Degradation of 2,4-Dichlorophenoxyacetic Acid (2,4-D) by Novel Photocatalytic Material of Tourmaline-Coated TiO2 Nanoparticles: Kinetic Study and Model
Abstract
:1. Introduction
2. Results and Discussion
2.1. Characterization of Tourmaline and Photocatalyst
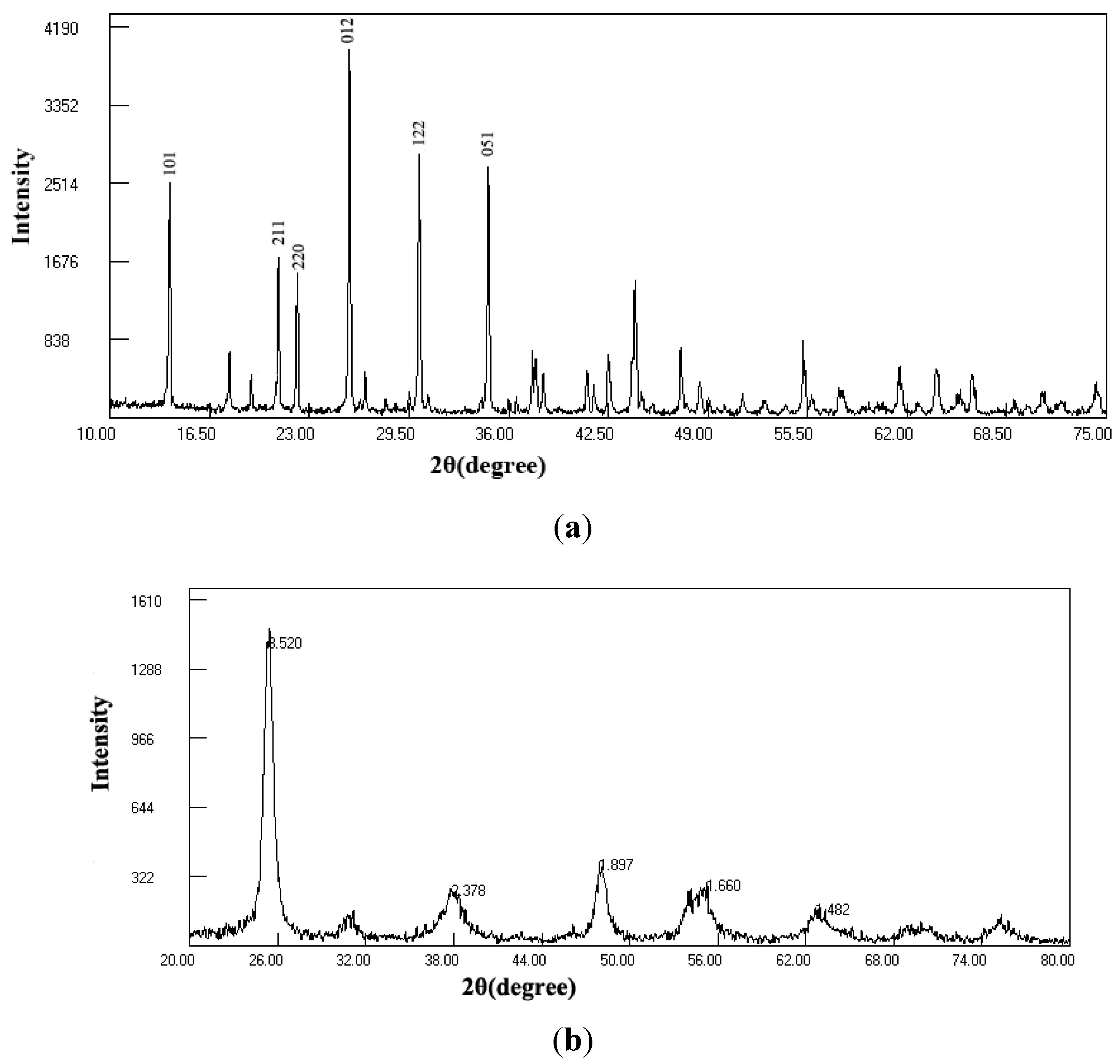
2.2. Photocatalytic Degradation

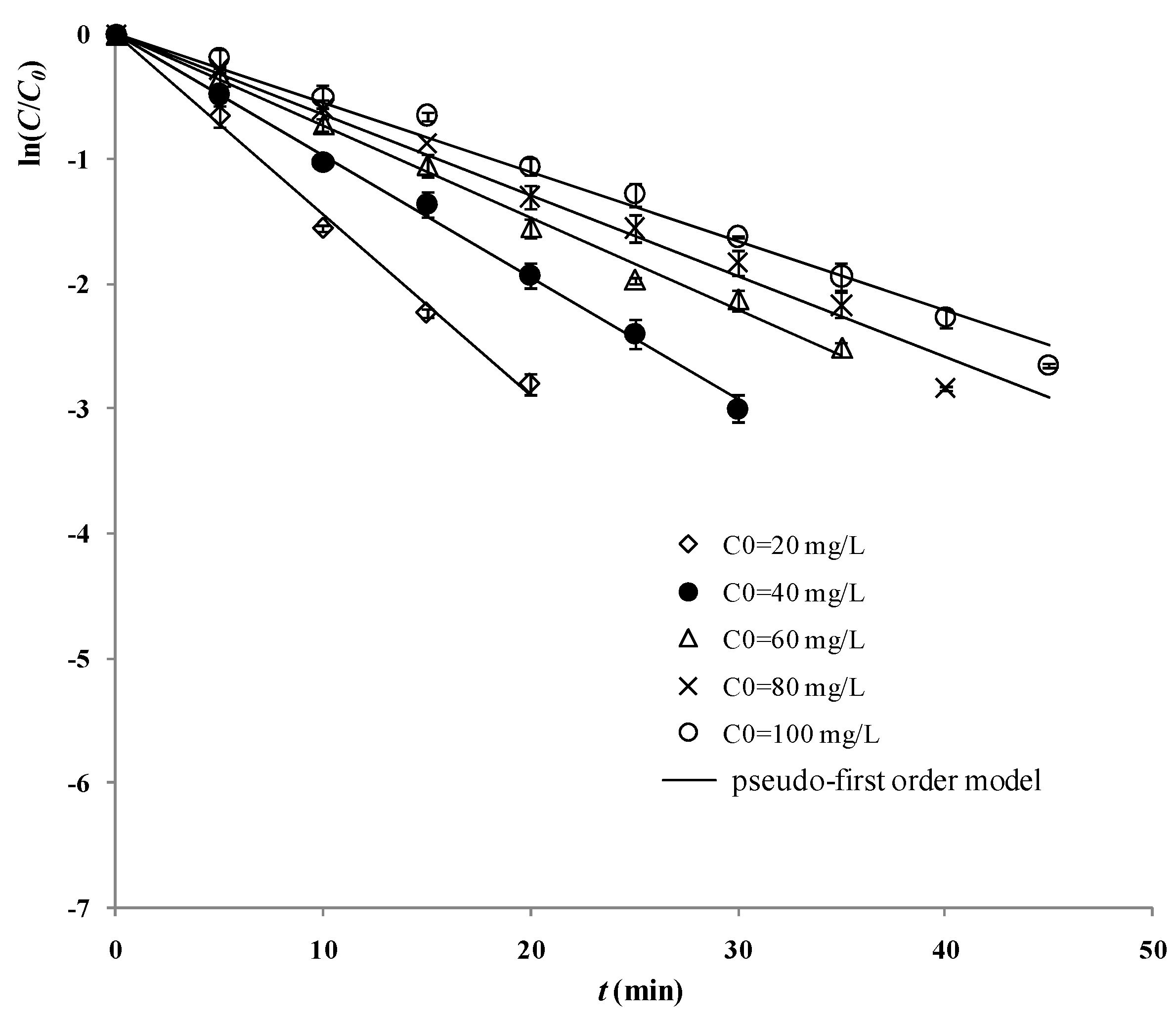
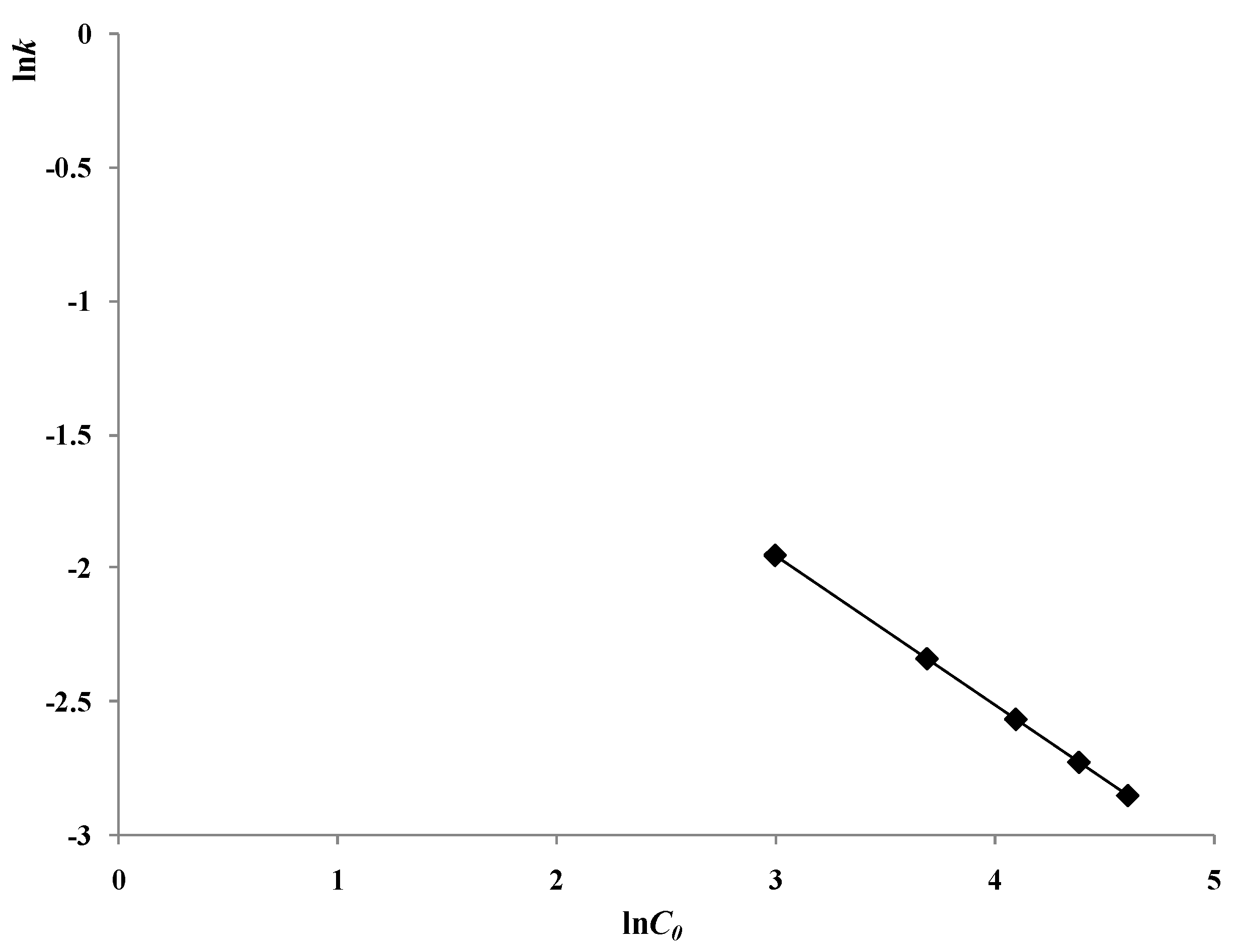
2.3. Influence of the Initial Concentration of 2,4-D
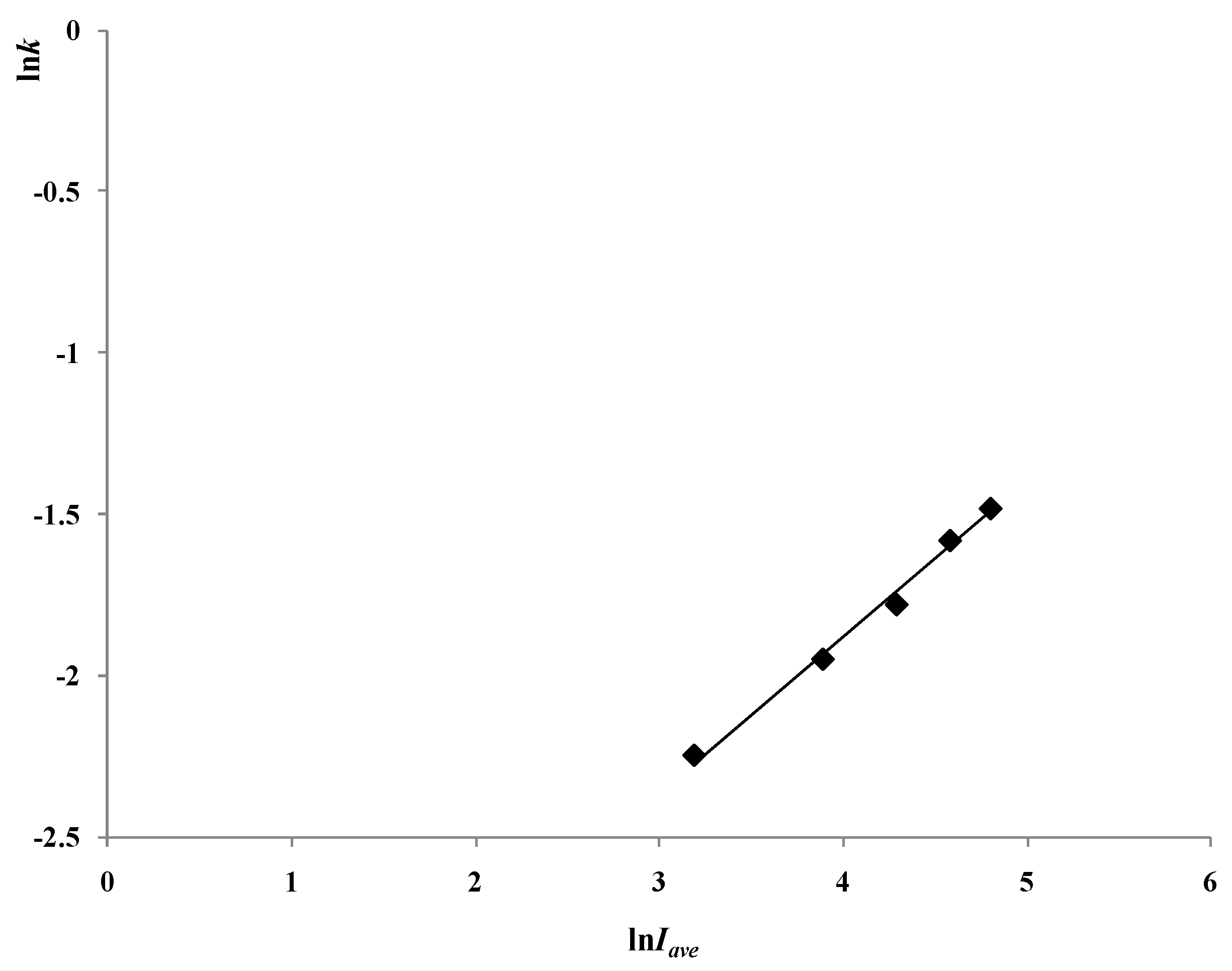
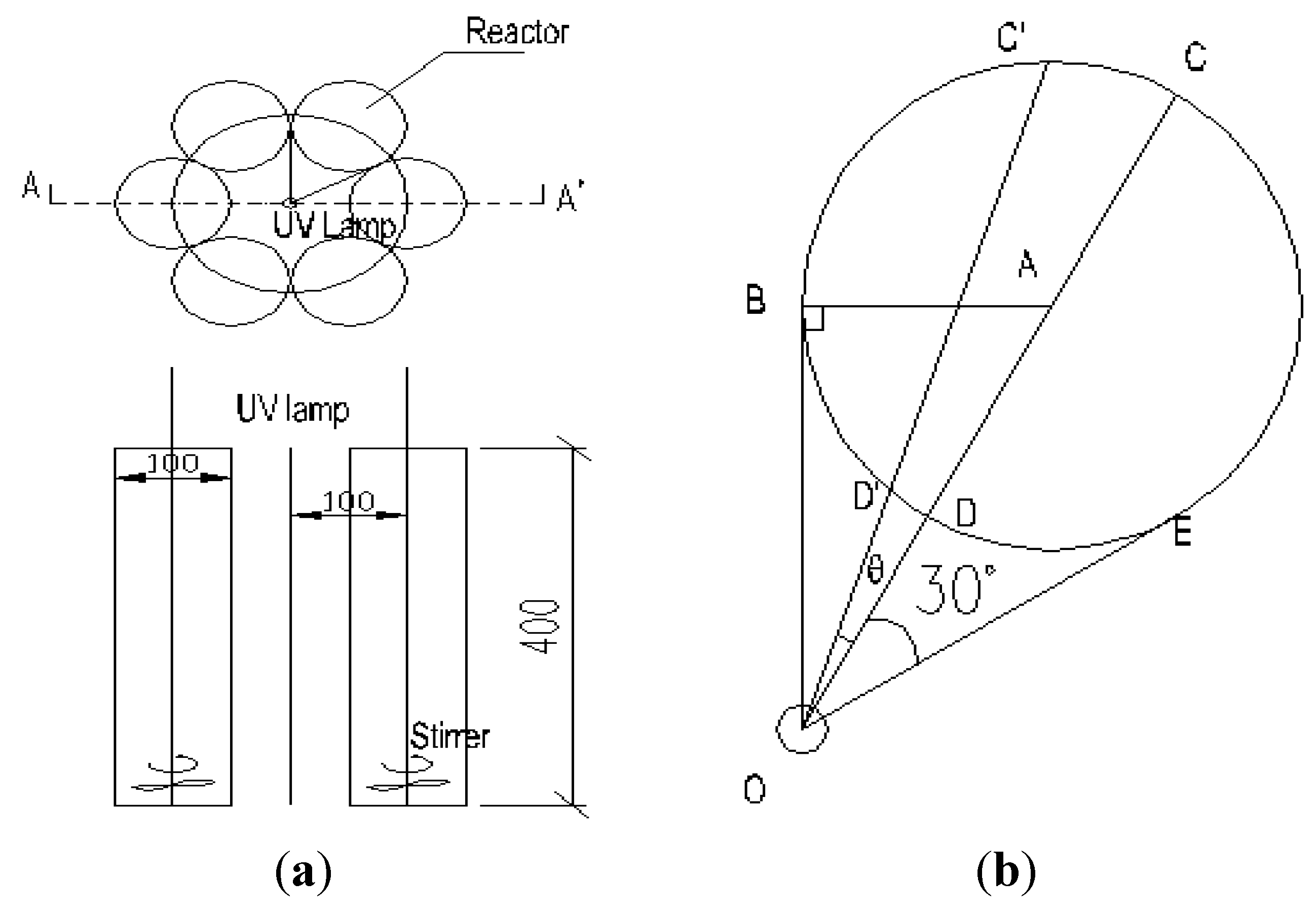
2.4. The Light Intensity Distribution Model and the Influence of UV Light Intensity
2.5. The Influence of the Concentration of Photocatalyst


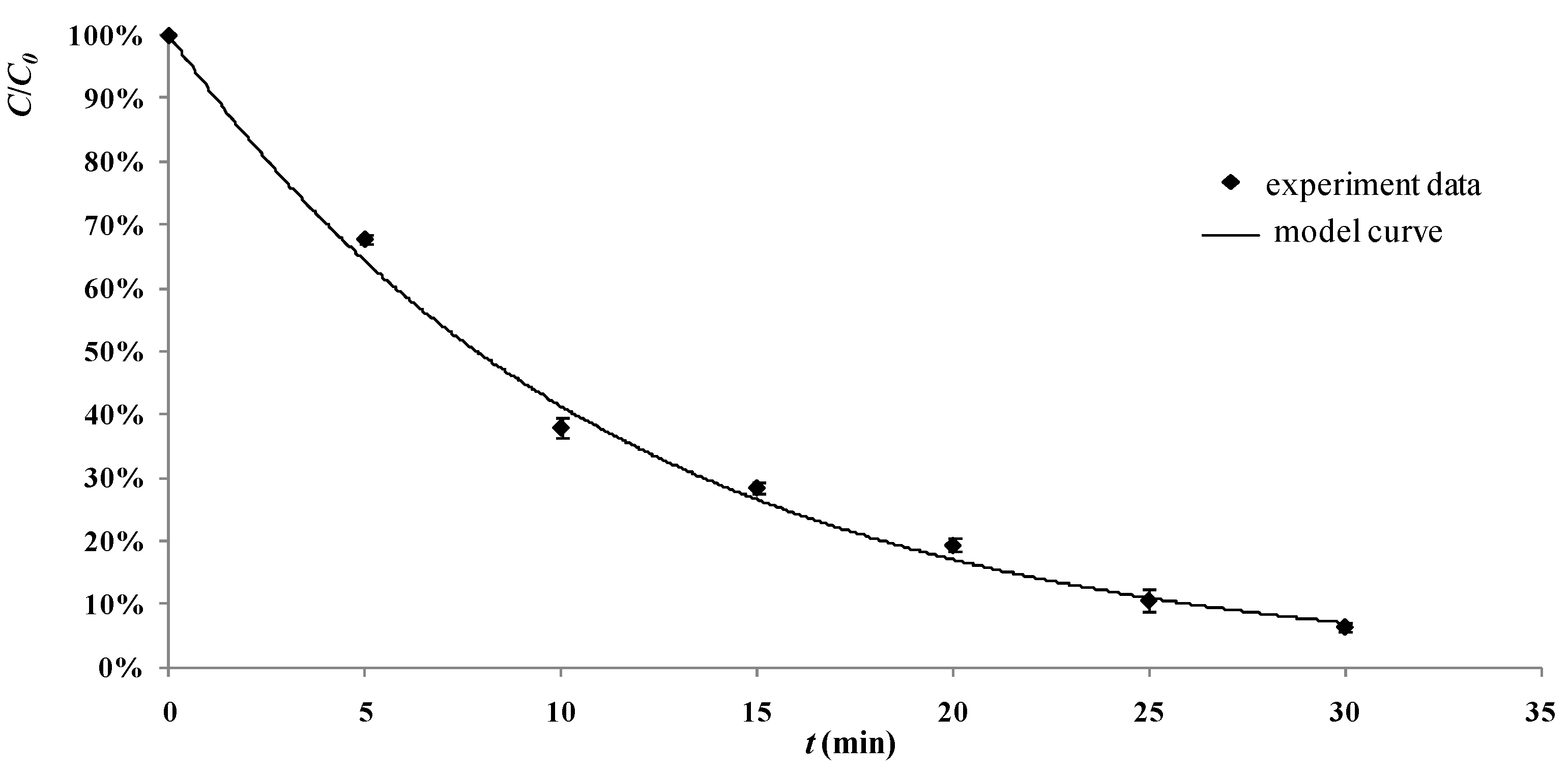
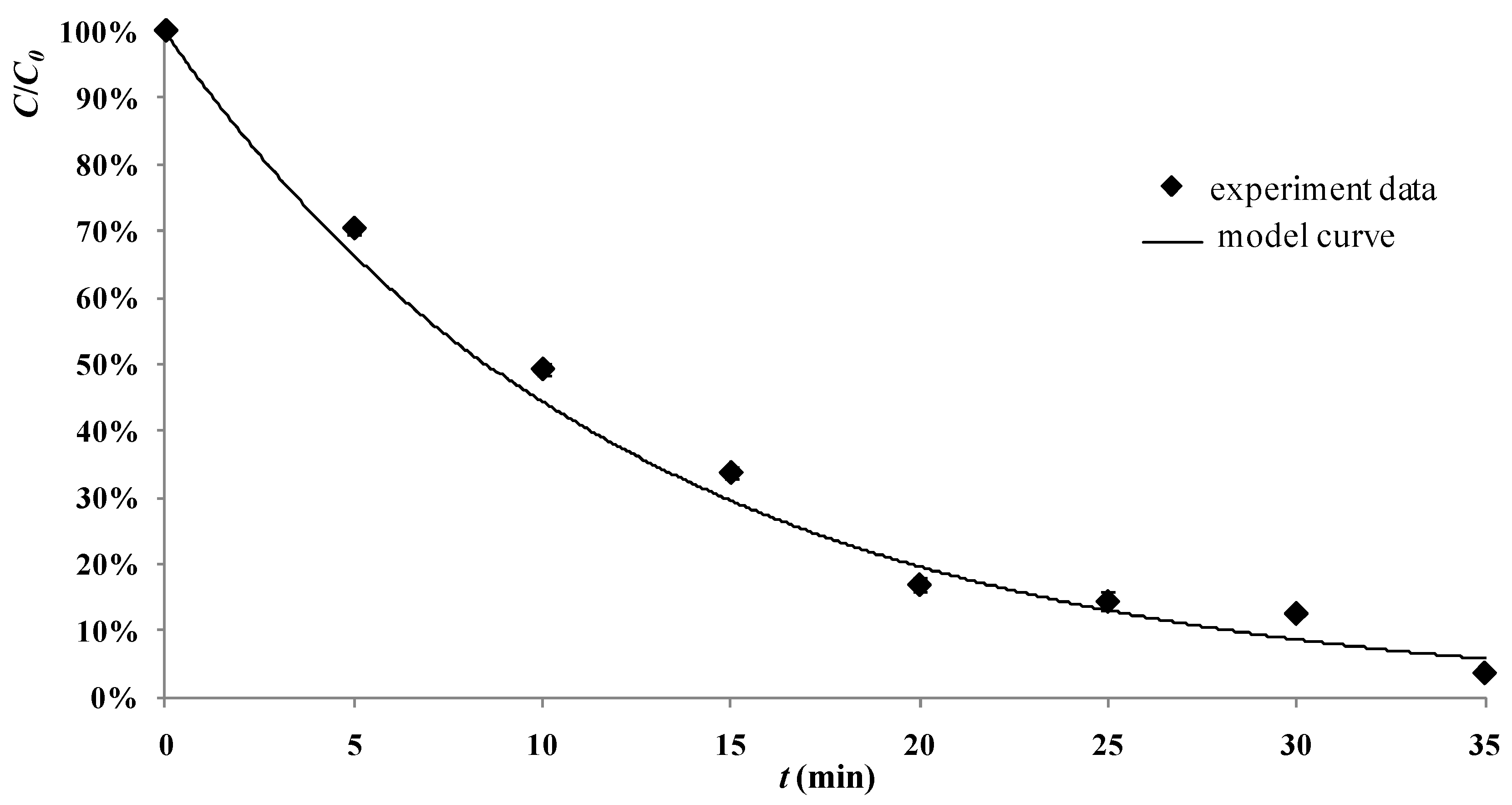
2.6. Development of the Model and Tests in the Real Process
3. Experimental Section
3.1. Materials and Instruments
3.2. Pretreatment of Tourmaline Powder
3.3. Synthesis of Tourmaline-Coated TiO2
3.4. Photocatalytic Degradation of 2,4-D by Tourmaline-Coated TiO2
3.5. Analysis and Characterization of Photocatalyst
4. Conclusions
References
- Jo, W.-K. Photocatalytic oxidation of low-level airborne 2-propanol and trichloroethylene over titania irradiated with bulb-type light-emitting diodes. Materials 2013, 6, 265–278. [Google Scholar] [CrossRef]
- Nakata, K.; Fujishima, A. TiO2 photocatalysis: design and applications. J. Photochem. Photobiol. C 2012, 13, 169–189. [Google Scholar] [CrossRef]
- Kabra, K.; Chaudhary, R.; Sawhney, R.L. Treatment of hazardous organic and inorganic compounds through aqueous-phase photocatalysis: A review. Ind. Eng. Chem. Res. 2004, 43, 7683–7696. [Google Scholar] [CrossRef]
- Colmenares, J.C.; Luque, R.; Campelo, J.M.; Colmenares, F.; Karpiński, Z.; Romero, A.A. Nanostructured photocatalysts and their applications in the photocatalytic transformation of lignocellulosic biomass: An overview. Materials 2009, 2, 2228–2258. [Google Scholar] [CrossRef]
- Zhang, A.; Zhou, M.; Han, L.; Zhou, Q. The combination of rotating disk photocatalytic reactor and TiO2 nanotube arrays for environmental pollutants removal. J. Hazard. Mater. 2011, 186, 1374–1383. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Prasad, M.; Liu, Y. Schorl: A novel catalyst in mineral-catalyzed Fenton-like system for dyeing wastewater discoloration. J. Hazard. Mater. 2009, 165, 1186–1192. [Google Scholar] [CrossRef] [PubMed]
- Tokumura, M.; Znad, H.T.; Kawase, Y. Modeling of an external light irradiation slurry photoreactor: UV light or sunlight-photoassisted Fenton discoloration of azo-dye Orange II with natural mineral tourmaline powder. Chem. Eng. Sci. 2006, 61, 6361–6371. [Google Scholar] [CrossRef]
- Muñoz, M.; Gullett, B.K.; Touati, A.; Font, R. Effect of 2,4-dichlorophenoxyacetic acid (2,4-D) on PCDD/F emissions from open burning of biomass. Environ. Sci. Technol. 2012, 46, 9308–9314. [Google Scholar] [CrossRef] [PubMed]
- Gehring, C.A.; Irving, H.R.; Parish, R.W. Effects of auxin and abscisic acid on cytosolic calcium and pH in plant cells. Proc. Natl. Acad. Sci. USA 1990, 87, 9645–9649. [Google Scholar] [CrossRef] [PubMed]
- Wilson, R.D.; Geronimo, J.; Armbruster, J.A. 2,4-D dissipation in field soils after applications of 2,4-D dimethylamine salt and 2,4-D 2-ethylhexyl ester. Environ. Toxicol. Chem. 1997, 16, 1239–1246. [Google Scholar] [CrossRef]
- Laganà, A.; Bacaloni, A.; Leva, I.D.; Faberi, A.; Fago, G.; Marino, A. Occurrence and determination of herbicides and their major transformation products in environmental waters. Anal. Chim. Acta 2002, 462, 187–198. [Google Scholar] [CrossRef]
- Wang, Y.; Wu, C.; Wang, X.; Zhou, S. The role of humic substances in the anaerobic reductive dechlorination of 2,4-dichlorophenoxyacetic acid by Comamonas. koreensis strain CY01. J. Hazard. Mater. 2009, 164, 941–947. [Google Scholar] [CrossRef]
- Nishioka, M.G.; Burlholder, H.M.; Brinkman, M.C.; Battelle, C.H. Trasnport of Lawn-Applied 2,4-D from Turf to Home: Assessing the Relative Importance of Transport Mechanisms and Exposure Pathways; National Exposure Research Laboratory, U.S. Environmental Protection Agency: Washington, DC, USA, 1999. [Google Scholar]
- Ang, C.; Meleady, K.; Wallace, L. Pesticide residues in drinking water in the North coast region of New South Wales, Australia, 1986–1987. Arch. Environ. Contam. Toxicol. 1989, 42, 595–602. [Google Scholar] [CrossRef]
- Fonseca, M.B.; Glusczak, L.; Moraes, B.S.; Menezes, C.C.; Pretto, A.; Tierno, M.A.; Zanella, R.; Gonçalves, F.F.; Loro, V.L. The 2,4-D herbicide effects on acetylcholinesterase activity and metabolic parameters of piava freshwater fish (Leporinus. obtusidens). Ecotoxicol. Environ. Saf. 2008, 69, 416–420. [Google Scholar] [CrossRef] [PubMed]
- Kaioumova, D.; Süsal, C.; Opelz, G. Induction of apoptosis in human lymphocytes by the herbicide 2,4-dichlorophenoxyacetic acid. Hum. Immunol. 2001, 62, 64–74. [Google Scholar] [CrossRef] [PubMed]
- Pochettino, A.A.; Bongiovanni, B.; Duffard, R.O.; Duffard, A.M.E. Oxidative stress in ventral prostate, ovary, and breast by 2,4-dichlorophenoxyacetic acid in pre- and postnatal exposed rats. Environ. Toxicol. 2013, 28, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Aly, O.M.; Faust, S.D. Herbicides in surface waters, studies on fate of 2,4-D and ester derivatives in natural surface waters. J. Agric. Food Chem. 1964, 12, 541–546. [Google Scholar] [CrossRef]
- Erne, K. Detection and determination of chlorophenoxyacetic acid derivatives in water. Acta Chem. Scand. 1963, 17, 1663–1676. [Google Scholar] [CrossRef]
- Robson, T.O. Some Studies of the Persistence of 2,4-D in Natural Surface Waters in Britain; British Crop Protection Council: Surrey, UK, 1966. [Google Scholar]
- Ayar, N.; Bilgin, B.; Atun, G. Kinetics and equilibrium studies of the herbicide 2,4-dichlorophenoxyacetic acid adsorption on bituminous shale. Chem. Eng. J. 2008, 138, 239–248. [Google Scholar] [CrossRef]
- Gupta, V.K.; Ali, I.; Suhas; Saini, V.K. Adsorption of 2,4-D and carbofuran pesticides using fertilizer and steel industry wastes. J. Colloid Interface Sci. 2006, 299, 556–563. [Google Scholar] [CrossRef] [PubMed]
- Cupples, A.M.; Sims, G.K. Identification of in situ 2,4-dichlorophenoxyacetic acid-degrading soil microorganisms using DNA-stable isotope probing. Soil Biol. Biochem. 2007, 39, 232–238. [Google Scholar] [CrossRef]
- Rivera-Utrilla, J.; Sánchez-Polo, M.; Abdel, M.M.; Ocampo-Pérez, R. Role of activated carbon in the photocatalytic degradation of 2,4-dichlorophenoxyacetic acid by the UV/TiO2/activated carbon system. Appl. Catal. B 2012, 126, 100–107. [Google Scholar] [CrossRef]
- Meng, J.; Liang, J.; Ou, X.; Ding, Y.; Liang, G. Effects of mineral tourmaline particles on the photocatalytic activity of TiO2 thin films. J. Nanosci. Nanotechnol. 2008, 8, 1279–1283. [Google Scholar] [PubMed]
- Yoshikawa, N.; Kimura, T.; Kawase, Y. Oxidative degradation of nonionic surfactants with TiO2 photocatalyst in a bubble column reactor. Can. J. Chem. Eng. 2003, 81, 719–724. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bian, X.; Chen, J.; Ji, R. Degradation of 2,4-Dichlorophenoxyacetic Acid (2,4-D) by Novel Photocatalytic Material of Tourmaline-Coated TiO2 Nanoparticles: Kinetic Study and Model. Materials 2013, 6, 1530-1542. https://doi.org/10.3390/ma6041530
Bian X, Chen J, Ji R. Degradation of 2,4-Dichlorophenoxyacetic Acid (2,4-D) by Novel Photocatalytic Material of Tourmaline-Coated TiO2 Nanoparticles: Kinetic Study and Model. Materials. 2013; 6(4):1530-1542. https://doi.org/10.3390/ma6041530
Chicago/Turabian StyleBian, Xuesen, Jianqiu Chen, and Rong Ji. 2013. "Degradation of 2,4-Dichlorophenoxyacetic Acid (2,4-D) by Novel Photocatalytic Material of Tourmaline-Coated TiO2 Nanoparticles: Kinetic Study and Model" Materials 6, no. 4: 1530-1542. https://doi.org/10.3390/ma6041530



