Self-Ordered Titanium Dioxide Nanotube Arrays: Anodic Synthesis and Their Photo/Electro-Catalytic Applications
Abstract
:1. Introduction
1.1. Electrochemical Anodization


1.1.1. Nanotube Formation Stages during Anodization

1.1.2. Formation of Complex Nanotube Geometries
1.1.3. Crystallization of Nanotubes
1.2. Surface & Bulk Properties of TiO2
1.3. Photocatalysis and Photoelectrochemistry
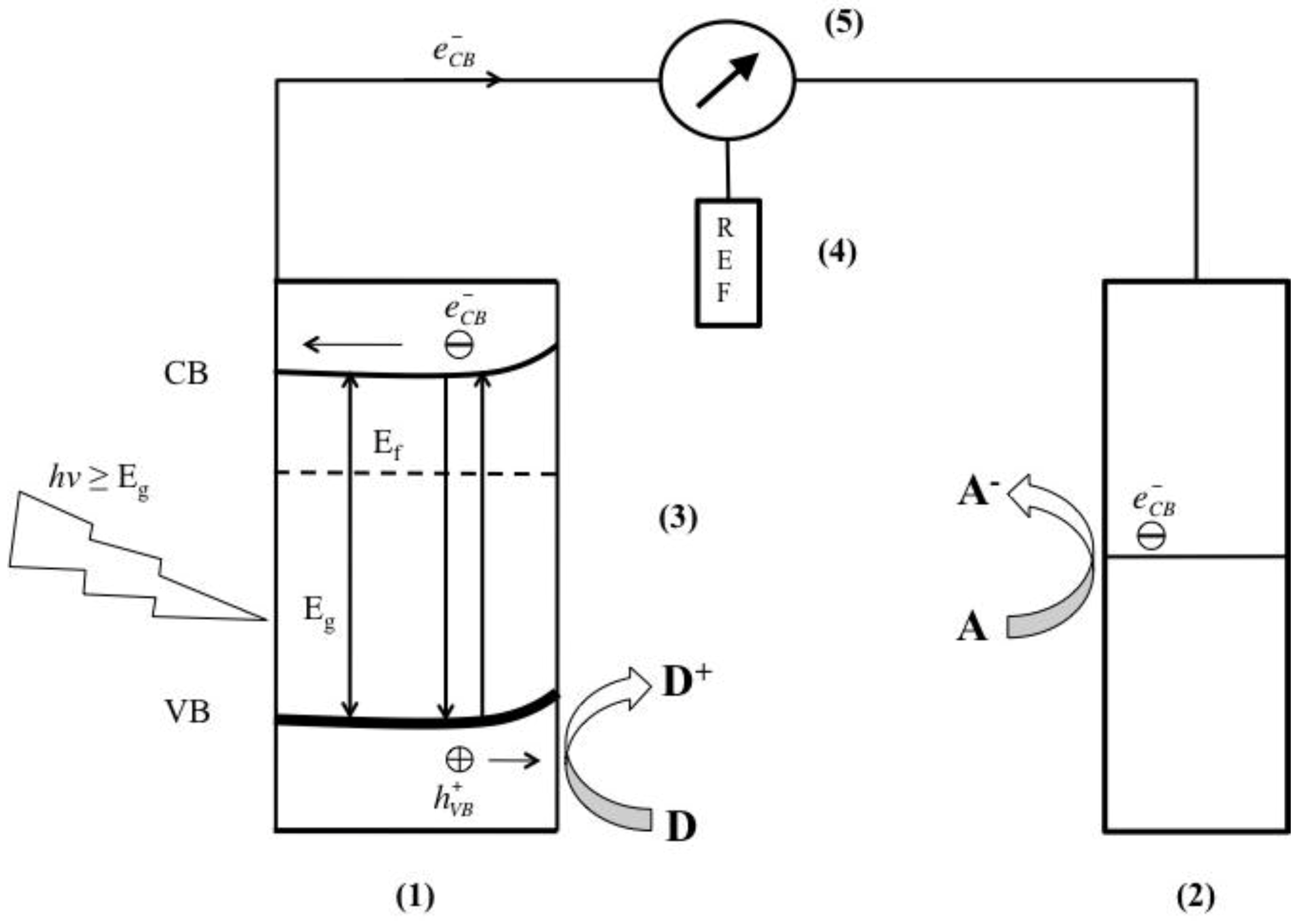
- The redox potential of the photogenerated VB hole should be sufficiently positive for the hole to act as an acceptor;
- The redox potential of the photogenerated CB electron should be sufficiently negative for the electron to act as a donor;
- Photocatayst should be economically available and be environmentally inert;
- Photocatalyst should be stable in a wide pH range and in a variety of electrolytes.
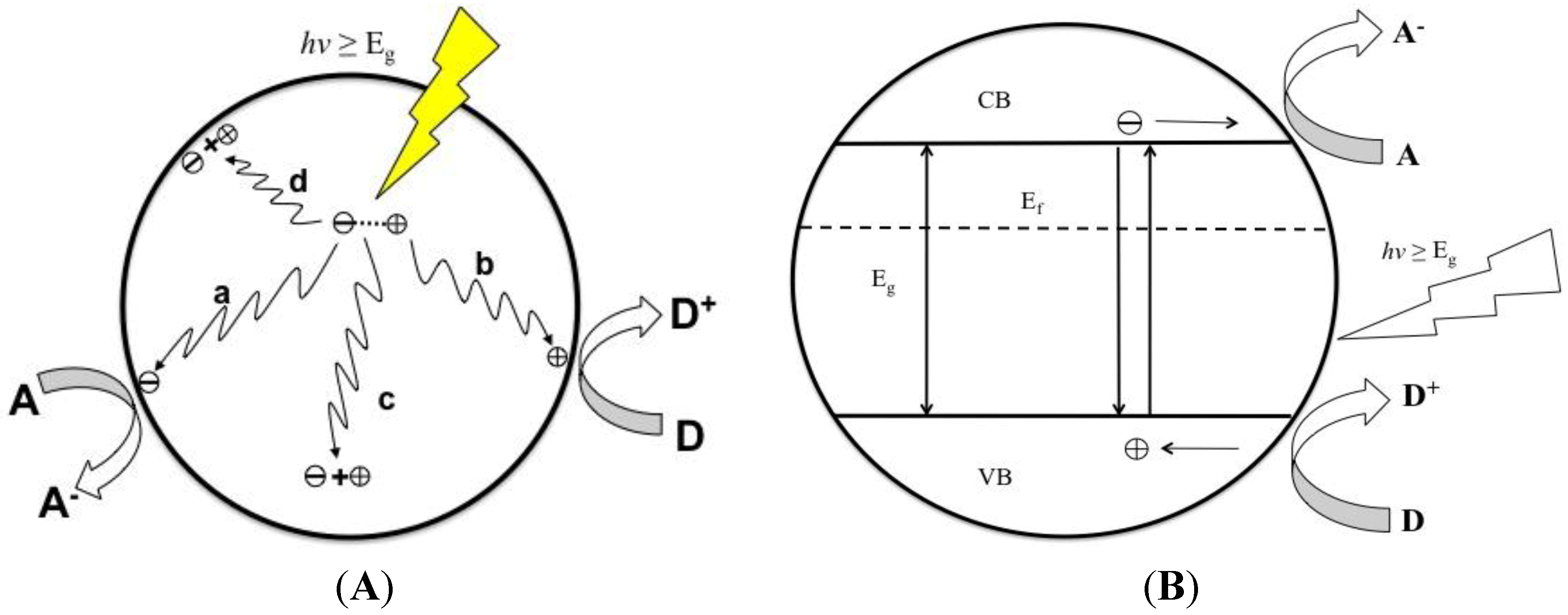
2. Aqueous Photocatalytic Mineralization
2.1. Organic Pollutants


2.2. Surface Charge
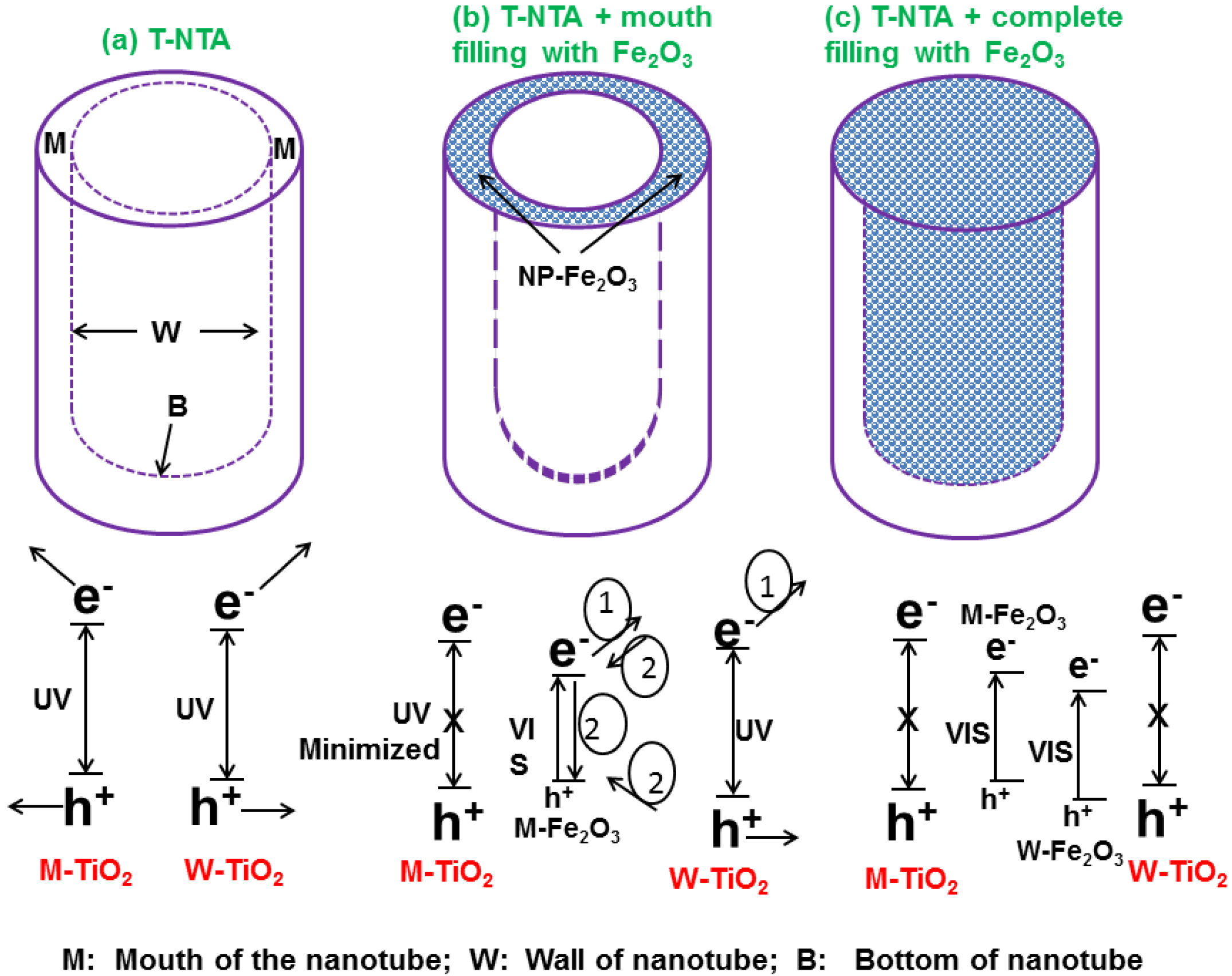
2.3. Nanotube Geometry
2.4. Photocataytic Inactivation of Microorganisms
2.5. Carbon Dioxide Reduction
3. Photoelectro-Catalystic Pollutant Degradation
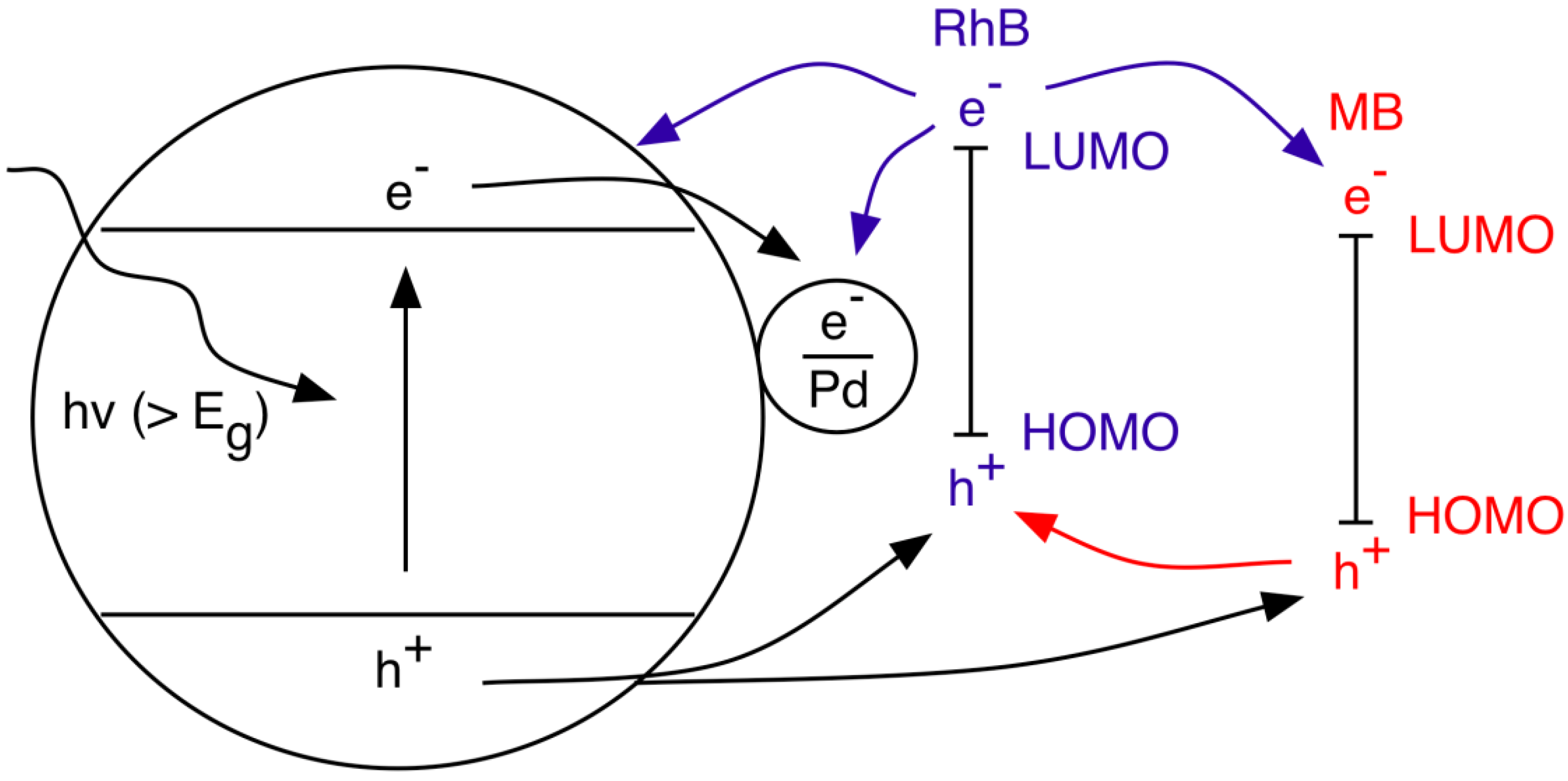
4. Catalyst Modification
4.1. Metal Nanoparticles
4.2. Non-Metal Doping
4.3. Sensitization/Heterostructure
Metal Oxides
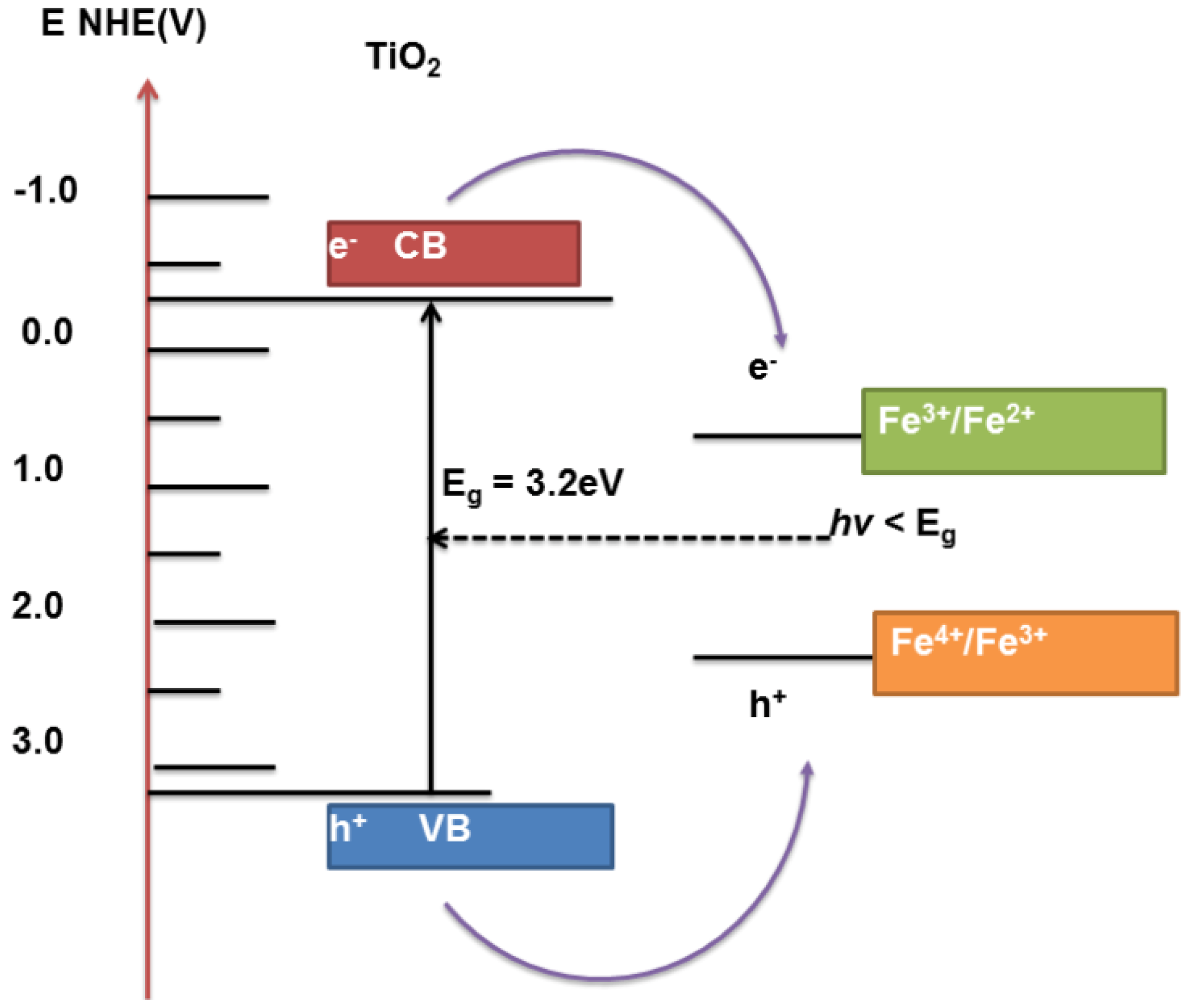
5. Theory
5.1. Computational Protocol
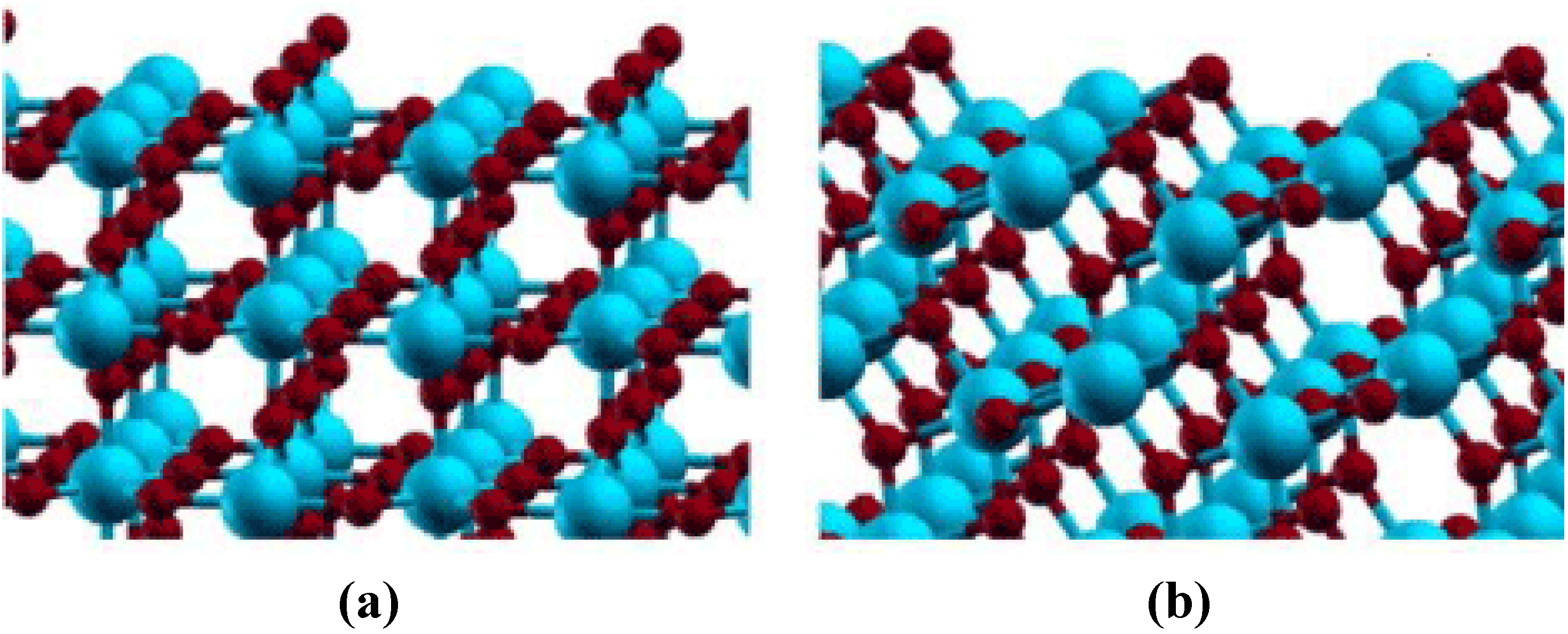
5.1.1. Clean Titania Surface
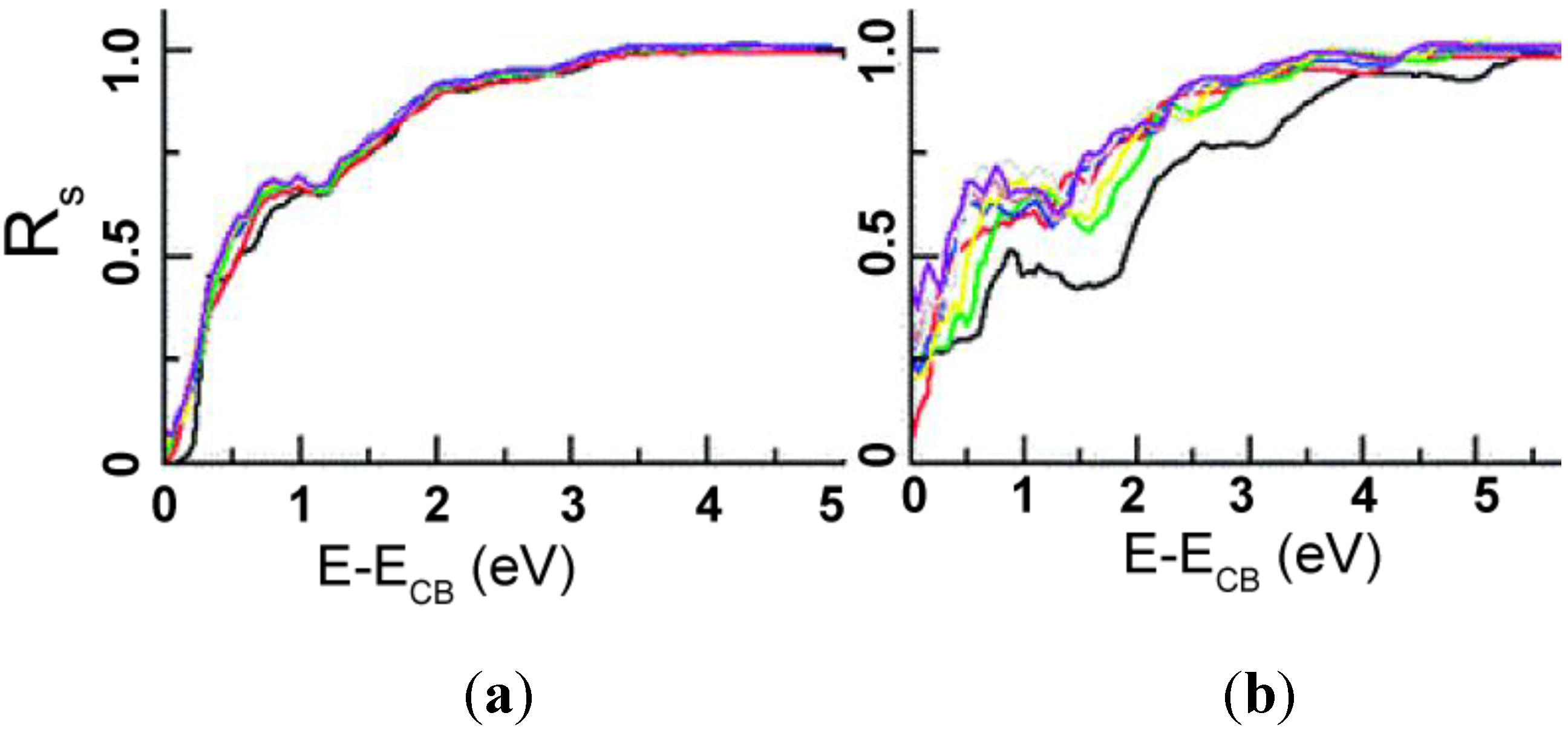
5.1.2. Photocatalysis

5.1.3. Photocatalysis of Organic Compounds
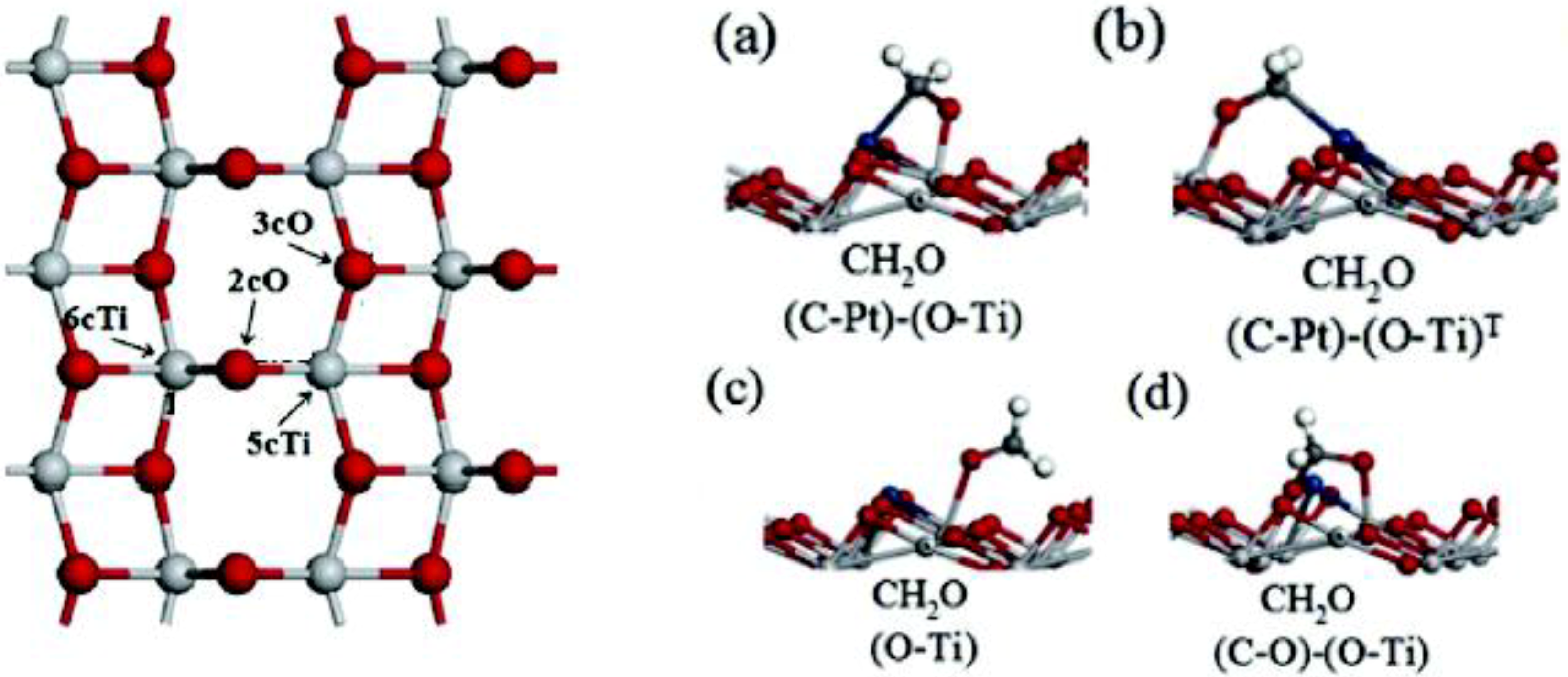
5.1.4. Dye-Sensitized Solar Cell

5.1.5. Chromophore Binding to Titania Surface
- the anchoring group should be present on the electron-accepting group;
- the HOMO and LUMO of the dye should be energetically computed above the VB edge and the CB edge of the oxide, respectively;
- the dye absorption spectrum should match the solar spectrum;
- high intensity transition in the photoconversion;
- the fluorescence lifetime should be sufficiently high (above the nanosecond time scale) to allow an electronic injection to the semiconductor from the excited state of the dye before quenching of the excited state by a radiative decay.
5.2. Electron Transfer Process
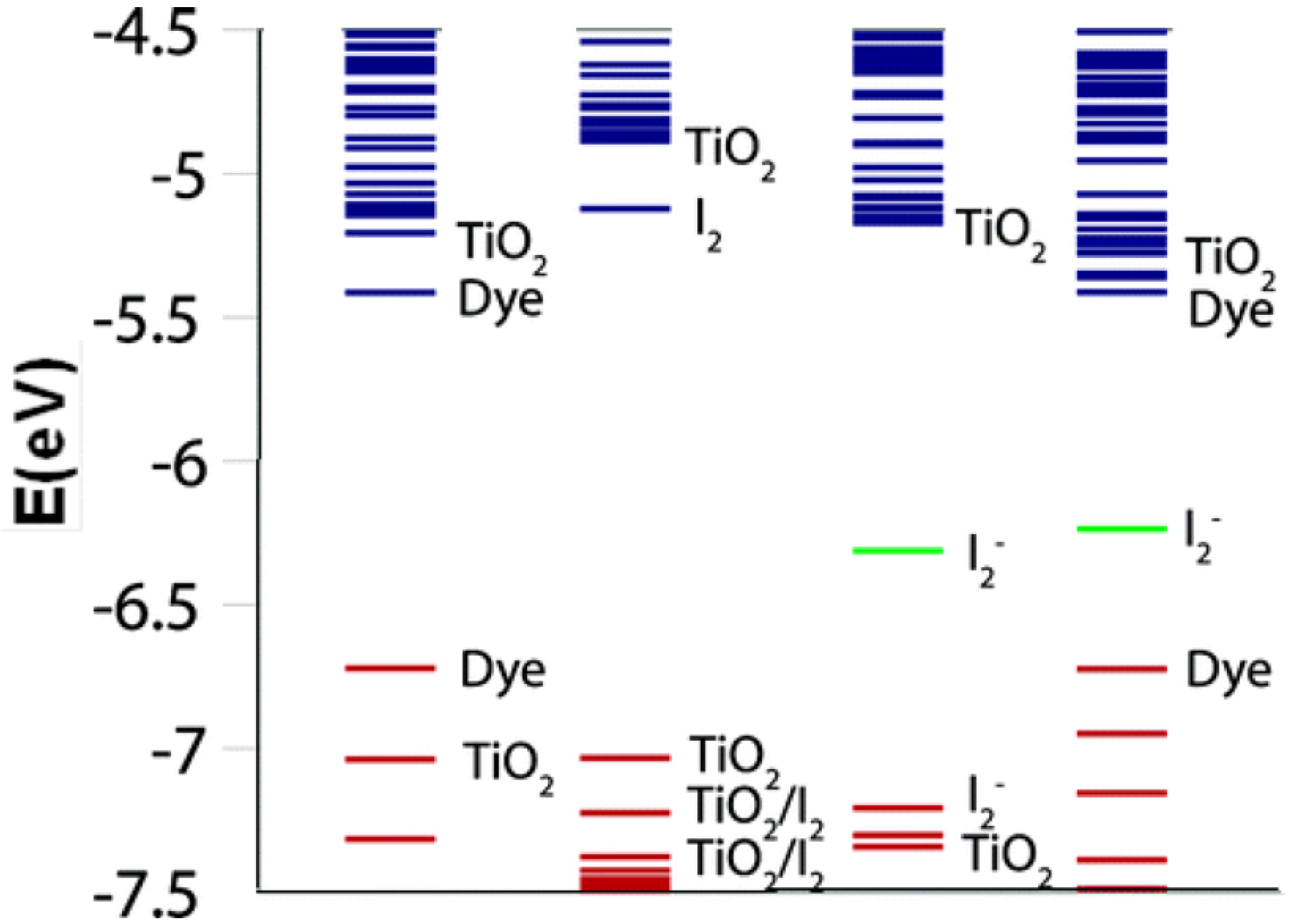
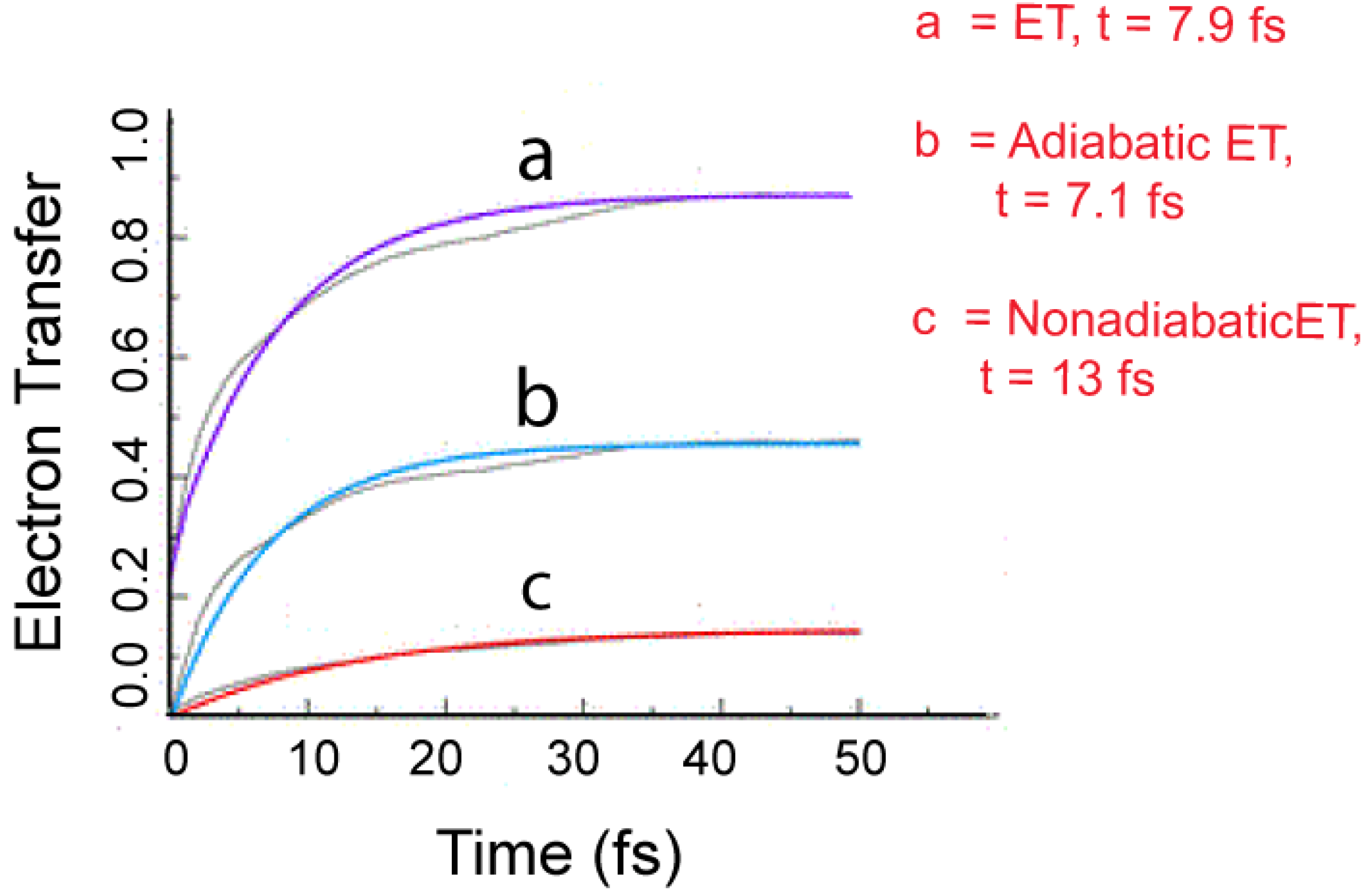

5.3. Future Aspects
6. Concluding Remarks
Acknowledgments
References
- Feynman, R.P. There’s plenty of room at the bottom. Eng. Sci. 1960, 23, 22–36. [Google Scholar]
- Taniguchi, N. On the basic Concept of “Nano-Technology”; The Japan Society of Precision Engineering: Tokyo, Japan, 1974; pp. 18–23. [Google Scholar]
- Devan, R.S.; Patil, R.A.; Lin, J.H.; Ma, Y.R. One-dimensional metal-oxide nanostructures: Recent developments in synthesis, characterization, and applications. Adv. Funct. Mater. 2012, 22, 3326–3370. [Google Scholar] [CrossRef]
- Therese, G.H.A.; Kamath, P.V. Electrochemical synthesis of metal oxides and hydroxides. Chem. Mater. 2000, 12, 1195–1204. [Google Scholar] [CrossRef]
- Wu, X.-J.; Zhu, F.; Mu, C.; Liang, Y.; Xu, L.; Chen, Q.; Chen, R.; Xu, D. Electrochemical synthesis and applications of oriented and hierarchically quasi-1D semiconducting nanostructures. Coordin. Chem. Rev. 2010, 254, 1135–1150. [Google Scholar] [CrossRef]
- Ghicov, A.; Schmuki, P. Self-ordering electrochemistry: A review on growth and functionality of TiO2 nanotubes and other self-aligned MOx structures. Chem. Commun. 2009, 20, 2791–2808. [Google Scholar] [CrossRef]
- Roy, P.; Berger, S.; Schmuki, P. TiO2 Nanotubes: Synthesis and Applications. Angew. Chem. Int. Ed. 2011, 50, 2904–2939. [Google Scholar] [CrossRef]
- Macak, J.M.; Tsuchiya, H.; Ghicov, A.; Yasuda, K.; Hahn, R.; Bauer, S.; Schmuki, P. TiO2 nanotubes: Self-organized electrochemical formation, properties and applications. Curr. Opin. Solid. St. M. 2007, 11, 3–18. [Google Scholar] [CrossRef]
- Misra, M.; Raja, K.S. Ordered Titanium Dioxide Nanotubular Arrays as Photoanodes for Hydrogen Generation. In On Solar Hydrogen & Nanotechnology; Vayssieres, L., Ed.; John Wiley & Sons: Chichester, UK, 2010; Volume 1, pp. 267–290. [Google Scholar]
- Roy, P.; Kim, D.; Lee, K.; Spiecker, E.; Schmuki, P. TiO2 nanotubes and their application in dye-sensitized solar cells. Nanoscale 2010, 2, 45–59. [Google Scholar] [CrossRef] [PubMed]
- Haring, A.; Morris, A.; Hu, M. Controlling morphological parameters of anodized titania nanotubes for optimized solar energy applications. Materials 2012, 5, 1890–1909. [Google Scholar] [CrossRef]
- Paramasivam, I.; Jha, H.; Liu, N.; Schmuki, P. A review of photocatalysis using self-organized TiO2 nanotubes and other ordered oxide nanostructures. Small 2012, 8, 3073–3103. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, V.R.; Sarker, S.; Yu, B.; Kar, A.; Sun, X.; Dey, S.K. TiO2 nanotubes and its composites: Photocatalytic and other photo-driven applications. J. Mater. Res. 2013, 28, 280–293. [Google Scholar] [CrossRef]
- Keller, F.; Hunter, M.S.; Robinson, D.L. Structural features of oxide coatings on aluminum. J. Electrochem. Soc. 1953, 100, 411–419. [Google Scholar] [CrossRef]
- Hebert, K.R.; Albu, S.P.; Paramasivam, I.; Schmuki, P. Morphological instability leading to formation of porous anodic oxide films. Nat. Mater. 2012, 11, 162–166. [Google Scholar] [CrossRef]
- Houser, J.E.; Hebert, K.R. The role of viscous flow of oxide in the growth of self-ordered porous anodic alumina films. Nat. Mater. 2009, 8, 415–420. [Google Scholar] [CrossRef] [PubMed]
- Su, Z.; Zhou, W.; Jiang, F.; Hong, M. Anodic formation of nanoporous and nanotubular metal oxides. J. Mater. Chem. 2012, 22, 535–544. [Google Scholar] [CrossRef]
- Raja, K.S.; Misra, M.; Paramguru, K. Formation of self-ordered nano-tubular structure of anodic oxide layer on titanium. Electrochim. Acta 2005, 51, 154–165. [Google Scholar] [CrossRef]
- Valota, A.; LeClere, D.J.; Skeldon, P.; Curioni, M.; Hashimoto, T.; Berger, S.; Kunze, J.; Schmuki, P.; Thompson, G.E. Influence of water content on nanotubular anodic titania formed in fluoride/glycerol electrolytes. Electrochim. Acta 2009, 18, 4321–4327. [Google Scholar] [CrossRef]
- Richter, C.; Wu, Z.; Panaitescu, E.; Willey, R.J.; Menon, L. Ultrahigh-aspect-ratio titania nanotubes. Adv. Mater. 2007, 19, 946–948. [Google Scholar] [CrossRef]
- Hahn, R.; Macak, J.M.; Schmuki, P. Rapid anodic growth of TiO2 and WO3 nanotubes in fluoride free electrolytes. Electrochem. Commun. 2007, 9, 947–952. [Google Scholar] [CrossRef]
- Zwilling, V.; Aucouturier, M.; Darque-Ceretti, E. Anodic oxidation of titanium and TA6V alloy in chromic media. An electrochemical approach. Electrochim. Acta 1999, 45, 921–929. [Google Scholar] [CrossRef]
- Zwilling, V.; Darque-Ceretti, E.; Boutry-Forveille, A.; David, D.; Perrin, M.Y.; Ancouturier, M. Structure and physicochemistry of anodic oxide films on titanium and TA6V alloy. Surf. Interface Anal. 1999, 27, 629–637. [Google Scholar] [CrossRef]
- Gong, D.; Grimes, C.A.; Varghese, O.K.; Hua, W.; Singh, R.S.; Chen, Z.; Dickey, E.C. Titanium oxide nanotube arrays prepared by anodic oxidation. J. Mater. Res. 2001, 16, 3331–3334. [Google Scholar] [CrossRef]
- Macak, J.M.; Sirotna, K.; Schmuki, P. Self-organized porous titanium oxide prepared in Na2SO4/NaF electrolytes. Electrochim. Acta 2005, 50, 3679–3684. [Google Scholar] [CrossRef]
- Macak, J.M.; Tsuchiya, H.; Schmuki, P. High-aspect-ratio TiO2 nanotubes by anodization of titanium. Angew. Chem. Int. Ed. 2005, 44, 2100–2102. [Google Scholar] [CrossRef]
- Raja, K.S.; Gandhi, T.; Misra, M. Effect of water content of ethylene glycol as electrolyte for synthesis of ordered titania nanotubes. Electrochem. Commun. 2007, 9, 1069–1076. [Google Scholar] [CrossRef]
- Mor, G.K.; Varghese, O.K.; Paulose, M.; Grimes, C.A. Transparent highly ordered TiO2 nanotube arrays via anodization of titanium thin films. Adv. Funct. Mater. 2005, 15, 1291–1296. [Google Scholar] [CrossRef]
- Yu, X.; Li, Y.; Ge, W.; Yang, Q.; Zhu, N.; Zadeh, K.K. Formation of nanoporous titanium oxide films on silicon substrates using an anodization process. Nanotechnology 2006, 17, 808–814. [Google Scholar] [CrossRef]
- Macak, J.M.; Tsuchiya, H.; Berger, S.; Bauer, S.; Fujimoto, S.; Schmuki, P. On wafer TiO2 nanotube-layer formation by anodization of Ti-films on Si. Chem. Phys. Lett. 2006, 428, 421–425. [Google Scholar] [CrossRef]
- Chappanda, K.N.; Smith, Y.R.; Mohanty, S.K.; Rieth, L.W.; Tathireddy, P.; Misra, M. Growth and characterization of TiO2 nanotubes from sputtered Ti film on Si substrate. Nanoscale. Res. Lett. 2012, 7, 388–395. [Google Scholar] [CrossRef] [PubMed]
- Chappanda, K.N.; Smith, Y.R.; Misra, M.; Mohanty, S.K. Site-specific and patterned growth of TiO2 nanotube arrays from e-beam evaporated thin titanium film on Si wafer. Nanotechnology 2012, 23. [Google Scholar] [CrossRef]
- Kar, A.; Smith, Y.R.; Subramanian, V.R. Improved photocatalytic degradation of textile dye using titanium dioxide nanotubes formed over titanium wires. Environ. Sci. Technol. 2009, 43, 3260–3265. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Misra, M. Dye-sensitized photovoltaic wires using highly ordered TiO2 nanotube arrays. ACS Nano 2010, 4, 2196–2200. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Hu, Z.; Huang, H.; Zhang, Q.; Zhang, T.; Zhai, J.; Jiang, L. Heterogeneous 3-D nanotubular arrays of CdS-TiO2: Efficient collections of reflection light for enhanced photoelectric output. J. Mater. Chem. 2012, 22, 22120–22125. [Google Scholar] [CrossRef]
- Liu, Z.; Subramanian, V.; Misra, M. Vertically oriented TiO2 nanotube arrays grown on Ti meshes for flexible dye-sensitized solar cells. J. Phys. Chem. C 2009, 113, 14028–14033. [Google Scholar] [CrossRef]
- Smith, Y.R.; Subramanain, V. Heterostructural composites of TiO2 mesh-TiO2 nanoparticles photosensitized with CdS: A new flexible photoanode for solar cells. J. Phys. Chem. C 2011, 115, 8376–8385. [Google Scholar] [CrossRef]
- Liu, Z.; Zhang, Q.; Zhao, T.; Zhai, J.; Jiang, L. 3-D vertical arrays of TiO2 nanotubes on Ti meshes: Efficient photoanodes for water photoelectrolysis. J. Mater. Chem. 2011, 21, 10354–10358. [Google Scholar] [CrossRef]
- Chen, B.; Lu, K. Influence of patterned concave depth and surface curvature on anodization of titania nanotubes and alumina nanopores. Langmuir 2011, 27, 12179–12185. [Google Scholar] [CrossRef] [PubMed]
- Mohapatra, S.K.; John, S.E.; Banerjee, S.; Misra, M. Water Photooxidation by smooth and ultrathin α-Fe2O3 nanotube arrays. Chem. Mater. 2009, 21, 3048–3055. [Google Scholar] [CrossRef]
- Rangaraju, R.R.; Panday, A.; Raja, K.S.; Misra, M. Nanostructured anodic iron oxide film as photoanode for water oxidation. J. Phys. D Appl. Phys. 2009, 42. [Google Scholar] [CrossRef]
- Rangaraju, R.R.; Raja, K.S.; Panday, A.; Misra, M. Low-cost photoelectrocatalyst based on a nanoporous oxide layer of low-carbon steel. J. Phys. D Appl. Phys. 2010, 43. [Google Scholar] [CrossRef]
- Banerjee, S.; Mohapatra, S.K.; Misra, M. Synthesis of TaON nanotube arrays by sonoelectrochemical anodization followed by nitridation: A novel catalyst for photoelectrochemical hydrogen generation from water. Chem. Commun. 2009, 46, 7137–7139. [Google Scholar] [CrossRef]
- Kim, D.; Fujimoto, S.; Schmuki, P.; Tsuchiy, H. Nitrogen doped anodic TiO2 nanotubes grown from nitrogen-containing Ti alloys. Electrochem. Commun. 2008, 10, 910–913. [Google Scholar] [CrossRef]
- Allam, N.K.; Poncheri, A.J.; El-Sayed, M.A. Vertically oriented Ti–Pd mixed oxynitride nanotube arrays for enhanced photoelectrochemical water splitting. ACS Nano 2011, 5, 5056–5066. [Google Scholar] [CrossRef] [PubMed]
- Das, C.; Paramasivam, I.; Liu, N.; Schmuki, P. Photoelectrochemical and photocatalytic activity of tungsten doped TiO2 nanotube layers in the near visible region. Electrochim. Acta 2011, 56, 10557–10561. [Google Scholar] [CrossRef]
- Roy, P.; Das, C.; Lee, K.; Hahn, R.; Ruff, T.; Moll, M.; Schmuki, P. Oxide nanotubes on Ti−Ru alloys: Strongly enhanced and stable photoelectrochemical activity for water splitting. J. Am. Chem. Soc. 2011, 133, 5629–5631. [Google Scholar] [CrossRef] [PubMed]
- Allam, N.K.; Alamgir, F.; El-Sayed, M.A. Enhanced photoassisted water electrolysis using vertically oriented anodically fabricated Ti−Nb−Zr−O mixed oxide nanotube arrays. ACS Nano 2010, 4, 5819–5826. [Google Scholar] [CrossRef] [PubMed]
- Taveira, L.V.; Macak, J.M.; Sirotna, K.; Dick, L.F.P.; Schmuki, P. Voltage oscillations and morphology during the galvanostatic formation of self-organized TiO2 nanotubes. J. Electrochem. Soc. 2006, 153, B137–B143. [Google Scholar] [CrossRef]
- Chanmanee, W.; Watcharenwong, A.; Chenthamarakshan, C.R.; Kajitvichyanukul, P.; Tacconi, N.R.D.; Rajeshwar, K. Formation and characterization of self-organized TiO2 nanotube arrays by pulse anodization. J. Am. Chem. Soc. 2008, 130, 965–974. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Tovar, R.; Lee, K.; García-Antón, J.; Schmuki, P. Formation of anodic TiO2 nanotube or nanosponge morphology determined by the electrolyte hydrodynamic conditions. Electrochem. Commun. 2013, 26, 1–4. [Google Scholar] [CrossRef]
- Sánchez-Tovar, R.; Paramasivam, I.; Lee, K.; Schmuki, P. Influence of hydrodynamic conditions on growth and geometry of anodic TiO2 nanotubes and their use towards optimized DSSCs. J. Mater. Chem. 2012, 22, 12792–12795. [Google Scholar] [CrossRef]
- Mohapatra, S.K.; Misra, M.; Mahajan, V.K.; Raja, K.S. A novel method for the synthesis of titania nanotubes using sonoelectrochemical method and its application for photoelectrochemical splitting of water. J. Catal. 2007, 246, 362–369. [Google Scholar] [CrossRef]
- Sohn, Y.S.; Smith, Y.R.; Raja, K.S.; Subramanian, V.; Misra, M. Sonoelectrochemical Synthesis of Low Band Gap Titania Nanotubes for Photoelectrochemical Generation of Hydrogen. In Solar Hydrogen and Nanotechnology II; Guo, J., Ed.; SPIE: San Diego, CA, USA, 2007. [Google Scholar]
- Sohn, Y.S.; Smith, Y.R.; Subramanian, V.R.; Misra, M. Electrochemically assisted photocatalytic degradation of methyl orange using anodized titanium dioxide nanotubes. Appl. Catal. B Environ. 2008, 84, 372–378. [Google Scholar] [CrossRef]
- Smith, Y.R.; Sarma, B.; Mohanty, S.K.; Misra, M. Light-assisted anodized TiO2 nanotube arrays. ACS Appl. Mater. Interfaces 2012, 4, 5883–5890. [Google Scholar] [CrossRef] [PubMed]
- Macak, J.M.; Hildebrand, H.; Marten-Jahns, U.; Schmuki, P. Mechanistic aspects and growth of large diameter self-organized TiO2 nanotubes. J. Electroanal. Chem. 2008, 621, 254–266. [Google Scholar] [CrossRef]
- Crawford, G.A.; Chawla, N. Tailoring TiO2 nanotube growth during anodic oxidation by crystallographic orientation of Ti. Scripta. Mater. 2009, 60, 874–877. [Google Scholar] [CrossRef]
- Mohammadpour, A.; Waghmare, P.; Mitra, S.; Shankar, K. Anodic growth of large-diameter multipodal TiO2 nanotubes. ACS Nano 2010, 4, 7421–7430. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Lu, K. Hierarchically branched titania nanotubes with tailored diameters and branch numbers. Langmuir 2012, 28, 2937–2943. [Google Scholar] [CrossRef] [PubMed]
- Mohapatra, S.; Misra, M.; Mahanjan, V.; Raja, K. Synthesis of y-branched TiO2 nanotubes. Mater. Lett. 2008, 62, 1772–1774. [Google Scholar] [CrossRef]
- Lin, C.; Chen, S.; Cao, L. Anodic formation of aligned and bamboo-type TiO2 nanotubes at constant low voltages. Mater. Sci. Semicond. Proc. 2013, 16, 154–159. [Google Scholar] [CrossRef]
- Endut, Z.; Hamdi, M.; Basirun, W. Supercapacitance of bamboo-type anoic titania nanotube arrays. Surf. Coat. Technol. 2012, 215, 75–78. [Google Scholar] [CrossRef]
- Kim, D.; Ghicov, A.; Albu, S.; Schmuki, P. Bamboo-type TiO2 nanotubes: Improved conversion efficiency in dye-sensitized solar cells. J. Am. Chem. Soc. 2008, 130, 16454–16455. [Google Scholar] [CrossRef] [PubMed]
- Albu, S.P.; Kim, D.; Schmuki, P. Growth of aligned TiO2 bamboo-type nanotubes and highly ordered nanolace. Angew. Chem. Int. Ed. 2008, 47, 1916–1919. [Google Scholar] [CrossRef]
- Ye, M.; Xin, X.; Lin, C.; Lin, Z. High efficiency dye-sensitized solar cells based on hierarchically structured nanotubes. Nano Lett. 2011, 11, 3214–3220. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Hossain, M.F.; Takahashi, T. Photoelectrochemical water splitting on highly smooth and ordered TiO2 nanotube arrays for hydrogen generation. Int. J. Hydrog. Energ. 2010, 35, 8528–8535. [Google Scholar] [CrossRef]
- Smith, Y.R.; Sarma, B.; Mohanty, S.K.; Misra, M. Single-step anodization for synthesis of hierarchical TiO2 nanotube arrays on foil and wire substrate for enhanced photoelectrochemical water splitting. Int. J. Hydrog. Energ. 2013, 38, 2062–2069. [Google Scholar] [CrossRef]
- John, S.; Mohapatra, S.; Misra, M. Double-wall anodic titania nanotube arrays for water photooxidation. Langmuir 2009, 25, 8240–8247. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, V.K.; Misra, M.; Raja, K.S.; Mohapatra, S.K. Self-organized TiO2 nanotubular arrays for photoelectrochemical hydrogen generation: Effect of crystallization and defect structures. J. Phy. D Appl. Phys. 2008, 41. [Google Scholar] [CrossRef]
- Oh, H.; Hock, R.; Schurr, R.; Holzing, A.; Chi, C. Phase transformation and photocatalytic characteristics of anodic TiO2 nanotublar film. J. Phys. Chem. Solids 2013, 74, 708–715. [Google Scholar] [CrossRef]
- Kang, X.; Chen, S. Photocatalytic reduction of methylene blue by TiO2 nanotube arrays: Effects of TiO2 crystalline phase. J. Mater. Sci. 2010, 45, 2696–2702. [Google Scholar] [CrossRef]
- Wang, D.; Liu, L.; Zhang, F.; Tao, K.; Pippel, E.; Domen, K. Spontaneous phase and morphology transformations of anodized titania nanotubes induced by water at room temperature. Nano Lett. 2011, 11, 3649–3655. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.; Albu, S.P.; Lee, K.; So, S.; Schmuki, P. Water annealing and other low temperature treatments of anodic TiO2 nanotubes: A comparison of properties and efficiencies in dye sensitized solar cells and for water splitting. Electrochim. Acta 2010, 82, 98–102. [Google Scholar] [CrossRef]
- Diebold, U. The surface science of titanium dioxide. Surf. Sci. Rep. 2003, 48, 53–229. [Google Scholar] [CrossRef]
- Penn, R.; Banfield, J. Formation of rutile nuclei at anatase {112} twin interfaces and the phase transformation mechanism in nanocrystalline titania. Am. Mineral. 1999, 84, 871–876. [Google Scholar]
- Gateshki, M.; Yin, S.; Ren, Y.; Petkov, V. Titania polymorphs by soft chemistry: Is there a common structural pattern? Chem. Mater. 2007, 19, 2512–2518. [Google Scholar] [CrossRef]
- Pradhan, S.; Mao, Y.; Wong, S.; Chupas, P.; Petkov, V. Atomic-scale structure of nanosized titania and titanate: Particles, wires, and tubes. Chem. Mater. 2007, 19, 6180–6186. [Google Scholar] [CrossRef]
- Barnard, A.; Xu, H. An environmentally sensitive phase map of titania nanocrystals. ACS Nano 2008, 2, 2237–2242. [Google Scholar] [CrossRef] [PubMed]
- Noguera, C. Physics and Chemistry at Oxide Surfaces; Cambridge University Press: New York, NY, USA, 1996. [Google Scholar]
- Mandzy, N.; Grulke, E.; Druffel, T. Breakage of TiO2 agglomerates in electrostatically stabilized aqueous dispersions. Power Technol. 2005, 160, 121–126. [Google Scholar] [CrossRef]
- Bullard, J.; Cima, M. Orientation dependence of the isoelectric point of TiO2 (Rutile) surfaces. Langmuir 2006, 22, 10264–10271. [Google Scholar] [CrossRef] [PubMed]
- Kitano, M.; Wada, E.; Nakajima, K.; Hayashi, S.; Miyazaki, S.; Kobayashi, H.; Hara, M. Protonated titanate nanotubes with Lewis and Bronsted acidity: Relationship between nanotube structure and catalytic activity. Chem. Mater. 2013, 25, 385–393. [Google Scholar] [CrossRef]
- Becquerel, A.E. Recherches sur les effect de la resiation chimique de la lumiere solaire, su moyen des courants electriques (in French). C.R. Acad. Sci. 1839, 9, 145–149. [Google Scholar]
- Gerischer, H. Advances in Electrochemistry and Electrochemistry Engineering; Interscience Publishing: New York, NY, USA, 1961; Volume 1. [Google Scholar]
- Memming, R.; Schwandt, G. Electrochemical properties of gallium phosphide in aqueous solutions. Electrochim. Acta 1968, 13, 1299–1310. [Google Scholar] [CrossRef]
- Williams, R. Becquerel photovoltaic effect in binary compounds. J. Chem. Phys. 1960, 32, 1505–1515. [Google Scholar] [CrossRef]
- Fujishima, A.; Honda, K. Electrochemical evidence for the mechanism of the primary stage of photosynthesis. Bull. Chem. Soc. Jpn. 1971, 44, 1148–1150. [Google Scholar] [CrossRef]
- Fujishima, A.; Honda, K. Electrochemical photolysis of water at a semiconductor electrode. Nature 1972, 238, 37–38. [Google Scholar] [CrossRef] [PubMed]
- Nozik, A.J.; Memming, R. Physical chemistry of semiconductor-liquid interfaces. J. Phys. Chem. C 1996, 100, 13061–13078. [Google Scholar] [CrossRef]
- Lewis, N.S.; Nocera, D.G. Powering the planet: Chemical challenges in solar energy utilization. Proc. Natl. Acad. Sci. USA 2006, 103, 15729–15735. [Google Scholar] [CrossRef] [PubMed]
- Gratzel, M. Photoelectrochemical cells. Nature 2001, 414, 338–344. [Google Scholar] [CrossRef] [PubMed]
- Gratzel, M. Dye-sensitized solar cells. J. Photochem. Photobiol. C 2003, 4, 145–153. [Google Scholar] [CrossRef]
- O’Regan, B.; Gratzel, M. A low-cost, high-efficiency solar cell based on dye-sensitized colloidal TiO2 films. Nature 1991, 353, 737–740. [Google Scholar] [CrossRef]
- Ollis, D.F.; Pelizzetti, E.; Serpone, N. Photocatalyzed destruction of water contaminants. Environ. Sci. Technol. 1991, 25, 1522–1529. [Google Scholar] [CrossRef]
- Rajeshwar, K.; Osugi, M.E.; Chanmanee, W.; Chenthamarakshan, C.R.; Zanoni, M.V.B.; Kajitvichyanukul, P.; Krishnan-Ayer, R. Heterogeneous photocatalytic treatment of organic dyes in air and aqueous media. J. Photochem. Photobiol. C 2008, 9, 171–192. [Google Scholar] [CrossRef]
- Wang, R.; Hashimoto, K.; Fujishima, A.; Chikuni, M.; Kojima, E.; Kitamura, A.; Shimohigoshi, M.; Watanabe, T. Light-induced amphiphilic surfaces. Nature 1997, 388, 431–432. [Google Scholar] [CrossRef]
- Wolfrum, E.J.; Huang, J.; Blake, D.M.; Maness, P.-C.; Huang, Z.; Fiest, J. Photocatalytic oxidation of bacteria, bacterial and fungal spores, and model biofilm components to carbon dioxide on titanium dioxide-coated surfaces. Environ. Sci. Technol. 2002, 36, 3412–3419. [Google Scholar] [CrossRef] [PubMed]
- Indrakanti, V.P.; Kubickib, J.D.; Schobert, H.H. Photoinduced activation of CO2 on Ti-based heterogeneous catalysts: Current state, chemical physics-based insights and outlook Energ. Environ. Sci. 2009, 2, 745–758. [Google Scholar]
- Smith, Y.R. Physicochemical and Geometrical Factors that Influences the Photocatalytic Degradation Kinetics of A Model Water Contaminate; University of Nevada: Reno, NV, USA, 2011. [Google Scholar]
- Bard, A.J. Phtotelectrochemistry and heterogeneous photocatalysis at semiconductors. J. Photochem. 1979, 10, 59–75. [Google Scholar] [CrossRef]
- Dijkstra, M.; Michorius, A.; Buwalda, H.; Panneman, H.; Winkelman, J.; Beenackers, A. Comparison of the efficiency of immobilized and suspended systems in photocatalytic degradation. Catal. Today 2001, 66, 487–494. [Google Scholar] [CrossRef]
- Subramanian, V.; Kamat, P.; Wolf, E. Mass-transfer and kinetic studies during the photocatalytic degradation of an azo dye on optically trasnparent electrode thin film. Ind. Eng. Chem. Res. 2003, 42, 2131–2138. [Google Scholar] [CrossRef]
- Mascolo, G.; Comparelli, R.; Curri, M.; Lovecchio, G.; Lopez, A.; Agostiano, A. Photocatalytic degradation of methyl red by TiO2: Comparison of the efficiency of immobilized nanoparticles versus conventional suspended catalyst. J. Hazard. Mater. 2007, 142, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Carneiro, P.; Osugi, M.; Sene, J.; Anderson, M.; Zanoni, M. Evaluation of color removal and degradation of a reactive textile azo dye on nanoporous TiO2 thin-film electrodes. Electrochim. Acta 2004, 49, 3807–3820. [Google Scholar] [CrossRef]
- Guaraldo, T.; Pulchinelli, S.; Zanoni, M. Influence of particle size on the photoactivity of Ti/TiO2 thin film electrodes, and enhanced photoelectrocatalytic degradation of indigo carmine dye. J. Photoch. Photobiol. A 2011, 217, 259–266. [Google Scholar] [CrossRef]
- Noorjahan, M.; Reddy, M.; Kumari, V.; Lavedrine, B.; Boule, P.; Subrahmanyam, M. Photocatalytic degradation of H-acid over a novel TiO2 thin film fixed bed reactor and in aqueous suspensions. J. Photoch. Photobiol. A 2003, 156, 179–187. [Google Scholar] [CrossRef]
- Rachel, A.; Subrahmanyam, M.; Boule, P. Comparison of photocatalytic efficiencies of TiO2 in suspended and immobilised form for the photocatalytic degradation of nitrobenzenesulfonic acids. Appl. Catal. B Environ. 2002, 37, 301–308. [Google Scholar] [CrossRef]
- Zlamal, M.; Macak, J.; Schmuki, P.; Krysa, J. Electrochemically assisted photocatalysis on self-organized TiO2 nanotubes. Electrochem. Commun. 2007, 9, 2822–2826. [Google Scholar] [CrossRef]
- Hashimoto, K.; Irie, H.; Fujishima, A. TiO2 photocatalysis: A histroical overview and future prospects. Jpn. J. Appl. Phys. 2005, 44, 8269–8285. [Google Scholar] [CrossRef]
- Mills, A.; LeHunte, S. An overview of semiconductor photocatalysis. J. Photoch. Photobiol. A 1997, 108, 1–35. [Google Scholar] [CrossRef]
- Konstantinou, I.; Albanis, T. TiO2-assisted photocatalytic degradation of azo dyes in aqueous solution: Kinetic and mechanistic investigations. A review. Appl. Catal. B Environ. 2004, 49, 1–14. [Google Scholar] [CrossRef]
- Ollis, D. Kinetics of liquid phase photocatalyzed reactions: An illuminating approach. J. Phys. Chem. B 2005, 109, 2439–2444. [Google Scholar] [CrossRef] [PubMed]
- Beranek, R. (Photo)electrochemical methods for the determination of the band edge positions of TiO2-based nanomaterials. Adv. Phys. Chem. 2011. [Google Scholar] [CrossRef]
- Kumar, S.; Devi, L. Review on modified TiO2 photocatalysis under UV/Visible light: Selected results and related mechanisms on interfacial charge carrier transfer dynamics. J. Phys. Chem. A 2011, 115, 13211–13241. [Google Scholar] [CrossRef] [PubMed]
- Brugnera, M.; Rajeshwar, K.; Cardoso, J.; Zanoni, M. Bisphenol A removal from wastewater using self-organized TiO2 nanotublar array electrodes. Chemosphere 2010, 78, 569–575. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Yu, Y.; Wang, P. Hierarchical top-porous/bottom-tubular TiO2 nanostructures decorated with Pd nanoparticles for efficient photoelectrocatalytic decomposition of synergistic pollutants. ACS Appl. Mater. Interfaces 2012, 4, 990–996. [Google Scholar] [CrossRef] [PubMed]
- Ku, Y.; Lee, Y.; Wang, W. Photocatalytic Decomposition of 2-chlorophenol in aqueous solution by UV/TiO2 process with applied external bias voltage. J. Hazard. Mater. 2006, 138, 350–356. [Google Scholar] [CrossRef] [PubMed]
- Liang, H.; Li, X.; Yang, Y.; Sze, K. Effects of dissolved oxygen, pH, and anions on the 2,3-dichlorophenol degradation by photocatalytic reaction with anodic TiO2 nanotube films. Chemosphere 2008, 73, 805–812. [Google Scholar] [CrossRef] [PubMed]
- Liao, J.; Lin, S.; Zhang, L.; Pan, N.; Cao, X.; Li, J. Photocatalytic degradation of methyl orange using a TiO2/Ti mesh electrode with 3D nanotube arrays. ACS Appl. Mater. Interfaces 2012, 4, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Baram, N.; Starosvetsky, D.; Starosvetsky, J.; Epshtein, M.; Armon, R.; Ein-Eli, Y. Photocatalytic inactivation of microorganisms using nanotublar TiO2. Appl. Catal. B Environ. 2011, 101, 212–219. [Google Scholar] [CrossRef]
- Liang, H.; Li, X. Effects of structure of anodic TiO2 nanotube arrays on photocatalytic activity for the degradation of 2,3-dichlorophenol in aqueous solution. J. Hazard. Mater. 2009, 162, 1415–1422. [Google Scholar] [CrossRef] [PubMed]
- Oh, H.; Lee, J.; Kim, Y.; Suh, S.; Lee, J.; Chi, C. Synthesis of effective titania nanotubes for wastewater purification. Appl. Catal. B Environ. 2008, 84, 142–147. [Google Scholar] [CrossRef]
- Smith, Y.R.; Kar, A.; Subramanian, V.R. Investigation of physicochemical parameters that influence photocatalytic degradation of methyl orange over TiO2 nanotubes. Ind. Eng. Chem. Res. 2009, 48, 10268–10276. [Google Scholar] [CrossRef]
- Lv, X.; Zhang, H.; Chang, H. Improved photocatalytic activity of highly ordered TiO2 nanowire arrays for methylene blue degradation. Mater. Chem. Phys. 2012, 136, 789–795. [Google Scholar] [CrossRef]
- Jaeger, V.; Wilson, W.; Subramanian, V.R. Photodegradation of methyl orange and 2,3-butanedione on titanium-dioxide nanotube arrays efficiently synthesized on titanium coils. Appl. Catal. B Gen. 2011, 110, 6–13. [Google Scholar] [CrossRef]
- Benabbou, A.; Derriche, Z.; Felix, C.; Lejeune, P.; Guillard, C. Photocatalytic inactivation of Escherischia coli. Effect of concentration of TiO2 and microorganism, nature, and intensity of UV irradiation. Appl. Catal. B Environ. 2007, 76, 257–263. [Google Scholar] [CrossRef]
- Hou, Y.; Li, X.; Zhao, Q.; Chen, G.; Raston, C. Role of hydroxyl radicals and mechaniam of escherichia coli inactivation of Ag/AgBr/TiO2 nanotube array electrode under visible light irradiation. Environ. Sci. Technol. 2012, 46, 4042–4050. [Google Scholar] [CrossRef] [PubMed]
- Sun, D.; Tay, J.; Tan, K. Photocatalytic degradation of E. coliform in water. Water Res. 2003, 37, 3452–3462. [Google Scholar] [CrossRef] [PubMed]
- Matsunaga, T.; Tomada, R.; Nakajima, T.; Wake, H. Photochemical sterilization of microbial cells by semiconductor powders. FEMS Microbiol. Lett. 1985, 29, 211–214. [Google Scholar] [CrossRef]
- Kang, Q.; Lu, Q.; Liu, S.; Yang, L.; Wen, L.; Luo, S.; Cai, Q. A ternary hybrid CdS/Pt-TiO2 nanotube structure for photoelectrocatalytic bactericidal effects on Escherichia Coli. Biomaterials 2012, 31, 3317–3326. [Google Scholar] [CrossRef]
- Melillo, J.M.; McGuire, A.D.; Kicklighter, D.W.; Moore, B.; Vorosmarty, C.J.; Schloss, A.L. Global climate-change and terrestrial net primary production. Nature 1993, 363, 234–240. [Google Scholar] [CrossRef]
- Schimel, D.S. Terrestrial ecosystems and the carbon-cycle. Glob. Change Biol. 1995, 1, 77–91. [Google Scholar] [CrossRef]
- Allen, M.R.; Frame, D.J.; Huntingford, C.; Jones, C.D.; Lowe, J.A.; Meinshausen, M.; Meinshausen, N. Warming caused by cumulative carbon emissions towards the trillionth tonne. Nature 2009, 458, 1163–1166. [Google Scholar] [CrossRef] [PubMed]
- Meinshausen, M.; Meinshausen, N.; Hare, W.; Raper, S.C.B.; Frieler, K.; Knutti, R.; Frame, D.J.; Allen, M.R. Greenhouse-gas emission targets for limiting global warming to 2 °C. Nature 2009, 458, 1158–1163. [Google Scholar] [CrossRef] [PubMed]
- Xiaoding, X.; Moulijn, J.A. Mitigation of CO2 by chemical conversion: Plausible chemical reactions and promising products. Energ. Fuel 1996, 10, 305–325. [Google Scholar] [CrossRef]
- Halmann, M. Photoelectrochemical reduction of aqueous carbon dioxide on p-type gallium phosphide in liquid junction solar calls. Nature 1978, 275, 115–116. [Google Scholar] [CrossRef]
- Inoue, T.; Fujishima, A.; Konishi, S.; Honda, K. Photoelectrocatalytic reduction of carbon dioxide in aqueous suspension of semiconductor powders. Nature 1979, 277, 637–638. [Google Scholar] [CrossRef]
- Canfield, D.; Frese, K.W. Reduction of carbon-dioxide to methanol on n-GaAs and p-GaAs and p-InP-effect of crystal face, electrolyte and current density. J. Electrochem. Soc. 1983, 130, 1772–1773. [Google Scholar] [CrossRef]
- Taniguchi, I.; Aurianblajeni, B.; Brockris, J.O.M. The reduction of carbon-dioxide at illuminated p-type semiconductor electrodes in nonaqueous media. Electrochim. Acta 1984, 29, 923–932. [Google Scholar] [CrossRef]
- Hinogami, R.; Mori, T.; Yae, S.J.; Nakato, Y. Effcient photoelectrochemical reduction of carbon-dioxide on p-type silicon (p-Si) electrode modified with very small copper particles. Chem. Lett. 1994, 9, 1725–1728. [Google Scholar] [CrossRef]
- Hinogami, R.; Nakamura, Y.; Yae, S.; Nakato, Y. An approach to ideal semiconductor electrodes for efficient photoelectrochemical reduction of carbon dioxide by modification with small metal particles. J. Phys. Chem. B 1998, 102, 974–980. [Google Scholar] [CrossRef]
- Raja, K.S.; Smith, Y.R.; Kondamudi, N.; Manivannan, A.; Misra, M.; Subramanian, V. CO2 photoreduction in the liquid phase over Pd-supported on TiO2 nanotube and bismuth titanate photocatalysts. Electroc. Sol. State Lett. 2011, 14, F5–F8. [Google Scholar] [CrossRef]
- Bradley, M.G.; Tysak, T. P-Type silicon based photo-electrochemical cells for optical-energy conversion-electrochemistry of tetraazomacrocyclic metal-complexes at illuminated p-type silicon semiconducting electrodes. J. Electroanal. Chem. 1982, 135, 153–157. [Google Scholar] [CrossRef]
- Bradley, M.G.; Tysak, T.; Graves, D.J.; Vlachopoulos, N.A. Electrocatalytic reduction of carbon-dioxide at illuminated para-type silicon semiconducting electrodes. J. Chem. Soc. Chem. Commun. 1983, 7, 349–350. [Google Scholar] [CrossRef]
- Liu, B.-J.; Torimoto, T.; Matsumoto, H.; Yoneyama, H. Effect of solvents on photocatalytic reduction of carbon dioxide using TiO2 nanocrystal photocatalyst embedded in SiO2 matrices. J. Photochem. Photobio. A 1998, 108, 187–192. [Google Scholar] [CrossRef]
- Koci, K.; Obalova, L.; Placha, D.; Lacny, Z. Effect of temperature, pressure and volume of reacting phase pn photocatalytic CO2 reduction on suspended nanocrystalline TiO2. Collect. Czech. Chem. Commun. 2008, 73, 1192–1204. [Google Scholar] [CrossRef]
- Yoneyama, H.; Sugimura, K.; Kuwabata, S. Effecrs of electrolytes on the photoelectrochemical reduction of carbon dioxide at illuminated p-type cadmium telluride and p-type indium phosphide electrodes in aqueous solutions. J. Electroanal. Chem. 1988, 249, 143–153. [Google Scholar] [CrossRef]
- Varghese, O.K.; Paulose, M.; LaTempa, T.J.; Grimes, C.A. High-rate solar photocatalytic conversion of CO2 and water vapor to hydrocarbon fuels. Nano Lett. 2009, 9, 731–737. [Google Scholar] [CrossRef] [PubMed]
- Schulte, K.L.; DeSario, P.A.; Gray, K.A. Effect of crystal phase composition on the reductive and oxidative abilities of TiO2 nanotubes under UV and visible light. Appl. Catal. B Environ. 2010, 97, 354–360. [Google Scholar] [CrossRef]
- Smith, Y.R.; Subramanian, V.; Viswanathan, B. Photo-electrochemical and Photo-catalytic Conversion of Carbon Dioxide. In Photo-Electrochemistry & Photo-Biology for the Sustainability; Kaneco, S., Viswanathan, B., Katsumata, H., Eds.; Union Press: Osaka, Japan, 2012; Volume 1, pp. 155–182. [Google Scholar]
- Indrakanti, V.P.; Kubicki, J.D.; Schobert, H.H. Quantum chemical modeling of ground states of CO2 chemisorbed on anatase (001), (101), and (010) TiO2 surfaces. Energy Fuel 2008, 22, 2611–2618. [Google Scholar] [CrossRef]
- Bai, J.; Liu, Y.; Li, J.; Zhou, B.; Zheng, Q.; Cai, W. A novel thin-layer photoelectrocatalytic (PEC) reactor with double-faced titania nanotube arrays electrode for effective degradation of tetracycline. Appl. Catal. B Environ. 2010, 98, 154–160. [Google Scholar] [CrossRef]
- Tian, M.; Wu, G.; Adams, B.; Wen, J.; Chen, A. Kinetics of photoelectrocatalytic degrdation of nitrophenols on nanostructured TiO2 electrodes. J. Phys. Chem C 2008, 112, 825–831. [Google Scholar] [CrossRef]
- Zhang, Z.; Yuan, Y.; Shi, G.; Fang, Y.; Liang, L.; Ding, H.; Jin, L. Photoelectrocatalytic activity of highly ordered TiO2 nanotube arrays electrode for azo dye degradation. Environ. Sci. Technol. 2007, 41, 6259–6263. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Liu, G.; Fan, J.; Zhou, Q.; Zhou, H.; Zhang, N.; Hou, Z.; Zhang, M.; He, Z. Photoelectrocatalytic degradation of 4,4'-dibromobiphenyl in aqueous solution on TiO2 and doped TiO2 nanotube arrays. Chemosphere 2011, 82, 43–47. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Quan, X.; Cui, Q.; Ma, M.; Chen, S.; Wang, Z. Ecotoxicaological characterization of photoelectrocatalytic process for degradation of pentachlorophenol on titania nanotubes electrode. Ecotoc. Environ. Safe. 2008, 71, 267–273. [Google Scholar] [CrossRef]
- Li, S.; Zhang, G.; Guo, D.; Yu, L.; Zhang, W. Andization fabrication of highly ordered TiO2 nanotubes. J. Phys. Chem. C 2009, 113, 12759–12765. [Google Scholar] [CrossRef]
- Yao, Y.; Li, K.; Chen, S.; Jia, J.; Wang, Y.; Wang, H. Decolorization of Rhodamine B in a thin-film photoelectrocatalytic (PEC) reactor with slant-placed TiO2 nanotubes electrode. Chem. Eng. J. 2012, 187, 29–35. [Google Scholar] [CrossRef]
- Li, L.; Zhou, Z.; Lei, J.; He, J.; Zhang, S.; Pan, F. HIghly ordered anodic TiO2 nanotube arrays and their stabilities as photo(electro)catalysts. Appl. Surf. Sci. 2012, 258, 3647–3651. [Google Scholar] [CrossRef]
- Baram, N.; Ein-Eli, Y. Electrochemical impedance spectroscopy of porous TiO2 for photocatalytic applications. J. Phys. Chem. C 2010, 114, 9781–9790. [Google Scholar] [CrossRef]
- Chai, S.; Zhao, G.; Li, P.; Lei, Y.; Zhang, Y.; Li, D. Novel sieve-like SnO2/TiO2 nanotubes with integrated photoelectrocatalysis: Fabrication and application for efficient toxicity elimination of nitrophenol wastewater. J. Phys. Chem. C 2011, 115, 18261–18269. [Google Scholar] [CrossRef]
- Yu, L.; Wang, Z.; Shi, L.; Yuan, S.; Zhao, Y.; Fang, J.; Deng, W. Photoelectrocatalytic performance of TiO2 nanoparticles incorporated TiO2 nanotube arrays. Appl. Catal. B Environ. 2012, 113–114, 318–325. [Google Scholar] [CrossRef]
- Fox, M.A.; Dulay, M.T. Heterogeneous photocatalysis. Chem. Rev. 1993, 93, 341–357. [Google Scholar] [CrossRef]
- Primo, A.; Corma, A.; García, H. Titania supported gold nanoparticles as photocatalyst. Phys. Chem. Chem. Phys. 2011, 13, 886–910. [Google Scholar] [CrossRef] [PubMed]
- Mohapatra, S.; Kondamudi, N.; Banerjee, S.; Misra, M. Functionalization of self-organized TiO2 nanotubes with Pd nanoparticles for photocatalytic decomposition of dyes under solar light illumination. Langmuir 2008, 24, 11276–11281. [Google Scholar] [CrossRef] [PubMed]
- Wender, H.; Feil, A.; Diaz, L.; Riberio, C.; Machado, G.; Migowski, P.; Weibel, D.; Dupont, J.; Teixeira, S. Self-organized TiO2 nanotube arrays: Synthesis by anodization in an ionic liquid and assessment of photocatalytic properties. ACS Appl. Mater. Interfaces 2011, 3, 1359–1365. [Google Scholar] [CrossRef] [PubMed]
- Xiao, F. Layer-by-layer self-assemly construction of highly ordered metal-TiO2 nanotube arrays heterostructures (M/TNTs, M = Au, Ag, Pt) with tunable catalytic activities. J. Phys. Chem. C 2012, 116, 16487–16498. [Google Scholar] [CrossRef]
- Yu, J.; Dai, G.; Cheng, B. Effect of crystallization methods on morphology and photocatalytic activity of anodized TiO2 nanotube array films. J. Phys. Chem. C 2010, 114, 19378–19385. [Google Scholar] [CrossRef]
- Yu, J.; Dai, G.; Huang, B. Fabrication and characterization of visible-ligh-driven plasmonic photocatalyst Ag/AgCl/TiO2 nanotube arrays. J. Phys. Chem. C 2009, 113, 16394–16401. [Google Scholar] [CrossRef]
- Yang, L.; Xiao, Y.; Zeng, G.; Luo, S.; Kuang, S.; Cai, Q. Fabrication and characterization of Pt/C-TiO2 nanotube arrays as anode materials for methanol electrocatalytic oxidation. Energy Fuel 2009, 23, 3134–3138. [Google Scholar] [CrossRef]
- Hou, Y.; Li, X.; Zou, X.; quan, X.; Chen, G. Photoelectrocatalytic activity of a Cu2O-loaded self-organized highly oriented TiO2 nanotube array electrode for 4-chlorophenol degradation. Environ. Sci. Technol 2009, 43, 858–863. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Luo, S.; Li, Y.; Xiao, Y.; Kang, Q.; Cai, Q. High efficient photocatalytic degradation of p-nitrophenol on a unique Cu2O/TiO2 p-n heterojunction network catalyst. Environ. Sci. Technol. 2010, 44, 7641–7646. [Google Scholar] [CrossRef] [PubMed]
- Gong, J.; Pu, W.; Yang, C.; Zhang, J. A simple electrochemical oxidation method to prepare highly ordered Cr-doped titania nanotube arrays with promoted photoelectrochemical property. Electrochim. Acta 2012, 68, 178–183. [Google Scholar] [CrossRef]
- He, H.; Xiao, P.; Zhou, M.; Zhang, Y.; Lou, Q.; Dong, X. Boosting catalytic activity with a p-n junction: Ni/TiO2 nanotube arrays composite catalyst for methanol oxidation. Int. J. Hydrog. Energy 2012, 37, 4967–4973. [Google Scholar] [CrossRef]
- Muramatsu, Y.; Jin, Q.; Fujishima, M.; Tada, H. Visible-light-activation of TiO2 nanotube array by the molecular iron oxide surface modification. Appl. Catal. B Environ. 2012, 119–120, 74–80. [Google Scholar] [CrossRef]
- Nie, J.; Mo, Y.; Zheng, B.; Yuan, H.; Xiao, D. Electrochemical fabrication of lanthanum-doped TiO2 nanotube array electrode and investigation of its photoelectrochemical capability. Electrochim. Acta 2013, 90, 589–596. [Google Scholar] [CrossRef]
- Yun, D.; Cho, H.; Jang, J.; Park, J. Nano zero-valent iron impregnated on titanium dioxide nanotube array film for both oxidation and reduction of methyl orange. Water Res. 2013, 47, 1858–1866. [Google Scholar] [CrossRef] [PubMed]
- Kamat, P. Quantum dot solar cells. Semiconductor nanocrystals at light harvesters. J. Phys. Chem. C 2008, 112, 18737–18753. [Google Scholar] [CrossRef]
- Subramanian, V.; Wolf, E.; Kamat, P. Semiconductor-metal composite nanostructures. To what extent do metal nanoparticles imrpove the photocatalytic activity of TiO2 films? J. Phys. Chem. B 2001, 105, 11439–11446. [Google Scholar] [CrossRef]
- Takai, A.; Kamat, P. Capture, store, and discharge. Shuttling photogenerated electrons across TiO2–silver interface. ACS Nano 2011, 5, 7369–7376. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.; Chen, W.; Kamat, P. Know thy nano neighbor. Plasmonic versus electron charging effects of metal nanoparticles in dye-sensitized solar cells. ACS Nano 2012, 6, 4418–4427. [Google Scholar] [CrossRef] [PubMed]
- Qi, J.; Dang, X.; Hammond, P.; Belcher, A. Highly efficient plasmon-enhanced dye-sensitized solar cells through metal@oxide core-shell nanostructure. ACS Nano 2011, 5, 7108–7116. [Google Scholar] [CrossRef] [PubMed]
- Peng, S.; McMahon, J.; Schatz, G.; Gray, S.; Sun, Y. Reversing the size-dependence of surface plasmon resonances. Proc. Natl. Acad. Sci. USA 2010, 107, 14530–14534. [Google Scholar] [CrossRef] [PubMed]
- Cong, Y.; Li, Z.; Zhang, Y.; Wang, Q.; Xu, Q. Synthesis of α-Fe2O3/TiO2 nanotube arrays for photoelectro-Fenton degradation of phenol. Chem. Eng. J. 2012, 191, 356–363. [Google Scholar] [CrossRef]
- Kontos, A.; Likodimos, V.; Stergiopoulos, T.; Tsoukleris, D.; Falaras, P. Self-organized anodic TiO2 nanotube arrays functionalized by iron oxide nanoparticles. Chem. Mater. 2009, 21, 662–672. [Google Scholar] [CrossRef]
- Asahi, R.; Morikawa, T.; Ohwaki, T.; Aoki, K.; Taga, Y. Visible-light photocatalysis in nitrogen-doped titanium oxides. Science 2001, 293, 269–271. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Ao, Y.; Chen, M.; Fu, D. Photoelectrochemical property and photocatalytic activity of N-doped TiO2 nanotube arrays. Appl. Surf. Sci. 2010, 256, 4397–4401. [Google Scholar] [CrossRef]
- Wu, H.; Zhang, Z. High photoelectrochemical water splitting performance on nitrogen doped double-wall TiO2 nanotube array electrodes. Int. J. Hydrog. Energy 2011, 36, 13481–13487. [Google Scholar] [CrossRef]
- Sang, L.-X.; Zhi, Z.; Guang, B.; Chun, D.; Chong, M. A photoelectrochemical investigation of the hydrogen-evolving doped TiO2 nanotube arrays electrode. Int. J. Hydrog. Energy 2012, 37, 854–859. [Google Scholar] [CrossRef]
- Murphy, A.B. Does carbondoping of TiO2 allow water splitting in visible light? Comments on “Nanotube enhanced photoresponse of carbon modified (CM)-n-TiO2 for efficient water splitting”. Sol. Energy Mater. Sol. C 2008, 92, 363–367. [Google Scholar] [CrossRef]
- Park, J.H.; Kim, S.; Bard, A.J. Novel carbon-doped TiO2 nanotube arrays with high aspect ratios for efficient solar water splitting. Nano Lett. 2006, 6, 24–28. [Google Scholar] [CrossRef] [PubMed]
- Raja, K.S.; Mahajan, V.K.; Misra, M. Determination of photo conversion efficiency of nanotubular titanium oxide photo-electrochemical cell for solar hydrogen generation. J. Power Sources 2006, 159, 1258–1265. [Google Scholar] [CrossRef]
- Raja, K.S.; Misra, M.; Mahajan, V.K.; Gandhi, T.; Pillai, P.; Mohapatra, S.K. Photo-electrochemical hydrogen generation using bandgap modified nanotubular titanium oxide in solar light. J. Power Sources 2006, 161, 1450–1457. [Google Scholar] [CrossRef]
- Mohapatra, S.K.; Misra, M.; Mahajan, V.K.; Raja, K.S. Design of a highly efficient photoelectrolytic cell for hydrogen generation by water splitting: Application of TiO2-xCx nanotubes as a photoanode and Pt/TiO2 nanotubes as a cathode. J. Phys. Chem. C 2007, 111, 8677–8685. [Google Scholar] [CrossRef]
- Tang, Y.; Luo, S.; Teng, Y.; Liu, C.; Xu, X.; Zhang, X. Efficient removal of herbicide 2,4-dichlorophenoxyacetic acid from water using Ag/reduced graphene oxide co-decorated TiO2 nanotube arrays. J. Hazard. Mater. 2012, 241–242, 323–330. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Luo, S.; Liu, S.; Cai, Q. Graphitized carbon nanotubes formed in TiO2 nanotube arrays: A novel functional material with tube-in-tube nanostructure. J. Phys. Chem. C 2008, 112, 8939–8943. [Google Scholar] [CrossRef]
- Kar, P.; Raja, K.S.; Misra, M.; Agasanapur, B.N. Formation and stability of anatase phase of phosphate incorporated and carbon doped titania nanotubes. Mater. Res. Bull. 2009, 44, 398–402. [Google Scholar] [CrossRef]
- Asapu, R.; Palla, V.M.; Wang, B.; Guo, Z.; Sadu, R.; Chen, D.H. Phosphorus-doped titania nanotubes with enhanced photocatalytic activity. J. Photochem. Photobiol. A 2011, 225, 81–87. [Google Scholar] [CrossRef]
- Takeshita, K.; Yamashita, A.; Ishibashi, T.; Onishi, H.; Nishijima, K.; Ohno, T. Transient IR absorption study of charge carriers photogenerated in sulfur-doped TiO2J. Photochem. Photobiol. A 2006, 177, 269–275. [Google Scholar] [CrossRef]
- Yu, Y.; Wu, H.H.; Zhu, B.L.; Wang, S.R.; Huang, W.P.; Wu, S.H.; Zhang, S.M. Preparation, characterization and photocatalytic activities of F-doped TiO2 Nanotubes. Cataly. Lett. 2007, 121, 165–171. [Google Scholar] [CrossRef]
- Lai, Y.K.; Huang, J.Y.; Zhang, H.F.; Subramaniam, V.P.; Tang, Y.X.; Gong, D.G.; Sundar, L.; Sun, L.; Chen, Z.; Lin, C.J. Nitrogen-doped TiO2 nanotube array films with enhanced photocatalytic activity under various light sources. J. Hazard. Mater. 2010, 184, 855–863. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Hanedaa, H.; Hishitaa, S.; Ohashia, N.; Labhsetwar, N.K. Fluorine-doped TiO2 powders prepared by spray pyrolysis and their improved photocatalytic activity for decomposition of gas-phase acetaldehyde. J. Fluor. Chem. 2005, 126, 69–77. [Google Scholar] [CrossRef]
- Su, Y.; Xiao, Y.; Fu, X.; Deng, Y.; Zhang, F. Photocatalytic properties and electronic structures of iodine-doped TiO2 nanotubes. Mater. Res. Bull. 2009, 44, 2169–2173. [Google Scholar] [CrossRef]
- Vogel, R.; Hoyer, P.; Weller, H. Quantum-sized PbS, CdS, Ag2S, Sb2S3, and Bi2S3 particles as sensitizers for various nanoporous wide-bandgap semiconductors. J. Phys. Chem. C 1994, 98, 3183–3188. [Google Scholar] [CrossRef]
- Tvrdy, K.; Frantsuzovc, P.A.; Kamat, P.V. Photoinduced electron transfer from semiconductor quantum dots to metal oxide nanoparticles. Proc. Natl. Acad. Sci. USA 2010, 108, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Hodes, G. Comparison of dye- and semiconductor-sensitized porous nanocrystalline liquid junction solar cells. J. Phys. Chem. C 2008, 112, 17778–17787. [Google Scholar] [CrossRef]
- Kongkanand, A.; Tvrdy, K.; Takechi, K.; Kuno, M.; Kamat, P.V. Quantum dot solar cells. Tuning photoresponse through sizeand shape control of CdSe-TiO2 architecture. J. Am. Chem. Soc. 2008, 130, 4007–4015. [Google Scholar] [CrossRef] [PubMed]
- Santra, P.K.; Kamat, P.V. Tandem-layered quantum dot solar cells: Tuning the photovoltaic response with luminescent ternary cadmium chalcogenides. J. Am. Chem. Soc. 2013, 135, 877–885. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.C.; Lee, Y.L.; Chang, C.H.; Shen, Y.J.; Yang, Y.M. Quantum-dot-sensitized solar cells: Assembly of CdS-quantum-dots coupling techniques of self-assembled monolayer and chemical bath deposition. Appl. Phys. Lett. 2007, 90, 143517:1–143517:3. [Google Scholar]
- Sun, W.T.; Yu, Y.; Pan, H.Y.; Gao, X.F.; Chen, Q.; Peng, L.M. CdS quantum dots sensitized TiO2 nanotube-array photoelectrodes, J. Am. Chem. Soc. 2008, 130, 1124–1125. [Google Scholar] [CrossRef] [PubMed]
- Plass, R.; Pelet, S.; Krueger, J.; Grätzel, M.; Bach, U. Quantum dot sensitization of organic inorganic hybrid solar cells. J. Phys. Chem. B 2002, 106, 7578–7580. [Google Scholar] [CrossRef]
- Peter, L.M.; Wijayantha, K.G.U.; Riley, D.J.; Waggett, J.P. Band-edge tuning in selfassembled layers of Bi2S3 nanoparticles used to photosensitize nanocrystalline TiO2. J. Phys. Chem. B 2003, 107, 8378–8381. [Google Scholar] [CrossRef]
- Shen, Q.; Sato, T.; Hashimoto, M.; Chen, C.; Toyoda, T. Photoacoustic and photoelectrochemical characterization of CdSe-sensitized TiO2 electrodes composed of nanotubes and nanowires. Thin Solid Films 2006, 499, 299–305. [Google Scholar] [CrossRef]
- Robel, I.; Subramanian, V.; Kuno, M.; Kamat, P.V. Quantum dot solar cells. Harvesting light energy with CdSe nanocrystals molecularly linked to mesoscopic TiO2 films. J. Am. Chem. Soc. 2006, 128, 2385–2393. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Yang, X.; Chi, L.; Cui, M. Photoelectrochemical performance of CdTe sensitized TiO2 nanotube array photoelectrodes. Electrochim. Acta 2013, 91, 330–336. [Google Scholar] [CrossRef]
- Feng, H.T.T.; Chen, L.; Yuan, L.; Cai, Q. Visible light-induced efficienctly oxidative decomposition of p-Nitrophenol by CdTe/TiO2 nanotube arrays. Chem. Eng. J. 2013, 215–216, 591–599. [Google Scholar] [CrossRef]
- Zaban, A.; Mićić, O.I.; Gregg, B.A.; Nozik, A.J. Photosensitization of nanoporous TiO2 electrodes with InP quantum dots. Langmuir 1998, 14, 3153–3156. [Google Scholar] [CrossRef]
- Banerjee, S.; Mohapatra, S.K.; Das, P.P.; Misra, M. Synthesis of coupled semiconductor by filling 1D TiO2 nanotubes with CdS. Chem. Mater. 2008, 20, 6784–6791. [Google Scholar] [CrossRef]
- Liu, Y.; Zhou, H.; Zhou, B.; Li, J.; Chen, H.; Wang, J.; Bai, J.; Shangguan, W.; Cai, W. Highly stable CdS-modified short TiO2 nanotube array electrode for efficient visible-light hydrogen generation. Int. J. Hydrog. Energy 2011, 36, 167–174. [Google Scholar] [CrossRef]
- Wilson, W.; Manivannan, A.; Subramanian, V. Heterogeneous photocatalytic degradation of recalcitrant pollutants over CdS-TiO2 nanotubes: Boosting effect of TiO2 nanoparticles at nanotube-CdS interface. Appl. Catal. A Gen. 2012, 441–442, 1–9. [Google Scholar] [CrossRef]
- Hossain, M.F.; Biswas, S.; Zhang, Z.H.; Takahashi, T. Bubble-like CdSe nanoclusters sensitized TiO2 nanotube arrays for improvement in solar cell. J. Photochem. Photobiol. A 2011, 217, 68–75. [Google Scholar] [CrossRef]
- Mukherjee, B.; Wilson, W.; Subramanian, V.R. TiO2 nanotube (T_NT) surface treatment revisited: Implications of ZnO, TiCl4, and H2O2 treatment on the photoelectrochemical properties of T_NT and T_NT–CdSe. Nanoscale 2013, 5, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, B.; Smith, Y.R.; Subramanian, V.R. CdSe Nanocrystal assemblies on anodized TiO2 nanotubes: Optical, surface, and photoelectrochemical properties. J. Phys. Chem. C 2012, 116, 15175–15184. [Google Scholar] [CrossRef]
- Gao, X.F.; Sun, W.T.; Ai, G.; Peng, L.M. Photoelectric performance of TiO2 nanotube array photoelectrodes cosensitized with CdS/CdSe quantum dots. Appl. Phys. Lett. 2010, 96, 153104:1–153104:3. [Google Scholar]
- Zhang, H.; Chen, G.; Bahnemann, D.W. Photoelectrocatalytic materials for environmental applications. J. Mater. Chem. 2009, 19, 5089–5121. [Google Scholar] [CrossRef]
- Sarma, B.; Smith, Y.R.; Mohanty, S.K.; Misra, M. Electrochemical deposition of CdO on anodized TiO2 nanotube arrays for enhanced photoelectrochemical properties. Mater. Lett. 2012, 85, 33–36. [Google Scholar] [CrossRef]
- Smith, Y.R.; Sarma, B.; Mohanty, S.K.; Misra, M. Formation of TiO2-WO3 nanotubular composite via single-step anodization and its application in photoelectrochemical hydrogen generation. Electrochem. Commun. 2012, 19, 131–134. [Google Scholar] [CrossRef]
- Lai, C.W.; Sreekantan, S. Incorporation of WO3 species into TiO2 nanotubes via wet impregnation and their water-splitting performance. Electrochim. Acta 2013, 87, 294–302. [Google Scholar] [CrossRef]
- Jeon, T.H.; Choi, W.; Park, H. Photoelectrochemical and photocatalytic behaviors of hematite-decorated titania nanotube arrays: Energy level mismatch versus surface specific reactivity. J. Phys. Chem C 2011, 115, 7134–7142. [Google Scholar] [CrossRef]
- Mohapatra, S.K.; Banerjee, S.; Misra, M. Synthesis of Fe2O3/TiO2 nanorod–nanotube arrays by filling TiO2 nanotubes with Fe. Nanotechnology 2008, 19. [Google Scholar] [CrossRef]
- Sun, L.; Li, J.; Wang, C.L.; Li, S.F.; Chen, H.B.; Lin, C.J. An electrochemical strategy of doping Fe3+ into TiO2 nanotube array films for enhancement in photocatalytic activity. Sol. Energy Mater. Sol. C 2009, 93, 1875–1880. [Google Scholar] [CrossRef]
- Shrestha, N.K.; Yang, M.; Nah, Y.C.; Paramasivam, I.; Schmuki, P. Self-organized TiO2 nanotubes: Visible light activation by Ni oxide nanoparticle decoration. Electrochem. Commun. 2010, 12, 254–257. [Google Scholar] [CrossRef]
- Hou, L.-R.; Yuan, C.-Z.; Peng, Y. Synthesis and photocatalytic property of SnO2/TiO2 nanotubes composites. J. Hazard. Mater. 2007, 139, 310–315. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Li, Q.; Zong, L.; Wang, X.; Yang, J. BaTiO3/TiO2 heterostructure nanotube arrays for improved photoelectrochemical and photocatalytic activity. Electrochim. Acta 2013, 91, 30–35. [Google Scholar] [CrossRef]
- Umebayashi, T.; Yamaki, T.; Itoh, H.; Asai, K. Analysis of electronic structures of 3D transition metal-doped TiO2 based on band calculations. J. Phys. Chem. Solids 2002, 63, 1909–1920. [Google Scholar] [CrossRef]
- Tuan Vu, A.; Tuan Nguyen, Q.; Linh Bui, T.H.; Cuong Tran, M.; Phuong Dang, T.; Hoa Tran, T.K. Synthesis and characterization of TiO2 photocatalyst doped by transition metal ions (Fe3+ , Cr3+ and V5+). Adv. Nat. Sci. Nanosci. Nanotechnol. 2010, 1. [Google Scholar] [CrossRef]
- Wu, Q.; Ouyang, J.; Xie, K.; Sun, L.; Wang, M.; Lin, C. Ultrasound-assisted synthesis and visible-light-driven photocatalytic activity of Fe-incorporated TiO2 nanotube array photocatalysts. J. Hazard. Mater. 2012, 199–200, 410–417. [Google Scholar] [CrossRef] [PubMed]
- Hussain, S.T.; Siddiqa, A. Iron and chromium doped titanium dioxide nanotubes for the degradation of environmental and industrial pollutants. Int. J. Environ. Sci. Technol. 2011, 8, 351–362. [Google Scholar] [CrossRef]
- Duncan, W.R.; Craig, C.F.; Prezhdo, O.V. Time-domain ab initio study of charge relaxation and recombination in dye-sensitized TiO2. J. Am. Chem. Soc. 2007, 129, 8528–8543. [Google Scholar] [CrossRef] [PubMed]
- Vittadini, A.; Casarin, M.; Selloni, A. Chemistry of and on TiO2-anatase surfaces by DFT calculations: A partial review. Theor. Chem. Acc. 2006, 117, 663–671. [Google Scholar] [CrossRef]
- Thomas, A.G.; Syres, K.L. Adsorption of organic molecules on rutile TiO2 and anatase TiO2 single crystal surfaces. Chem. Soc. Rev. 2012, 41, 4207–4217. [Google Scholar] [CrossRef] [PubMed]
- Duncan, W.R.; Prezhdo, O.V. Theoretical studies of photoinduced electron transfer in dye-sensitized TiO2. Annu. Rev. Phys. Chem. 2007, 58, 143–184. [Google Scholar] [CrossRef] [PubMed]
- Carp, O. Photoinduced reactivity of titanium dioxide. Prog. Solid State Chem. 2004, 32, 33–177. [Google Scholar] [CrossRef]
- Dovesi, R.; Civalleri, B.; Roetti, C.; Saunders, V.R.; Orlando, R. Ab Initio Quantum Simulation in Solid State Chemistry. In Reviews in Computational Chemistry; Lipkowitz, K.B., Larter, R., Cundari, T.R., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2005; Volume 21. [Google Scholar]
- Kresse, G.; Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 1996, 54, 11169–11186. [Google Scholar] [CrossRef]
- Segall, M.D.; Lindan, P.J.D.; Probert, M.J.; Pickard, C.J.; Hasnip, P.J.; Clark, S.J.; Payne, M.C. First-principles simulation: Ideas, illustrations and the CASTEP code. J. Phys. Condens. Matter 2002, 14, 2717–2744. [Google Scholar] [CrossRef]
- Gonze, X.; Amadon, B.; Anglade, P.M.; Beuken, J.M.; Bottin, F.; Boulanger, P.; Bruneval, F.; Caliste, D.; Caracas, R.; Cote, M.; et al. ABINIT: First-principles approach of materials and nanosystem properties. Comput. Phys. Commun. 2009, 180, 2582–2615. [Google Scholar] [CrossRef]
- Giannozzi, P.; Baroni, S.; Bonini, N.; Calandra, M.; Car, R.; Cavazzoni, C.; Ceresoli, D.; Chiarotti, G.L.; Cococcioni, M.; Dabo, I.; et al. QUANTUM ESPRESSO: A modular and open-source software project for quantum simulations of materials. J. Phys. Condens. Matter 2009, 21. [Google Scholar] [CrossRef]
- Dovesi, R.; Saunders, V.R.; Roetti, C.; Orlando, R.; Zicovich-Wilson, C.M.; Pascale, F.; Civalleri, B.; Doll, K.; Harrison, N.M.; Bush, I.J.; D’Arco, P.; Llunell, M. CRYSTAL09 User’s Manual; University of Torino: Torino, Italy, 2009. [Google Scholar]
- Soler, J.M.; Artacho, E.; Gale, J.D.; García, A.; Junquera, J.; Ordejón, P.; Sánchez-Portal, D. The SIESTA method for ab initio order-N materials simulation. J. Phys. Condens. Matter 2002, 14, 2745–2779. [Google Scholar] [CrossRef]
- Jacquemin, D.; Perpète, E.A.; Ciofini, I.; Adamo, C. Accurate simulation of optical properties in dyes. Acc. Chem. Res. 2009, 42, 326–334. [Google Scholar] [CrossRef] [PubMed]
- Becke, A.D. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 1993, 98, 5648–5644. [Google Scholar] [CrossRef]
- Adamo, C.; Barone, V. Toward reliable density functional methods without adjustable parameters: The PBE0 model. J. Chem. Phys. 1999, 110, 6158–6171. [Google Scholar] [CrossRef]
- Marsman, M.; Paier, J.; Stroppa, A.; Kresse, G. Hybrid functionals applied to extended systems. J. Phys. Condens. Matter 2008, 20. [Google Scholar] [CrossRef]
- Yanai, T.; Tew, D.P.; Handy, N.C. A new hybrid exchange–correlation functional using the Coulomb-attenuating method (CAM-B3LYP). Chem. Phys. Lett. 2004, 393, 51–57. [Google Scholar] [CrossRef]
- Shirley, E.L.; Martin, R.M. GW quasiparticle calculations in atoms. Phys. Rev. B 1993, 47, 15404–15412. [Google Scholar] [CrossRef]
- Tomasi, J.; Mennucci, B.; Cammi, R. Quantum mechanical continuum solvation models. Chem. Rev. 2005, 105, 2999–3093. [Google Scholar] [CrossRef] [PubMed]
- Dapprich, S.; Komaromi, I.; Byun, K.S.; Morokuma, K.; Frisch, M.J. A New ONIOM implementation in Gaussian 98. Part 1. The calculation of energies, gradients and vibrational frequencies and electric field derivatives. J. Mol. Struct. Theochem. 1999, 1, 461–462. [Google Scholar]
- Runge, E.G.; Gross, E.K.U. Density-functional theory for time-dependent systems. Phys. Rev. Lett. 1984, 52, 997–1000. [Google Scholar] [CrossRef]
- Wang, L.; May, V. Laser pulse control of ultrafast heterogeneous electron transfer: A computational study. J. Chem. Phys. 2004, 121, 8039–8049. [Google Scholar] [CrossRef] [PubMed]
- Ramakrishna, S.; Willig, F.; May, V.; Knorr, A. Femtosecond spectroscopy of heterogeneous electron transfer: Extraction of excited-state population dynamics from pump-probe signals. J. Phys. Chem. B 2003, 107, 607–611. [Google Scholar] [CrossRef]
- Rego, L.G.C.; Batista, V.S. Quantum dynamics simulations of interfacial electron transfer in sensitized TiO2 semiconductors. J. Am. Chem. Soc. 2003, 125, 7989–7997. [Google Scholar] [CrossRef] [PubMed]
- Stier, W.; Prezhdo, O.V. Nonadiabatic molecular dynamics simulation of light-induced electron transfer from an anchored molecular electron donor to a semiconductor acceptor. J. Phys. Chem. B 2002, 106, 8047–8054. [Google Scholar] [CrossRef]
- Duncan, W.R.; Stier, W.M.; Prezhdo, O.V. Ab initio nonadiabatic molecular dynamics of the ultrafast electron injection across the alizarin-TiO2 interface. J. Am. Chem. Soc. 2005, 127, 7941–7951. [Google Scholar] [CrossRef] [PubMed]
- Prezhdo, O.V.; Duncan, W.R.; Prezhdo, V.V. Dynamics of the photoexcited electron at the chromophore-semiconductor interface. Acc. Chem. Res. 2008, 41, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Gong, X.Q.; Selloni, A.; Batzill, M.; Diebold, U. Steps on anatase TiO2 (101). Nat. Mater. 2006, 5, 665–670. [Google Scholar] [CrossRef] [PubMed]
- Li, S.-C.; Losovyj, Y.; Diebold, U. Adsorption-site-dependent electronic structure of catechol on the anatase TiO2 (101) surface. Langmuir 2011, 27, 8600–8604. [Google Scholar] [CrossRef] [PubMed]
- Ambrosio, F.; Martsinovich, N.; Troisi, A. Effect of the anchoring group on electron injection: Theoretical study of phosphonated dyes for dye-sensitized solar cells. J. Phys. Chem. C 2012, 116, 2622–2629. [Google Scholar] [CrossRef]
- Scaranto, J.; Giorgianni, S. A DFT study of CO adsorbed on the clean and hydroxylated anatase TiO2 (001) surfaces. J. Mol. Str. Theochem. 2008, 858, 72–76. [Google Scholar] [CrossRef]
- Gong, X.; Selloni, A. Role of steps in the reactivity of the anatase TiO2 (101) surface. J. Catal. 2007, 249, 134–139. [Google Scholar] [CrossRef]
- Labat, F.; Baranek, P.; Domain, C.; Minot, C.; Adamo, C. Density functional theory analysis of the structural and electronic properties of TiO2 rutile and anatase polytypes: Performances of different exchange-correlation functionals. J. Chem. Phys. 2007, 126, 154703:1–154703:12. [Google Scholar] [CrossRef]
- Asahi, R.T.Y.; Mannstadt, W.; Freeman, A.J. Electronic and optical properties of anatase TiO2. Phys. Rev. B 2000, 61, 7459–7465. [Google Scholar] [CrossRef]
- Martsinovich, N.; Jones, D.R.; Troisi, A. Electronic structure of TiO2 surfaces and effect of molecular adsorbates using different DFT implementations. J. Phys. Chem. C 2010, 114, 22659–22670. [Google Scholar] [CrossRef]
- Hagfeldt, A.; Boschloo, G.; Sun, L.; Kloo, L.; Pettersson, H. Dye-sensitized solar cells. Chem. Rev. 2010, 110, 6595–6663. [Google Scholar] [CrossRef] [PubMed]
- Sousa, C.; Tosoni, S.; Illas, F. Theoretical approaches to excited-state-related phenomena in oxide surfaces. Chem. Rev. 2012, 113, 4465–4495. [Google Scholar]
- Tada, H.; Jin, Q.; Kobayashi, H. Prediction of the main route in the TiO2-photocatalyzed degradation of organic compounds in water by density functional calculations. Chem. Phys. Chem. 2012, 13, 3457–3461. [Google Scholar] [PubMed]
- Henderson, M.A. An HREELS and TPD study of water on TiO2 (110): The extent of molecular versus dissociative adsorption. Surf. Sci. 1996, 355, 151–166. [Google Scholar] [CrossRef]
- Bandura, A.V.; Sykes, D.G.; Shapovalov, V.; Troung, T.N.; Kubicki, J.D. Adsorption of water on the TiO2 (Rutile) (110) surface: A comparison of periodic and embedded cluster calculations. J. Phys. Chem. A 2004, 2, 7844–7853. [Google Scholar] [CrossRef]
- Harris, L.; Quong, A. Molecular chemisorption as the theoretically preferred pathway for water adsorption on ideal rutile TiO2 (110). Phys. Rev. Lett. 2004, 93, 086105:1–086015:8. [Google Scholar] [CrossRef]
- Shapovalov, V.; Stefanovich, E.V.; Truong, T.N. Nature of the excited states of the rutile TiO2 (110) surface with adsorbed water. Surf. Sci. 2002, 498, 1–6. [Google Scholar] [CrossRef]
- Jedidi, A.; Markovits, A.; Minot, C.; Bouzriba, S.; Abderraba, M. Modeling localized photoinduced electrons in rutile-TiO2 using periodic DFT + U methodology. Langmuir 2010, 26, 16232–16238. [Google Scholar] [CrossRef] [PubMed]
- Lun Pang, C.; Lindsay, R.; Thornton, G. Chemical reactions on rutile TiO2 (110). Chem. Soc. Rev. 2008, 37, 2328–2353. [Google Scholar] [CrossRef] [PubMed]
- Onishi, H.; Aruga, T.; Iwasawa, Y. Catalytic reactions on a metal oxide single crystal: Switchover of the reaction paths in formic acid decomposition on titanium dioxide TiO2 (110). J. Am. Chem. Soc. 1993, 115, 10460–10461. [Google Scholar] [CrossRef]
- Ahdjoudj, J.; Minot, C. A theoretical study of HCO2H adsorption on TiO2 (110). Catal. Lett. 1997, 46, 83–91. [Google Scholar] [CrossRef]
- Vittadini, A.; Selloni, A.; Rotzinger, F.P.; Gra, M. Formic acid adsorption on dry and hydrated TiO2 anatase (101) surfaces by DFT calculations. J. Phys. Chem. B 2000, 104, 1300–1306. [Google Scholar] [CrossRef]
- Li, S.; Lu, X.; Guo, W.; Zhu, H.; Li, M.; Zhao, L.; Li, Y.; Shan, H. Formaldehyde oxidation on the Pt/TiO2 (101) surface: A DFT investigation. J. Organomet. Chem. 2012, 704, 38–48. [Google Scholar] [CrossRef]
- Yamazaki-Nishida, S.; Cervera-March, S.; Nagano, K.J.; Anderson, M.A.; Hori, K. An experimental and theoretical study of the reaction mechanism of the photoassisted catalytic degradation of trichloroethylene in the gas phase. J. Phys. Chem. 1995, 99, 15814–15821. [Google Scholar] [CrossRef]
- Labat, F.; Bahers, T.; Ciofini, I.; Adamo, C. First-principles modeling of dye-sensitized solar cells: Challenges and perspectives. Acc. Chem. Res. 2012, 45, 1268–1277. [Google Scholar] [CrossRef] [PubMed]
- Asbury, J.B.; Hao, E.; Wang, Y.; Ghosh, H.N.; Lian, T. Ultrafast electron transfer dynamics from molecular adsorbates to semiconductor. J. Phys. Chem. B 2001, 105, 4545–4557. [Google Scholar] [CrossRef]
- Ai, X.; Anderson, N.A.; Guo, J.; Lian, T. Electron injection dynamics of Ru polypyridyl complexes on SnO2 nanocrystalline thin films. J. Phys. Chem. B 2005, 109, 7088–7094. [Google Scholar] [CrossRef] [PubMed]
- Odelius, M.; Persson, P.; Lunell, S. Bi-isonicotinic acid on rutile (110): Calculated molecular and electronic structure. Surf. Sci. 2003, 529, 47–58. [Google Scholar] [CrossRef]
- Zabri, H.; Gillaizeau, I.; Bignozzi, C.A.; Caramori, S.; Charlot, M.-F.; Cano-Boquera, J.; Odobel, F. Synthesis and comprehensive characterizations of new cis-RuL(2)X(2) (X = Cl, CN, and NCS) sensitizers for nanocrystalline TiO2 solar cell using Bis-phosphonated bipyridine ligands (L). Inorg. Chem. 2003, 42, 6655–6666. [Google Scholar] [CrossRef] [PubMed]
- Patthey, L.; Rensmo, H.; Persson, P.; Westermark, K.; Vayssieres, L.; Stashans, A.; Petersson, A.; Brühwiler, P.A.; Siegbahn, H.; Lunell, S.; Mårtensson, N. Adsorption of bi-isonicotinic acid on rutile TiO2 (110). J. Chem. Phys. 1999, 110, 5913:1–5913:6. [Google Scholar] [CrossRef]
- Kondov, I.; Thoss, M.; Wang, H. Computational study of titanium (IV) complexes with organic chromophores. Int. J. Quantum Chem. 2006, 106, 1291–1303. [Google Scholar] [CrossRef]
- Wang, H.; Thoss, M. Multilayer formulation of the multiconfiguration time-dependent Hartree theory. J. Chem. Phys. 2003, 119, 1289–l300. [Google Scholar] [CrossRef]
- Rensmo, H.; Södergren, S.; Patthey, L.; Westermark, K.; Vayssieres, L.; Kohle, O.; Brühwiler, P.A.; Hagfeldt, A.; Siegbahn, H. The electronic structure of the cis-bis(4,4-dicarboxy-2,2-bipyridine)-bis(isothiocyanato ruthenium(II) complex and its ligand 2,2-bipyridyl-4,4-dicarboxylic acid studied with electron spectroscopy. Chem. Phys. Lett. 1997, 274, 51–57. [Google Scholar] [CrossRef]
- Duncan, W.R.; Prezhdo, O.V. Electronic structure and spectra of catechol and alizarin in the gas phase and attached to titanium. J. Phys. Chem. B 2005, 109, 365–373. [Google Scholar] [CrossRef] [PubMed]
- Long, R.; English, N.J.; Prezhdo, O.V. Photo-induced charge separation across the graphene-TiO2 interface is faster than energy losses: A time-domain ab initio analysis. J. Am. Chem. Soc. 2012, 134, 14238–14248. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Liang, W. Real-time propagation of the reduced one-electron density matrix in atom-centered orbitals: Application to electron injection dynamics in dye-sensitized TiO2 clusters. J. Phys. Chem. C 2008, 112, 16655–16662. [Google Scholar] [CrossRef]
- Kondov, I.; Čížek, M.; Benesch, C.; Wang, H.; Thoss, M. Quantum dynamics of photoinduced electron-transfer reactions in dye-semiconductor systems: First-principles description and application to coumarin 343-TiO2. J. Phys. Chem. C 2007, 111, 11970–1191. [Google Scholar] [CrossRef]
- Wang, L.; Ernstorfer, R.; Willig, F.; May, V. Absorption spectra related to heterogeneous electron transfer reactions: The perylene TiO2 system. J. Phys. Chem. B 2005, 109, 9589–9595. [Google Scholar] [CrossRef] [PubMed]
- Nelson, J. Continuous-time random-walk model of electron transport in nanocrystalline TiO2 electrodes. Phys. Rev. B 1999, 59, 374–380. [Google Scholar] [CrossRef]
- Nelson, J.; Chandler, R.E. Random walk models of charge transfer and transport in dye sensitized systems. Coord. Chem. Rev. 2004, 248, 1181–1194. [Google Scholar] [CrossRef]
- Walker, A.B.; Peter, L.M.; Cass, M.J.; Cameron, P.J.; Martínez, D. Multi-timescale monte carlo method for simulating electron transport in dye-sensitized nanocrystalline solar cells. J. Mater. Chem. 2005, 15, 2253–2256. [Google Scholar] [CrossRef]
- Li, J.; Kondov, I.; Wang, H.; Thoss, M. Theoretical study of photoinduced electron-transfer processes in the dye−semiconductor system alizarin−TiO2. J. Phys. Chem. C 2010, 114, 18481–18493. [Google Scholar] [CrossRef]
© 2013 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Smith, Y.R.; Ray, R.S.; Carlson, K.; Sarma, B.; Misra, M. Self-Ordered Titanium Dioxide Nanotube Arrays: Anodic Synthesis and Their Photo/Electro-Catalytic Applications. Materials 2013, 6, 2892-2957. https://doi.org/10.3390/ma6072892
Smith YR, Ray RS, Carlson K, Sarma B, Misra M. Self-Ordered Titanium Dioxide Nanotube Arrays: Anodic Synthesis and Their Photo/Electro-Catalytic Applications. Materials. 2013; 6(7):2892-2957. https://doi.org/10.3390/ma6072892
Chicago/Turabian StyleSmith, York R., Rupashree S. Ray, Krista Carlson, Biplab Sarma, and Mano Misra. 2013. "Self-Ordered Titanium Dioxide Nanotube Arrays: Anodic Synthesis and Their Photo/Electro-Catalytic Applications" Materials 6, no. 7: 2892-2957. https://doi.org/10.3390/ma6072892




