Corrosion Behavior of Titanium in Artificial Saliva by Lactic Acid
Abstract
:1. Introduction
2. Results and Discussion
2.1. pH Results
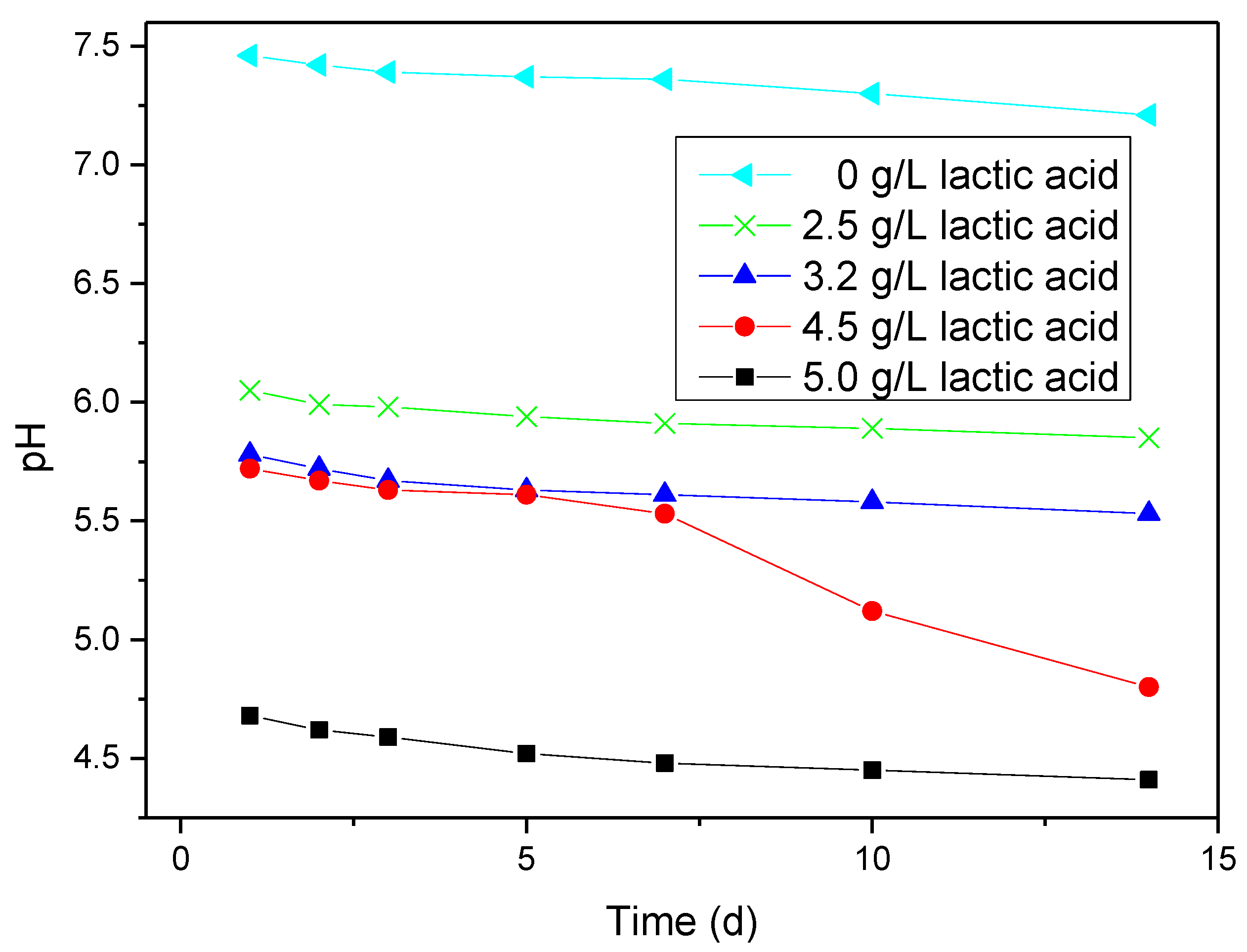
2.2. Open-Circuit Potential (OCP) Measurements
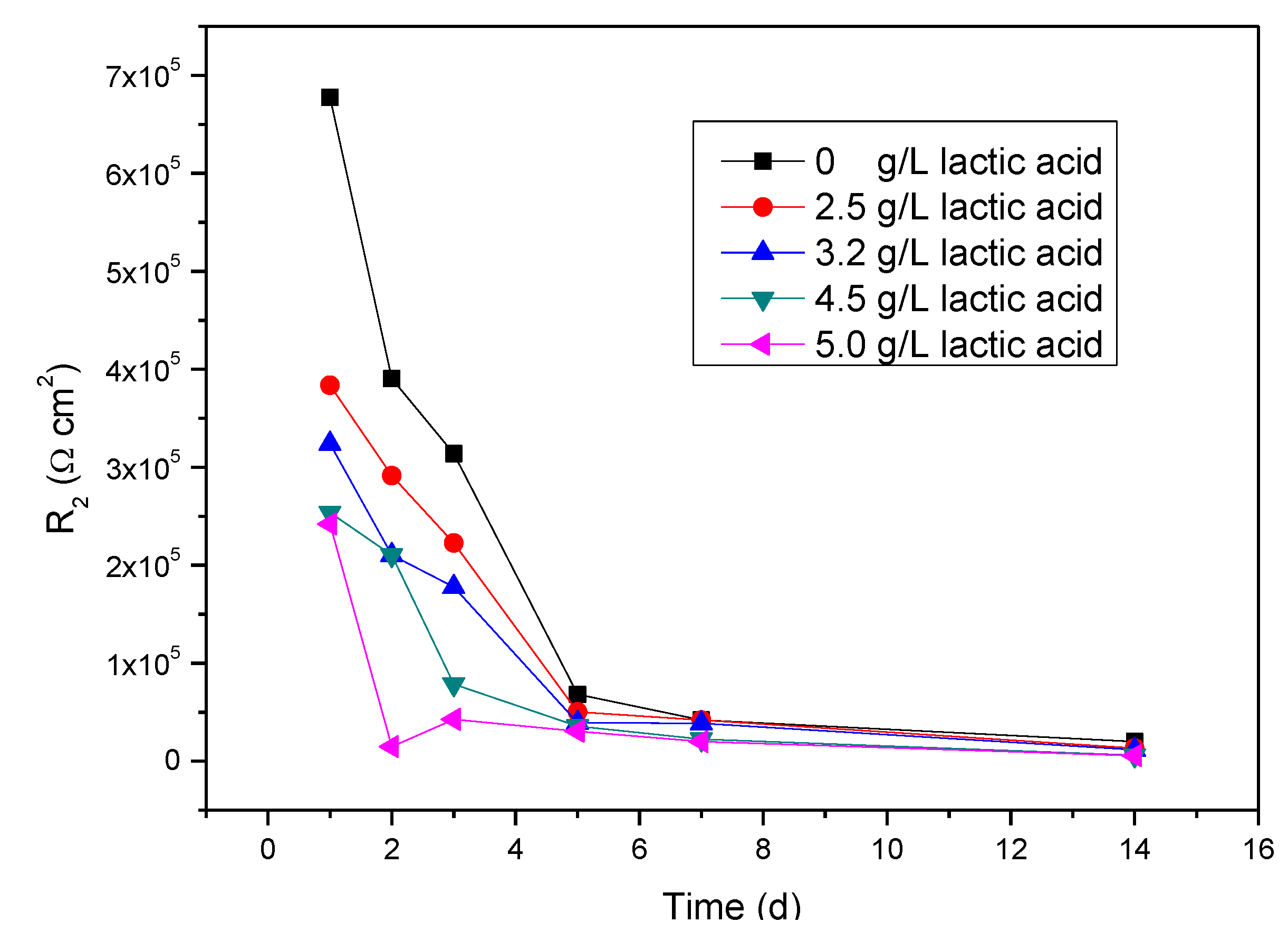
2.3. Electrochemical Impedance Spectroscopy (EIS) Studies



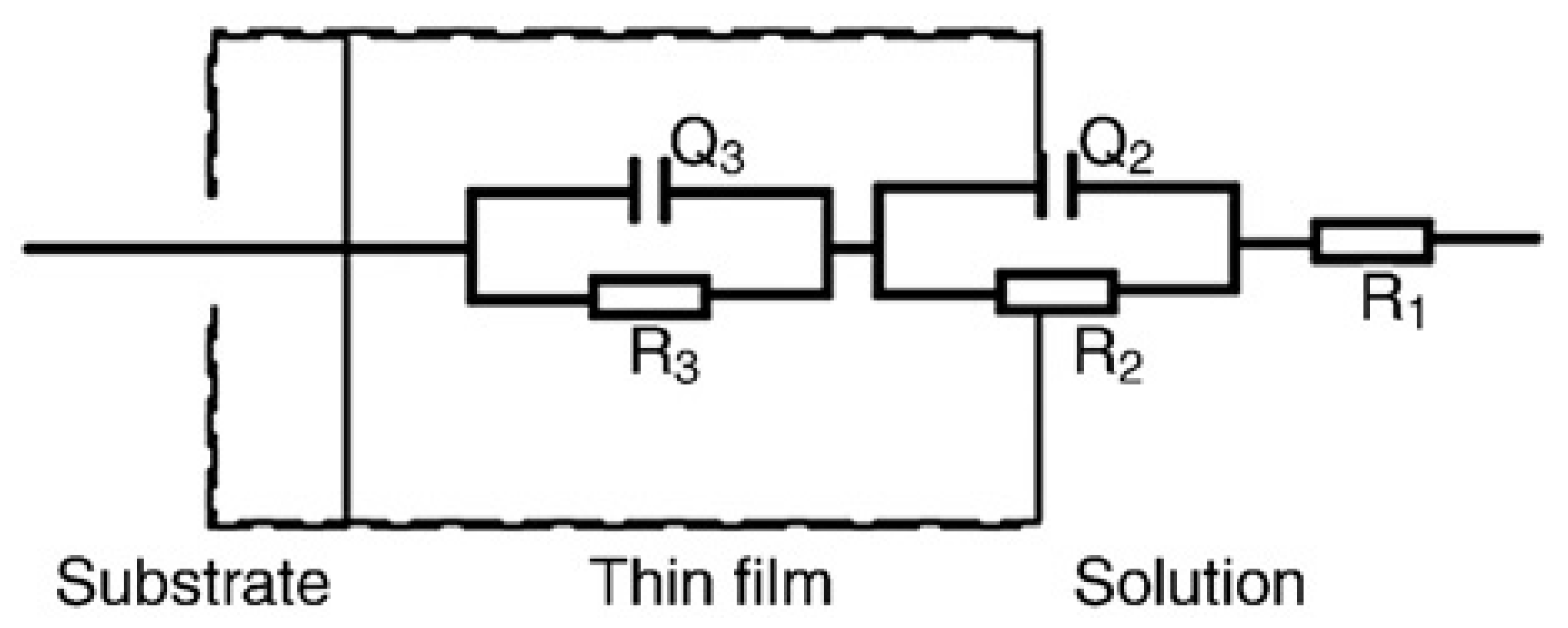
| Lactic acid | Time | R1 | Q2 | n1 | R3 | Q3 | n2 | R2 |
|---|---|---|---|---|---|---|---|---|
| g/L | days | Ω·cm2 | Ω−1·cm−2·sn | Ω·cm2 | Ω−1·cm−2·sn | Ω·cm2 | ||
| 0.0 | 1 | 17.45 | 360.5 | 1.000 | 98.14 | 97.96 | 1.000 | 6.776 × 105 |
| 2 | 13.18 | 224.9 | 0.8957 | 150.9 | 96.00 | 0.8950 | 3.904 × 105 | |
| 3 | 6.328 | 390.7 | 0.8525 | 444.1 | 91.99 | 0.8889 | 3.139 × 105 | |
| 5 | 5.841 | 205.6 | 0.8342 | 653.1 | 303.3 | 0.8714 | 6.829 × 104 | |
| 7 | 5.741 | 124.8 | 0.7561 | 980.3 | 920.4 | 0.8191 | 4.209 × 104 | |
| 14 | 5.370 | 229.3 | 0.7335 | 988.2 | 803.3 | 0.6076 | 1.999 × 104 | |
| 2.5 | 1 | 10.01 | 923.5 | 0.9906 | 6.283 | 91.42 | 0.9216 | 3.837 × 105 |
| 2 | 9.681 | 131.7 | 0.8798 | 8.922 | 85.96 | 0.8169 | 2.914 × 105 | |
| 3 | 8.423 | 397.1 | 0.8458 | 10.53 | 104.1 | 0.8316 | 2.227 × 105 | |
| 5 | 7.605 | 381.6 | 0.8359 | 30.83 | 956.0 | 0.8012 | 5.030 × 104 | |
| 7 | 6.794 | 507.2 | 0.8123 | 61.64 | 872.3 | 0.7169 | 4.1998 × 104 | |
| 14 | 5.025 | 773.6 | 0.7737 | 8.940 | 796.2 | 0.6574 | 1.336 × 104 | |
| 3.2 | 1 | 6.896 | 928.8 | 0.9959 | 7.392 | 94.92 | 0.8924 | 3.240 × 105 |
| 2 | 6.021 | 566.3 | 0.9378 | 8.028 | 20.16 | 0.8854 | 2.103 × 105 | |
| 3 | 5.106 | 793.8 | 0.8991 | 1487 | 15.67 | 0.8871 | 1.779 × 105 | |
| 5 | 4.705 | 96.5 | 0.8086 | 973.2 | 11.63 | 0.8655 | 3.944 × 104 | |
| 7 | 3.182 | 62.3 | 0.5844 | 424.7 | 946.8 | 0.7378 | 3.877 × 104 | |
| 14 | 2.737 | 134.7 | 0.5987 | 366.3 | 134.0 | 0.6570 | 1.172 × 104 | |
| 4.5 | 1 | 6.875 | 152.3 | 0.9129 | 9.001 | 98.98 | 0.8812 | 2.538 × 105 |
| 2 | 6.001 | 55.63 | 0.9108 | 8.128 | 22.16 | 0.8354 | 1.903 × 105 | |
| 3 | 5.549 | 125.7 | 0.8694 | 4.900 | 19.05 | 0.7465 | 7.584 × 104 | |
| 5 | 5.738 | 33.75 | 0.8648 | 48.89 | 96.72 | 0.7126 | 3.574 × 104 | |
| 7 | 4.922 | 50.97 | 0.8040 | 11.19 | 944.5 | 0.6515 | 2.253 × 104 | |
| 14 | 3.901 | 129.0 | 0.5053 | 121.6 | 182.3 | 0.6427 | 6,124 | |
| 5.0 | 1 | 5.191 | 281.5 | 0.8100 | 8.315 | 98.34 | 0.8390 | 2.421 × 104 |
| 2 | 4.240 | 882.8 | 0.7788 | 10.13 | 40.44 | 0.8770 | 1.483 × 104 | |
| 3 | 3.280 | 7.823 | 0.7588 | 5.109 | 38.48 | 0.7883 | 4.274 × 104 | |
| 5 | 2.980 | 44.64 | 0.7395 | 1546 | 34.10 | 0.7279 | 3.054 × 104 | |
| 7 | 2.380 | 224.2 | 0.5377 | 90.03 | 790.1 | 0.6072 | 1.998 × 104 | |
| 14 | 2.481 | 81.19 | 0.5219 | 45.68 | 117.9 | 0.5094 | 5,726 |
2.4. Polarization Curves Tests
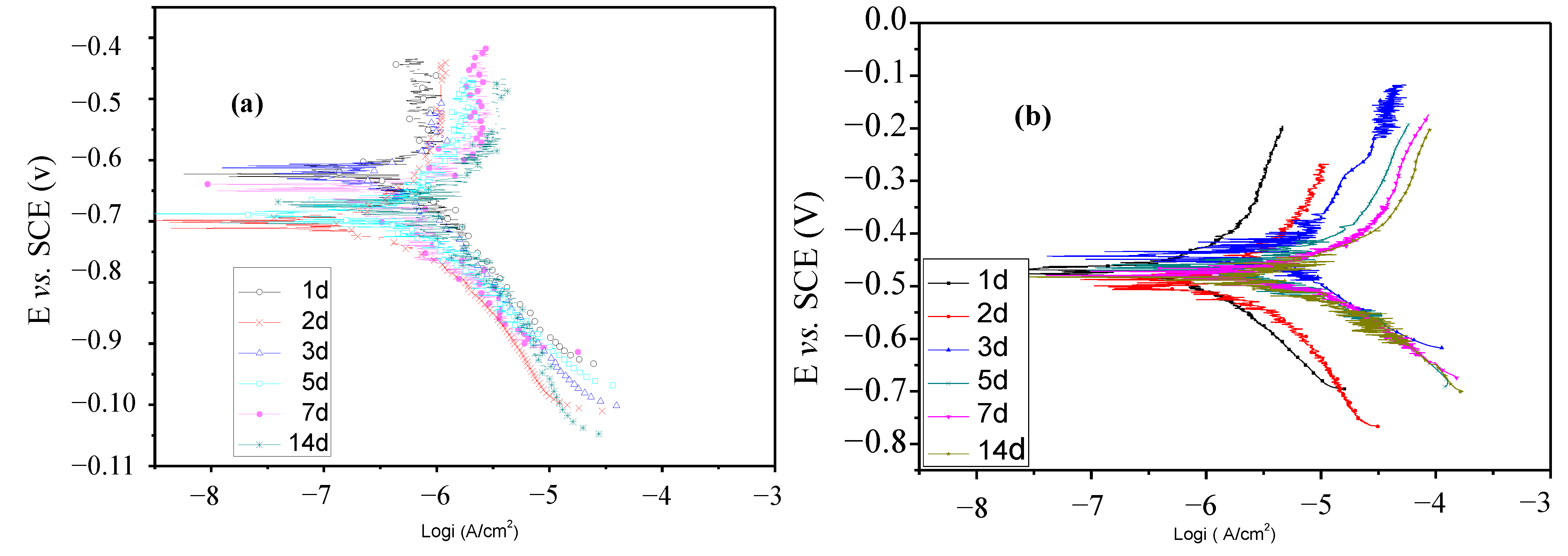
| Lactic acid g/L | Time d | Ecorr mV | Icorr μA/cm2 | ba mV/dec | bc mV/dec |
|---|---|---|---|---|---|
| 0.0 | 1 | −560.452 | 0.221 | 166.573 | 194.813 |
| 2 | −615.427 | 0.464 | 156.569 | 523.672 | |
| 3 | −623.942 | 0.664 | 247.394 | 279.867 | |
| 5 | −689.747 | 0.727 | 188.126 | 460.020 | |
| 7 | −672.599 | 1.367 | 325.928 | 247.620 | |
| 14 | −701.093 | 1.597 | 323.461 | 167.816 | |
| 2.5 | 1 | −558.415 | 0.483 | 243.754 | 832.327 |
| 2 | −589.474 | 0.727 | 188.126 | 460.020 | |
| 3 | −603.838 | 0.856 | 222.604 | 480.795 | |
| 5 | −661.228 | 1.270 | 257.742 | 752.736 | |
| 7 | −688.779 | 1.957 | 256.943 | 127.068 | |
| 14 | −692.844 | 2.957 | 112.101 | 390.369 | |
| 3.2 | 1 | −367.863 | 0.605 | 113.954 | 193.862 |
| 2 | −362.064 | 0.940 | 145.262 | 181.065 | |
| 3 | −436.498 | 1.185 | 177.877 | 154.509 | |
| 5 | −437.044 | 1.718 | 156.346 | 298.723 | |
| 7 | −438.890 | 2.374 | 98.356 | 221.833 | |
| 14 | −458.169 | 3.602 | 119.909 | 254.598 | |
| 4.5 | 1 | −309.275 | 1.158 | 317.385 | 331.341 |
| 2 | −369.804 | 1.959 | 285.187 | 620.453 | |
| 3 | −440.754 | 2.587 | 204.845 | 337.687 | |
| 5 | −433.871 | 3.216 | 117.898 | 250.677 | |
| 7 | −478.407 | 4.373 | 129.221 | 225.129 | |
| 14 | −479.760 | 5.003 | 150.240 | 193.490 | |
| 5.0 | 1 | −368.589 | 2.226 | 310.458 | 760.726 |
| 2 | −393.751 | 3.420 | 304.961 | 405.529 | |
| 3 | −443.304 | 3.979 | 134.820 | 276.343 | |
| 5 | −466.912 | 4.680 | 143.859 | 226.051 | |
| 7 | −477.709 | 5.615 | 136.394 | 233.153 | |
| 14 | −483.366 | 7.788 | 160.194 | 236.892 |
2.5. Scanning Electron Microscopy Analysis

2.6. Corrosion Mechanism
3. Experimental Section
3.1. Preparation of the Specimens
3.2. Medium
3.3. pH Tests
3.4. Electrochemical Examinations
3.5. Corrosion Morphology
4. Conclusions
- (1)
- The corrosion of titanium in artificial saliva will result in a slight decrease in the pH value of the solution. Additionally, the corrosion increases with increasing the immersion time.
- (2)
- The addition of lactic acid into artificial saliva solutions can distinctly accelerate the corrosion rate, and the corrosion of titanium is aggravated with increasing the amount of lactic acid.
- (3)
- SEM indicates that lactic acid can accelerate the pitting corrosion in artificial saliva.
- (4)
- Lactic acid is apt to form a chelate compound ([Ti(OH)3]·L), which dissolves in water. The formation of [Ti(OH)3]·L accelerates the dissolution of passivation film (TiO2) on titanium, and this causes the deficiency of the protective film, leading to a tendency of pitting corrosion.
- (5)
- The addition of lactic acid does change the mechanism, but accelerates the pitting corrosion.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Cremasco, A.; Osório, W.R.; Freire, C.M.A.; Garcia, A.; Caram, R. Electrochemical corrosion behavior of a Ti-35Nb alloy. Electrochim. Acta 2008, 53, 4867–4874. [Google Scholar] [CrossRef]
- Ohkubo, C.; Hanatani, S.; Hosoi, T. Present status of titanium removable dentures—A review of the literature. J. Oral. Rehabil. 2008, 35, 706–714. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, C.; Barbosa, M.A. Corrosion behavior of titanium in biofluids containing H2O2 studied by electrochemical impedance spectroscopy. Corros. Sci. 2001, 43, 547–559. [Google Scholar] [CrossRef]
- Long, M.; Rack, H.J. Titanium alloys in total joint replacement—A materials science perspective. Biomaterials 1998, 19, 1621–1639. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.F. Biocompatibility of clinical implant materials. In Biocompatibility of Clinical Implant Materials; Williams, D.F., Ed.; Volume 1, CRC Press: Boca Raton, FL, USA, 1981; pp. 9–44. [Google Scholar]
- Pan, J.; Thierry, D.; Leygraf, C. Electrochemical impedance spectroscopy study of the passive oxide film on titanium for implant application. Electrochim. Acta 1996, 41, 1143–1153. [Google Scholar] [CrossRef]
- Kovacs, P.; Davidson, J.A. Chemical and electrochemical aspects of the biocompatibility of titanium and its alloys. In ASTM STP 1272 The Material and Biological Issues; ASTM International: West Conshohocken, PA, USA, 1996; pp. 163–178. [Google Scholar]
- Mareci, D.; Ungureanu, G.; Aelenei, D.; Mirza Rosca, J.C. Electrochemical characteristics of titanium based biomaterials in artificial saliva. Mater. Corros. 2007, 11, 848–856. [Google Scholar] [CrossRef]
- Strietzel, R.; Hosch, A.; Kalbfleisch, H.; Buch, D. In vitro corrosion of titanium. Biomaterials 1998, 19, 1495–1499. [Google Scholar] [CrossRef] [PubMed]
- Hanawa, T.; Kon, M.; Ohkawa, S.; Asaoka, K. Diffusion of elements in porcelain into titanium oxide. Dent. Mater. J. 1994, 13, 164–173. [Google Scholar] [CrossRef] [PubMed]
- Esposito, M.; Lausmaa, J.; Hirsch, J.M.; Thomsen, P. Surface analysis of failed oral titanium implants. J. Biomed. Mater. Res. 1999, 48, 559–568. [Google Scholar] [CrossRef] [PubMed]
- Lopez, M.F.; Jimenez, J.A.; Gutierrez, A. Corrosion study of surface-modified vanadium-free titanium alloys. Electrochim. Acta 2003, 48, 1395–1401. [Google Scholar] [CrossRef]
- Dearnley, P.A.; Dahm, K.L.; Cimenoǧlu, H. The corrosion-wear behavior of thermally oxidised CP-Ti and Ti–6Al–4V. Wear 2004, 256, 469–479. [Google Scholar] [CrossRef]
- Velten, D.; Biehl, V.; Aubertin, F.; Valeske, B.; Possart, W.; Breme, J. Preparation of TiO2 layers on cp-Ti and Ti6Al4V by thermal and anodic oxidation and by sol-gel coating techniques and their characterization. J. Biomed. Mater. Res. 2002, 59, 18–28. [Google Scholar] [CrossRef] [PubMed]
- Merritt, K.; Rodrigo, J.J. Immune response to synthetic materials. Sensitization of patients receiving orthopaedic implants. Clin. Orthop. 1996, 326, 71–79. [Google Scholar]
- Urban, R.M.; Jacobs, J.J.; Tomlinson, M.J.; Gavrilovic, J.; Black, J.; Peoch, M. Dissemination of wear particles to the liver, spleen, and abdominal lymph nodes of patients with hip or knee replacement. J. Bone Joint. Surg. Am. 2000, 82, 457–476. [Google Scholar] [CrossRef] [PubMed]
- Lalor, P.A.; Revell, P.A.; Gray, A.B.; Wright, S.; Railton, G.T.; Freeman, M.A. Sensitivity to titanium. A cause of implant failure? J. Bone Joint. Surg. Br. 1991, 73B, 25–28. [Google Scholar]
- Abdallah, H.I.; Balsara, R.K.; O’Riordan, A.C. Pacemaker contact sensitivity: clinical recognition and management. Ann. Thorac. Surg. 1994, 57, 1017–1018. [Google Scholar] [CrossRef] [PubMed]
- Yamauchi, R.; Morita, A.; Tsuji, T. Pacemaker dermatitis from titanium. Contact Dermatitis 2000, 42, 52–53. [Google Scholar] [PubMed]
- Basketter, D.A.; Whittle, E.; Monk, B. Possible allergy to complex titanium salt. Contact Dermatitis 2000, 42, 310–311. [Google Scholar] [CrossRef] [PubMed]
- Pohler, O.E.M. Unalloyed titanium for implants in bone surgery. Injury 2000, 31, D7–D13. [Google Scholar] [CrossRef]
- Green, T.; Machtei, E.; Horwitz, J.; Peled, M. Fracture of dental implants: Literature review and report of a case. Implant. Dent. 2002, 11, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Grimsdottir, M.R.; Hensten-Pettersen, A. Cytotoxic and antibacterial effects of orthodontic appliances. Scand. J. Dent. Res. 1993, 101, 229–231. [Google Scholar] [PubMed]
- Mabilleau, G.; Bourdon, S.; Joly-Guillou, M.L.; Filmon, R.; Baslé, M.F.; Chappard, D. Influence of fluoride, hydrogen peroxide and lactic acid on the corrosion resistance of commercially pure titanium. Acta Biomater. 2006, 2, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Koike, M.; Nakamura, S.; Fujii, H. In vitro assessment of release from titanium by immersion tests. J. Jpn. Prosthodont. Soc. 1997, 41, 675–679. [Google Scholar] [CrossRef]
- Koike, M.; Fujii, H. In vitro assessment of corrosive properties of titanium as a biomaterial. J. Oral Rehabil. 2001, 28, 540–548. [Google Scholar] [CrossRef] [PubMed]
- Masatoshi, T.; Masafumi, K.; Yukyo, T. Corrosion behavior of Ti-Ag alloys used in dentistry in lactic acid solution. Met. Mater. Int. 2011, 17, 175–179. [Google Scholar] [CrossRef]
- Koike, M.; Fujii, H. The corrosion resistance of pure titanium in organic acids. Biomaterials 2001, 22, 2931–2936. [Google Scholar] [CrossRef] [PubMed]
- Mansfeld, F.; Kending, M.W.; Tsai, S. Recording and analysis of A.C. impedance data for corrosion studies. I. Background and methods of analysis. Corrosion 1981, 37, 301–307. [Google Scholar]
- Tanno, K.; Itoh, M.; Sekiya, H.; Yashiro, H.; Kumagai, N. The corrosion inhibition of carbon steel in lithium bromide solution by hydroxide and molybdate at moderate temperatures. Corros. Sci. 1993, 34, 1453–1461. [Google Scholar] [CrossRef]
- Mansfeld, F. The interaction of bacteria and metal surfaces. Electrochim. Acta 2007, 52, 7670–7680. [Google Scholar] [CrossRef]
- Tian, B.; Xie, D.B.; Wang, F.H. Corrosion behavior of TiN and TiN/Ti composite films on Ti6Al4V alloy in Hank’s solution. J. Appl. Electrochem. 2009, 39, 447–453. [Google Scholar] [CrossRef]
- Balakrishnan, A.; Lee, B.C.; Kim, T.N.; Panigrahi, B.B. Corrosion behaviour of ultra fine grained titanium in simulated body fluid for implant application. Trends Biomater. Artif. Organs. 2008, 22, 58–64. [Google Scholar]
- Fekry, A.M. The influence of chloride and sulphate ions on the corrosion behavior of Ti and Ti-6Al-4V in oxalic acid. Electrochem. Acta 2009, 54, 3480–3489. [Google Scholar] [CrossRef]
- Chen, Y.J. Effect of Streptococcus Sanguis and Lactic Acid on the Corrosion Behavior of Titanium. Ph.D. Thesis, Yunnan University, Kunming, China, 2013. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Qu, Q.; Wang, L.; Chen, Y.; Li, L.; He, Y.; Ding, Z. Corrosion Behavior of Titanium in Artificial Saliva by Lactic Acid. Materials 2014, 7, 5528-5542. https://doi.org/10.3390/ma7085528
Qu Q, Wang L, Chen Y, Li L, He Y, Ding Z. Corrosion Behavior of Titanium in Artificial Saliva by Lactic Acid. Materials. 2014; 7(8):5528-5542. https://doi.org/10.3390/ma7085528
Chicago/Turabian StyleQu, Qing, Lei Wang, Yajun Chen, Lei Li, Yue He, and Zhongtao Ding. 2014. "Corrosion Behavior of Titanium in Artificial Saliva by Lactic Acid" Materials 7, no. 8: 5528-5542. https://doi.org/10.3390/ma7085528




