1. Introduction
Light weight lithium ion batteries (LIBs) have received increasing interest in recent years due to their high energy densities, long cycle lives, and environmental friendliness. They have become important power sources in consumer electronic products, such as cellular phones and laptop computers. Currently carbon materials are generally used as anode materials for LIBs due to their small surface change, structural stability during cycling, high energy density, and abundance [
1]. Carbon materials can be classified in several ways. They can be divided into graphitizable carbons and non-graphitizable carbons, according to differences in the graphitization process, or they can be distinguished as soft carbons, hard carbons, and graphite with different degrees of graphitization [
2,
3]. Among these carbons, artificial graphite and modified natural graphite are the most commonly chosen carbon anode materials for commercial applications due to their high reversibility. Despite their commercial success, the use of LIBs as a source of electricity for power tools and in the automotive industries is still quite limited, due at least in part to stress generation that results in fracturing/disintegration of the electrode during Lithium ion (Li
+) intercalation and de-intercalation.
The crystalline layered structure of graphitic carbon enables the insertion of Li
+ to achieve high capacities. However, fully exploiting the potential of crystalline graphite in LIBs remains problematic, due to the capacity decrease that accompanies repeated high C-rate charge-discharging. It has been suggested that the slow solid-state diffusion of Li
+ within the graphitic anode materials limits the high C-rate capability of both LIBs [
4], and amorphous carbon materials (e.g., coke), by having a small crystalline size and a wide interlayer distance, that results in a higher charge-discharge rate, due to the fast diffusion of Li
+ within their disordered structures [
5,
6]. However, amorphous carbon materials also show poor capacity retention during their cycle life due to pulverization of the particles, variations in surface morphology, large voids, disordered non-crystallized sites, random lattice structures and contamination from metallic impurities that causes compositional changes during cycling. To reduce the disadvantages of these two types of carbon materials,
i.e., graphitic and amorphous [
7,
8], appropriate combinations, comprising both crystalline and disordered materials, were investigated as potential anode materials for high power applications [
9].
In this work, mesophase soft carbon (MSC); mesophase graphite (SMG), prepared from mesocarbon microbeads (MCMBs); and a commercial hard carbon (HC) were selected as high C-rate LIBs anode materials. The three anode materials were assembled in coin-type cells for capacity retention studies using various C-rates for charging and discharging. The results obtained were correlated with the physical properties of the carbonaceous materials obtained from XRD, scanning and transmission electron microscopic (SEM and TEM), and Raman studies.
3. Results and Discussion
SEM and TEM images of SMG, HC and MSC are shown in
Figure 1.
Figure 1.
(a) SEM and (b) TEM images of SMG, HC, and MSC.
Figure 1.
(a) SEM and (b) TEM images of SMG, HC, and MSC.
From the SEM images, it is apparent that MSC and SMG are spherically shaped with average diameters of 8 to 9 μm; however, there are more fine fragments on the surfaces of the carbonized MSC materials than on the high-temperature graphitized SMG. In contrast to the spherical shapes observed for MSC and SMG, the HC sample shows polyhedral flake plates with a lot of small fragments with dimensions of 1–2 μm. This suggests the reasons for the higher SSA (3.4 m
2·g
−1), compared to those for the spherical SMC and MSG samples (
i.e., 2.63 and 2.10 m
2·g
−1, respectively), see
Table 1. From the TEM images, obvious and long-range layers are observed with SMG, whereas less distinct, short, layered structures are found on the MSC particles’ surfaces. This suggests that the SMG sample has a higher crystallinity and higher-ordered layered structures with unified orientation, compared to MSC. The TEM image of hard carbon reveals that the sample consists of short-layered grains with staggered stacking. The high true and tap densities for SMG (
Table 1) can be attributed to the highly crystalline structure that results from high-temperature heating process.
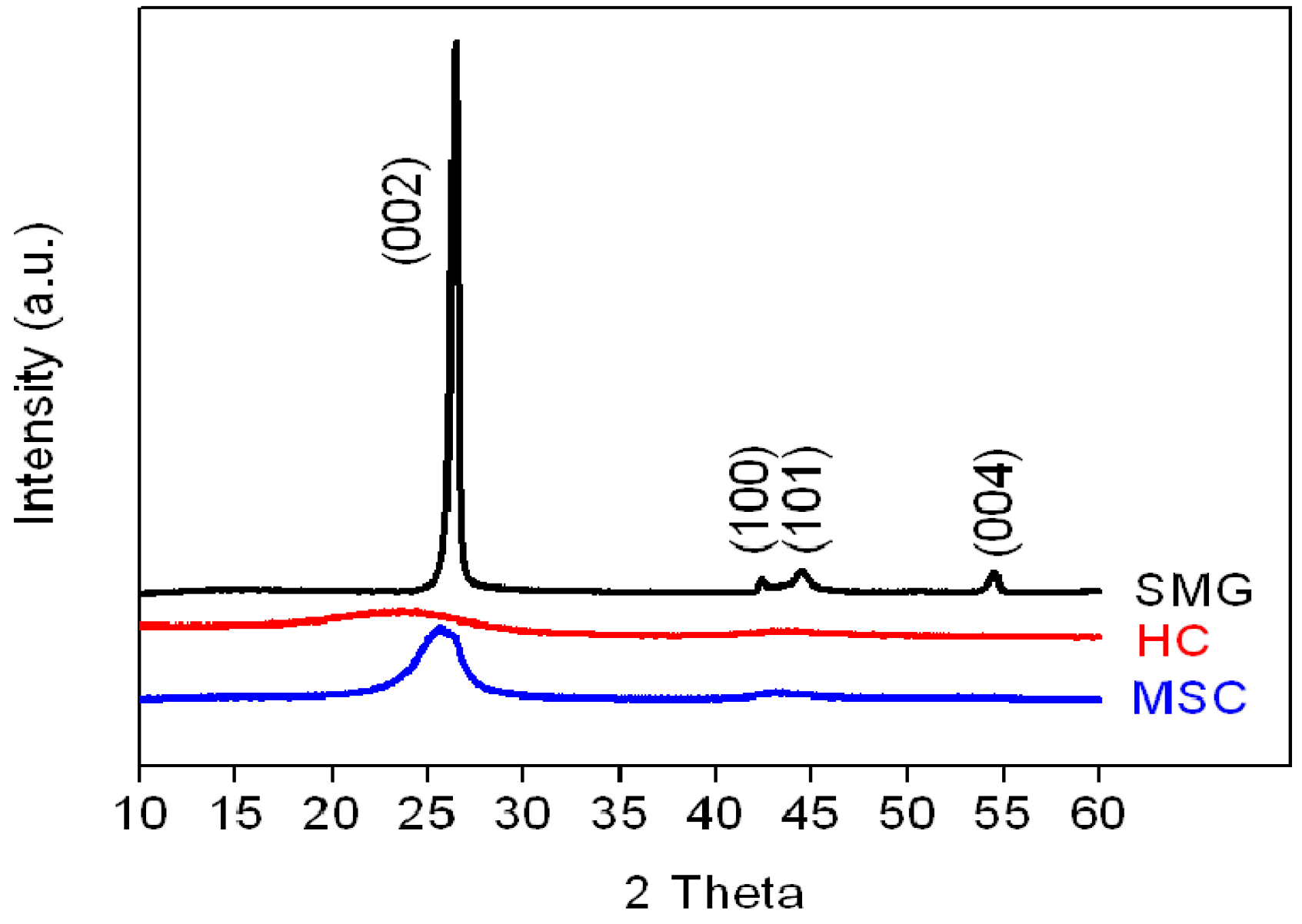
The X-ray diffraction patterns of the three anode materials are shown in
Figure 2.
Figure 2.
XRD patterns of SMG, HC, and MSC anode materials.
Figure 2.
XRD patterns of SMG, HC, and MSC anode materials.
Compared to the weak and broad (002) peaks of the HC and MSC samples at 2θ = 26.8°, the corresponding diffraction peak of SMG has a relatively high intensity, indicating that high-temperature heat treatment at 3000 °C markedly increases the crystallinity of the SMG. The calculated interlayer spacings (d002 value) of SMG (0.3368 nm) are smaller than those of MSC and HC (0.3478 and 0.3794 nm, respectively, as listed in
Table 2). For the HC and MSC materials, the weak and broad (002) and (100) diffraction peaks suggest the presence of small domains of coherent and parallel stacked graphene sheets.
Table 2.
Structural parameters of the SMG, HC and MSC anode materials estimated from XRD patterns and Raman spectroscopy.
Table 2.
Structural parameters of the SMG, HC and MSC anode materials estimated from XRD patterns and Raman spectroscopy.
| Sample | d(002) (nm) a | Lc (nm) b | La (nm) c | R value d |
|---|
| SMG | 0.3368 | 20.7 | 45.1 | 0.20 |
| HC | 0.3794 | 1.0 | 4.0 | 1.09 |
| MSC | 0.3478 | 3.2 | 5.1 | 0.90 |
The values of the Lc parameters, evaluated from the line widths of the (002) peak (used to determine the relative degree of crystallinity of SMG, HC and MSC), are 20.7 nm, 1.0 nm and 3.2 nm, respectively. The large Lc value for SMG can be attributed to its highly stacked parallel crystallinity, which is consistent with its ordered structure; while the graphene layers of MSC are observed in broad and curved structural units. These results are consistent with the TEM images for SMG and MSC, see
Figure 3a,b.
Figure 3.
TEM images of (a) SMG and (b) MSC.
Figure 3.
TEM images of (a) SMG and (b) MSC.
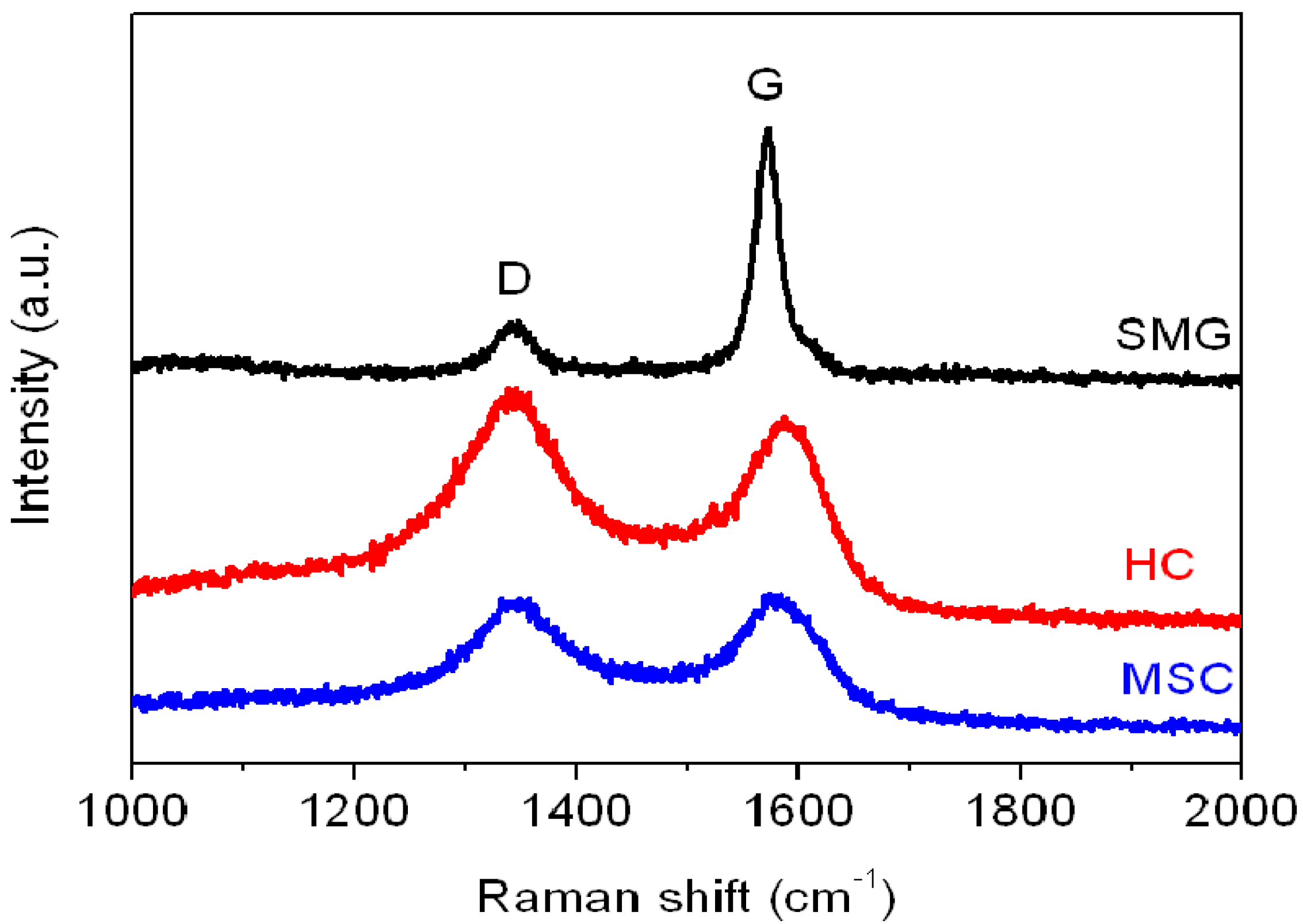
The in-plane characteristics of the anode materials were characterized from the Raman spectra, as shown in
Figure 4.
Figure 4.
Raman spectra plot of SMG, HC, and MSC anode materials.
Figure 4.
Raman spectra plot of SMG, HC, and MSC anode materials.
The average ID/IG ratio (
R value in
Table 2) evaluated from the integrated intensities of the D-band (defect-induced mode, near 1350 cm
−1) and G-band (graphite-related mode, at 1550–1605 cm
−1) were used to determine the degree of crystallinity of the anode samples. The high-temperature graphitized SMG shows a relatively lower
R (0.20) compared to HC and MSC (1.09 and 0.9, respectively). The low
R value is a characteristic of a well-developed (high crystallinity) graphite structure.
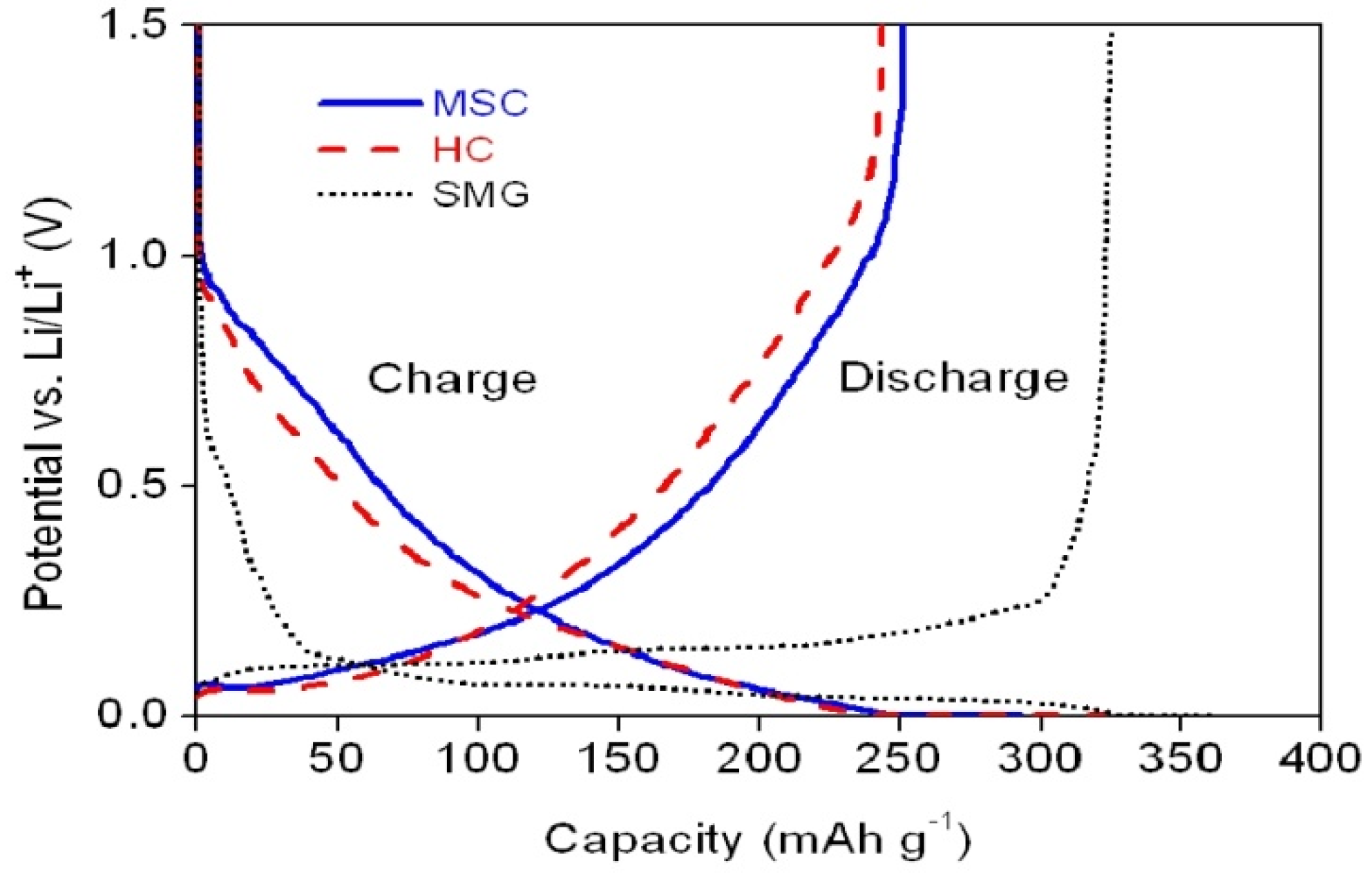
The initial charge and discharge profiles of the coin-type cells prepared with SMG, MSC and HC as anode materials are shown in
Figure 5.
Figure 5.
Initial charge/discharge curves of the coin-type cells with various anode materials.
Figure 5.
Initial charge/discharge curves of the coin-type cells with various anode materials.
During the course of charging, the intercalation of Li
+ ion in the cell with SMG anode starts at approximately 0.5–0.8 V
vs. Li/Li
+, due to the irreversible formation of a solid electrolyte interface layer on the carbon surface resulting from electrolyte decomposition and subsequent reduction with Li
+ ions [
10,
11]. There follows a long plateau, due to Li
+ ions intercalating at voltages lower than 0.2 V
vs. Li/Li
+. The cells with MSC and HC anodes show sloping potential profiles starting as 1 V
vs. Li/Li
+. This behavior is attributed to Li
+ ion intercalation into the highly distorted structure and variously distributed active sites [
12,
13]. The SMG cell exhibits both a higher initial discharge capacity (325.1 mAh·g
−1) and a greater coulombic efficiency (90%) than either the MSC and HC cells. Although the MSC and HC cells show very similar charge/discharge characteristics, the MSC anode has both a slightly higher initial capacity (248.5 mAh g
−1) and a greater coulombic efficiency (85.2%) than the HC anode (243.5 mAh g
−1 and 75.4%, respectively), as shown in
Table 3.
Table 3.
Initial charge/discharge capacities and coulombic efficiencies of the cells with various anode materials.
Table 3.
Initial charge/discharge capacities and coulombic efficiencies of the cells with various anode materials.
| Anode materials | Initial charge capacity a (mAh g−1) | Initial discharge capacity a (mAh g−1) | Initial coulombic efficiency b (%) |
|---|
| MSC | 291.6 | 248.5 | 85.2 |
| HC | 322.9 | 243.5 | 75.4 |
| SMG | 361.2 | 325.1 | 90.0 |
The discharge curves of carbonaceous anodes have been separated into 3 regions [
14,
15,
16]; the discharge plateau between 0 V and 0.12 V attributed to Li
+ de-intercalation from micropores among the stacked graphene layers; the sloping curve between 0.12 V and 0.8 V, due to the de-intercalation of Li
+ intercalated between turbostratically disordered carbonaceous interlayers; and the sloped curve at a potential higher than 0.8 V, which corresponds to the de-intercalation of Li
+ covalently bound to the edge sites of the graphene layer or large numbers of disordered micropores [
17,
18]. HC has the lowest initial coulombic efficiency among the samples, which may be due to the irreversible incorporation of Li
+ in non-crystallized sites, or defect structural voids [
19], while MSC shows a lower charge and a higher discharge capacity than HC in the initial cycle.
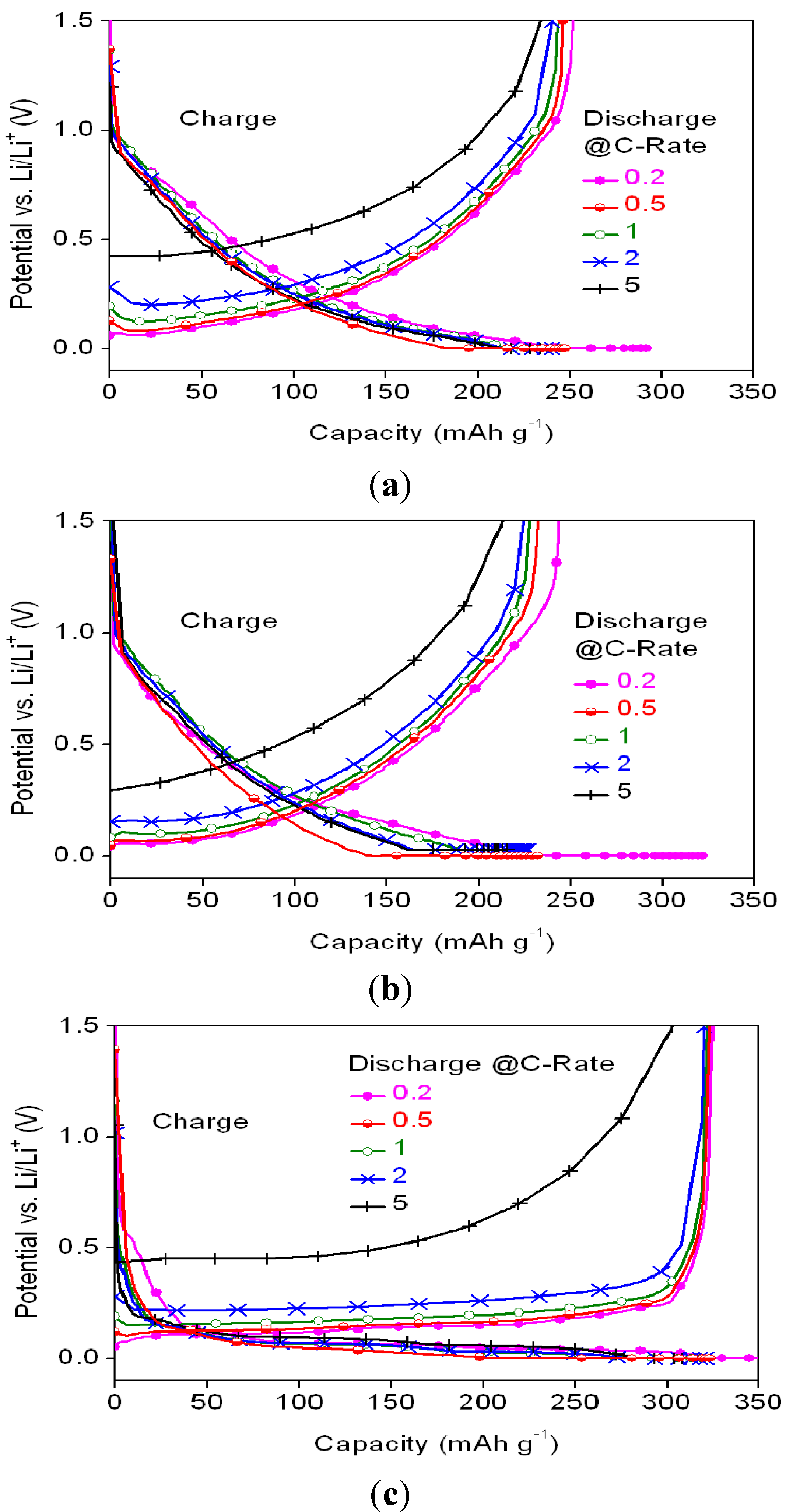
The charge/discharge profiles of different samples are shown in
Figure 6, while the ratios of the discharge capacities at various C-rates (relative to discharge at 0.2 C) for the various anode materials are listed in
Table 4.
Figure 6.
Potential profiles of the cells with (a) MSC, (b) HC, and (c) SMG anode materials at 0.2 C charge rate and various discharge C-rates.
Figure 6.
Potential profiles of the cells with (a) MSC, (b) HC, and (c) SMG anode materials at 0.2 C charge rate and various discharge C-rates.
Table 4.
Discharge capability ratios of the MSC, HC and SMG anode materials at various C-rates.
Table 4.
Discharge capability ratios of the MSC, HC and SMG anode materials at various C-rates.
| Samples | Discharge capability ratio relative to 0.2 C (%) |
|---|
| 0.5 C | 1 C | 2 C | 5 C |
|---|
| MSC | 98.0 | 96.8 | 95.1 | 93.2 |
| HC | 95.1 | 93.5 | 92.1 | 87.4 |
| SMG | 99.4 | 99.1 | 98.5 | 93.1 |
Figure 7.
Potential profiles of the cell with (a) MSC, (b) HC, and (c) SMG anode materials at various charge C-rates and 0.2 C discharge rate.
Figure 7.
Potential profiles of the cell with (a) MSC, (b) HC, and (c) SMG anode materials at various charge C-rates and 0.2 C discharge rate.
SMG and MSC show higher discharge rate capabilities than HC. The irreversible capacity loss of HC at voltages lower than 0.04 V
vs. Li
+/Li was attributed to lithium plating on the carbon surface and into the macropores [
20]. Of the three samples, SMG shows the highest reversible capacities at various discharge C-rates, with most of its capacity being seen with voltages lower than 0.5 V.
In addition to the discharge capability ratios of the MSC, HC and SMG at various C-rates, the charge capacity ratios of the anode materials at various C-rates were also determined. The charge/discharge profiles are shown in
Figure 7, while the CC-stage charging capacity and the capacity ratios at various C-rates are listed in
Figure 8 and
Table 5, respectively.
Figure 8.
Plots of CC-stage charging capacity of the MSC, HC and SMG anode materials at various C-rates.
Figure 8.
Plots of CC-stage charging capacity of the MSC, HC and SMG anode materials at various C-rates.
Table 5.
CC-stage charging capability ratio for the MSC, HC and SMG anode materials at various C-rates.
Table 5.
CC-stage charging capability ratio for the MSC, HC and SMG anode materials at various C-rates.
| Sample | CC-stage charge capacity at 0.2 C (mAh g−1) | Charging capability ratio relative to 0.2 C (%) |
|---|
| 0.5 C | 1 C | 2 C | 5 C |
|---|
| MSC | 251.5 | 81.2 | 74.4 | 61.2 | 42.0 |
| HC | 242.4 | 72.8 | 65.7 | 58.2 | 33.8 |
| SMG | 329.0 | 84.3 | 63.5 | 26.7 | 7.4 |
Of the three anode materials, SMG shows the greatest charging capacity decrease at the CC-stage. Most of its charging capacity loss occurs when the charging C-rate is higher than 2 C. Though the loss can be compensated for during the CV-stage, the charge capacity in the CV region cannot be fully utilized in the real cell because the potential of the CV-stage is close to the reduction potential of Li
+, thereby promoting lithium-dendrite deposition during charging [
21].