Utilization of an Ionic Liquid/Urea Mixture as a Physical Coupling Agent for Agarose/Talc Composite Films
Abstract
:1. Introduction
2. Results and Discussion
2.1. Mechanical Testing

2.2. Morphology Examination
2.3. DSC Analysis

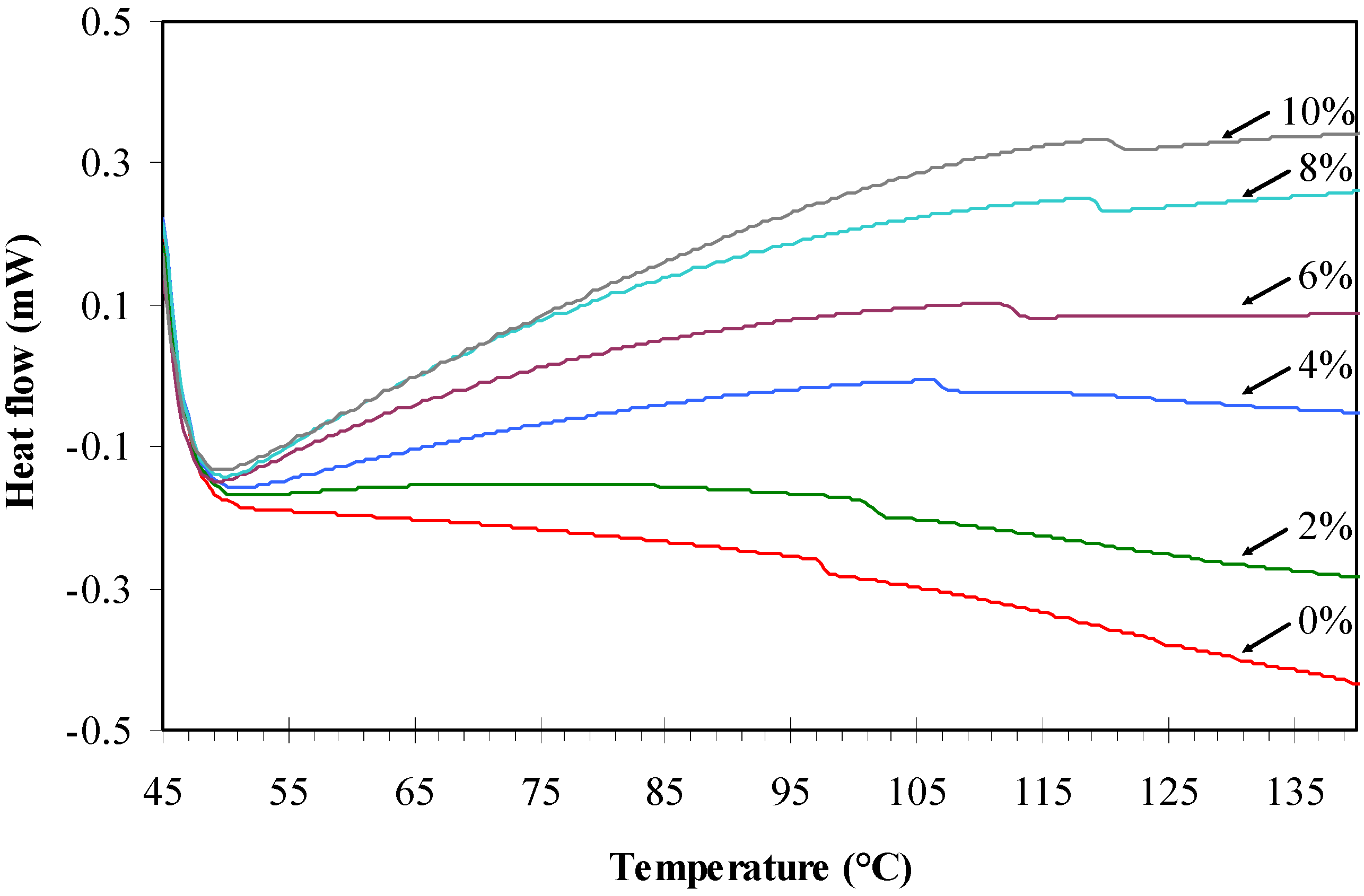
| Agarose/talc (wt %) | BmimCl/Urea (wt %) | Tg (°C) |
|---|---|---|
| 100 | 0 | 98.9 |
| 98 | 2 | 102.4 |
| 96 | 4 | 108.5 |
| 94 | 6 | 113.5 |
| 92 | 8 | 120.6 |
| 90 | 10 | 122.1 |
2.4. TGA Characterization
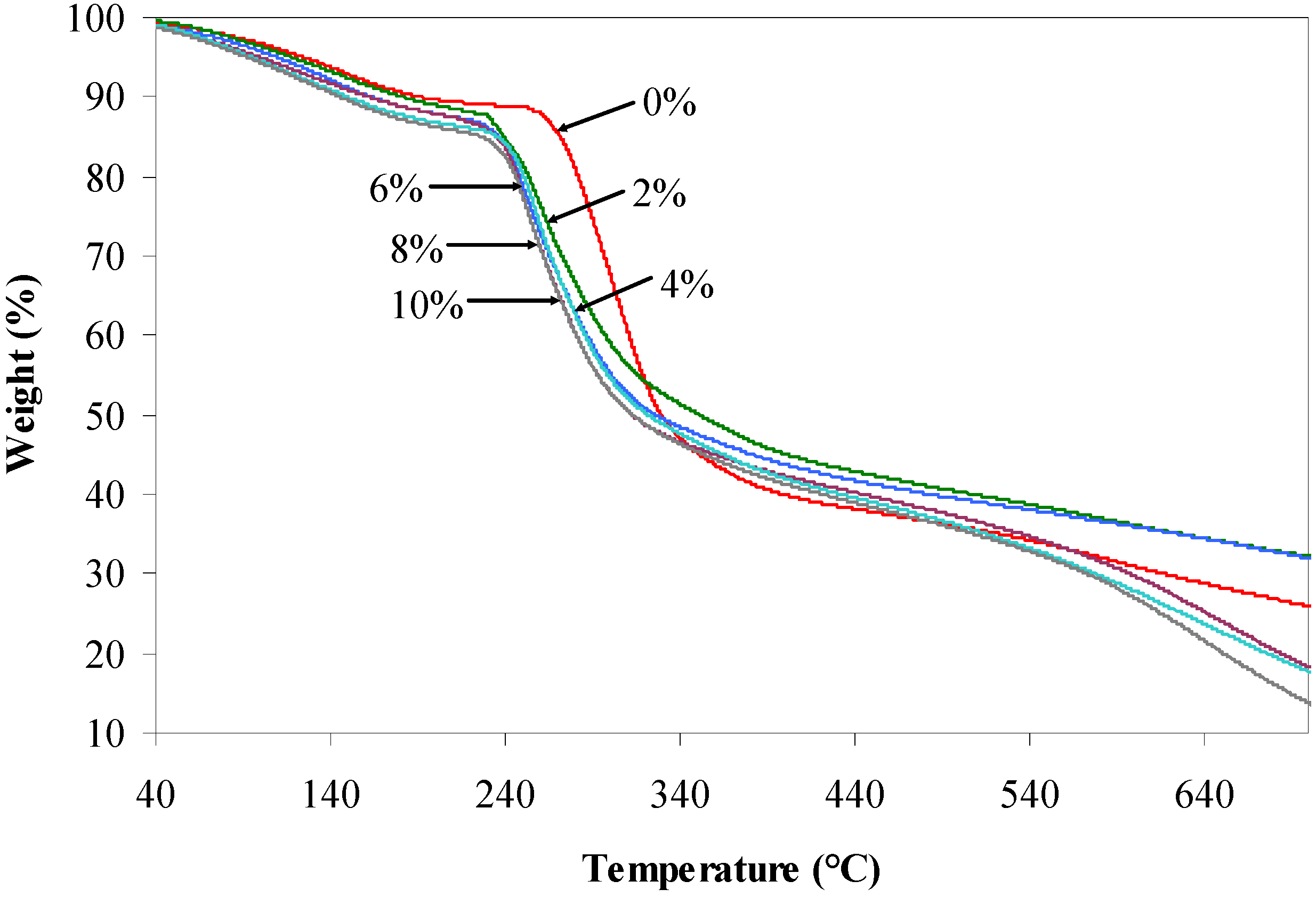
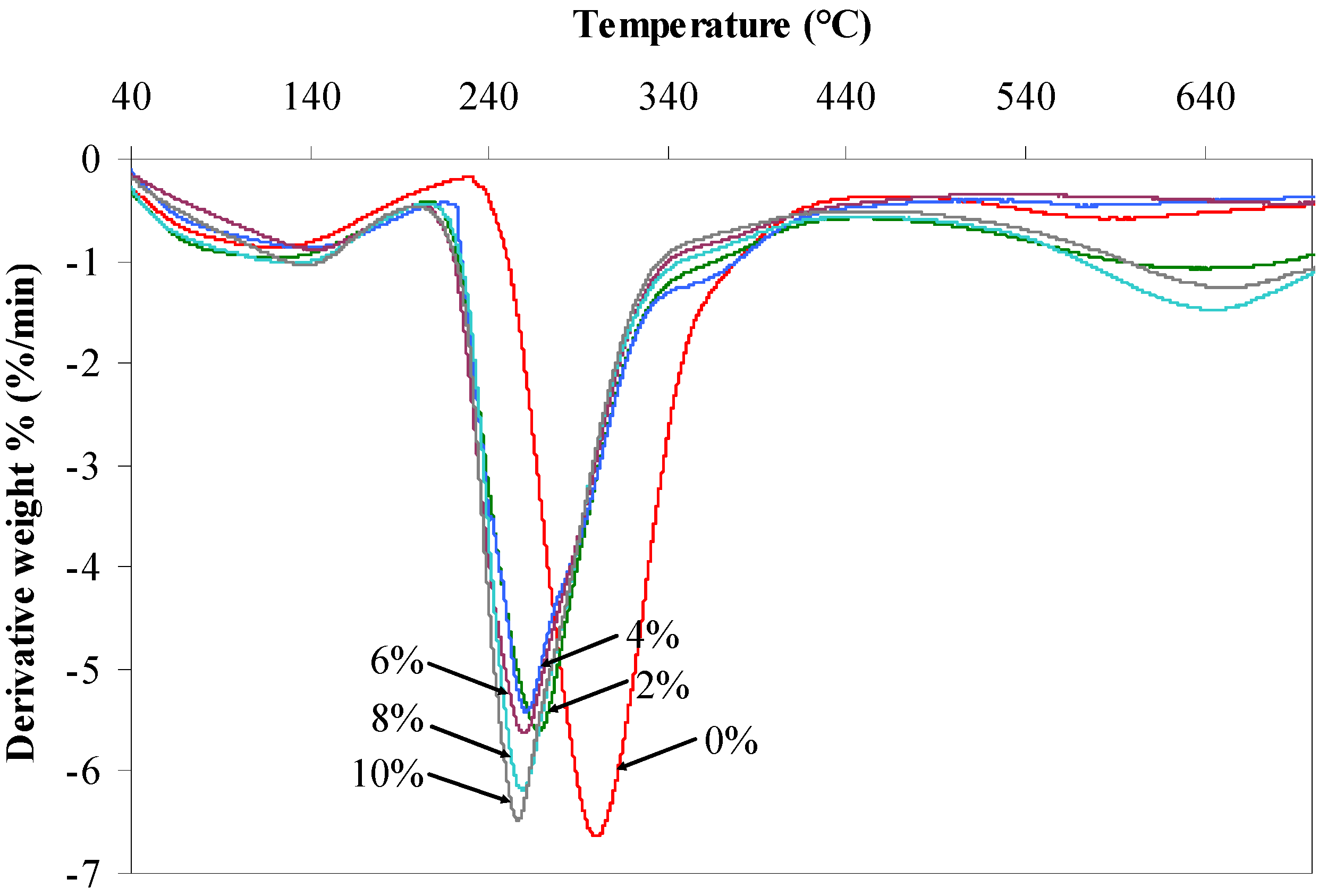
| Agarose/talc (wt %) | BmimCl/Urea (wt %) | Initial decomposition temperature (°C) | Maximum decomposition temperature (°C) |
|---|---|---|---|
| 100 | 0 | 192.7 | 300.1 |
| 98 | 2 | 182.1 | 267.8 |
| 96 | 4 | 163.5 | 260.6 |
| 94 | 6 | 163.4 | 259.7 |
| 92 | 8 | 150.4 | 258.9 |
| 90 | 10 | 146.0 | 256.2 |
2.5. FTIR Analysis
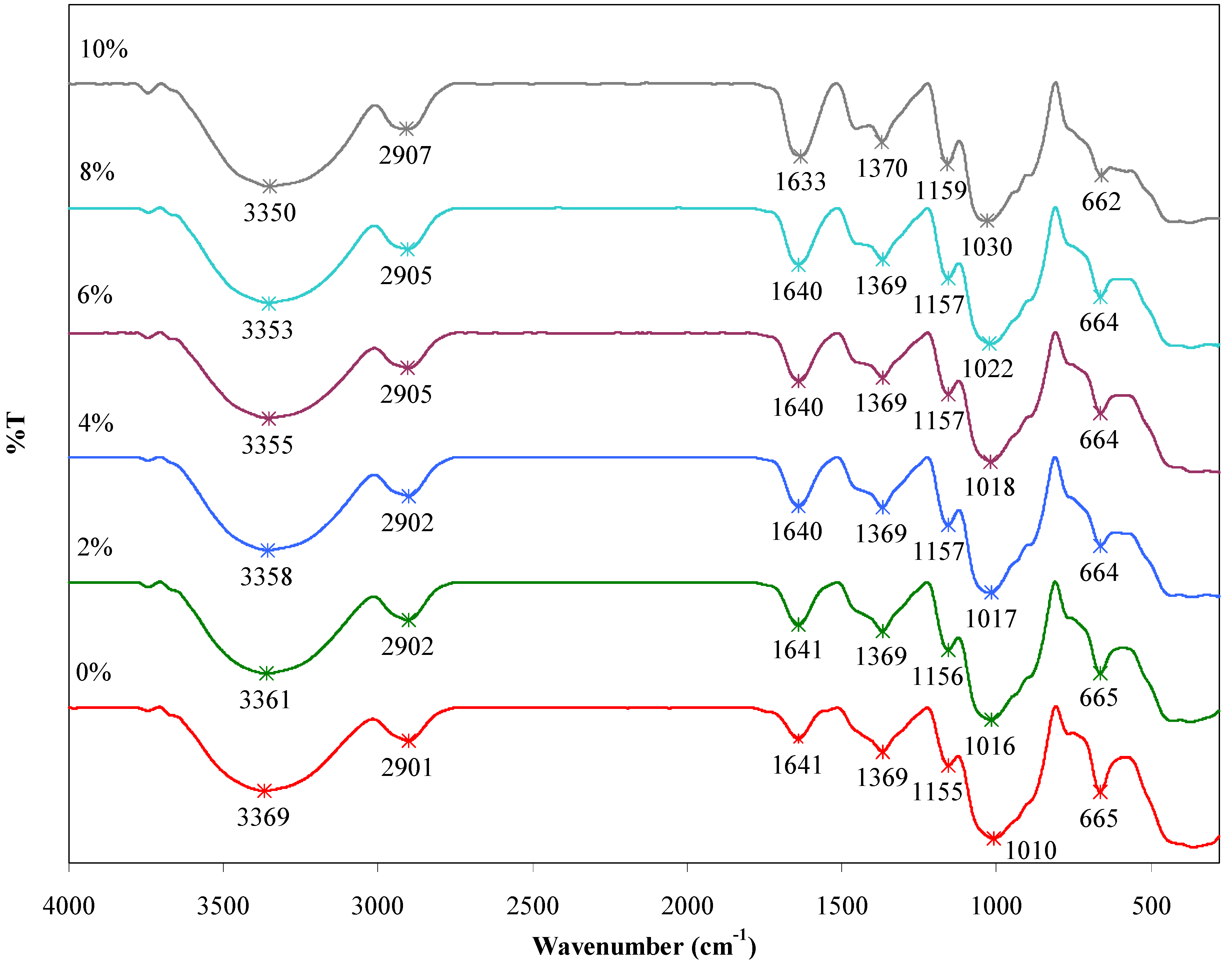
| BmimCl/Urea (wt %) | Wavenumber (cm−1) | |||||
|---|---|---|---|---|---|---|
| O–H stretching | C–H stretching | CH3 bending | C–O–C stretching | Si–O–Si stretching | Si–O–Mg stretching | |
| 0 | 3369 | 2901 | 1369 | 1155 | 1010 | 665 |
| 2 | 3361 | 2902 | 1369 | 1156 | 1016 | 665 |
| 4 | 3358 | 2902 | 1369 | 1157 | 1017 | 664 |
| 6 | 3355 | 2905 | 1369 | 1157 | 1018 | 664 |
| 8 | 3353 | 2905 | 1369 | 1157 | 1022 | 664 |
| 10 | 3350 | 2907 | 1370 | 1159 | 1030 | 662 |

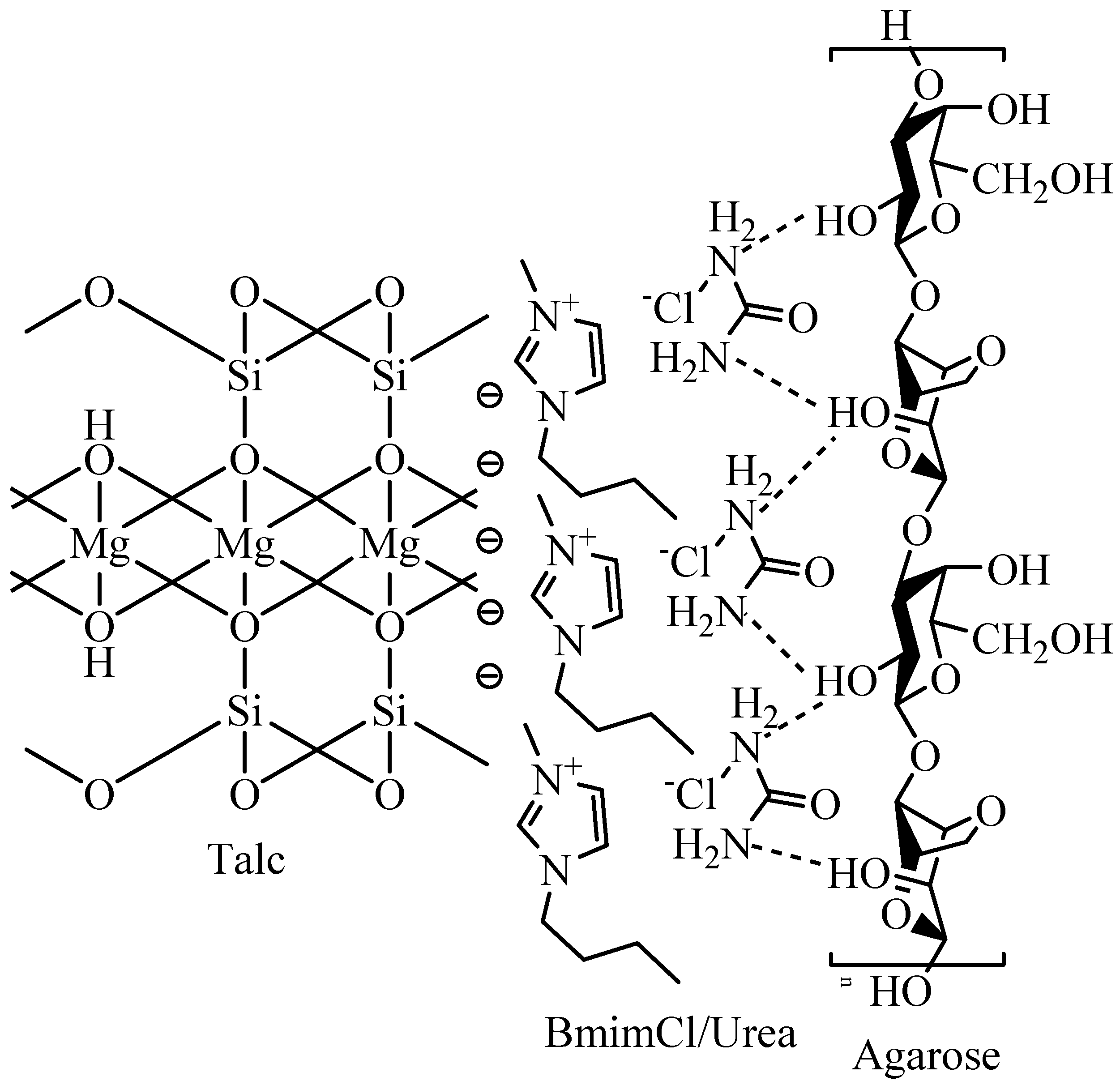
2.6. FT-Raman Study
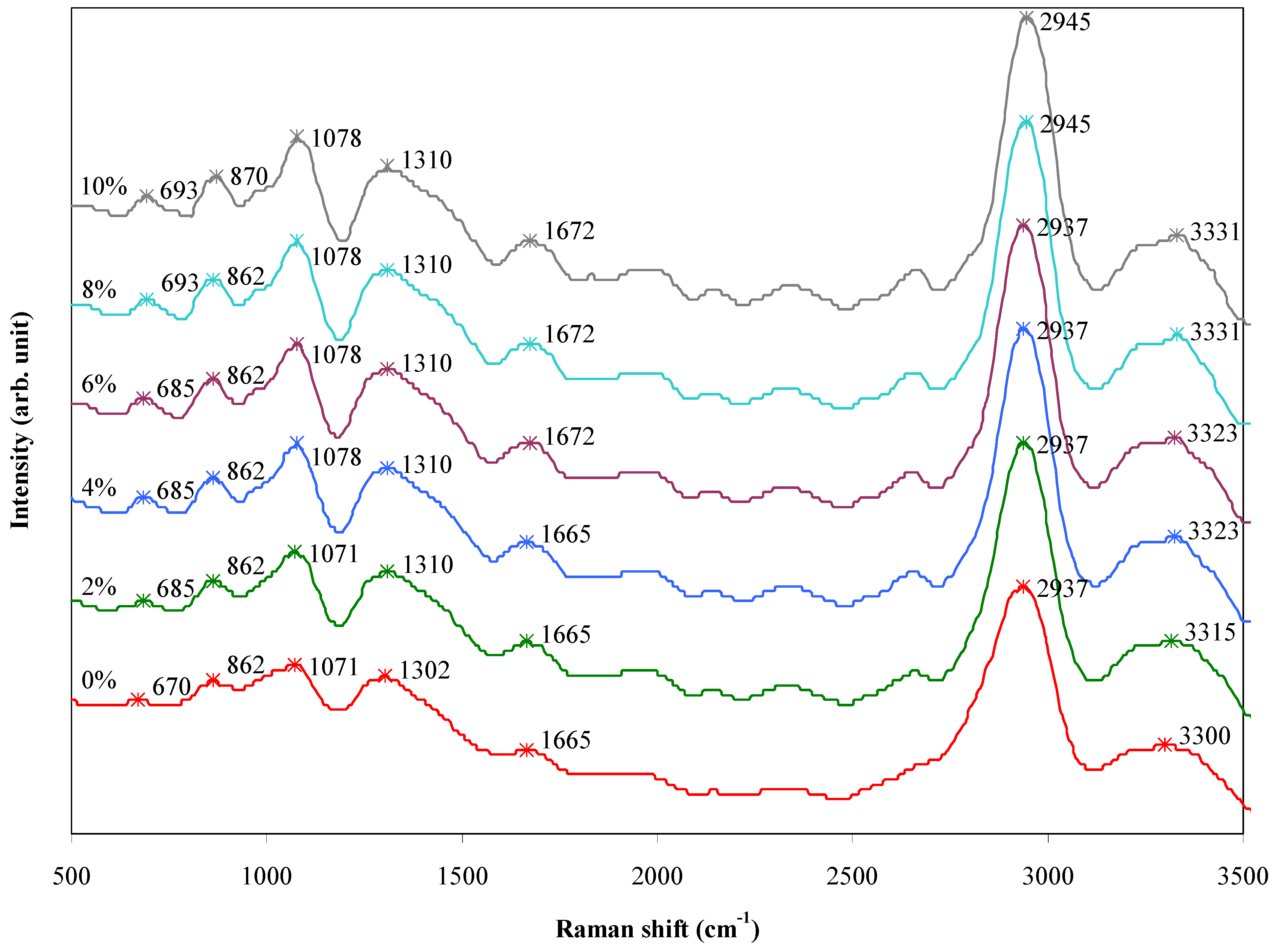
| Raman shift (cm−1) | Assignment | Functional group |
|---|---|---|
| 670–693 | SiO4 stretching | Silica |
| 862–870 | C–H bending | Alkane |
| 1071–1078 | C–O stretching | Alcohol |
| 1302–1310 | CH3 bending | Alkane |
| 2937–2945 | C–H stretching | Alkane |
| 3300–3331 | O–H stretching | Alcohol |
3. Experimental Section
3.1. Materials
3.2. Preparation of BmimCl/Urea Mixture and Agarose/Talc Composite Films
3.3. Characterization
4. Conclusions
Acknowledgments
References
- Leong, Y.W.; Abu Bakar, M.B.; Ishak, Z.A.M.; Ariffin, A.; Pukanszky, B. Comparison of the mechanical properties and interfacial interactions between talc, kaolin, and calcium carbonate filled polypropylene composites. J. Appl. Polym. Sci. 2004, 91, 3315–3326. [Google Scholar] [CrossRef]
- Zhou, Y.; Rangari, V.; Mahfuz, H.; Jeelani, S.; Mallick, P.K. Experimental study on thermal and mechanical behavior of polypropylene, talc/polypropylene and polypropylene/clay nanocomposites. Mater. Sci. Eng. A 2005, 402, 109–117. [Google Scholar] [CrossRef]
- Wang, J.; Somasundaran, P. Mechanisms of ethyl(hydroxyethyl) cellulose–solid interaction: Influence of hydrophobic modification. J. Colloid Interface Sci. 2006, 293, 322–332. [Google Scholar] [CrossRef] [PubMed]
- Flaris, V. Talc. In Functional Fillers for Plastics, 1st ed.; Xanthos, M., Ed.; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2005; Volume 1, pp. 207–220. [Google Scholar]
- Kord, B. Evaluation on the effect of wood flour and coupling agent content on the hygroscopic thickness swelling rate of polypropylene composites. BioResources 2011, 6, 3055–3065. [Google Scholar]
- Sarang, S.; Misra, R.D.K. Strain rate sensitive behavior of wollastonite-reinforced ethylene–propylene copolymer composites. Mater. Sci. Eng. A 2004, 381, 259–272. [Google Scholar] [CrossRef]
- González, J.; Albano, C.; Ichazo, M.; Diaz, B. Effects of coupling agents on mechanical and morphological behavior of the PP/HDPE blend with two different CaCO3. Eur. Polym. J. 2002, 38, 2465–2475. [Google Scholar] [CrossRef]
- Shamsuri, A.A.; Abdullah, D.K. Protonation and Complexation Approaches for Production of Protic Eutectic Ionic Liquids. J. Phys. Sci. 2010, 21, 15–28. [Google Scholar]
- Shamsuri, A.A.; Abdullah, D.K. A Preliminary Study of Oxidation of Lignin from Rubber Wood to Vanillin in Ionic Liquid Medium. Oxid. Commun. 2012, 35, 767–775. [Google Scholar]
- Łuczak, J.; Joskowska, M.; Hupka, J. Imidazolium ionic liquids in mineral processing. Physicochem. Probl. Miner. Process. 2008, 42, 223–236. [Google Scholar]
- Das, A.; Stöckelhuber, K.W.; Jurk, R.; Fritzsche, J.; Klüppel, M.; Heinrich, G. Coupling activity of ionic liquids between diene elastomers and multi-walled carbon nanotubes. Carbon 2009, 47, 3313–3321. [Google Scholar] [CrossRef]
- Donato, R.K.; Benvegnú, M.A.; Furlan, L.G.; Mauler, R.S.; Schrekker, H.S. Imidazolium salts as liquid coupling agents for the preparation of polypropylene-silica composites. J. Appl. Polym. Sci. 2010, 116, 304–307. [Google Scholar]
- Donato, R.K.; Matějka, L.; Schrekker, H.S.; Pleštil, J.; Jigounov, A.; Brus, J.; Šlouf, M. The multifunctional role of ionic liquids in the formation of epoxy-silica nanocomposites. J. Mater. Chem. 2011, 21, 13801–13810. [Google Scholar] [CrossRef]
- Shamsuri, A.A.; Abdullah, D.K.; Daik, R. Fabrication of agar/biopolymer blend aerogels in ionic liquid and co-solvent mixture. Cell. Chem. Technol. 2012, 46, 45–52. [Google Scholar]
- Shamsuri, A.A.; Daik, R. Plasticizing effect of choline chloride/urea eutectic-based ionic liquid on physicochemical properties of agarose films. BioResources 2012, 7, 4760–4775. [Google Scholar] [CrossRef]
- Burdukova, E.; Becker, M.; Bradshaw, D.J.; Laskowski, J.S. Presence of negative charge on the basal planes of New York talc. J. Colloid Interface Sci. 2007, 315, 337–342. [Google Scholar] [CrossRef] [PubMed]
- Radford, D.W.; Grabher, A.; Bridge, J. Inorganic polymer matrix composite strength related to interface condition. Materials 2009, 2, 2216–2227. [Google Scholar] [CrossRef]
- Song, Z.; Hou, X.; Zhang, L.; Wu, S. Enhancing crystallinity and orientation by hot-stretching to improve the mechanical properties of electrospun partially aligned polyacrylonitrile (PAN) nanocomposites. Materials 2011, 4, 621–632. [Google Scholar] [CrossRef]
- Schaber, P.M.; Colson, J.; Higgins, S.; Thielen, D.; Anspach, B.; Brauer, J. Thermal decomposition (pyrolysis) of urea in an open reaction vessel. Thermochim. Acta 2004, 424, 131–142. [Google Scholar] [CrossRef]
- Fang, H.L.; DaCosta, H.F. Thermolysis characterization of urea-SCR. In Proceeding of the 8th Annual Conference of the Engine-Efficiency and Emissions Research (DEER), San Diego, CA, USA, 25–29 August 2002.
- Roohpour, N.; Wasikiewicz, J.; Moshaverinia, A.; Paul, D.; Rehman, I.; Vadgama, P. Isopropyl myristate-modified polyether-urethane coatings as protective barriers for implantable medical devices. Materials 2009, 2, 719–733. [Google Scholar] [CrossRef]
- Sprynskyy, M.; Gadzała-Kopciuch, R.; Nowak, K.; Buszewski, B. Removal of zearalenone toxin from synthetics gastric and body fluids using talc and diatomite: A batch kinetic study. Colloids Surf. B Biointerfaces 2012, 94, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Du, C.; Hu, Y.; Jin, S.; Yang, W.; Tang, A.; Avvakumov, E.G. Preparation of porous material from talc by mechanochemical treatment and subsequent leaching. Appl. Clay Sci. 2006, 31, 290–297. [Google Scholar] [CrossRef]
- Wang, Z.; Latonen, R.; Kvarnström, C.; Ivaska, A.; Niu, L. Preparation of multi-walled carbon nanotube/amino-terminated ionic liquid arrays and their electrocatalysis towards oxygen reduction. Materials 2010, 3, 672–681. [Google Scholar] [CrossRef]
- Magniez, K.; Chaffraix, T.; Fox, B. Toughening of a carbon-fibre composite using electrospun poly(hydroxyether of bisphenol a) nanofibrous membranes through inverse phase separation and inter-domain etherification. Materials 2011, 4, 1967–1984. [Google Scholar] [CrossRef]
- Lu, Z. Chemical Coupling in Wood-Polymer Composite. Ph.D. Thesis, Louisiana State University, Baton Rouge, LA, USA, December 2003. [Google Scholar]
- de Veij, M.; Vandenabeele, P.; de Beer, T.; Remon, J.P.; Moens, L. Reference database of Raman spectra of pharmaceutical excipients. J. Raman Spectros. 2009, 40, 297–307. [Google Scholar] [CrossRef]
- Pereira, L.; Sousa, A.; Coelho, H.; Amado, A.M.; Ribeiro-Claro, P.J.A. Use of FTIR, FT-Raman and 13C-NMR spectroscopy for identification of some seaweed phycocolloids. Biomol. Eng. 2003, 20, 223–228. [Google Scholar] [CrossRef] [PubMed]
- Kizil, R.; Irudayaraj, J. Discrimination of irradiated starch gels using FT-Raman spectroscopy and chemometrics. J. Agric. Food Chem. 2006, 54, 13–18. [Google Scholar] [CrossRef] [PubMed]
- American Society for Testing and Materials. ASTM D882-91: Standard Test Method for Tensile Properties of Thin Plastic Sheeting; ASTM: Philadelphia, PA, USA, 1992. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Shamsuri, A.A.; Daik, R. Utilization of an Ionic Liquid/Urea Mixture as a Physical Coupling Agent for Agarose/Talc Composite Films. Materials 2013, 6, 682-698. https://doi.org/10.3390/ma6020682
Shamsuri AA, Daik R. Utilization of an Ionic Liquid/Urea Mixture as a Physical Coupling Agent for Agarose/Talc Composite Films. Materials. 2013; 6(2):682-698. https://doi.org/10.3390/ma6020682
Chicago/Turabian StyleShamsuri, Ahmad Adlie, and Rusli Daik. 2013. "Utilization of an Ionic Liquid/Urea Mixture as a Physical Coupling Agent for Agarose/Talc Composite Films" Materials 6, no. 2: 682-698. https://doi.org/10.3390/ma6020682




