Long-Term Stability of a Cellulose-Based Glucose Oxidase Membrane
Abstract
: A cellulose-based glucose oxidase membrane was prepared on a glassy carbon (GC) electrode. The current response of the electrode to glucose was measured by applying a potential of 1.0 V vs. Ag/AgCl on the base GC and was proportional to the concentration of glucose up to 1 mM. The long-term stability of the electrode was examined by measuring the daily glucose response. Over four months, the response magnitude was maintained and then gradually decreased. After 11 months, though the response magnitude decreased to 50% of the initial value, the linear response range did not change. Therefore, the electrode could be used as a glucose biosensor even after 11 months of use. The entrapment of the enzyme in the cellulose matrix promoted the stability of the enzyme, as revealed by data on the enzyme activity after the enzyme electrode was immersed in urea. Therefore, the cellulose matrix may be used to improve the performance of biosensors, bioreactors and bio-fuel cells.1. Introduction
Numerous studies have focused on improving the stability of enzyme membranes for use in biosensors and bioreactors [1], because increasing the long-term stability of enzyme membranes [2,3] may extend the lifetime of biosensors and bioreactors.
To increase the long-term stability, various factors that decrease the stability should be eliminated. Such factors may include [4]: (1) the lifetime of the immobilized enzymes and membrane materials; (2) strength of immobilization or entrapment and (3) fouling and contamination of the samples. Typically, removing more than one factor should be addressed to increase the stability of the enzyme membrane.
Many studies have been conducted regarding the solid immobilization of enzyme membranes and it has been reported that covalent bonding or cross-linking between the base material and enzyme facilitates strong immobilization [5–8]. However, the required reagents may alter the enzyme. To avoid this issue, enzyme entrapment has been employed for the immobilization [9–16]. Furthermore, our group has developed enzyme entrapment methods, which avoid enzyme activity loss including the polyion complex membrane [3,14,15] and cellulose-based membrane [4,16].
For cellulose-based enzyme membranes, excellent long-term stability was achieved as the response magnitude did not change up to four months after preparation [16]. Furthermore, cellulose was stable against chemical and biological contamination and the lifetime of the enzymes in the membranes was extended. In this study, the electrode responses during long-term usage were examined.
2. Results and Discussion
2.1. Current Response to Glucose on Cellulose-Based Glucose Oxidase Membrane Electrode
As described in the experimental section, glucose oxidase (GOD) was immobilized in the cellulose membrane on the glassy carbon (GC) electrode. The current response of the electrode to glucose was measured. The characteristics of the electrode were similar to those described in a previous report [16] as the response time was ca. 10 s. Because the response was not observed when the electrode without GOD was utilized, the increase in the oxidation current was caused by the oxidation of hydrogen peroxide produced by the enzymatic reaction.
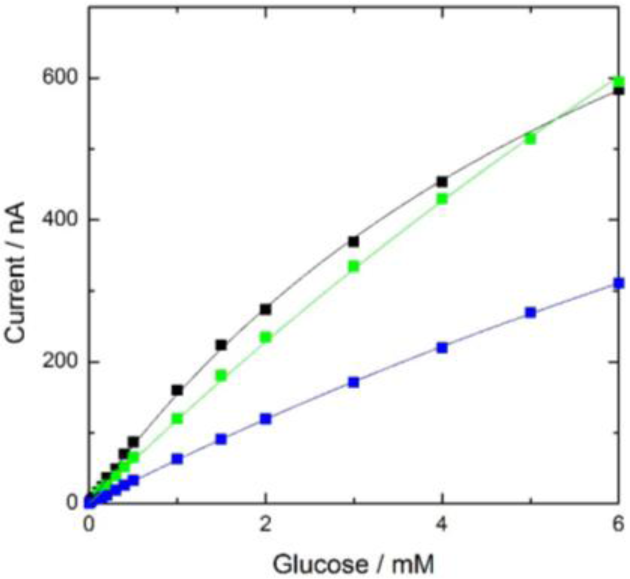
The response current was plotted against glucose concentration (black line in Figure 1) and was proportional to the glucose concentration, up to 1 mM. The lower detection limit was 10 μM glucose, which was similar to that in a previous report [16]. Thus, the electrode can be used as a sensitive glucose biosensor and the preparation of the cellulose-based GOD membrane is easy and inexpensive.
2.2. Long-Term Stability of Cellulose-Based GOD Membrane
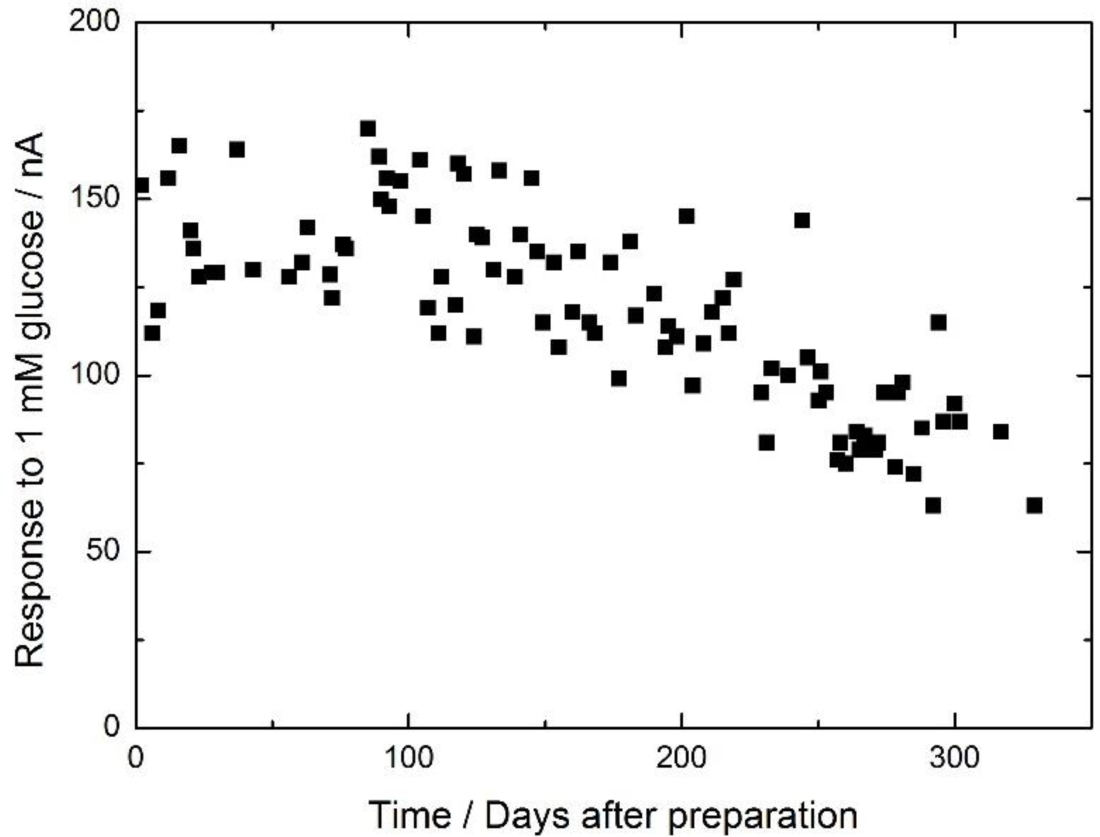
The long-term stability of the enzyme membrane was examined by measuring the current response of the electrode to 1 mM glucose daily for 11 months (Figure 2). During the first four months after preparation, the magnitude of the current response was almost the same as the initial value. The calibration curve of the electrode on the 117th day after preparation (green plot in Figure 1) was the same as the initial curve (black plot in Figure 1). After four months, the current gradually decreased and eventually reached ca. 50% of the initial value after 11 months. However, the calibration curve on the 329th day after preparation (blue plot in Figure 1) had a similar shape to the initial curve, but a different magnitude, meaning the proportional response range against the glucose concentration had not changed. The magnitude of the response to 1 mM glucose on the 2nd, 117th and 329th day were determined to be 161 ± 10, 119 ± 6 and 63 ± 5 nA (number of the measurements, n = 5), respectively, i.e., the magnitude of the error was within several percent of the signal. Therefore, the electrode can be used as a biosensor for 11 months.
The long-term stability is likely related to the characteristics of the cellulose matrix, enzyme capture, and stabilizer effects. The cellulose membrane could be penetrated by small molecules such as l-ascorbate and acetoaminophene [16]. On the other hand, the enzyme entrapped in the cellulose membrane remained within the membrane for four months, likely because of the structure of the cellulose matrix. The enzyme electrode could be used as a biosensor even after 11 months of electrode use because of the capture and stabilization of the enzyme. Though the measurement error was less than 10%, the fluctuation of the stability data was larger, likely because of the lack of temperature control during the measurements.
2.3. Effect of a Denaturant on Electrode Response
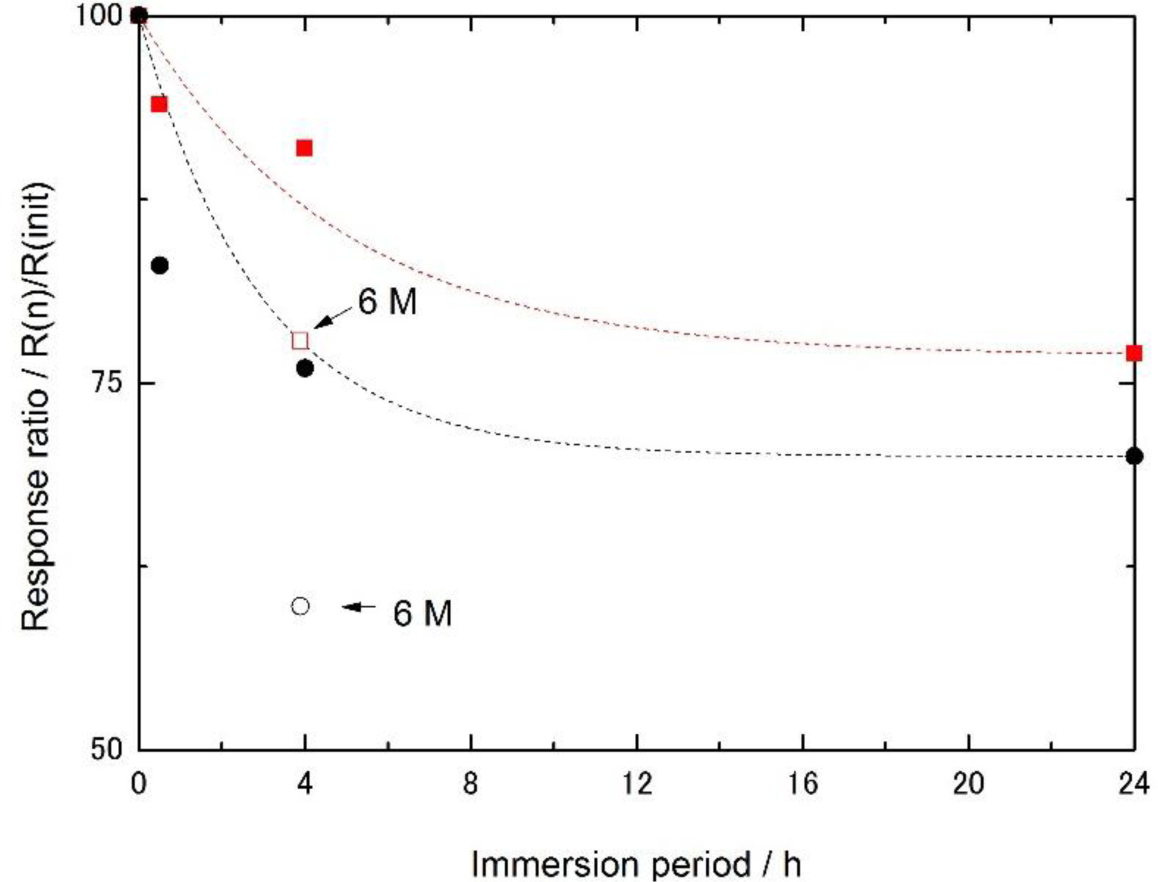
The effect of urea, a denaturant, on the electrode response to glucose was measured by immersing the enzyme electrode in a 3 M solution of urea. The ratio of the current response before and after the immersion is plotted in Figure 3. The response of the cellulose-based enzyme electrode gradually decreased with the immersion time (red squares in Figure 3). The response of the polyion complex membrane containing the enzyme was also measured (black circles in Figure 3). Open marks represent the results obtained when the electrodes were immersed in a 6 M solution of urea. Notably, neither membrane was altered after immersion in urea. A comparison of these two matrices revealed that the enzyme in the cellulose matrix retained a higher activity than the enzyme in the polyion complex membrane matrix. Cellulose appeared to stabilize the entrapped enzyme and thus enhance its activity.
Based on the permeation characteristics of the two membranes, urea could easily penetrate the membranes [14,16]. Nevertheless, the immobilized enzymes were not denatured to the same degree, which may be related to the different characteristics of the matrix materials. In particular, cellulose is a hydrophilic material, whereas the polyion complex is hydrophobic [3,17]. The environment was altered by the type of matrix materials and thus the surroundings of the enzyme were also altered. As a result, the cellulose matrix was more suitable to increase the immobilized enzyme lifetime. A more detailed investigation is now in progress.
3. Experimental Section
3.1. Materials
GOD (203 U·mg−1; EC 1.1.3.4; obtained from Aspergillus niger), cellulose solution (5 wt%) in ionic liquid (1-ethyl-3-methylimidazolium acetate), poly-l-lysine (average MW 100,000), and polystyrene sulfonate (average MW 70,000) were obtained from Aldrich (St. Louis, MO, USA) and were used as received. All other reagents were of analytical reagent grade.
A GC electrode (disk, 3 mm in diameter) was purchased from Bioanalytical Systems (West Lafayette, IN, USA).
3.2. Preparation of Cellulose-based Enzyme Membrane
A modified version of a reported procedure [16] was carried out. Briefly, GOD was dissolved in water at 5 wt%, and the GOD solution (20 μL) was dropped on a GC electrode. The electrode was allowed to dry for 2 h. Then, 20 μL of cellulose solution in an ionic liquid was spread on the electrode for coating. The electrode remained stationary for 1 min to allow the formation of uniform layer, and then the electrode was immersed in water for 5 min to remove the ionic liquid. After drying for 4 h, the electrode was used for the experiments.
3.3. Measurement of Current Response to Glucose
The electrode was immersed in a 0.1 M citrate buffer solution (pH 5.5, 15 mL), and a potential of 1.0 V vs. Ag/AgCl was applied to the base electrode to measure the oxidation current of hydrogen peroxide produced by the enzymatic reaction. The current response to glucose was then measured by the addition of a solution of glucose to the buffer. After the measurement, the electrode was stored in a refrigerator at 4 °C.
The long-term stability of the electrode was evaluated based on the daily current response to 1 mM glucose.
The effect of urea on the electrode response was also measured. The current responses of 1 mM glucose were measured before and after the immersion of the electrode into a 0.1 M citrate buffer solution (pH 5.5) containing urea.
All measurements were performed at room temperature (23 ± 2 °C).
3.4. Preparation of GOD-Entrapped Polyion Complex Membrane
A GOD-entrapped polyion complex membrane was prepared by the successively dropping polystyrene sulfonate (20 μL, 20 mM monomeric unit), GOD (20 μL, 5 wt%), poly-l-lysine (20 μL, 20 mM monomeric unit) solutions on the GC electrode. After drying for 4 h, the electrode was used for subsequent experiments.
4. Conclusions
A cellulose-based GOD membrane was prepared on a GC electrode. The current response of the electrode to glucose was measured by applying a potential of 1.0 V vs. Ag/AgCl on the base GC. The current response to glucose was proportional to glucose concentration up to 1 mM. The long-term stability of the electrode was examined by measuring the daily glucose response. During the first four months, the response magnitude did not change, and after this period, the response gradually decreased. After 11 months, although the response magnitude decreased to 50% of the initial value, the linear response range had not changed. Thus, the electrode could be used as a glucose biosensor even after being used for 11 months.
The long-term stability may be due to the enzyme entrapment by the cellulose matrix. The entrapment in the cellulose matrix promotes the stability of the enzyme, as confirmed by data regarding enzyme activity after the enzyme electrode was immersed in urea. Thus, the cellulose matrix might improve the performance of biosensors, bioreactors, and bio-fuel cells.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Wang, Y.; Hasebe, Y. Methylene blue-induced stabilization effect of adsorbed glucose oxidase on a carbon-felt surface for bioelectrocatalytic activity. J. Electrochem. Soc 2012, 159, F110–F118. [Google Scholar]
- Reddy, K.R.C.; Kayasth, A.M. Improved stability of urease upon coupling to alkylamine and arylamine glass and its analytical use. J. Mol. Catal. B 2006, 38, 104–112. [Google Scholar]
- Yabuki, S. Polyelectrolyte complex membranes for immobilizing biomolecules, and their applications to bio-sensing. Anal. Sci 2011, 27, 695–702. [Google Scholar]
- Yabuki, S. Supporting materials that improve the stability of enzyme membranes. Anal. Sci 2014. in press. [Google Scholar]
- Bora, U.; Kannan, K.; Nahar, P. A simple method for functionalization of cellulose membrane for covalent immobilization of biomolecules. J. Membr. Sci 2005, 205, 215–222. [Google Scholar]
- Jamal, K.; Worsfold, O.; McCormac, T.; Dempsey, E. A stable and selective electrochemical biosensor for the liver enzyme alanine aminotransferase (ALT). Biosens. Bioelectron 2009, 24, 2926–2930. [Google Scholar]
- Huang, X.J.; Ge, D.; Xu, Z.K. Preparation and characterization of stable chitosan nanofibrous membrane for lipase immobilization. Euro. Polym. J 2007, 43, 3710–3718. [Google Scholar]
- Yabuki, S.; Mizutani, F. Preparation of amperometric glucose sensor based on electrochemically polymerized films of indole derivatives. Sens. Actuators B 2005, 108, 651–653. [Google Scholar]
- Kawanami, Y.; Yamasaki, S.; Yamada, S.; Takehara, K. Immobilization of bacterial luciferase into poly(N-isopropylacrylamide) film for electrochemical control of a bioluminescence reaction. Anal. Sci 2012, 28, 1013–1015. [Google Scholar]
- Nakamura, T.; Ji, X. P.; Endo, K.; Takano, D. Enhanced enzymatic reaction of tyrosinase-immobilized polyacrylamide–(γ-cyclodextrin) membrane coated on a platinum disk electrode in acetonitrile. Chem. Lett 2007, 36, 506–507. [Google Scholar]
- Sharma, S. K.; Suman; Pundir, C.S.; Sehgal, N.; Kumar, A. Galactose sensor based on galactose oxidase immobilized in polyvinyl formal. Sens. Actuators B 2006, 119, 15–19. [Google Scholar]
- Yabuki, S.; Shinohara, H.; Aizawa, M. Electro-conductive enzyme membrane. J. Chem. Soc. Chem. Commun 1989, 1989, 945–946. [Google Scholar]
- Yabuki, S.; Shinohara, H.; Ikariyama, Y.; Aizawa, M. Electrical activity controlling system for a mediator-coexisted alcohol dehydrogenase-NAD conductive membrane. J. Electroanal. Chem 1990, 277, 179–187. [Google Scholar]
- Mizutani, F.; Yabuki, S.; Hirata, Y. Amperometric l-lactate-sensing electrode based on a polyion complex layer containing lactate oxidase. Application to serum and milk samples. Anal. Chim. Acta 1995, 314, 233–239. [Google Scholar]
- Yabuki, S.; Mizutani, F.; Hirata, Y. Glucose-sensing electrode based on glucose oxidase-attached polyion complex membrane containing peroxidase and ferrocene. Electroanalysis 2001, 13, 380–383. [Google Scholar]
- Yabuki, S.; Hirata, Y.; Sato, Y.; Iijima, S. Preparation of a cellulose-based enzyme membrane using ionic liquid to lengthen the duration of enzyme stability. Anal. Sci 2012, 28, 373–377. [Google Scholar]
- Yabuki, S.; Fujii, S.; Mizutani, F.; Hirata, Y. Permeation regulation of charged species by the component change of polyion complex membranes. Anal. Biochem 2008, 375, 141–143. [Google Scholar]
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yabuki, S.; Iwamoto, M.; Hirata, Y. Long-Term Stability of a Cellulose-Based Glucose Oxidase Membrane. Materials 2014, 7, 899-905. https://doi.org/10.3390/ma7020899
Yabuki S, Iwamoto M, Hirata Y. Long-Term Stability of a Cellulose-Based Glucose Oxidase Membrane. Materials. 2014; 7(2):899-905. https://doi.org/10.3390/ma7020899
Chicago/Turabian StyleYabuki, Soichi, Miho Iwamoto, and Yoshiki Hirata. 2014. "Long-Term Stability of a Cellulose-Based Glucose Oxidase Membrane" Materials 7, no. 2: 899-905. https://doi.org/10.3390/ma7020899



