, 878–885. [Google Scholar] [CrossRef]
- Frey, B.R.; Lieffers, V.J.; Landhäusser, S.M.; Comeau, P.G.; Greenway, K.J. An analysis of sucker regeneration of trembling aspen. Can. J. For. Res. 2003, 33, 1169–1179. [Google Scholar] [CrossRef]
- Lennie, A.D.; Landhäusser, S.M.; Ieffers, V.J.; Sidders, D. Regeneration of aspen following partial and strip understory. For Chron. 2009, 85, 631–638. [Google Scholar]
- Paré, D.; Bergeron, Y.; Longpré, M.-H. Potential productivity of aspen cohorts originating from fire, harvesting, and tree-fall gaps on two deposit types in northwestern Quebec. Can. J. For. Res. 2001, 31, 1067–1073. [Google Scholar] [CrossRef]
- Moulinier, J.; Lorenzetti, F.; Bergeron, Y. Gap dynamics in aspen stands of the clay belt of northwestern Quebec following a forest tent caterpillar outbreak. Can. J. For. Res. 2011, 41, 1606–1617. [Google Scholar] [CrossRef]
- Thorpe, H.C.; Thomas, S.C.; Caspersen, J.P. Tree mortality following partial harvest is determined by skidding proximity. Ecol. Appl. 2008, 18, 1652–1663. [Google Scholar] [CrossRef]
- Rongzhou Man, R.; Rice, J.A. Response of aspen stands to forest tent caterpillar defoliation and subsequent overstory mortality in northeastern Ontario, Canada. For. Ecol. Manag. 2010, 260, 1853–1860. [Google Scholar] [CrossRef]
- Drapeau, P.; Nappi, A.; Imbeau, L.; Saint-Germain, M. Standing deadwood for keystone bird species in the eastern boreal forest: Managing for snag dynamics. For. Chron. 2009, 85, 227–234. [Google Scholar]
- Kebli, H.; Drouin, P.; Brais, S.; Kernaghan, G. Species composition of saproxylic fungal communities on decaying logs in the boreal forest. Microb. Ecol. 2011, 61, 898–910. [Google Scholar] [CrossRef]
- Brais, S.; Drouin, P. Interactions between deadwood and soil characteristics in a natural boreal trembling aspen—Jack pine stand. Can. J. For. Res. 2012, 42, 1456–1466. [Google Scholar] [CrossRef]
- Bauhus, J.; Puettmann, K.; Messier, C. Silviculture for old-growth attributes. For. Ecol. Manag. 2009, 258, 525–537. [Google Scholar] [CrossRef]
- Stokland, J.N.; Siitonen, J.; Jonsson, B.G. Biodiversity in Dead Wood; Cambridge University Press: New York, NY, USA, 2012; ISBN 978-0-521-71703. [Google Scholar]
- Work, T.T.; Brais, S.; Harvey, B.D. Reductions in downed deadwood from biomass harvesting alter composition of spiders and ground beetle assemblages in jack-pine forests of Western Quebec. For. Ecol. Manag. 2013. submitted. [Google Scholar]
- Niemelä, J.; Koivula, M.; Kotze, D.J. The effects of forestry on carabid beetles (Coleoptera:Carabidae) in boreal forests. J. Insect Conserv. 2007, 11, 5–18. [Google Scholar] [CrossRef]
- Paradis, S.; Work, T.T. Partial cutting does not maintain spider assemblages within the observed range of natural variability in eastern Canadian black spruce forests. For. Ecol. Manag. 2011, 262, 2079–2093. [Google Scholar] [CrossRef]
- Pinzon, J.; Spence, J.R.; Langor, D. Responses of ground-dwelling spiders (Araneae) to variable retention haversting practices in the boreal forest. For. Ecol. Manag. 2013, 266, 42–53. [Google Scholar] [CrossRef]
- Klimaszewski, J.; Langor, D.W.; Work, T.T.; Pelletier, G.; Hammond, H.E.J.; Germaine, C. The effects of patch harvesting and site preparation on ground beetles (Coleoptera:Carabidae) in yellow birch dominated forests of southeastern Quebec. Can. J. For. Res. 2005, 35, 2616–2628. [Google Scholar] [CrossRef]
- Raymond, P.; Bédard, S.; Roy, V.; Larouche, C.; Tremblay, S. The irregular shelterwood system: Review, classification and potential application to forests affected by partial disturbances. J. For. 2009, 107, 405–413. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Brais, S.; Work, T.T.; Robert, É.; O'Connor, C.D.; Strukelj, M.; Bose, A.; Celentano, D.; Harvey, B.D. Ecosystem Responses to Partial Harvesting in Eastern Boreal Mixedwood Stands. Forests 2013, 4, 364-385. https://doi.org/10.3390/f4020364
Brais S, Work TT, Robert É, O'Connor CD, Strukelj M, Bose A, Celentano D, Harvey BD. Ecosystem Responses to Partial Harvesting in Eastern Boreal Mixedwood Stands. Forests. 2013; 4(2):364-385. https://doi.org/10.3390/f4020364
Chicago/Turabian StyleBrais, Suzanne, Timothy T. Work, Émilie Robert, Christopher D. O'Connor, Manuella Strukelj, Arun Bose, Danielle Celentano, and Brian D. Harvey. 2013. "Ecosystem Responses to Partial Harvesting in Eastern Boreal Mixedwood Stands" Forests 4, no. 2: 364-385. https://doi.org/10.3390/f4020364
APA StyleBrais, S., Work, T. T., Robert, É., O'Connor, C. D., Strukelj, M., Bose, A., Celentano, D., & Harvey, B. D. (2013). Ecosystem Responses to Partial Harvesting in Eastern Boreal Mixedwood Stands. Forests, 4(2), 364-385. https://doi.org/10.3390/f4020364




