Blends and Nanocomposite Biomaterials for Articular Cartilage Tissue Engineering
Abstract
:1. Introduction
| Polymers | Examples |
|---|---|
| Natural polymers | Proteins: collagen [13], gelatin [14], fibrin glue [15] Polysaccarides: Agarose [16], alginate [17], cellulose [18], chitosan [19], chondroitin sulphate [20], and hyaluronic acid [21] |
| Synthetic polymers | poly(α-hydroxy esters): Poly(L-lactic-co-glycolic acid) [22], poly(ε-caprolactone) [23], Poly(NiPAAm) [24], poly(vinyl alcohol) [25], Polyurethane [26] |
2. Articular Cartilage Tissue Engineering
2.1. Structure-Property Relationships of Native Articular Cartilage
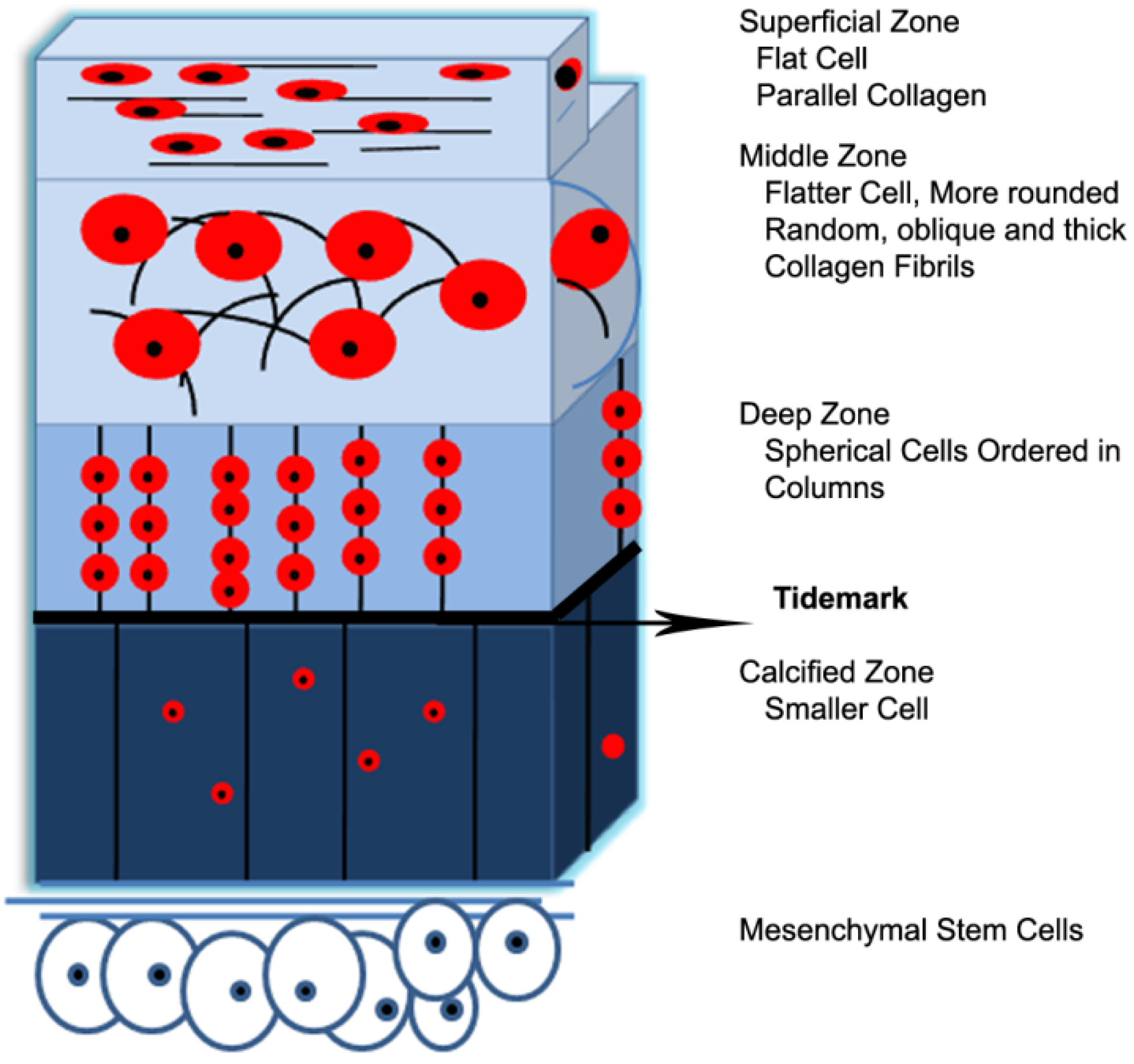
| Mechanical Properties | Articular Cartilage |
|---|---|
| Tensile Modulus (at 10% ε) | 5–25 MPa [62,74] |
| Equilibrium Relaxation Modulus | 6.5–45 MPa [63] |
| Elongation to Break | 80% [32] |
| Ultimate Tensile Stress | 15–35 MPa [75] |
| Equilibrium Compressive Aggregate Modulus a | 0.1–2.0 MPa [37] |
| Hydraulic Permeability | 0.5–5.0 × 10−15 m4 N−1∙s−1 [37] |
| Intrinsic, Equilibrium Young’s Modulus in Compression b | 0.4–0.8 MPa [56] |
| Compressive Strength | 14–59 MPa [76] |
| Equilibrium Shear Modulus | 0.05–0.25 MPa [77] |
2.2. Joint Disease and Medical Interventions
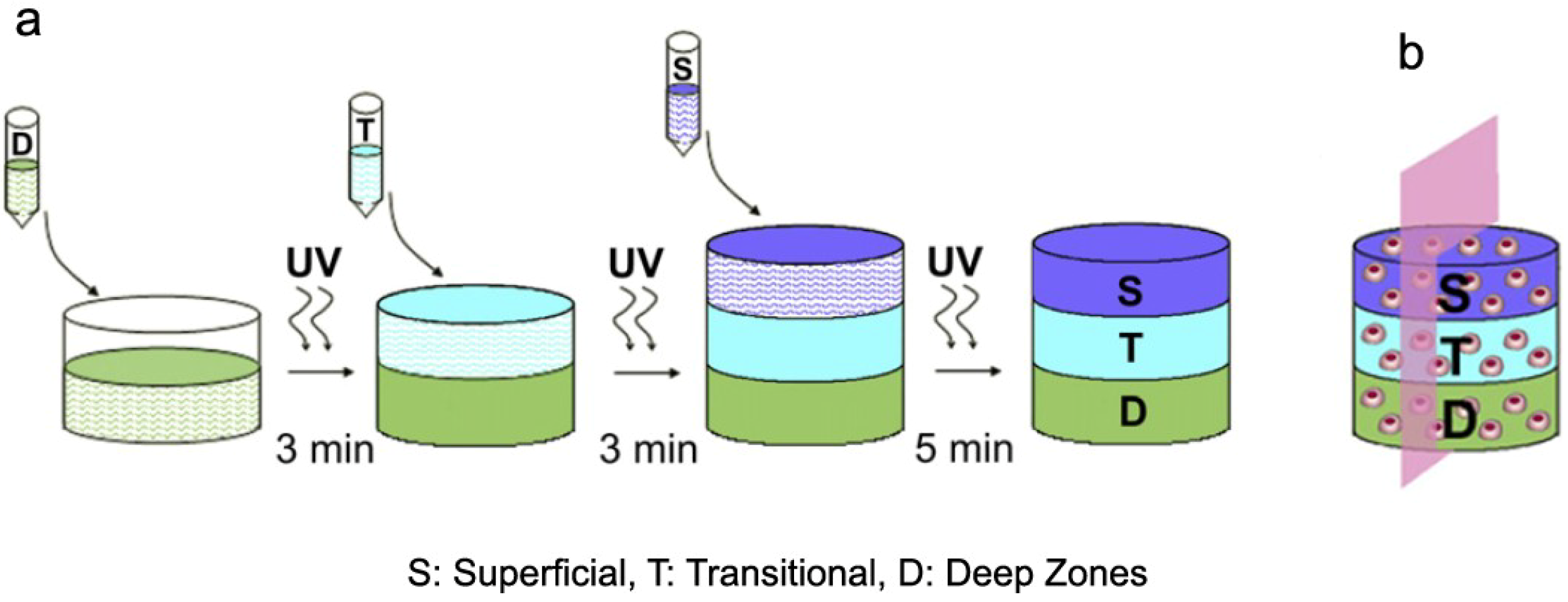
2.3. Tissue Engineering of Cartilage and Scaffold Requirements
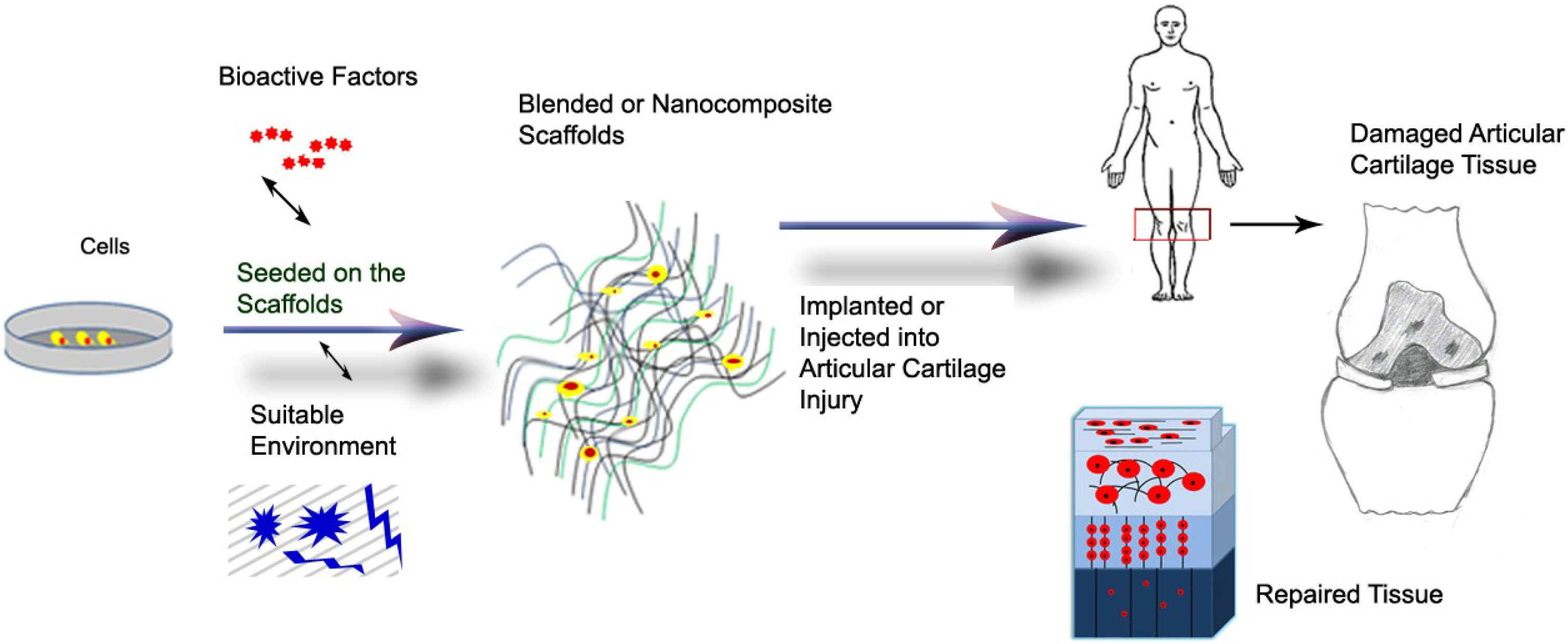
3. Biomaterial Blends
| Polymers | Disadvantages | Advantages |
|---|---|---|
| Chitosan | Low tensile and compressive properties, low processability. | Antibacterial activity, low toxicity, good cell interaction, good biocompatibility, renewability, water solubility, stability to variations of pH. |
| Collagen | Low tensile and compressive properties, high degradation rate. | Low antigenicity, good cell adhesion, biological signaling, biodegradability. |
| Hyaluronic acid | Not support thermodynamically cell attachment. Hydrophilic surface. | No immunogenicity, good cell interaction. |
| Alginates | Hard processability, low tensile properties. | Injectable polymers, easily crosslinking under mild condition, high and tunable porosity scaffold, high diffusion rates of macromolecules, good cell incorporation. |
| Poly(ε-caprolactone) | Long term degradation application due to slow degradation rate, susceptible to undergo auto-catalyzed bulk hydrolysis, hydrophobic surface then no cell interaction. | FDA approval, easily processable. |
| Polyurethane | Acidic degradation byproducts in poly(esther urethanes) causing autocatalyzed degradation and in vivo inflammation. | Good tensile and compressive properties and also biological properties such as cell attachment, incorporation and supporting chondrocyte phenotype, and low infection. |
| PLGA | Low biological properties such as cell attachment, incorporation and supporting chondrocyte phenotype, releasing acidic degradation byproducts caused inflammatory response. | FDA approval, tailorable physicomechanical properties. |
3.1. Blends with Collagen


3.2. Blends with Chondroitin Sulphate
3.3. Blends with Chitosan
3.4. Blends with PVA
4. Biomaterial Nanocomposites
4.1. Polymer-Polymer Nanofiber Composites
4.2. Polymer-Silica Nanoparticle Composites
4.3. Polymer-Hydroxyapatite Nanoparticle Composites
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kohane, D.S.; Langer, R. Polymeric biomaterials in tissue engineering. Pediatr. Res. 2008, 63, 487–491. [Google Scholar] [CrossRef]
- Cheung, H.-Y.; Lau, K.-T.; Lu, T.-P.; Hui, D. A critical review on polymer-based bio-engineered materials for scaffold development. Compos. B 2007, 38, 291–300. [Google Scholar] [CrossRef]
- Thomson, R.C.; Wake, M.C.; Yaszemski, M.J.; Mikos, A.G. Biodegradable polymer scaffolds to regenerate organs. In Biopolymers II; Peppas, N.A., Langer, R.S., Eds.; Springer: Berlin/Heidelberg, Germany, 1995; pp. 245–274. [Google Scholar]
- Temenoff, J.S.; Mikos, A.G. Injectable biodegradable materials for orthopedic tissue engineering. Biomaterials 2000, 21, 2405–2412. [Google Scholar] [CrossRef]
- Middleton, J.C.; Tipton, A.J. Synthetic biodegradable polymers as orthopedic devices. Biomaterials 2000, 21, 2335–2346. [Google Scholar] [CrossRef]
- Puppi, D.; Chiellini, F.; Piras, A.M.; Chiellini, E. Polymeric materials for bone and cartilage repair. Prog. Polym. Sci. 2010, 35, 403–440. [Google Scholar] [CrossRef]
- Nesic, D.; Whiteside, R.; Brittberg, M.; Wendt, D.; Martin, I.; Mainil-Varlet, P. Cartilage tissue engineering for degenerative joint disease. Adv. Drug Deliv. Rev. 2006, 58, 300–322. [Google Scholar] [CrossRef]
- Ratner, B.D.; Bryant, S.J. Biomaterials: Where we have been and where we are going. Annu. Rev. Biomed. Eng. 2004, 6, 41–75. [Google Scholar] [CrossRef]
- Muzzarelli, R.A.A.; Greco, F.; Busilacchi, A.; Sollazzo, V.; Gigante, A. Chitosan, hyaluronan and chondroitin sulfate in tissue engineering for cartilage regeneration: A review. Carbohydr. Polym. 2012, 89, 723–739. [Google Scholar] [CrossRef]
- Gunatillake, P.A.; Adhikari, R. Biodegradable synthetic polymers for tissue engineering. Eur. Cells Mater. 2003, 5, 1–16. [Google Scholar]
- Patterson, J.; Martino, M.M.; Hubbell, J.A. Biomimetic materials in tissue engineering. Mater. Today 2012, 13, 14–22. [Google Scholar] [CrossRef]
- Aizawa, Y.; Owen, S.C.; Shoichet, M.S. Polymers used to influence cell fate in 3D geometry: New trends. Prog. Polym. Sci. 2012, 37, 645–658. [Google Scholar] [CrossRef]
- Mimura, T.; Imai, S.; Kubo, M.; Isoya, E.; Ando, K.; Okumura, N.; Matsusue, Y. A novel exogenous concentration-gradient collagen scaffold augments full-thickness articular cartilage repair. Osteoarthr. Cartil. 2008, 16, 1083–1091. [Google Scholar] [CrossRef]
- Lien, S.-M.; Chien, C.-H.; Huang, T.-J. A novel osteochondral scaffold of ceramic-gelatin assembly for articular cartilage repair. Mater. Sci. Eng. C 2009, 29, 315–321. [Google Scholar] [CrossRef]
- Könst, Y.E.; Benink, R.J.; Veldstra, R.; van der Krieke, T.J.; Helder, M.N.; van Royen, B.J. Treatment of severe osteochondral defects of the knee by combined autologous bone grafting and autologous chondrocyte implantation using fibrin gel. Knee Surg. Sports Traumatol. Arthrosc. 2012, 20, 2263–2269. [Google Scholar] [CrossRef]
- Buschmann, M.D.; Gluzband, Y.A.; Grodzinsky, A.J.; Hunziker, E.B. Mechanical compression modulates matrix biosynthesis in chondrocyte/agarose culture. J. Cell Sci. 1995, 108, 1497–1508. [Google Scholar]
- Fragonas, E.; Valente, M.; Pozzi-Mucelli, M.; Toffanin, R.; Rizzo, R.; Silvestri, F.; Vittur, F. Articular cartilage repair in rabbits by using suspensions of allogenic chondrocytes in alginate. Biomaterials 2000, 21, 795–801. [Google Scholar] [CrossRef]
- Lopes, J.L.; Machado, J.M.; Castanheira, L.; Granja, P.L.; Gama, F.M.; Dourado, F.; Gomes, J.R. Friction and wear behaviour of bacterial cellulose against articular cartilage. Wear 2011, 271, 2328–2333. [Google Scholar] [CrossRef] [Green Version]
- Alves da Silva, M.L.; Crawford, A.; Mundy, J.M.; Correlo, V.M.; Sol, P.; Bhattacharya, M.; Hatton, P.V.; Reis, R.L.; Neves, N.M. Chitosan/polyester-based scaffolds for cartilage tissue engineering: Assessment of extracellular matrix formation. Acta Biomater. 2010, 6, 1149–1157. [Google Scholar] [Green Version]
- Basalo, I.M.; Chahine, N.O.; Kaplun, M.; Chen, F.H.; Hung, C.T.; Ateshian, G.A. Chondroitin sulfate reduces the friction coefficient of articular cartilage. J. Biomech. 2007, 40, 1847–1854. [Google Scholar] [CrossRef]
- Toh, W.S.; Lee, E.H.; Guo, X.-M.; Chan, J.K.Y.; Yeow, C.H.; Choo, A.B.; Cao, T. Cartilage repair using hyaluronan hydrogel-encapsulated human embryonic stem cell-derived chondrogenic cells. Biomaterials 2010, 31, 6968–6980. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, F.; Liu, K.; Shen, H.; Zhu, Y.; Zhang, W.; Liu, W.; Wang, S.; Cao, Y.; Zhou, G. The impact of PLGA scaffold orientation on in vitro cartilage regeneration. Biomaterials 2012, 33, 2926–2935. [Google Scholar] [CrossRef]
- Neves, S.C.; Moreira Teixeira, L.S.; Moroni, L.; Reis, R.L.; van Blitterswijk, C.A.; Alves, N.M.; Karperien, M.; Mano, J.F. Chitosan/Poly(e-caprolactone) blend scaffolds for cartilage repair. Biomaterials 2011, 32, 1068–1079. [Google Scholar] [CrossRef] [Green Version]
- Bartlett Ii, R.L.; Sharma, S.; Panitch, A. Cell-penetrating peptides released from thermosensitive nanoparticles suppress pro-inflammatory cytokine response by specifically targeting inflamed cartilage explants. Nanomedicine 2013, 9, 419–427. [Google Scholar] [CrossRef]
- Noguchi, T.; Yamamuro, T.; Oka, M.; Kumar, P.; Kotoura, Y.; Hyonyt, S.-H.; Ikada, Y. Poly (vinyl alcohol) hydrogel as an artificial articular cartilage: Evaluation of biocompatibility. J. Appl. Biomater. 1991, 2, 101–107. [Google Scholar] [CrossRef]
- Lee, C.R.; Grad, S.; Gorna, K.; Gogolewski, S.; Goessl, A.; Alini, M. Fibrin-polyurethane composites for articular cartilage tissue engineering: A preliminary analysis. Tissue Eng. 2005, 11, 1562–1573. [Google Scholar] [CrossRef]
- Sionkowska, A. Current research on the blends of natural and synthetic polymers as new biomaterials: Review. Prog. Polym. Sci. 2011, 36, 1254–1276. [Google Scholar] [CrossRef]
- Zhu, M.; Zhu, Y.; Zhang, L.; Shi, J. Preparation of chitosan/mesoporous silica nanoparticle composite hydrogels for sustained co-delivery of biomacromolecules and small chemical drugs. Sci. Technol. Adv. Mater. 2013, 14. [Google Scholar] [CrossRef]
- Berthod, F.; Saintigny, G.; Chretien, F.; Hayek, D.; Collombel, C.; Damour, O. Optimization of thickness, pore size and mechanical properties of a biomaterial designed for deep burn coverage. Clin. Mater. 1994, 15, 259–265. [Google Scholar] [CrossRef]
- Tamai, N.; Myoui, A.; Hirao, M.; Kaito, T.; Ochi, T.; Tanaka, J.; Takaoka, K.; Yoshikawa, H. A new biotechnology for articular cartilage repair: Subchondral implantation of a composite of interconnected porous hydroxyapatite, synthetic polymer (PLA-PEG), and bone morphogenetic protein-2 (rhBMP-2). Osteoarthr Cartil. 2005, 13, 405–417. [Google Scholar] [CrossRef]
- Bodugoz-Senturk, H.; Macias, C.E.; Kung, J.H.; Muratoglu, O.K. Poly(vinyl alcohol)-acrylamide hydrogels as load-bearing cartilage substitute. Biomaterials 2009, 30, 589–596. [Google Scholar] [CrossRef]
- Corkhill, P.H.; Trevett, A.S.; Tighe, B.J. The potential of hydrogels as synthetic articular cartilage. Proc. Inst. Mech. Eng. H 1990, 204, 147–155. [Google Scholar] [CrossRef]
- Utracki, L.A. History of commercial polymer alloys and blends (from a perspective of the patent literature). Polym. Eng. Sci. 1995, 35, 2–17. [Google Scholar] [CrossRef]
- Ma, P.X. Scaffolds for tissue fabrication. Mater. Today 2004, 7, 30–40. [Google Scholar]
- Khademhosseini, A.; Vacanti, J.; Langer, R. Progress in tissue engineering. Sci. Am. 2009, 300, 64–71. [Google Scholar] [CrossRef]
- Duarte Campos, D.F.; Drescher, W.; Rath, B.; Tingart, M.; Fischer, H. Supporting biomaterials for articular cartilage repair. Cartilage 2012, 3, 205–221. [Google Scholar] [CrossRef]
- Hunziker, E.B. Articular cartilage repair: Basic science and clinical progress. A review of the current status and prospects. Osteoarthr. Cartil. 2002, 10, 432–463. [Google Scholar] [CrossRef]
- Chung, C.; Burdick, J.A. Engineering cartilage tissue. Adv. Drug Deliv. Rev. 2008, 60, 243–262. [Google Scholar] [CrossRef]
- Athanasiou, K.A.; Darling, E.M.; Hu, J.C. Articular cartilage tissue engineering. In Synthesis Lectures on Tissue Engineering; Athanasiou, K.A., Ed.; Morgan & Claypool Publishers: San Rafael, CA, USA, 2009. [Google Scholar]
- Mankin, H.; Mow, V.; Buckwalter, J.; Iannotti, J.; Ratcliffe, A. Articular cartilage structure, composition, and function. In Orthopaedic Basic Science: Biology and Biomechanics of the Musculoskeletal System, 2nd ed.; Buckwalter, J., Einhorn, T., Simon, S., Eds.; American Academy of Orthopaedic Surgeons Publishers: Rosemont, IL, USA, 2000; pp. 443–470. [Google Scholar]
- Mow, V.C.; Holmes, M.H.; Lai, W.M. Fluid transport and mechanical properties of articular cartilage: A review. J. Biomech. 1984, 17, 377–394. [Google Scholar] [CrossRef]
- Mow, V.C.; Lai, W.M. Mechanics of animal joints. Annu. Rev. Fluid. Mech. 1979, 11, 247–288. [Google Scholar] [CrossRef]
- Heinegard, D.; Oldberg, A. Structure and biology of cartilage and bone matrix noncollagenous macromolecules. FASEB J. 1989, 3, 2042–2051. [Google Scholar]
- Buckwalter, J.A.; Mankin, H.J. Articular cartilage: Tissue design and chondrocyte-matrix interactions. Instr. Course Lect. 1998, 47, 477–486. [Google Scholar]
- Temenoff, J.S.; Mikos, A.G. Review: Tissue engineering for regeneration of articular cartilage. Biomaterials 2000, 21, 431–440. [Google Scholar] [CrossRef]
- Poole, C.A. Articular cartilage chondrons: Form, function and failure. J. Anat. 1997, 191, 1–13. [Google Scholar]
- Weiss, C.; Rosenberg, L.; Helfet, A.J. An ultrastructural study of normal young adult human articular cartilage. J. Bone Joint Surg. Am. 1968, 50, 663–674. [Google Scholar]
- Mohammadi, H.; Mequanint, K.; Herzog, W. Computational aspects in mechanical modeling of the articular cartilage tissue. Proc. Inst. Mech. Eng. H 2013, 227, 402–420. [Google Scholar] [CrossRef]
- Herzog, W.; Diet, S.; Suter, E.; Mayzus, P.; Leonard, T.R.; Müller, C.; Wu, J.Z.; Epstein, M. Material and functional properties of articular cartilage and patellofemoral contact mechanics in an experimental model of osteoarthritis. J. Biomech. 1998, 31, 1137–1145. [Google Scholar] [CrossRef]
- Hunziker, E.B.; Michel, M.; Studer, D. Ultrastructure of adult human articular cartilage matrix after cryotechnical processing. Microsc. Res. Tech. 1997, 37, 271–284. [Google Scholar] [CrossRef]
- Trippel, S.B.; Henry, M.J. Articular cartilage injury and repair. In Traumatic Disorders of the Knee; Siliski, J.M., Ed.; Springer-Verlag: New York, NY, USA, 1994; pp. 19–36. [Google Scholar]
- Buckwalter, J.A.; Rosenberg, L.; Hunziker, E.B. Articular cartilage: Composition, structure, response to injury, and methods of facilitating repair. In Articular Cartilage and Knee Joint Function: Basic Science and Arthroscopy; Ewing, J.W., Ed.; Raven Press: New York, NY, USA, 1990; pp. 19–56. [Google Scholar]
- Klein, T.J.; Malda, J.; Sah, R.L.; Hutmacher, D.W. Tissue engineering of articular cartilage with biomimetic zones. Tissue Eng. B Rev. 2009, 15, 143–157. [Google Scholar] [CrossRef]
- Hung, C.T.; Lima, E.G.; Mauck, R.L.; Taki, E.; LeRoux, M.A.; Lu, H.H.; Stark, R.G.; Guo, X.E.; Ateshian, G.A. Anatomically shaped osteochondral constructs for articular cartilage repair. J. Biomech. 2003, 36, 1853–1864. [Google Scholar] [CrossRef]
- Nguyen, L.H.; Kudva, A.K.; Saxena, N.S.; Roy, K. Engineering articular cartilage with spatially-varying matrix composition and mechanical properties from a single stem cell population using a multi-layered hydrogel. Biomaterials 2011, 32, 6946–6952. [Google Scholar] [CrossRef]
- Mow, V.C.; Guo, X.E. Mechano-electrochemical properties of articular cartilage: Their inhomogeneities and anisotropies. Annu. Rev. Biochem. 2002, 4, 175–209. [Google Scholar]
- Stockwell, R.A. The interrelationship of cell density and cartilage thickness in mammalian articular cartilage. J. Anat. 1971, 109, 411–421. [Google Scholar]
- Flik, K.R.; Verma, N.; Cole, B.; Bach, B., Jr. Articular cartilage. In Cartilage Repair Strategies; Williams, R., III, Ed.; Humana Press: Totowa, NJ, USA, 2007; pp. 1–12. [Google Scholar]
- Linn, F.C.; Sokoloff, L. Movement and composition of interstitial fluid of cartilage. Arthritis Rheum. 1965, 8, 481–494. [Google Scholar] [CrossRef]
- Mak, A.F. The apparent viscoelastic behavior of articular cartilage—The contributions from the intrinsic matrix viscoelasticity and interstitial fluid flows. J. Biomech. Eng. 1986, 108, 123–130. [Google Scholar] [CrossRef]
- Cohen, N.P.; Foster, R.J.; Mow, V.C. Composition and dynamics of articular cartilage: Structure, function, and maintaining healthy state. J. Orthop. Sport Phys. 1998, 28, 203–215. [Google Scholar] [CrossRef]
- Moutos, F.T.; Freed, L.E.; Guilak, F. A biomimetic three-dimensional woven composite scaffold for functional tissue engineering of cartilage. Nat. Mater. 2007, 6, 162–167. [Google Scholar] [CrossRef]
- Mow, V.C.; Kuei, S.C.; Lai, W.M.; Armstrong, C.G. Biphasic creep and stress relaxation of articular cartilage in compression: Theory and experiments. J. Biomech. Eng. 1980, 102, 73–84. [Google Scholar] [CrossRef]
- Federico, S.; Herzog, W. On the anisotropy and inhomogeneity of permeability in articular cartilage. Biomech. Model. Mechanobiol. 2008, 7, 367–378. [Google Scholar] [CrossRef]
- Lai, W.M.; Hou, J.S.; Mow, V.C. A triphasic theory for the swelling and deformation behaviors of articular cartilage. J. Biomech. Eng. 1991, 113, 245–258. [Google Scholar] [CrossRef]
- Hukins, D.W.L.; Aspden, R.M. Fiber reinforcing in articular cartilage. In Material Properties and Stress Analysis in Biomechanics; Yettram, A.L., Ed.; Manchester University Press: New York, NY, USA, 1989; pp. 44–59. [Google Scholar]
- Kovach, I.S. A molecular theory of cartilage viscoelasticity. Biophys. Chem. 1996, 59, 61–73. [Google Scholar] [CrossRef]
- Maroudas, A.; Bullough, P.; Swanson, S.A.V.; Freeman, M.A.R. The permeability of articular cartilage. J. Bone Joint Surg. Br. 1968, 5B, 166–177. [Google Scholar]
- Huyghe, J.M.; Wilson, W.; Malakpoor, K. On the thermodynamical admissibility of the triphasic theory of charged hydrated tissues. J. Biomech. Eng. 2009, 131. [Google Scholar] [CrossRef]
- Ateshian, G.A.; Chahine, N.O.; Basalo, I.M.; Hung, C.T. The correspondence between equilibrium biphasic and triphasic material properties in mixture models of articular cartilage. J. Biomech. 2004, 37, 391–400. [Google Scholar] [CrossRef]
- Moutos, F.T.; Guilak, F. Composite scaffolds for cartilage tissue engineering. Biorheology 2008, 45, 501–512. [Google Scholar]
- Akizuki, S.; Mow, V.C.; Müller, F.; Pita, J.C.; Howell, D.S.; Manicourt, D.H. Tensile properties of human knee joint cartilage: I. Influence of ionic conditions, weight bearing, and fibrillation on the tensile modulus. J. Orthop. Res. 1986, 4, 379–392. [Google Scholar] [CrossRef]
- Cohen, B.; Lai, W.M.; Mow, V.C. A transversely isotropic biphasic model for unconfined compression of growth plate and chondroepiphysis. J. Biomech. Eng. 1998, 120, 491–496. [Google Scholar] [CrossRef]
- Woo, S.L.Y.; Akeson, W.H.; Jemmott, G.F. Measurements of nonhomogeneous, directional mechanical properties of articular cartilage in tension. J. Biomech. 1976, 9, 785–791. [Google Scholar] [CrossRef]
- Kempson, G.E.; Freeman, M.A.R.; Swanson, S.A.V. Tensile properties of articular cartilage. Nature 1968, 220, 1127–1128. [Google Scholar]
- Kerin, A.J.; Wisnom, M.R.; Adams, M.A. The compressive strength of articular cartilage. Proc. Inst. Mech. Eng. H 1998, 212, 273–280. [Google Scholar] [CrossRef]
- Buckley, M.R.; Gleghorn, J.P.; Bonassar, L.J.; Cohen, I. Mapping the depth dependence of shear properties in articular cartilage. J. Biomech. 2008, 41, 2430–2437. [Google Scholar] [CrossRef]
- Chiang, H.; Jiang, C.-C. Repair of articular cartilage defects: Review and perspectives. J. Formos. Med. Assoc. 2009, 108, 87–101. [Google Scholar] [CrossRef]
- Buckwalter, J.A.; Mankin, H.J. Instructional course lectures, the American Academy of orthopaedic surgeons-articular cartilage. Part II: Degeneration and osteoarthrosis, repair, regeneration, and transplantation. J. Bone Joint Surg. 1997, 79, 612–632. [Google Scholar]
- Bera, B. Development of artificial articular cartilage. Sadhana 2009, 34, 823–831. [Google Scholar] [CrossRef]
- Brittberg, M.; Peterson, L.; Sjouml, E.; Tallheden, T.; Lindahl, A. Articular cartilage engineering with autologous chondrocyte transplantation. A review of recent developments. J. Bone Joint Surg. 2003, 85, 109–115. [Google Scholar]
- Ikada, Y. Challenges in tissue engineering. J. R. Soc. Interface 2006, 3, 589–601. [Google Scholar] [CrossRef]
- Meretoja, V.V.; Dahlin, R.L.; Wright, S.; Kasper, F.K.; Mikos, A.G. The effect of hypoxia on the chondrogenic differentiation of co-cultured articular chondrocytes and mesenchymal stem cells in scaffolds. Biomaterials 2013, 34, 4266–4273. [Google Scholar] [CrossRef]
- Mauck, R.L.; Soltz, M.A.; Wang, C.C.B.; Wong, D.D.; Chao, P.H.; Valhmu, W.B.; Hung, C.T.; Ateshian, G.A. Functional tissue engineering of articular cartilage through dynamic loading of chondrocyte-seeded agarose gels. J. Biomech. Eng. 2000, 122, 252–260. [Google Scholar] [CrossRef]
- Li, W.-J.; Tuli, R.; Okafor, C.; Derfoul, A.; Danielson, K.G.; Hall, D.J.; Tuan, R.S. A three-dimensional nanofibrous scaffold for cartilage tissue engineering using human mesenchymal stem cells. Biomaterials 2005, 26, 599–609. [Google Scholar] [CrossRef]
- Van der Kraan, P.M.; Buma, P.; van Kuppevelt, T.; van den Berg, W.B. Interaction of chondrocytes, extracellular matrix and growth factors: Relevance for articular cartilage tissue engineering. Osteoarthr. Cartil. 2002, 10, 631–637. [Google Scholar] [CrossRef]
- Mauck, R.L.; Nicoll, S.B.; Seyhan, S.L.; Ateshian, G.A.; Hung, C.T. Synergistic action of growthfactors and dynamic loading for articular cartilage tissue engineering. Tissue Eng. 2003, 9, 597–611. [Google Scholar] [CrossRef]
- Johnstone, B.; Yoo, J.U. Autologous mesenchymal progenitor cells in articular cartilage repair. Clin. Orthop. Relat. Res. 1999, 367, 156–162. [Google Scholar] [CrossRef]
- Cancedda, R.; Dozin, B.; Giannoni, P.; Quarto, R. Tissue engineering and cell therapy of cartilage and bone. Matrix Biol. 2003, 22, 81–91. [Google Scholar] [CrossRef]
- Fortier, L.A.; Barker, J.U.; Strauss, E.J.; McCarrel, T.M.; Cole, B.J. The role of growth factors in cartilage repair. Clin. Orthop. Relat. Res. 2011, 469, 2706–2715. [Google Scholar] [CrossRef]
- Darling, E.M.; Athanasiou, K.A. Rapid phenotypic changes in passaged articular chondrocyte subpopulations. J. Orthop. Res. 2005, 23, 425–432. [Google Scholar] [CrossRef]
- Casper, M.E.; Fitzsimmons, J.S.; Stone, J.J.; Meza, A.O.; Huang, Y.; Ruesink, T.J.; O’Driscoll, S.W.; Reinholz, G.G. Tissue engineering of cartilage using poly-e-caprolactone nanofiber scaffolds seeded in vivo with periosteal cells. Osteoarthr. Cartil. 2010, 18, 981–991. [Google Scholar] [CrossRef]
- Grande, D.A.; Halberstadt, C.; Naughton, G.; Schwartz, R.; Manji, R. Evaluation of matrix scaffolds for tissue engineering of articular cartilage grafts. J. Biomed. Mater. Res. 1997, 34, 211–220. [Google Scholar] [CrossRef]
- Amanpour, S.; Solouk, A.; Mirzadeh, H.; Mohagheghi, M.-A.; Rabbani, S.; Tirgari, F. In vitro and in vivo assays of cartilage repair by perforated polyurethane scaffold. Iran Polym. J. 2010, 19, 403–415. [Google Scholar]
- Getgood, A.; Brooks, R.; Fortier, L.; Rushton, N. Articular cartilage tissue engineering: Today’s research, tomorrow’s practice? J. Bone Joint Surg. Br. 2009, 91, 565–576. [Google Scholar]
- Toh, W.; Lee, E.; Cao, T. Potential of human embryonic stem cells in cartilage tissue engineering and regenerative medicine. Stem Cell Rev. Rep. 2011, 7, 544–559. [Google Scholar] [CrossRef]
- Eyre, D. Collagen of articular cartilage. Arthritis Res. 2002, 4, 30–35. [Google Scholar] [CrossRef] [Green Version]
- Lien, S.-M.; Ko, L.-Y.; Huang, T.-J. Effect of pore size on ECM secretion and cell growth in gelatin scaffold for articular cartilage tissue engineering. Acta Biomater. 2009, 5, 670–679. [Google Scholar] [CrossRef]
- Kim, D.-D.; Kim, D.-H.; Son, Y.-J. Three-dimensional porous scaffold of hyaluronic acid for cartilage tissue engineering. Stud. Mechanobiol. Tissue Eng. Biomater. 2011, 8, 329–349. [Google Scholar]
- Martel-Pelletier, J.; Kwan Tat, S.; Pelletier, J.P. Effects of chondroitin sulfate in the pathophysiology of the osteoarthritic joint: A narrative review. Osteoarthr. Cartil. 2010, 18, 7–11. [Google Scholar] [CrossRef]
- Wang, C.-C.; Yang, K.-C.; Lin, K.-H.; Liu, H.-C.; Lin, F.-H. A highly organized three-dimensionalalginate scaffold for cartilage tissue engineering prepared by microfluidic technology. Biomaterials 2011, 32, 7118–7126. [Google Scholar] [CrossRef]
- Francis Suh, J.K.; Matthew, H.W.T. Application of chitosan-based polysaccharide biomaterials in cartilage tissue engineering: A review. Biomaterials 2000, 21, 2589–2598. [Google Scholar] [CrossRef]
- Seo, S.; Na, K. Mesenchymal stem cell-based tissue engineering for chondrogenesis. J. Biomed. Biotechnol. 2011, 2011, 1–8. [Google Scholar]
- Hashemi Doulabi, A.; Mirzadeh, H.; Imani, M. Interaction and miscibility study of fumarate-based oligomer macromers with chitosan. Mater. Chem. Phys. 2013, 139, 515–524. [Google Scholar] [CrossRef]
- Kobayashi, M.; Chang, Y.-S.; Oka, M. A two year in vivo study of polyvinyl alcohol-hydrogel (PVA-H) artificial meniscus. Biomaterials 2005, 26, 3243–3248. [Google Scholar] [CrossRef]
- Yan, L.-P.; Wang, Y.-J.; Ren, L.; Wu, G.; Caridade, S.G.; Fan, J.-B.; Wang, L.Y.; Ji, P.H.; Oliveira, J.T.; Mano, J.F.; et al. Genipin-cross-linked collagen/chitosan biomimetic scaffolds for articular cartilage tissue engineering applications. J. Biomed. Mater. Res. A 2010, 95, 465–475. [Google Scholar] [Green Version]
- Lin, Y.-C.; Tan, F.-J.; Marra, K.G.; Jan, S.-S.; Liu, D.-C. Synthesis and characterization of collagen/hyaluronan/chitosan composite sponges for potential biomedical applications. Acta Biomater. 2009, 5, 2591–2600. [Google Scholar] [CrossRef]
- Rhee, S.H.; Tanaka, J. Synthesis of a hydroxyapatite/collagen/chondroitin sulfate nanocomposite by a novel precipitation method. J. Am. Ceram. Soc. 2001, 84, 459–461. [Google Scholar] [CrossRef]
- Lu, Z.; Doulabi, B.Z.; Huang, C.; Bank, R.A.; Helder, M.N. Collagen type II enhances chondrogenesis in adipose tissue-derived stem cells by affecting cell shape. Tissue Eng. A 2009, 16, 81–90. [Google Scholar]
- Lee, C.H.; Singla, A.; Lee, Y. Biomedical applications of collagen. Int. J. Pharm. 2001, 221, 1–22. [Google Scholar] [CrossRef]
- Nair, L.S.; Laurencin, C.T. Biodegradable polymers as biomaterials. Prog. Polym. Sci. 2007, 32, 762–798. [Google Scholar] [CrossRef]
- Responte, D.J.; Natoli, R.M.; Athanasiou, K.A. Collagens of articular cartilage: Structure, function, and importance in tissue engineering. Crit. Rev. Biomed. Eng. 2007, 35, 363–411. [Google Scholar] [CrossRef]
- Lynn, A.K.; Yannas, I.V.; Bonfield, W. Antigenicity and immunogenicity of collagen. J. Biomed. Mater. Res. B 2004, 71, 343–354. [Google Scholar]
- Wakitani, S.; Goto, T.; Pineda, S.J.; Young, R.G.; Mansour, J.M.; Caplan, A.I.; Goldberg, V.M. Mesenchymal cell-based repair of large, full-thickness defects of articular cartilage. J. Bone Joint Surg. Am. 1994, 76, 579–592. [Google Scholar]
- Wakitani, S.; Kimura, T.; Hirooka, A.; Ochi, T.; Yoneda, M.; Yasui, N.; Owaki, H.; Ono, K. Repair of rabbit articular surfaces with allograft chondrocytes embedded in collagen gel. J. Bone Joint Surg. Br. 1989, 71, 74–80. [Google Scholar]
- Kawamura, S.; Wakitani, S.; Kimura, T.; Maeda, A.; Caplan, A.I.; Shino, K.; Ochi, T. Articular cartilage repair: Rabbit experiments with a collagen gel-biomatrix and chondrocytes cultured in it. Acta Orthop. 1998, 69, 56–62. [Google Scholar] [CrossRef]
- Yang, X.; Guo, L.; Fan, Y.; Zhang, X. Preparation and characterization of macromolecule cross-linked collagen hydrogels for chondrocyte delivery. Int. J. Biol. Macromol. 2013, 61, 487–493. [Google Scholar] [CrossRef]
- Taravel, M.N.; Domard, A. Relation between the physicochemical characteristics of collagen and its interactions with chitosan: I. Biomaterials 1993, 14, 930–938. [Google Scholar] [CrossRef]
- Sionkowska, A.; Wisniewski, M.; Skopinska, J.; Kennedy, C.J.; Wess, T.J. Molecular interactions in collagen and chitosan blends. Biomaterials 2004, 25, 795–801. [Google Scholar] [CrossRef]
- Abedi, G.; Sotoudeh, A.; Soleymani, M.; Shafiee, A.; Mortazavi, P.; Aflatoonian, M.R. A collagen-poly(vinyl alcohol) nanofiber scaffold for cartilage repair. J. Biomater. Sci. Polym. Ed. 2011, 22, 2445–2455. [Google Scholar] [CrossRef]
- Chen, G.; Sato, T.; Ushida, T.; Hirochika, R.; Shirasaki, Y.; Ochiai, N.; Tateishi, T. The use of a novel PLGA fiber/collagen composite web as a scaffold for engineering of articular cartilage tissue with adjustable thickness. J. Biomed. Mater. Res. A 2003, 67, 1170–1180. [Google Scholar]
- Chen, G.; Liu, D.; Tadokoro, M.; Hirochika, R.; Ohgushi, H.; Tanaka, J.; Tateishi, T. Chondrogenic differentiation of human mesenchymal stem cells cultured in a cobweb-like biodegradable scaffold. Biochem. Biophys. Res. Commun. 2004, 322, 50–55. [Google Scholar] [CrossRef]
- Athansou, N.A.; Puddle, B.; Sallie, B. Highly sulphated glycosaminoglycans in articular cartilage and other tissues containing beta 2 microglobulin dialysis amyloid deposits. Nephrol. Dial. Transplant. 1995, 10, 1672–1678. [Google Scholar]
- Nanda, S.; Sood, N.; Reddy, B.V.K.; Markandeywar, T.S. Preparation and characterization of poly(vinyl alcohol)-chondroitin sulphate hydrogel as scaffolds for articular cartilage regeneration. Indian J. Mater. Sci. 2013, 2013. [Google Scholar] [CrossRef]
- Yang, Y.-L.; Sun, C.; Wilhelm, M.E.; Fox, L.J.; Zhu, J.; Kaufman, L.J. Influence of chondroitin sulfate and hyaluronic acid on structure, mechanical properties, and glioma invasion of collagen I gels. Biomaterials 2011, 32, 7932–7940. [Google Scholar] [CrossRef]
- Ko, C.-S.; Huang, J.-P.; Huang, C.-W.; Chu, I.M. Type II collagen-chondroitin sulfate-hyaluronan scaffold cross-linked by genipin for cartilage tissue engineering. J. Biosci. Bioeng. 2009, 107, 177–182. [Google Scholar] [CrossRef]
- McMahon, L.; Reid, A.; Campbell, V.; Prendergast, P. Regulatory effects of mechanical strain on the chondrogenic differentiation of MSCs in a collagen-GAG scaffold: Experimental and computational analysis. Ann. Biomed. Eng. 2008, 36, 185–194. [Google Scholar] [CrossRef]
- Chen, Y.-L.; Lee, H.-P.; Chan, H.-Y.; Sung, L.-Y.; Chen, H.-C.; Hu, Y.-C. Composite chondroitin-6-sulfate/dermatan sulfate/chitosan scaffolds for cartilage tissue engineering. Biomaterials 2007, 28, 2294–2305. [Google Scholar] [CrossRef]
- Chou, C.-H.; Cheng, W.T.K.; Kuo, T.-F.; Sun, J.-S.; Lin, F.-H.; Tsai, J.-C. Fibrin glue mixed with gelatin/hyaluronic acid/chondroitin-6-sulfate tri-copolymer for articular cartilage tissue engineering: The results of real-time polymerase chain reaction. J. Biomed. Mater. Res. A 2007, 82, 757–767. [Google Scholar]
- Muzzarelli, R.A.A. Chitins and chitosans for the repair of wounded skin, nerve, cartilage and bone. Carbohydr. Polym. 2009, 76, 167–182. [Google Scholar] [CrossRef]
- Muzzarelli, R.A.A. Nanochitins and nanochitosans, paving the way to eco-friendly and energy-saving exploitation of marine resources. In Polymer Science: A Comprehensive Reference; Matyjaszewski, K., Moller, M., Eds.; Elsevier BV: Amsterdam: The Netherlands, 2012; pp. 153–164. [Google Scholar]
- Hashemi Doulabi, A.; Mirzadeh, H.; Imani, M.; Samadi, N. Chitosan/Polyethylene glycol fumarate blend film: Physical and antibacterial properties. Carbohydr. Polym. 2013, 92, 48–56. [Google Scholar] [CrossRef]
- Oprenyeszk, F.; Chausson, M.; Maquet, V.; Dubuc, J.E.; Henrotin, Y. Protective effect of a new biomaterial against the development of experimental osteoarthritis lesions in rabbit: A pilot study evaluating the intra-articular injection of alginate-chitosan beads dispersed in an hydrogel. Osteoarthr. Cartil. 2013, 21, 1099–1107. [Google Scholar] [CrossRef]
- Yan, J.; Li, X.; Liu, L.; Wang, F.; Zhu, T.W.; Zhang, Q. Potential use of collagen-chitosan-hyaluronan tri-copolymer scaffold for cartilage tissue engineering. Artif. Cells Blood Substit. Biotecnol. 2006, 34, 27–39. [Google Scholar] [CrossRef]
- Li, Z.; Zhang, M. Chitosan-alginate as scaffolding material for cartilage tissue engineering. J. Biomed. Mater. Res. A 2005, 75, 485–493. [Google Scholar]
- Häuselmann, H.J.; Aydelotte, M.B.; Schumacher, B.L.; Kuettner, K.E.; Gitelis, S.H.; Thonar, E.J.M.A. Synthesis and turnover of proteoglycans by human and bovine adult articular chondrocytes cultured in alginate beads. Matrix 1992, 12, 116–129. [Google Scholar] [CrossRef]
- Jung, Y.; Kim, S.H.; Kim, S.-H.; Kim, Y.H.; Xie, J.; Matsuda, T.; Min, B.G. Cartilaginous tissue formation using a mechano-active scaffold and dynamic compressive stimulation. J. Biomater. Sci. Polym. Ed. 2008, 19, 61–74. [Google Scholar] [CrossRef]
- Yang, Z.; Wu, Y.; Li, C.; Zhang, T.; Zou, Y.; Hui, J.H.; Ge, Z.; Lee, E.H. Improved mesenchymal stem cells attachment and in vitro cartilage tissue formation on chitosan-modified poly(l-lactide-co-epsilon-caprolactone) scaffold. Tissue Eng. A 2012, 18, 242–251. [Google Scholar] [CrossRef]
- Baker, M.I.; Walsh, S.P.; Schwartz, Z.; Boyan, B.D. A review of polyvinyl alcohol and its uses in cartilage and orthopedic applications. J. Biomed. Mater. Res. B 2012, 100, 1451–1457. [Google Scholar]
- Cho, S.H.; Lim, S.M.; Han, D.K.; Yuk, S.H.; Im, G.I.; Lee, J.H. Time-dependent alginate/polyvinyl alcohol hydrogels as injectable cell carriers. J. Biomater. Sci. Polym. Ed. 2009, 20, 863–876. [Google Scholar] [CrossRef]
- Cai, C.; Chen, C.; Chen, G.; Wang, F.; Guo, L.; Yin, L.; Feng, D.; Yang, L. Type I collagen and polyvinyl alcohol blend fiber scaffold for anterior cruciate ligament reconstruction. Biomed. Mater. 2013, 8. [Google Scholar] [CrossRef]
- Gough, J.E.; Scotchford, C.A.; Downes, S. Cytotoxicity of glutaraldehyde crosslinked collagen/poly(vinyl alcohol) films is by the mechanism of apoptosis. J. Biomed. Mater. Res. 2002, 61, 121–130. [Google Scholar] [CrossRef]
- Bray, J.C.; Merrill, E.W. Poly(vinyl alcohol) hydrogels for synthetic articular cartilage material. J. Biomed. Mater. Res. 1973, 7, 431–443. [Google Scholar] [CrossRef]
- Mohan, N.; Nair, P.D. Polyvinyl alcohol-poly(caprolactone) semi IPN scaffold with implication for cartilage tissue engineering. J. Biomed. Mater. Res. B 2008, 84, 584–594. [Google Scholar] [CrossRef]
- Mohan, N.; Nair, P.D.; Tabata, Y. Growth factor-mediated effects on chondrogenic differentiation of mesenchymal stem cells in 3D semi-IPN poly(vinyl alcohol)-poly(caprolactone) scaffolds. J. Biomed. Mater. Res. A 2010, 94, 146–159. [Google Scholar] [CrossRef]
- Lee, S.-Y.; Pereira, B.P.; Yusof, N.; Selvaratnam, L.; Yu, Z.; Abbas, A.A.; Kamarul, T. Unconfined compression properties of a porous poly(vinyl alcohol)-chitosan-based hydrogel after hydration. Acta Biomater. 2009, 5, 1919–1925. [Google Scholar] [CrossRef]
- Ibrahim, N.S.; Krishnamurithy, G.; Rao Balaji Raghavendran, H.; Puvaneswary, S.; Wuey Min, N.; Kamarul, T. Novel HA-PVA/NOCC bilayered scaffold for osteochondral tissue-engineering applications—Fabrication, characterization, in vitro and in vivo biocompatibility study. Mater. Lett. 2013, 113, 25–29. [Google Scholar] [CrossRef]
- Ahn, E.S.; Gleason, N.J.; Nakahira, A.; Ying, J.Y. Nanostructure processing of hydroxyapatite-based bioceramics. Nano Lett. 2001, 1, 149–153. [Google Scholar] [CrossRef]
- Sardinha, V.M.; Lima, L.L.; Belangero, W.D.; Zavaglia, C.A.; Bavaresco, V.P.; Gomes, J.R. Tribological characterization of polyvinyl alcohol hydrogel as substitute of articular cartilage. Wear 2013, 301, 218–225. [Google Scholar] [CrossRef]
- Ma, R.; Xiong, D.; Miao, F.; Zhang, J.; Peng, Y. Novel PVP/PVA hydrogels for articular cartilage replacement. Mater. Sci. Eng. C 2009, 29, 1979–1983. [Google Scholar] [CrossRef]
- Ma, R.; Xiong, D.; Miao, F.; Zhang, J.; Peng, Y. Friction properties of novel PVP/PVA blend hydrogels as artificial cartilage. J. Biomed. Mater. Res. A 2010, 93, 1016–1019. [Google Scholar]
- Scholten, P.M.; Ng, K.W.; Joh, K.; Serino, L.P.; Warren, R.F.; Torzilli, P.A.; Maher, S.A. A semi-degradable composite scaffold for articular cartilage defects. J. Biomed. Mater. Res. A 2011, 97, 8–15. [Google Scholar]
- Bichara, D.A.; Zhao, X.; Hwang, N.S.; Bodugoz-Senturk, H.; Yaremchuk, M.J.; Randolph, M.A.; Muratoqlu, O.K. Porous poly (vinyl alcohol)-alginate gel hybrid construct for neocartilage formation using human nasoseptal cells. J. Surg. Res. 2010, 163, 331–336. [Google Scholar] [CrossRef]
- Engel, E.; Michiardi, A.; Navarro, M.; Lacroix, D.; Planell, J.A. Nanotechnology in regenerative medicine: The materials side. Trends Biotechnol. 2008, 26, 39–47. [Google Scholar] [CrossRef]
- Zhang, L.; Webster, T.J. Nanotechnology and nanomaterials: Promises for improved tissue regeneration. Nano Today 2009, 4, 66–80. [Google Scholar] [CrossRef]
- Coburn, J.M.; Gibson, M.; Monagle, S.; Patterson, Z.; Elisseeff, J.H. Bioinspired nanofibers support chondrogenesis for articular cartilage repair. Proc. Natl. Acad. Sci. USA 2012, 109, 10012–10017. [Google Scholar] [CrossRef]
- Wise, J.K.; Yarin, A.L.; Megaridis, C.M.; Cho, M. Chondrogenic differentiation of human mesenchymal stem cells on oriented nanofibrous scaffolds: Engineering the superficial zone of articular cartilage. Tissue Eng. A 2009, 15, 913–921. [Google Scholar] [CrossRef]
- Shin, H.J.; Lee, C.H.; Cho, I.H.; Kim, Y.-J.; Lee, Y.-J.; Kim, I.A.; Park, K.-D.; Yui, N.; Shin, J.-W. Electrospun PLGA nanofiber scaffolds for articular cartilage reconstruction: Mechanical stability, degradation and cellular responses under mechanical stimulation in vitro. J. Biomater. Sci. Polym. Ed. 2006, 17, 103–119. [Google Scholar] [CrossRef]
- Shirosaki, Y.; Okayama, T.; Tsuru, K.; Hayakawa, S.; Osaka, A. Synthesis and cytocompatibility of porous chitosan-silicate hybrids for tissue engineering scaffold application. Chem. Eng. J. 2008, 137, 122–128. [Google Scholar] [CrossRef]
- Buchtová, N.; Réthoré, G.; Boyer, C.; Guicheux, J.; Rambaud, F.; Vallé, K.; Belleville, P.; Sanchez, C.; Chauvet, O.; Weiss, P.; et al. Nanocomposite hydrogels for cartilage tissue engineering: Mesoporous silica nanofibers interlinked with siloxane derived polysaccharide. J. Mater. Sci. Mater. Med. 2013, 24, 1875–1884. [Google Scholar]
- Bhattarai, N.; Gunn, J.; Zhang, M. Chitosan-based hydrogels for controlled, localized drug delivery. Adv. Drug Deliv. Rev. 2010, 62, 83–99. [Google Scholar] [CrossRef]
- Lin, L.; Chow, K.L.; Leng, Y. Study of hydroxyapatite osteoinductivity with an osteogenic differentiation of mesenchymal stem cells. J. Biomed. Mater. Res. A 2009, 89, 326–335. [Google Scholar]
- Ducheyne, P.; Qiu, Q. Bioactive ceramics: The effect of surface reactivity on bone formation and bone cell function. Biomaterials 1999, 20, 2287–2303. [Google Scholar] [CrossRef]
- Kuo, Y.-C.; Leou, S.-N. Chondrogenesis of articular chondrocytes in hydroxyapatite/chitin/chitosan scaffolds supplemented with pituitary extract. Eng. Life Sci. 2010, 10, 65–74. [Google Scholar] [CrossRef]
- Spadaccio, C.; Rainer, A.; Trombetta, M.; Vadalá, G.; Chello, M.; Covino, E.; Denaro, V.; Toyoda, Y.; Genovese, J.A. Poly-l-Lactic acid/Hydroxyapatite electrospun nanocomposites induce chondrogenic differentiation of human MSC. Ann. Biomed. Eng. 2009, 37, 1376–1389. [Google Scholar] [CrossRef]
- Pan, Y.; Xiong, D.; Chen, X. Mechanical properties of nanohydroxyapatite reinforced poly(vinyl alcohol) gel composites as biomaterial. J. Mater. Sci. 2007, 42, 5129–5134. [Google Scholar] [CrossRef]
- Oliveira, J.M.; Rodrigues, M.T.; Silva, S.S.; Malafaya, P.B.; Gomes, M.E.; Viegas, C.A.; Dias, I.R.; Azevedo, J.T.; Mano, J.F.; Reis, R.L. Novel hydroxyapatite/chitosan bilayered scaffold for osteochondral tissue-engineering applications: Scaffold design and its performance when seeded with goat bone marrow stromal cells. Biomaterials 2006, 27, 6123–6137. [Google Scholar] [CrossRef] [Green Version]
- O’Shea, T.M.; Miao, X. Bilayered scaffolds for osteochondral tissue engineering. Tissue Eng. B Rev. 2008, 14, 447–464. [Google Scholar] [CrossRef]
- Qu, D.; Li, J.; Li, Y.; Khadka, A.; Zuo, Y.; Wang, H.; Liu, Y.; Cheng, L. Ectopic osteochondral formation of biomimetic porous PVA-n-HA/PA6 bilayered scaffold and BMSCs construct in rabbit. J. Biomed. Mater. Res. B 2011, 96, 9–15. [Google Scholar]
- Wang, X.; Grogan, S.P.; Rieser, F.; Winkelmann, V.; Maquet, V.; Berge, M.L.; Mainil-Varlet, P. Tissue engineering of biphasic cartilage constructs using various biodegradable scaffolds: An in vitro study. Biomaterials 2004, 25, 3681–3688. [Google Scholar] [CrossRef]
- Shi, H.-C.; Lu, D.; Wang, W.-P.; Lu, Y.; Zeng, Y.-J. N-carboxyethylchitosan/nanohydroxyapatite composites scaffold for tracheal cartilage tissue-engineering applications. Micro Nano Lett. 2012, 7, 76–79. [Google Scholar] [CrossRef]
- Gu, Z.-Q.; Xiao, J.-M.; Zhang, X.-H. The development of artificial articular cartilage-PVA-hydrogel. Biomed. Mater. Eng. 1998, 8, 75–81. [Google Scholar]
- Maiolo, A.S.; Amado, M.N.; Gonzalez, J.S.; Alvarez, V.A. Development and characterization of poly (vinyl alcohol) based hydrogels for potential use as an articular cartilage replacement. Mater. Sci. Eng. C 2012, 32, 1490–1495. [Google Scholar] [CrossRef]
- Pan, Y.; Xiong, D. Stress relaxation behavior of nano-hydroxyapatite reinforced poly(vinyl alcohol) gel composites as biomaterial. J. Mater. Sci. 2010, 45, 5495–5501. [Google Scholar] [CrossRef]
- Pan, Y.; Xiong, D. Friction properties of nano-hydroxyapatite reinforced poly(vinyl alcohol) gel composites as an articular cartilage. Wear 2009, 266, 699–703. [Google Scholar] [CrossRef]
- Pan, Y.; Xiong, D.; Gao, F. Viscoelastic behavior of nano-hydroxyapatite reinforced poly(vinyl alcohol) gel biocomposites as an articular cartilage. J. Mater. Sci. Mater. Med. 2008, 19, 1963–1969. [Google Scholar] [CrossRef]
- Zhang, L.; Hu, J.; Athanasiou, K.A. The role of tissue engineering in articular cartilage repair and regeneration. Crit. Rev. Biomed. Eng. 2009, 37, 1–57. [Google Scholar] [CrossRef]
- Ikada, Y.; Tsuji, H. Biodegradable polyesters for medical and ecological applications. Macromol. Rapid Commun. 2000, 21, 117–132. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Doulabi, A.H.; Mequanint, K.; Mohammadi, H. Blends and Nanocomposite Biomaterials for Articular Cartilage Tissue Engineering. Materials 2014, 7, 5327-5355. https://doi.org/10.3390/ma7075327
Doulabi AH, Mequanint K, Mohammadi H. Blends and Nanocomposite Biomaterials for Articular Cartilage Tissue Engineering. Materials. 2014; 7(7):5327-5355. https://doi.org/10.3390/ma7075327
Chicago/Turabian StyleDoulabi, Azadehsadat Hashemi, Kibret Mequanint, and Hadi Mohammadi. 2014. "Blends and Nanocomposite Biomaterials for Articular Cartilage Tissue Engineering" Materials 7, no. 7: 5327-5355. https://doi.org/10.3390/ma7075327





