A Corrosion Sensor for Monitoring the Early-Stage Environmental Corrosion of A36 Carbon Steel
Abstract
:1. Introduction
2. Results and Discussion
2.1. Iron Loss during Corrosion
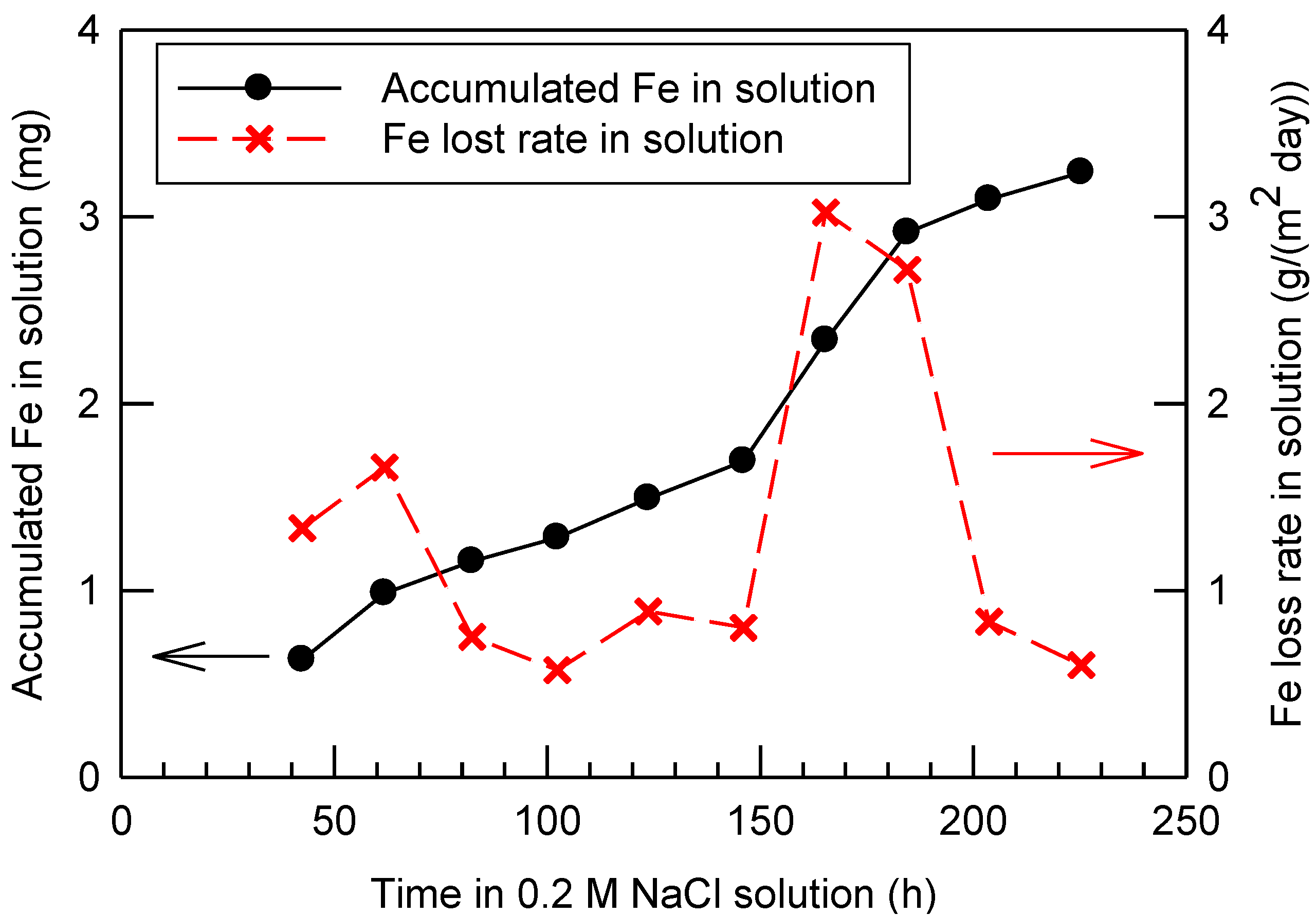
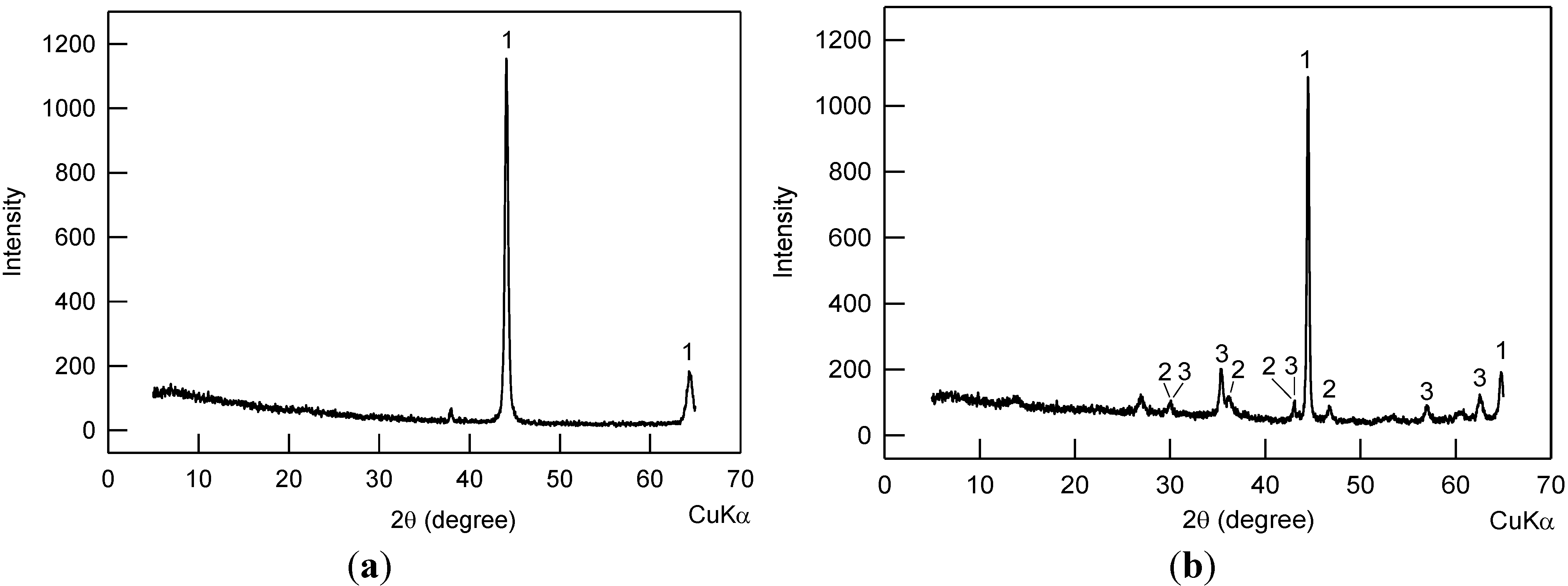
2.2. Compositions of the Rust
| Materials | α-Fe2O3 | γ-FeOOH | Fe3O4 | α-FeOOH | Amorphous Fe2O3 | Iron | Air |
|---|---|---|---|---|---|---|---|
| Electrical resistivity ρ (Ω·m) | (1.58–5.62) × 104 [25] | (0.20–0.80) × 105 [26] | 1.58 × 10−4 − 0.1 [27] | (1.30–2.33) × 105 [26] | 2.12 × 103 [28] | 1.0 × 10−7 | 4 × 1013 |
| Dielectric constant ε | 12 | 2.6 [29] | 20 | 11 [30] | 4.5 | – | 1 |
2.3. Electrical Resistance of the Sensor



2.4. Capacitance of the Sensor



3. Experimental Procedures
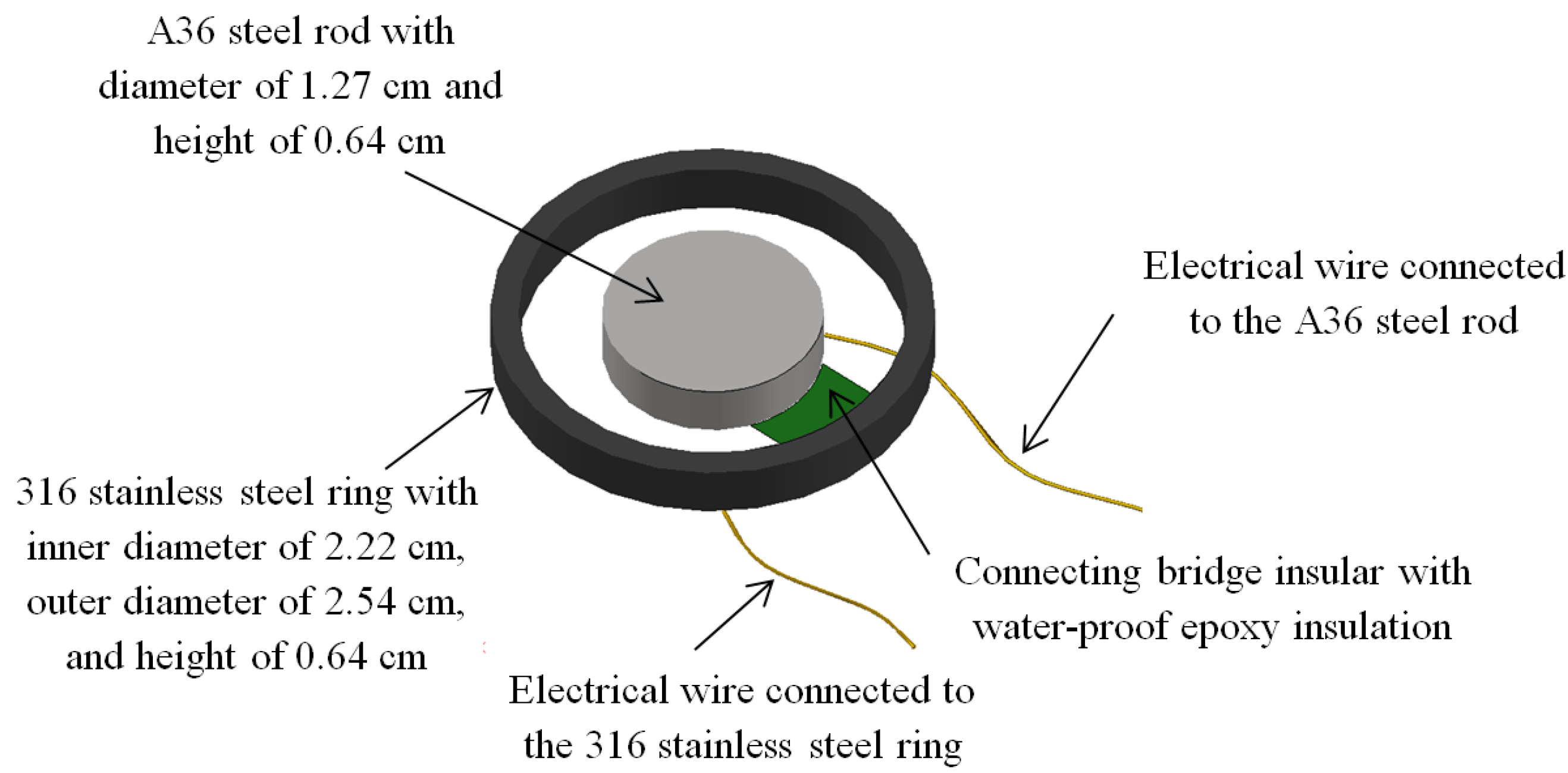
3.1. Cylindrical Capacitor
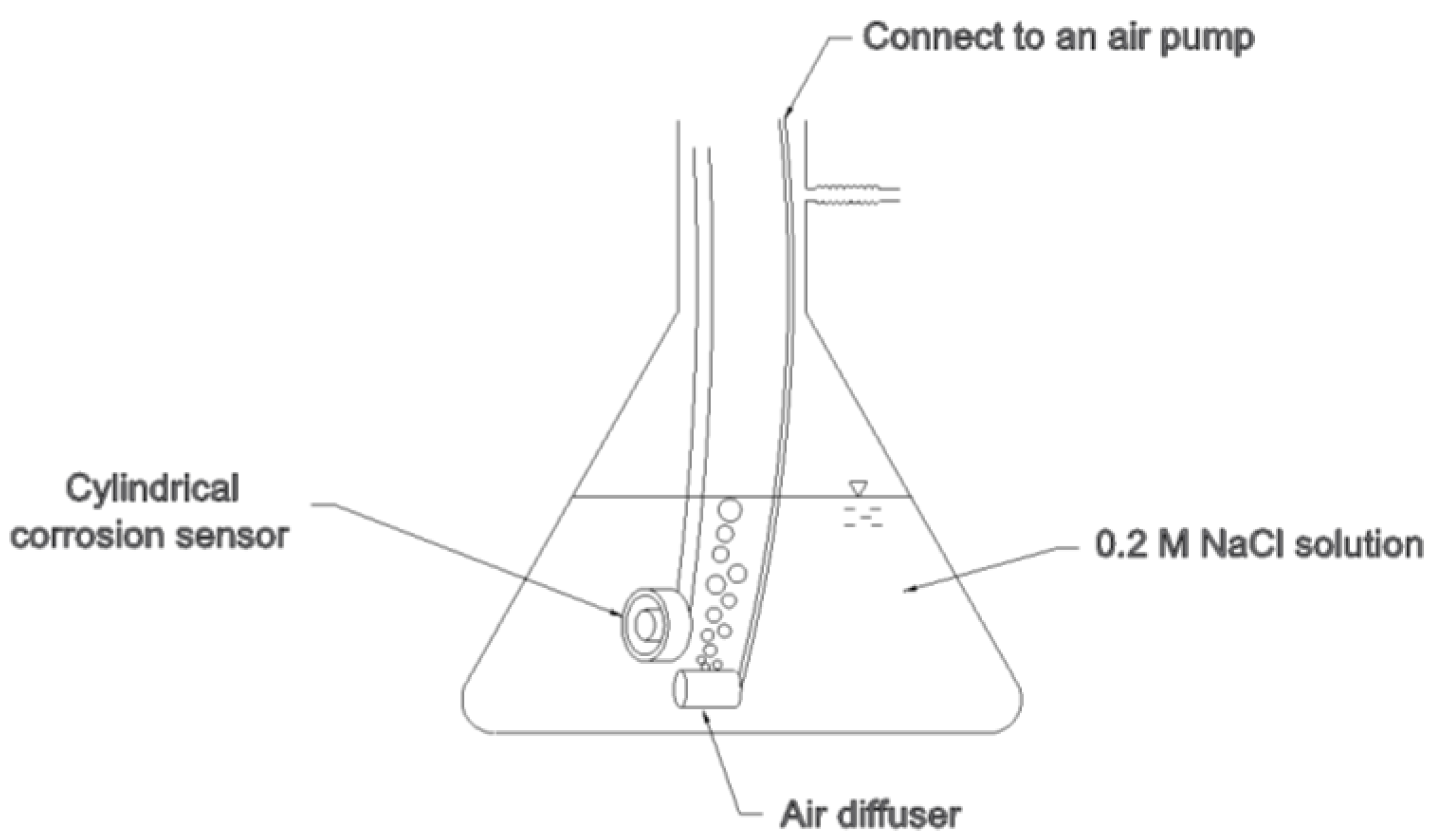
3.2. Corrosion Test
3.3. Sensor Measurements
3.4. Sample Analyses
4. Conclusions
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Nomenclature
| a | radius of the A36 steel rod (m) |
| b | inner radius of the 316 stainless steel ring (m) |
| C | sensor’s capacitance (pF) |
| h | height of the cylinder sensor (m) |
| R | electrical resistance of a material (Ω) |
| ε0 | free space permittivity, 8.85 pF/m |
| ε1 | equivalent dielectric constant of the porous iron rust on the A36 steel rod surface |
| ε2 | dielectric constant of the air gap between the iron rust on the A36 steel rod surface and the 316 stainless steel ring |
| ρ1 | equivalent electrical resistivity of the porous iron rust on the A36 steel rod surface (Ω·m) |
| ρ2 | electrical resistivity of the air gap between the iron rust on the A36 steel rod surface and the 316 stainless steel ring (Ω·m) |
| δ | average thickness of the iron rust on the A36 steel rod surface (m) |
| φ | average porosity of the iron rust on the A36 steel rod surface |
Conflicts of Interest
References
- Uhlig, H.H.; Revie, R.W. Corrosion and Corrosion Control: An Introduction to Corrosion Science and Engineering; John Wiley & Sons: New York, NY, USA, 1985. [Google Scholar]
- Borgard, B.; Warren, C.; Somayaji, S.; Heidersbach, R. In Corrosion Rates of Steel in Concrete; ASTM STP 1065. Berke, N.S., Chaker, V., Whiting, D., Eds.; American Society for Testing and Materials: Philadelphia, PA, USA, 1990; pp. 174–188. [Google Scholar]
- Fontana, M.G.; Greene, N.D. Corrosion Engineering; McGraw-Hill: New York, NY, USA, 1967; Chapter 1. [Google Scholar]
- Soares, C.G.; Garbatov, Y.; Zayed, A. Effect of environmental factors on steel plate corrosion under marine immersion conditions. Corros. Eng. Sci. Technol. 2011, 46, 524–541. [Google Scholar] [CrossRef]
- Montemor, M.F.; Simões, A.M.P.; Ferreira, M.G.S. Chloride-induced corrosion on reinforcing steel: From the fundamentals to the monitoring techniques. Cem. Concr. Compos. 2003, 25, 491–502. [Google Scholar] [CrossRef]
- Haleem, S.M.A.E.; Wanees, S.A.E.; Aal, E.E.A.E.; Diab, A. Environmental factors affecting the corrosion behavior of reinforcing steel II. Role of some anions in the initiation and inhibition of pitting corrosion of steel in Ca(OH)2 solutions. Corros. Sci. 2010, 52, 292–302. [Google Scholar] [CrossRef]
- Morris, W.; Vico, A.; Vázquez, M. Chloride induced corrosion of reinforcing steel evaluated by concrete resistivity measurements. Electrochim. Acta 2004, 49, 4447–4453. [Google Scholar] [CrossRef]
- Li, S.Y.; Kim, Y.G.; Jung, S.; Song, H.S.; Lee, S.M. Application of steel thin film electrical, resistance sensor for in situ corrosion monitoring. Sens. Actuators B 2007, 120, 368–377. [Google Scholar] [CrossRef]
- Zivica, V. Utilisation of electrical resistance method for the evaluation of the state of steel reinforcement in concrete and the rate of its corrosion. Constr. Build. Mater. 2000, 14, 351–358. [Google Scholar] [CrossRef]
- Reading, M.S.; Denzine, A.F. A critical comparison of corrosion monitoring techniques used in industrial applications. In Industrial Corrosion and Corrosion Control Technology; Shalaby, H.M., Al-Hashem, A., Lowther, M., Al-Besharah, J., Eds.; Kuwait Institute for Scientific Research: Kuwait City, Kuwait, 1996; pp. 511–519. [Google Scholar]
- Popova, A.; Veleva, S.; Raicheva, S. Kinetic approach to mild steel corrosion. React. Kinet. Catal. Lett. 2005, 85, 99–105. [Google Scholar] [CrossRef]
- Fontana, M.G. Corrosion Engineering, 3rd ed.; McGraw Hill: New York, NY, USA, 1987. [Google Scholar]
- Sen, R.; Liby, L.; Mullins, G. Strengthening steel bridge sections using CFRP laminates. Compos. Part B Eng. 2001, 32, 309–322. [Google Scholar] [CrossRef]
- Tavakkolizadeh, M.; Saadatmanesh, H. Fatigue strength of steel girders strengthened with carbon fiber reinforced polymer patch. J. Struct. Eng. ASCE 2003, 129, 186–196. [Google Scholar] [CrossRef]
- Evans, U.R.; Taylor, C.A.J. Mechanism of atmospheric rusting. Corros. Sci. 1972, 12, 227–246. [Google Scholar] [CrossRef]
- Haupt, S.; Strehblow, H.H. Corrosion, layer formation, and oxide reduction of passive iron in alkaline solution: A combined electrochemical and surface analytical study. Langmuir 1987, 3, 873–885. [Google Scholar] [CrossRef]
- Abd El-Maksoud, S.A. The effect of organic compounds on the electrochemical behavior of steel in acidic media. Int. J. Electrochem. Sci. 2008, 3, 528–555. [Google Scholar]
- Broomfield, J.P. Corrosion of Steel in Concrete: Understanding, Investigation and Repair; Taylor & Francis: London, UK, 2007; Chapter 2. [Google Scholar]
- Roberts, W.L.; Campbell, T.J.; Rapp, G.R., Jr. Encyclopedia of Minerals, 2nd ed.; Chapman & Hall: New York, NY, USA, 1990. [Google Scholar]
- In Mineral Powder Diffraction File: Data Book; JCPDS-International Centre for Diffraction Data. American Society for Testing and Materials: Swarthmore, PA, USA, 1986.
- Suzuki, I.; Masuko, N.; Hisamatsu, Y. Electrochemical properties of iron rust. Corros. Sci. 1979, 19, 521–535. [Google Scholar] [CrossRef]
- González, J.A.; Miranda, J.M.; Otero, E.; Feliu, S. Effect of electrochemically reactive rust layers on the corrosion of steel in a Ca(OH)2 solution. Corros. Sci. 2007, 49, 436–448. [Google Scholar] [CrossRef]
- Nishimura, N.; Katayama, H.; Noda, K.; Kodama, T. Electrochemical behavior of rust formed on carbon steel in a wet/dry environment containing chloride ions. Corrosion 2000, 56, 935–941. [Google Scholar] [CrossRef]
- Yuan, J.; Wu, X.; Wang, W.; Zhu, S.; Wang, F. The effect of surface finish on the scaling behavior of stainless steel in steam and supercritical water. Oxid. Met. 2013, 79, 541–551. [Google Scholar] [CrossRef]
- Varshney, D.; Yogi, A. Structural and electrical conductivity of Mn doped hematite (α-Fe2O3) phase. J. Mol. Struct. 2011, 995, 157–162. [Google Scholar] [CrossRef]
- Lair, V.; Antony, H.; Legrand, L.; Chaussé, A. Electrochemical reduction of ferric corrosion products and evaluation of galvanic coupling with iron. Corros. Sci. 2006, 48, 2050–2063. [Google Scholar] [CrossRef]
- Ltai, R.; Shibuya, M.; Matsumura, T.; Ishi, G. Electrical resistivity of magnetite anodes. J. Electrochem. Soc. 1971, 118, 1709–1711. [Google Scholar] [CrossRef]
- Akl, A.A. Microstructure and electrical properties of iron oxide thin films deposited by spray pyrolysis. Appl. Surf. Sci. 2004, 221, 319–329. [Google Scholar] [CrossRef]
- Glotch, T.D.; Rossman, G.R. Mid-infrared reflectance spectra and optical constants of six iron oxide/oxyhydroxide phases. Icarus 2009, 204, 663–671. [Google Scholar] [CrossRef]
- Filius, J.D.; Lumsdon, D.G.; Meeussen, J.C.L.; Hiemstra1, T.; van Riemsdijk, W.H. Adsorption of fulvic acid on goethite. Geochim. Cosmochim. Acta 2000, 64, 51–60. [Google Scholar] [CrossRef]
- Haynes, W.M. CRC Handbook of Chemistry and Physics,, 91st ed.; CRC Press: Boca Raton, FL, USA, 2010. [Google Scholar]
- Basu, B.N. Electromagnetic Theory and Applications in Beam-Wave Electronics; World Scientific: Singapore, Singapore, 1996. [Google Scholar]
- In Standard Specification for Carbon Structural Steel; ASTM Standard A36/A36M-08. ASTM International: West Conshohocken, PA, USA, 2008.
- Park, J.C.; Lee, M.S.; Han, D.W.; Lee, D.H.; Park, B.J.; Lee, I.S.; Uzawa, M.; Aihara, M.; Takatori, K. Inactivation of Vibrio parahaemolyticus in effluent seawater by alternating-current treatment. Appl. Environ. Microbiol. 2004, 70, 1833–1835. [Google Scholar] [CrossRef]
- Kortum, G. Treatise on Electrochemistry; Elsevier: London, UK, 1965. [Google Scholar]
- Fernandes, S.Z.; Mehendale, S.G.; Verkatachalam, S. Influence of frequency of alternating current on the electrochemical dissolution of mild steel and nickel. J. Appl. Electrochem. 1980, 10, 649–654. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chen, D.; Yen, M.; Lin, P.; Groff, S.; Lampo, R.; McInerney, M.; Ryan, J. A Corrosion Sensor for Monitoring the Early-Stage Environmental Corrosion of A36 Carbon Steel. Materials 2014, 7, 5746-5760. https://doi.org/10.3390/ma7085746
Chen D, Yen M, Lin P, Groff S, Lampo R, McInerney M, Ryan J. A Corrosion Sensor for Monitoring the Early-Stage Environmental Corrosion of A36 Carbon Steel. Materials. 2014; 7(8):5746-5760. https://doi.org/10.3390/ma7085746
Chicago/Turabian StyleChen, Dong, Max Yen, Paul Lin, Steve Groff, Richard Lampo, Michael McInerney, and Jeffrey Ryan. 2014. "A Corrosion Sensor for Monitoring the Early-Stage Environmental Corrosion of A36 Carbon Steel" Materials 7, no. 8: 5746-5760. https://doi.org/10.3390/ma7085746





