Design of Anion Exchange Membranes and Electrodialysis Studies for Water Desalination
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
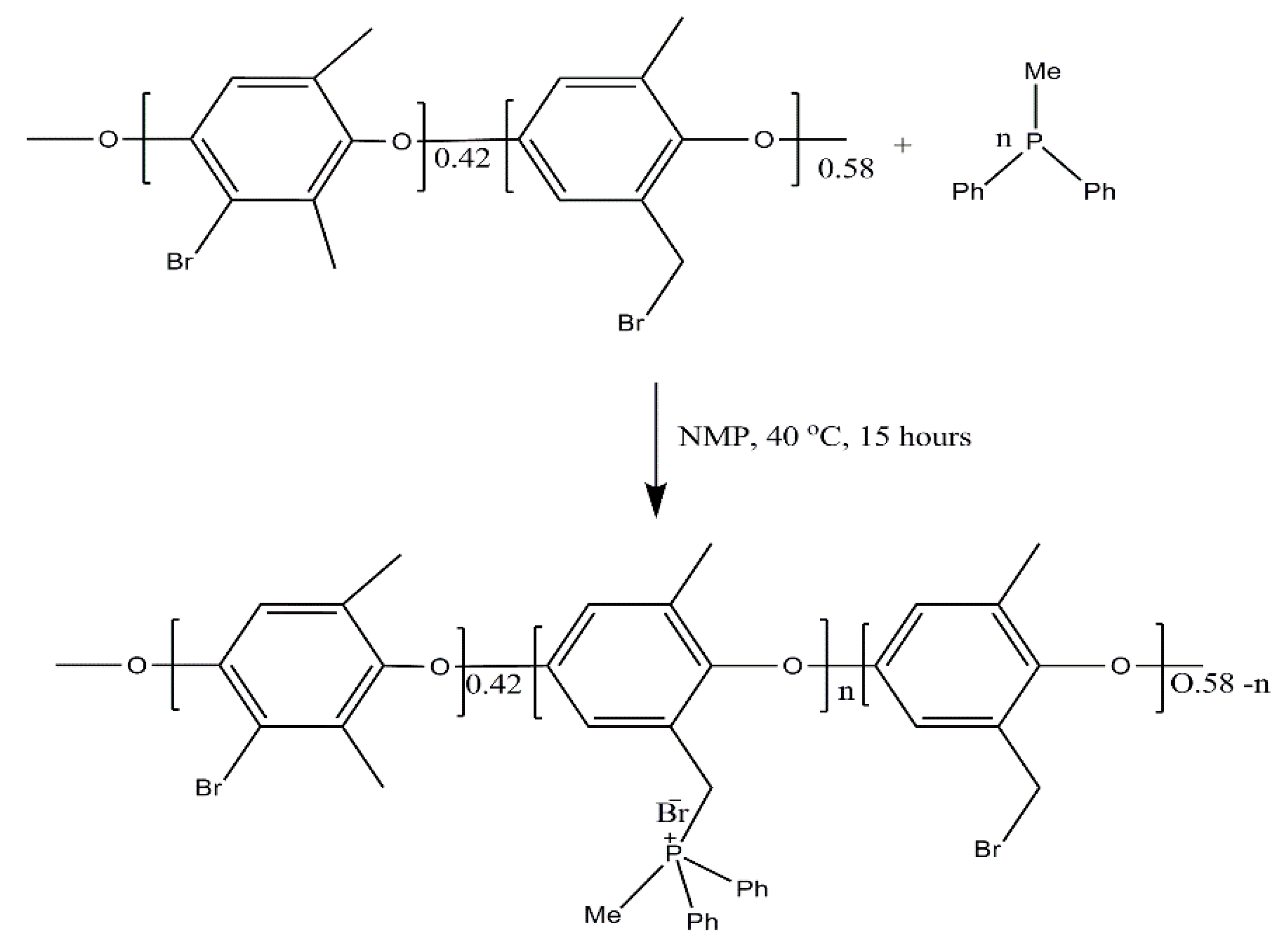
2.2. Membrane Preparation
2.3. Characterizations
2.3.1. FTIR Spectra and Thermal Stability
2.3.2. Ion Exchange Capacity
2.3.3. Microscopic Characterizations for AEMs
2.3.4. Water Uptake and Linear Expansion Ratio and Fixed Group Concentration
2.3.5. Mechanical Property
2.3.6. Membrane Area Resistance
2.3.7. Membrane Transport Number
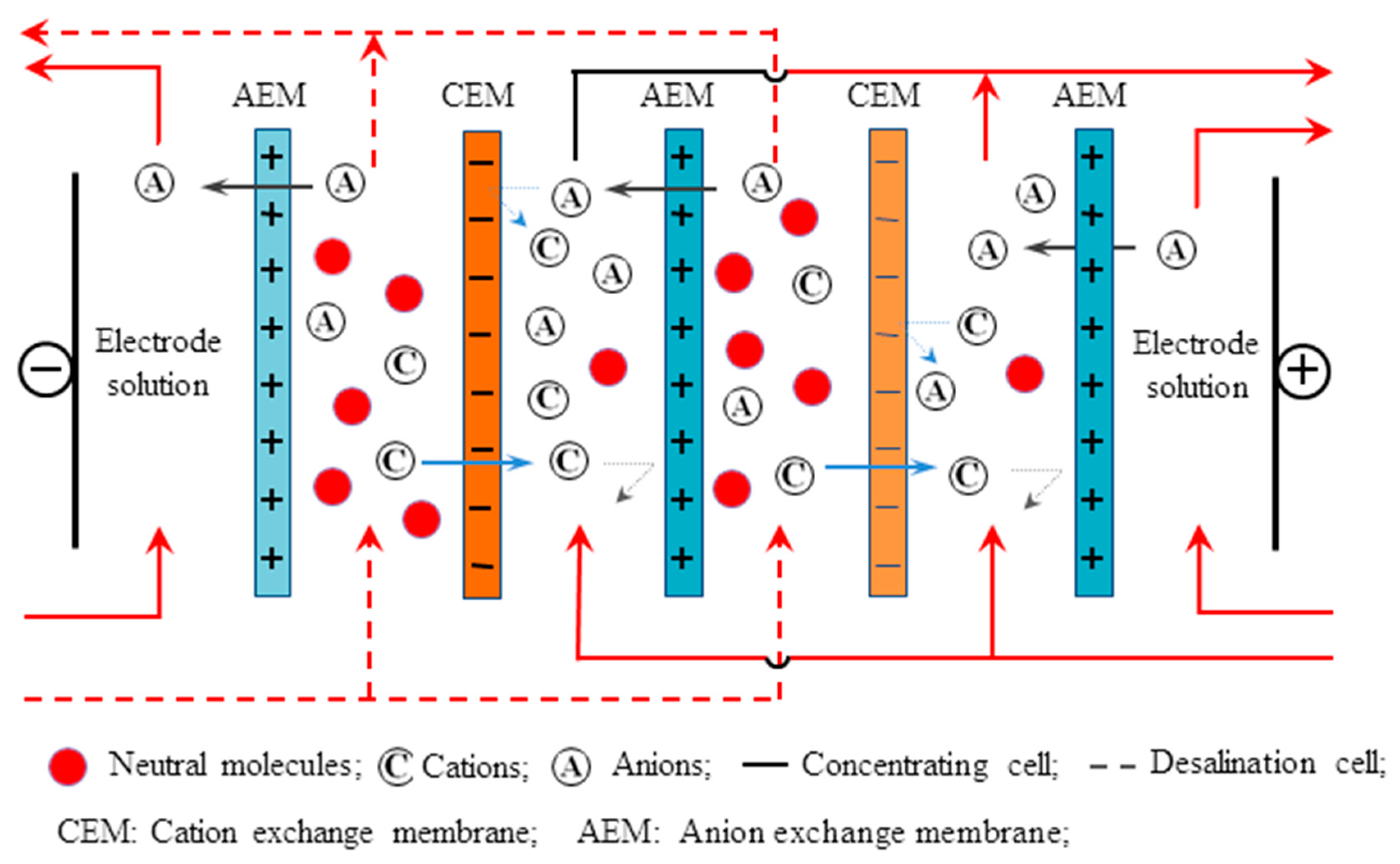
2.3.8. Electrodialysis Stack
2.3.9. Desalination rate and Water Recovery Rate
3. Results and Discussion
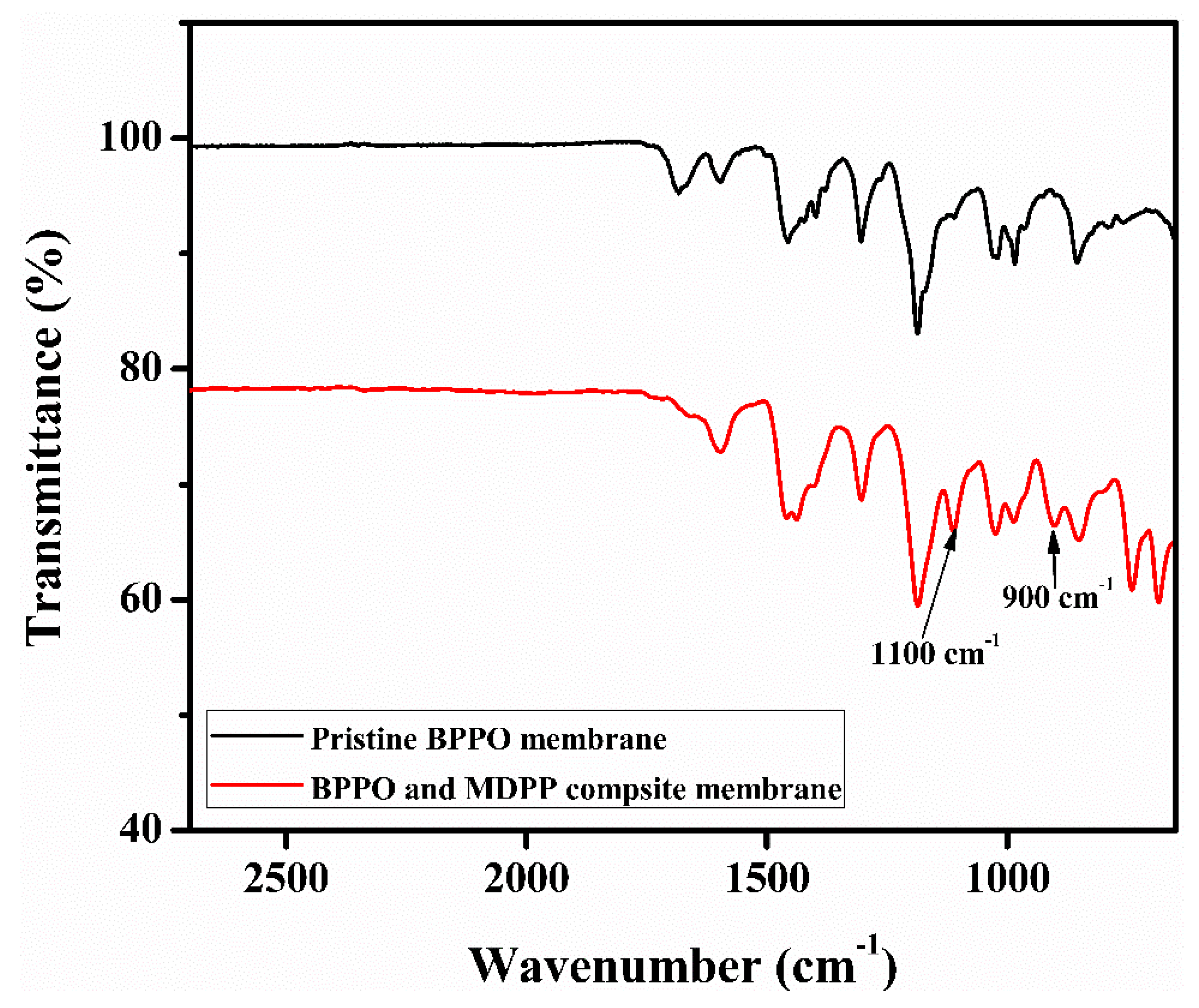
3.1. FTIR Analysis
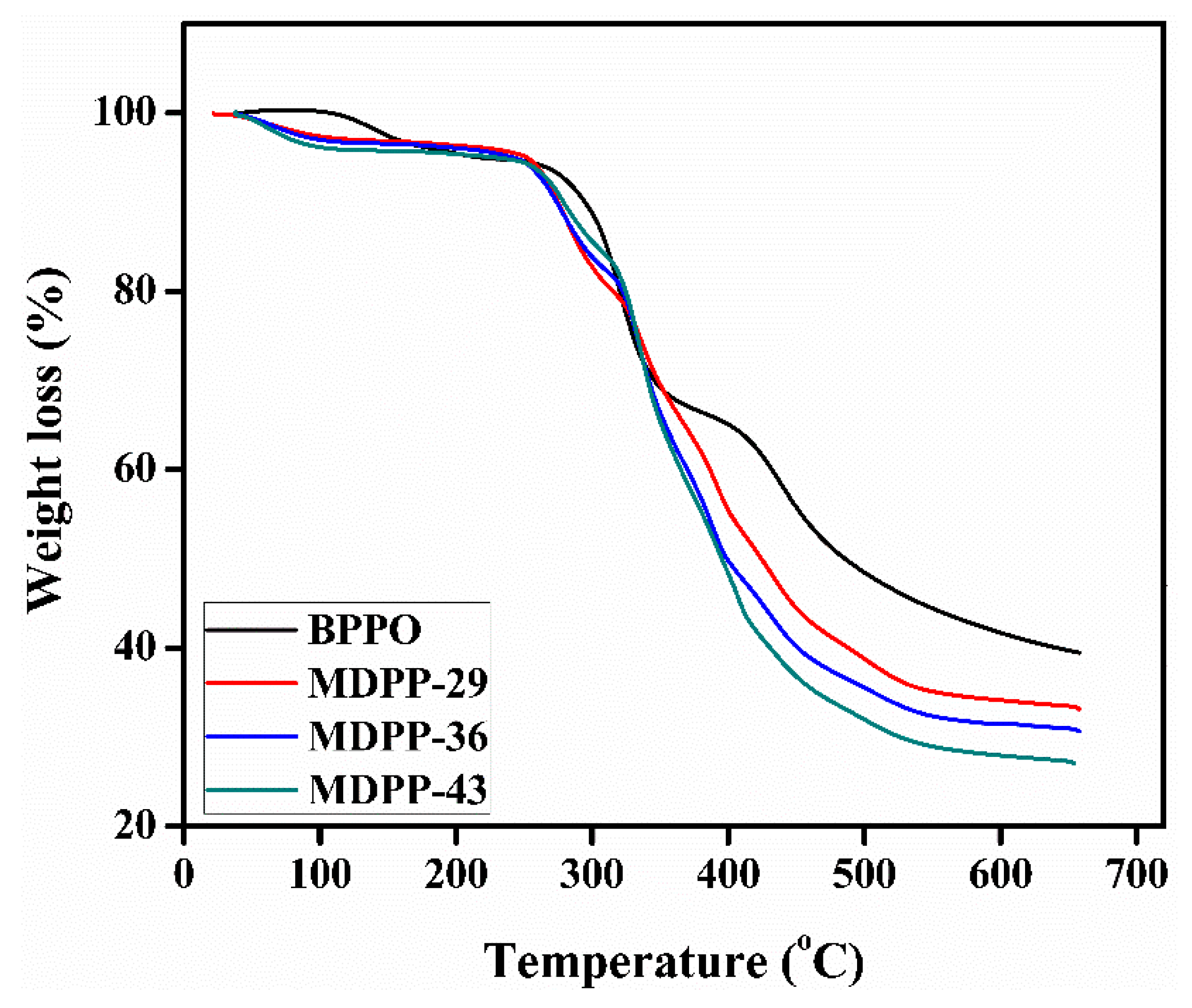
3.2. Thermal Stability
3.3. Morphology of Membranes
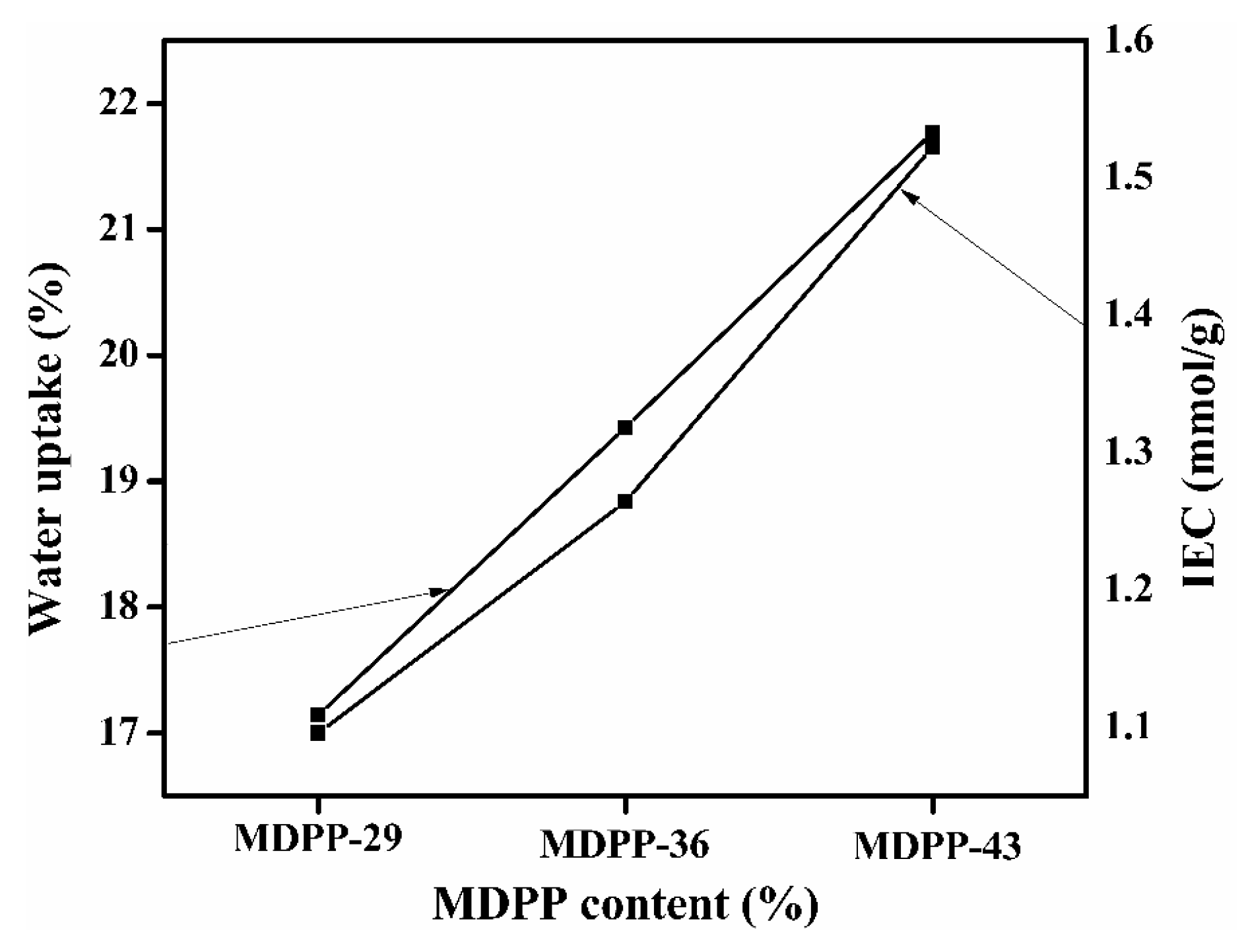
3.4. Water Uptake (WR), Ion Exchange Capacity and Linear Expansion Ratio (LER)
3.5. Mechanical Properties
3.6. Membrane Area Resistance and Transport Number
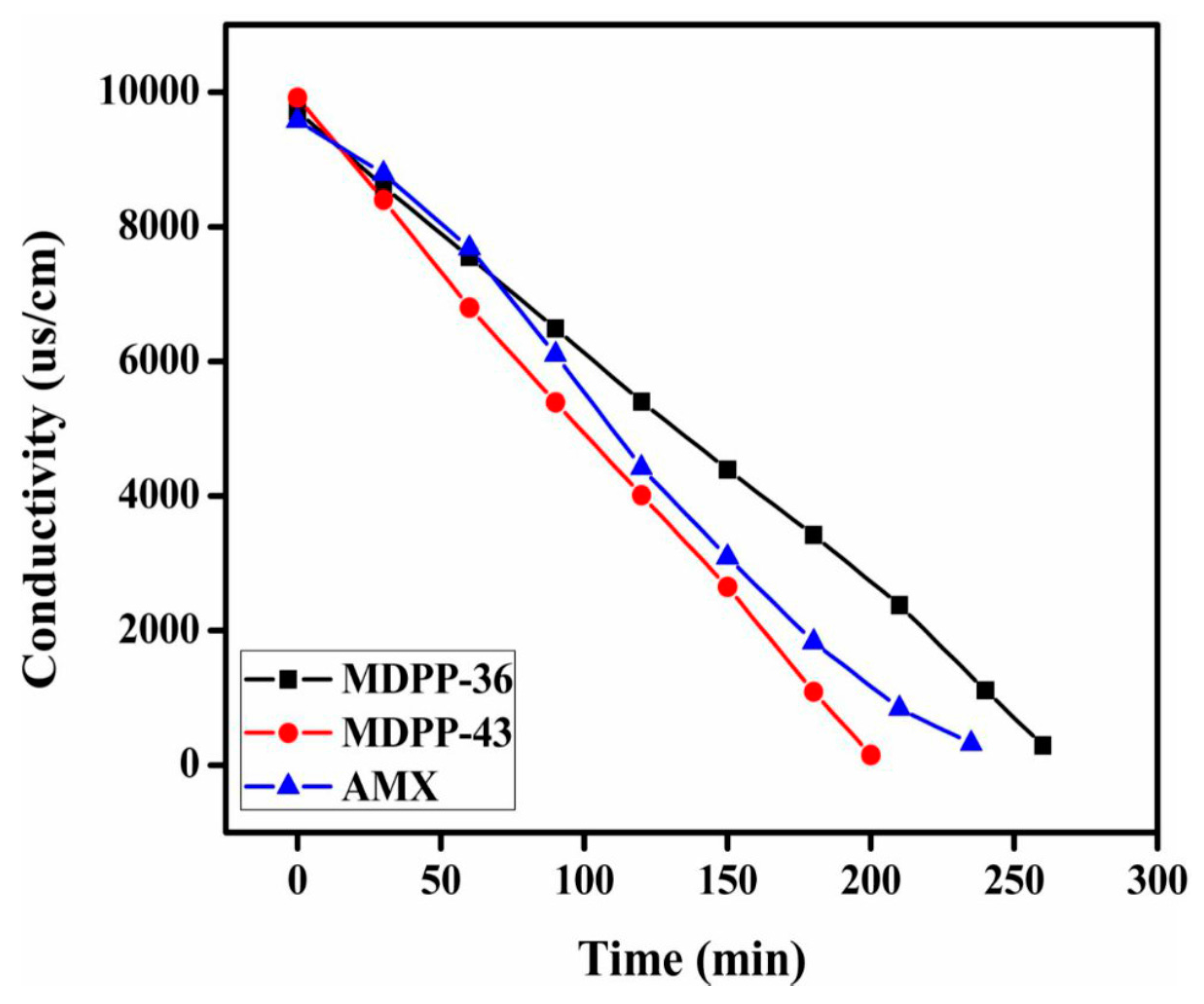
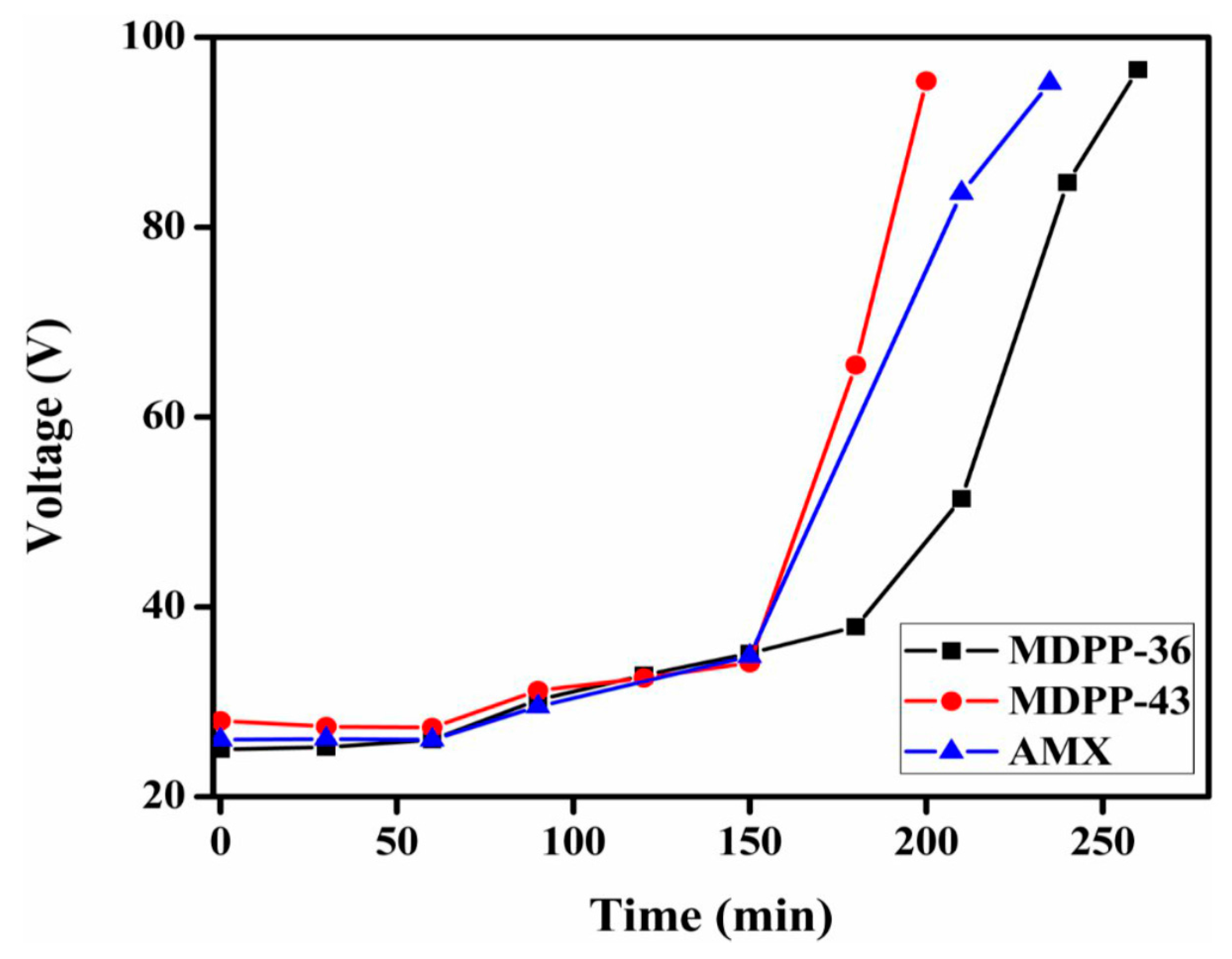
3.7. Membrane Performance
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| AEM | Anion exchange membrane |
| BPPO | Brominated poly(2,6-dimethyl-1,4-phenylene oxide) |
| MDPP | Methyl(diphenyl)phosphine |
| NMP | N-Methyl-2-pyrrolidone |
| WR | Water uptake |
| IEC | Ion exchange capacity |
| LER | Linear expension ration |
| AMX | Commercial anion exchange membrane |
| CMX | Commercial cation exchange membrane |
| R | Gas constant |
| RW | Desalination rate |
| SEM | Scanning electron microscopy |
| TS | Tensile strength |
| Eb | Elongation at break |
| ED | Electrodialysis |
References
- Juda, W.; McRae, W.A. Coherent ion-exchange gels and membranes. J. Am. Chem. Soc. 1950, 72, 1044. [Google Scholar] [CrossRef]
- Buonomenna, M.G. Membrane processes for a sustainable industrial growth. RSC Adv. 2013, 3, 5694–5740. [Google Scholar] [CrossRef]
- Kim, D.H. A review of desalting process techniques and economic analysis of the recovery of salts from retentates. Desalination 2011, 270, 1–8. [Google Scholar] [CrossRef]
- Khan, M.I.; Akhtar, S.; Zafar, S.; Shaheen, A.; Khan, M.A.; Luque, R. Removal of Congo Red from Aqueous Solution by Anion Exchange Membrane (EBTAC): Adsorption Kinetics and Themodynamics. Materials 2015, 8, 4147–4161. [Google Scholar] [CrossRef]
- Luo, J.; Wu, C.; Xu, T.; Wu, Y. Diffusion dialysis-concept, principle and applications. J. Membr. Sci. 2011, 366, 1–16. [Google Scholar] [CrossRef]
- Langevin, D.; Pinoche, M.; Se, E.; Me, M.; Roux, R. CO2 facilitated transport through functionalized cation-exchange membranes. J. Membr. Sci. 1993, 82, 51–63. [Google Scholar] [CrossRef]
- Adachi, K.; Hu, W.; Matsumoto, H.; Ito, K.; Tanioka, A. Permeation of n-butane, 1-butene and 1,3-butadiene through anhydrated Ag+-doped perfluorocarbon-type ion-exchange membranes. Polymer 1998, 39, 2315–2323. [Google Scholar] [CrossRef]
- Way, J.D.; Noble, R.D.; Reed, D.L.; Ginley, G.M.; Jarr, L.A. Facilitated transport of CO2 in ion exchange memberanes. AIChE J. 1987, 33, 480–487. [Google Scholar] [CrossRef]
- Sharaf, O.Z.; Orhan, M.F. An overview of fuel cell technology: Fundamentals and applications. Renew. Sustain. Energy Rev. 2014, 32, 810–853. [Google Scholar] [CrossRef]
- Varcoe, J.R.; Slade, R.C. Prospects for alkaline anion-exchange membranes in low temperature fuel cells. Fuel Cells 2005, 5, 187–200. [Google Scholar] [CrossRef] [Green Version]
- Yasuda, A.; Yamaga, N.; Doi, K.; Fujioka, T.; Kusanagi, S. Electrochemical characteristics of the planar electrochemical carbon monoxide sensor with a perfluorosulfonate ionomer film. Solid State Ion. 1990, 40, 476–479. [Google Scholar] [CrossRef]
- Strathmann, H.; Grabowski, A.; Eigenberger, G. Ion-exchange membranes in the chemical process industry. Ind. Eng. Chem. Res. 2013, 52, 10364–10379. [Google Scholar] [CrossRef]
- Góźdź, A.S.; Trochimczuk, W. Continuous modification of polyethylene with styrene and divinylbenzene in melt. J. Appl. Polym. Sci. 1980, 25, 947–950. [Google Scholar] [CrossRef]
- Pozniak, G.; Trochimczuk, W. Chloromethylation of the polyethylene/poly(styrene-co-divinylbenzene) system. J. Appl. Polym. Sci. 1982, 27, 1833–1838. [Google Scholar] [CrossRef]
- Poźniak, G.; Trochimczuk, W. Interpolymer anion exchange membranes, I. Properties of weak base membranes based on polyethylene modified by styrene and divinylbenzene in the presence of diluents. Angew. Makromol. Chem. 1980, 92, 155–168. [Google Scholar] [CrossRef]
- Wang, G.; Weng, Y.; Chu, D.; Chen, R.; Xie, D. Developing a polysulfone-based alkaline anion exchange membrane for improved ionic conductivity. J. Membr. Sci. 2009, 332, 63–68. [Google Scholar] [CrossRef]
- Wang, G.; Weng, Y.; Zhao, J.; Chen, R.; Xie, D. Preparation of a functional poly(ether imide) membrane for potential alkaline fuel cell applications: Chloromethylation. J. Appl. Polym. Sci. 2009, 112, 721–727. [Google Scholar] [CrossRef]
- Xu, T.; Liu, Z.; Li, Y.; Yang, W. Preparation and characterization of Type II anion exchange membranes from poly(2,6-dimethyl-1,4-phenylene oxide) (PPO). J. Membr. Sci. 2008, 320, 232–239. [Google Scholar] [CrossRef]
- Li, X.; Liu, Q.; Yu, Y.; Meng, Y. Quaternized poly(arylene ether) ionomers containing triphenyl methane groups for alkaline anion exchange membranes. J. Mater. Chem. A 2013, 1, 4324–4335. [Google Scholar] [CrossRef]
- Zhang, H.; Zhou, Z. Alkaline polymer electrolyte membranes from quaternized poly(phthalazinone ether ketone) for direct methanol fuel cell. J. Appl. Polym. Sci. 2008, 110, 1756–1762. [Google Scholar] [CrossRef]
- Yan, C.; Zhang, S.; Yang, D.; Jian, X. Preparation and characterization of chloromethylated/quaternized poly(phthalazinone ether sulfone ketone) for positively charged nanofiltration membranes. J. Appl. Polym. Sci. 2008, 107, 1809–1816. [Google Scholar] [CrossRef]
- Zeng, Q.H.; Liu, Q.L.; Broadwell, I.; Zhu, A.M.; Xiong, Y.; Tu, X.P. Anion exchange membranes based on quaternized polystyrene-block-poly(ethylene-ran-butylene)-block-polystyrene for direct methanol alkaline fuel cells. J. Membr. Sci. 2010, 349, 237–243. [Google Scholar] [CrossRef]
- Stamatialis, D.F.; Papenburg, B.J.; Gironés, M.; Saiful, S.; Bettahalli, S.N.; Schmitmeier, S.; Wessling, M. Medical applications of membranes: Drug delivery, artificial organs and tissue engineering. J. Membr. Sci. 2008, 308, 1–34. [Google Scholar] [CrossRef]
- Wang, N.; Wu, C.; Cheng, Y.; Xu, T. Organic–inorganic hybrid anion exchange hollow fiber membranes: A novel device for drug delivery. Int. J. Pharm. 2011, 408, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Kujawski, W.; Poźniak, G. Transport properties of ion-exchange membranes during pervaporation of water-alcohol mixtures. Sep. Sci. Technol. 2005, 40, 2277–2295. [Google Scholar] [CrossRef]
- Merrifield, R.B. Solid phase synthesis (Nobel lecture). Angew. Chem. Int. Ed. Engl. 1985, 24, 799–810. [Google Scholar] [CrossRef]
- Laskin, S.; Kuschner, M.; Drew, R.T.; Cappiello, V.P.; Nelson, N. Tumors of the respiratory tract induced by inhalation of bis(chloromethyl) ether. Arch. Environ. Health Int. J. 1971, 23, 135–136. [Google Scholar] [CrossRef]
- Pafford, M.M.; Reichl, J.A.; Rosenbaum, B.M. Method for Preparation of Anion Exchange Resins. U.S. Patent 4785020, 2004. [Google Scholar]
- Barrett, J.H.; Heights, C. Method of Preparing Anion Exchange Resins with Sulfuryl Chloride. U.S. Patent 3,812,061, 21 May 1972. [Google Scholar]
- Jeřábek, K.; Pulko, I.; Soukupova, K.; Štefanec, D.; Krajnc, P. Porogenic solvents influence on morphology of 4-vinylbenzyl chloride based PolyHIPEs. Macromolecules 2008, 41, 3543–3546. [Google Scholar] [CrossRef]
- Zhao, C.H.; Gong, Y.; Liu, Q.L.; Zhang, Q.G.; Zhu, A.M. Self-crosslinked anion exchange membranes by bromination of benzylmethyl-containing poly(sulfone)s for direct methanol fuel cells. Int. J. Hydrogen Energy 2012, 37, 11383–11393. [Google Scholar] [CrossRef]
- Yan, J.; Hickner, M.A. Anion exchange membranes by bromination of benzylmethyl-containing poly(sulfone)s. Macromolecules 2010, 43, 2349–2356. [Google Scholar] [CrossRef]
- Xu, T.; Yang, W. A novel positively charged composite membranes for nanofiltration prepared from poly(2,6-dimethyl-1,4-phenylene oxide) by in situ amines crosslinking. J. Membr. Sci. 2003, 215, 25–32. [Google Scholar]
- Lin, Z.; Xu, T.; Zhang, L. Radiation-induced grafting of N-isopropylacrylamide onto the brominated poly(2,6-dimethyl-1,4-phenylene oxide) membranes. Radiat. Phys. Chem. 2006, 75, 532–540. [Google Scholar] [CrossRef]
- Hibbs, M.R.; Fujimoto, C.H.; Cornelius, C.J. Synthesis and characterization of poly(phenylene)-based anion exchange membranes for alkaline fuel cells. Macromolecules 2009, 42, 8316–8321. [Google Scholar] [CrossRef]
- Liu, Z.; Li, X.; Shen, K.; Feng, P.; Zhang, Y.; Xu, X.; Hu, W.; Jiang, Z.; Liu, B.; Guiver, M.D. Naphthalene-based poly(arylene ether ketone) anion exchange membranes. J. Mater. Chem. A 2013, 1, 6481–6488. [Google Scholar] [CrossRef]
- Merle, G.; Wessling, M.; Nijmeijer, K. Anion exchange membranes for alkaline fuel cells: A review. J. Membr. Sci. 2011, 377, 1–35. [Google Scholar] [CrossRef]
- Kujawski, W.; Poźniak, G.; Nguyen, Q.T.; Néel, J. Properties of interpolymer PESS ion-exchange membranes in contact with solvents of different polarities. Sep. Sci. Technol. 1997, 32, 1657–1667. [Google Scholar] [CrossRef]
- Cheng, C.; Yang, Z.; Pan, J.; Tong, B.; Xu, T. Facile and cost effective PVA based hybrid membrane fabrication for acid recovery. Sep. Purif. Technol. 2014, 136, 250–257. [Google Scholar] [CrossRef]
- Wu, Y.; Luo, J.; Zhao, L.; Zhang, G.; Wu, C.; Xu, T. QPPO/PVA anion exchange hybrid membranes from double crosslinking agents for acid recovery. J. Membr. Sci. 2013, 428, 95–103. [Google Scholar] [CrossRef]
- Larchet, C.; Nouri, S.; Auclair, B.; Dammak, L.; Nikonenko, V. Application of chronopotentiometry to determine the thickness of diffusion layer adjacent to an ion-exchange membrane under natural convection. Adv. Colloid Interface 2008, 139, 45–61. [Google Scholar] [CrossRef] [PubMed]
- Lakshminarayanaiah, N. Transport Phenomena in Membranes; Academic Press: New York, NY, USA, 1969. [Google Scholar]
- Hossain, M.M.; Wu, L.; Li, Y.; Ge, L.; Xu, T. Preparation of porous poly(vinylidene fluoride) membranes with acrylate particles for electrodialysis application. Sep. Purif. Technol. 2015, 150, 102–111. [Google Scholar] [CrossRef]
- Jiang, C.; Wang, Y.; Zhang, Z.; Xu, T. Electrodialysis of concentrated brine from RO plant to produce coarse salt and freshwater. J. Membr. Sci. 2014, 450, 323–330. [Google Scholar] [CrossRef]
- Wu, C.; Wu, Y.; Luo, J.; Xu, T.; Fu, Y. Anion exchange hybrid membranes from PVA and multi-alkoxy silicon copolymer tailored for diffusion dialysis process. J. Membr. Sci. 2010, 356, 96–104. [Google Scholar] [CrossRef]
- Li, C.; Sun, G.; Ren, S.; Liu, J.; Wang, Q.; Wu, Z.; Sun, H.; Jin, W. Casting Nafion–sulfonated organosilica nano-composite membranes used in direct methanol fuel cells. J. Membr. Sci. 2006, 272, 50–57. [Google Scholar] [CrossRef]
- Spry, D.B.; Goun, A.; Glusac, K.; Moilanen, D.E.; Fayer, M.D. Proton Transport and the Water Environment in Nafion Fuel Cell Membranes and AOT Reverse Micelles. J. Am. Chem. Soc. 2007, 129, 8122–8130. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Wu, C.; Wu, Y.; Gu, J.; Xu, T. Polyelectrolyte complex/PVA membranes for diffusion dialysis. J. Hazard. Mater. 2013, 261, 114–122. [Google Scholar] [CrossRef] [PubMed]
- Chakrabarty, T.; Singh, A.K.; Shahi, V.K. Zwitterionic silica copolymer based crosslinked organic-inorganic hybrid polymer electrolyte membranes for fuel cell applications. RSC Adv. 2012, 2, 1949–1961. [Google Scholar] [CrossRef]
- Singh, S.; Jasti, A.; Kumar, M.; Shahi, V.K. A green method for the preparation of highly stable organic-inorganic hybrid anion-exchange membranes in aqueous media for electrochemical processes. Polym. Chem. 2010, 1, 1302–1312. [Google Scholar] [CrossRef]
- Cheng, J.-H.; Xiao, Y.-C.; Wu, C.; Chung, T.-S. Chemical modification of P84 polyimide as anion-exchange membranes in a free-flow isoelectric focusing system for protein separation. Chem. Eng. J. 2010, 160, 340–350. [Google Scholar] [CrossRef]
- Khan, M.A.; Kumar, M.; Alothman, Z.A. Preparation and characterization of organic–inorganic hybrid anion-exchange membranes for electrodialysis. J. Ind. Eng. Chem. 2015, 21, 723–730. [Google Scholar] [CrossRef]
- Klaysom, C.; Moon, S.-H.; Ladewig, B.P.; Lu, G.M.; Wang, L. The influence of inorganic filler particle size on composite ion-exchange membranes for desalination. J. Phys. Chem. C 2011, 115, 15124–15132. [Google Scholar] [CrossRef]

| Membranes | MDPP-29 | MDPP-36 | MDPP-43 | AMX |
|---|---|---|---|---|
| BPPO (g) | 0.8 | 0.8 | 0.8 | - |
| MDPP of BPPO (%) | 29 | 36 | 43 | - |
| LER (%) | 7.96 | 8.67 | 11.86 | 7.65 |
| Thickness (μm) | 110 | 83 | 84 | 134 [44] |
| Membranes | MDPP-29 | MDPP-36 | MDPP-43 | AMX |
|---|---|---|---|---|
| TS (MPa) | 30.77 | 23.97 | 16.66 | 40.59 |
| Eb (%) | 485.57 | 589.84 | 647.98 | 27.64 |
| Membranes | AMX | MDPP-29 | MDPP-36 | MDPP-43 |
|---|---|---|---|---|
| Area resistance (Ω·cm2) | 11.9 | 677 | 8.3 | 2.9 |
| Transport number | 0.99 | 0.92 | 0.94 | 0.95 |
| Membranes | Operating Current (0.2 A) | |||
|---|---|---|---|---|
| Desalting Ratio (%) | Current Efficiency η (%) | Energy Consumption (P) (Kw·h·kg−1 water) | Flux (mole·m−2·h−1) | |
| MPDD-36 | 84.6 | 45.2 | 41.45 | 4.82 |
| MPDD-43 | 98.5 | 59.4 | 29.52 | 6.33 |
| AMX | 96.7 | 49.6 | 40.55 | 5.29 |
| Membranes | Water Recovery Rate (%) | Desalination Rate (%) |
|---|---|---|
| MPDD-36 | 95 | 96.0 |
| MPDD-43 | 98 | 98.49 |
| AMX | 97 | 97.0 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, M.I.; Luque, R.; Akhtar, S.; Shaheen, A.; Mehmood, A.; Idress, S.; Buzdar, S.A.; Ur Rehman, A. Design of Anion Exchange Membranes and Electrodialysis Studies for Water Desalination. Materials 2016, 9, 365. https://doi.org/10.3390/ma9050365
Khan MI, Luque R, Akhtar S, Shaheen A, Mehmood A, Idress S, Buzdar SA, Ur Rehman A. Design of Anion Exchange Membranes and Electrodialysis Studies for Water Desalination. Materials. 2016; 9(5):365. https://doi.org/10.3390/ma9050365
Chicago/Turabian StyleKhan, Muhammad Imran, Rafael Luque, Shahbaz Akhtar, Aqeela Shaheen, Ashfaq Mehmood, Sidra Idress, Saeed Ahmad Buzdar, and Aziz Ur Rehman. 2016. "Design of Anion Exchange Membranes and Electrodialysis Studies for Water Desalination" Materials 9, no. 5: 365. https://doi.org/10.3390/ma9050365
APA StyleKhan, M. I., Luque, R., Akhtar, S., Shaheen, A., Mehmood, A., Idress, S., Buzdar, S. A., & Ur Rehman, A. (2016). Design of Anion Exchange Membranes and Electrodialysis Studies for Water Desalination. Materials, 9(5), 365. https://doi.org/10.3390/ma9050365








