The Synthesis of α-MoO3 by Ethylene Glycol
Abstract
:1. Introduction
2. Results and Discussion
2.1. Effect of the Content of the Ethylene Glycol
| 0.1 M of ammonium molybdate tetrahydrate solution (mL) | Ethylene glycol (mL) | Mo (ppm) | R (%) |
|---|---|---|---|
| 250 | – | 85,960 | – |
| 25 | 41,920 | 51.2 | |
| 50 | 30,764 | 64.2 | |
| 100 | 49,840 | 42 | |
| 125 | 70,630 | 17.8 |
2.2. Effect of Sintering Temperatures

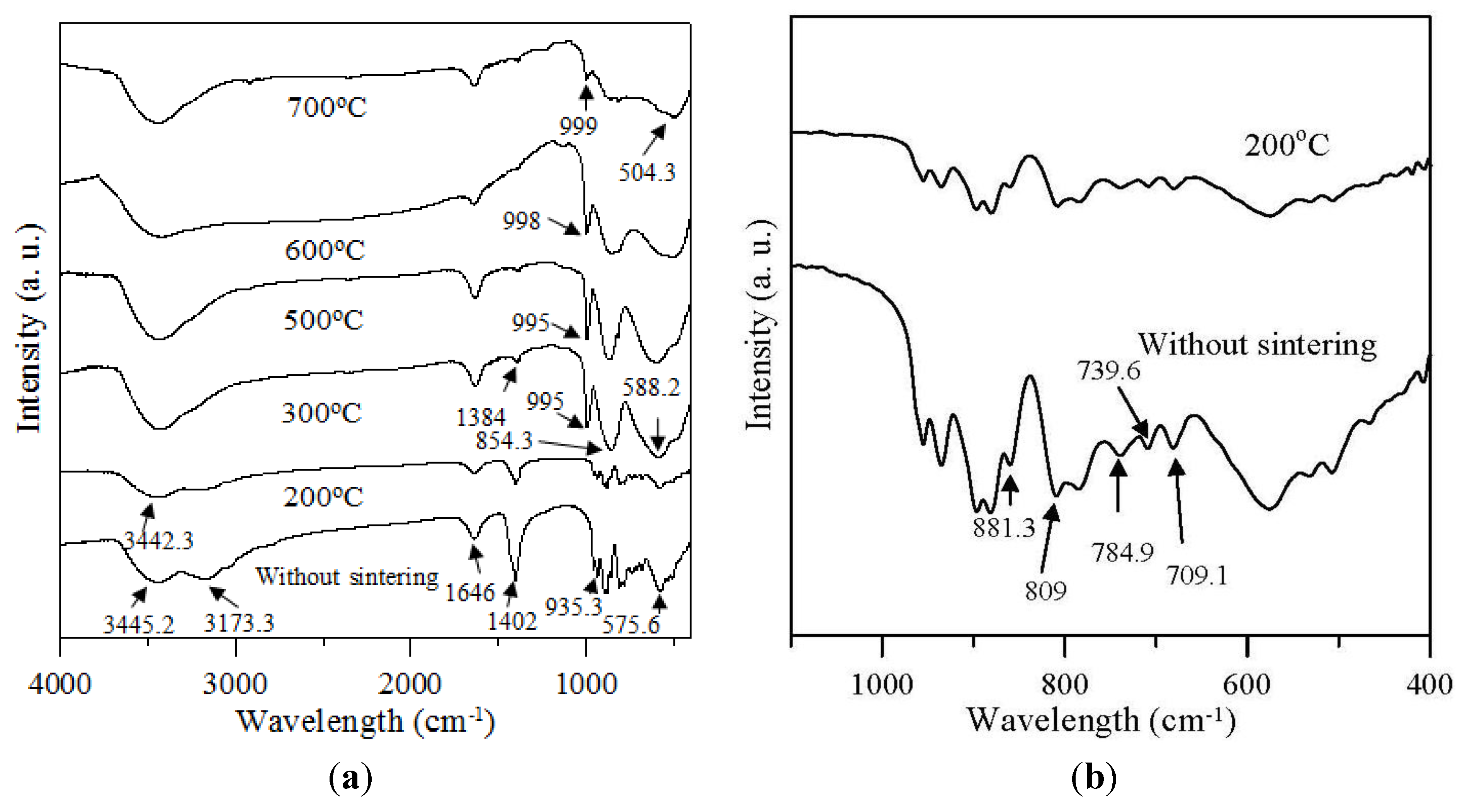


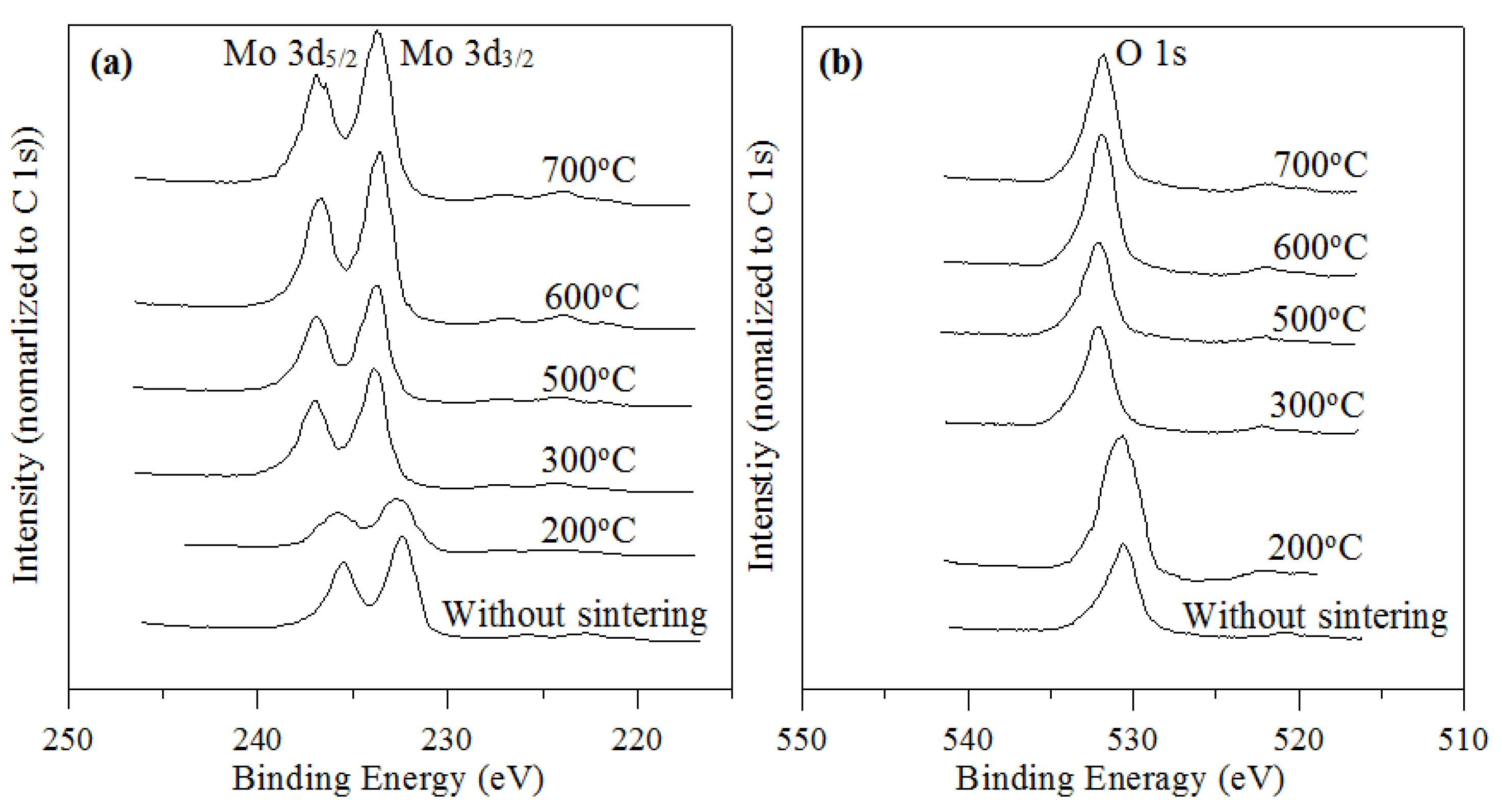
| Sintering Temperature (°C) | Mo 3d5/2 (eV) | FWHM (eV) | Mo 3d3/2 (eV) | FWHM (eV) | ΔMo 3d (eV) | O 1s (eV) | FWHM (eV) | O/Mo atomic ratio | intensity ratio |
|---|---|---|---|---|---|---|---|---|---|
| Before sintering | 232.46 | 1.57 | 235.49 | 1.42 | 3.03 | 530.59 | 2.19 | 4.1 | 0.62 |
| 200 | 232.69 | 1.95 | 235.87 | 1.9 | 3.27 | 530.67 | 2.57 | 4.9 | 0.62 |
| 300 | 233.65 | 1.41 | 236.92 | 1.41 | 3.27 | 532.08 | 2.04 | 3.6 | 0.61 |
| 500 | 233.72 | 1.45 | 236.93 | 1.39 | 3.21 | 532.08 | 1.94 | 3.4 | 0.68 |
| 600 | 233.55 | 1.5 | 236.77 | 1.5 | 3.22 | 531.85 | 1.94 | 3.3 | 0.57 |
| 700 | 233.61 | 1.58 | 236.93 | 1.64 | 3.32 | 531.81 | 2.08 | 3.1 | 0.6 |
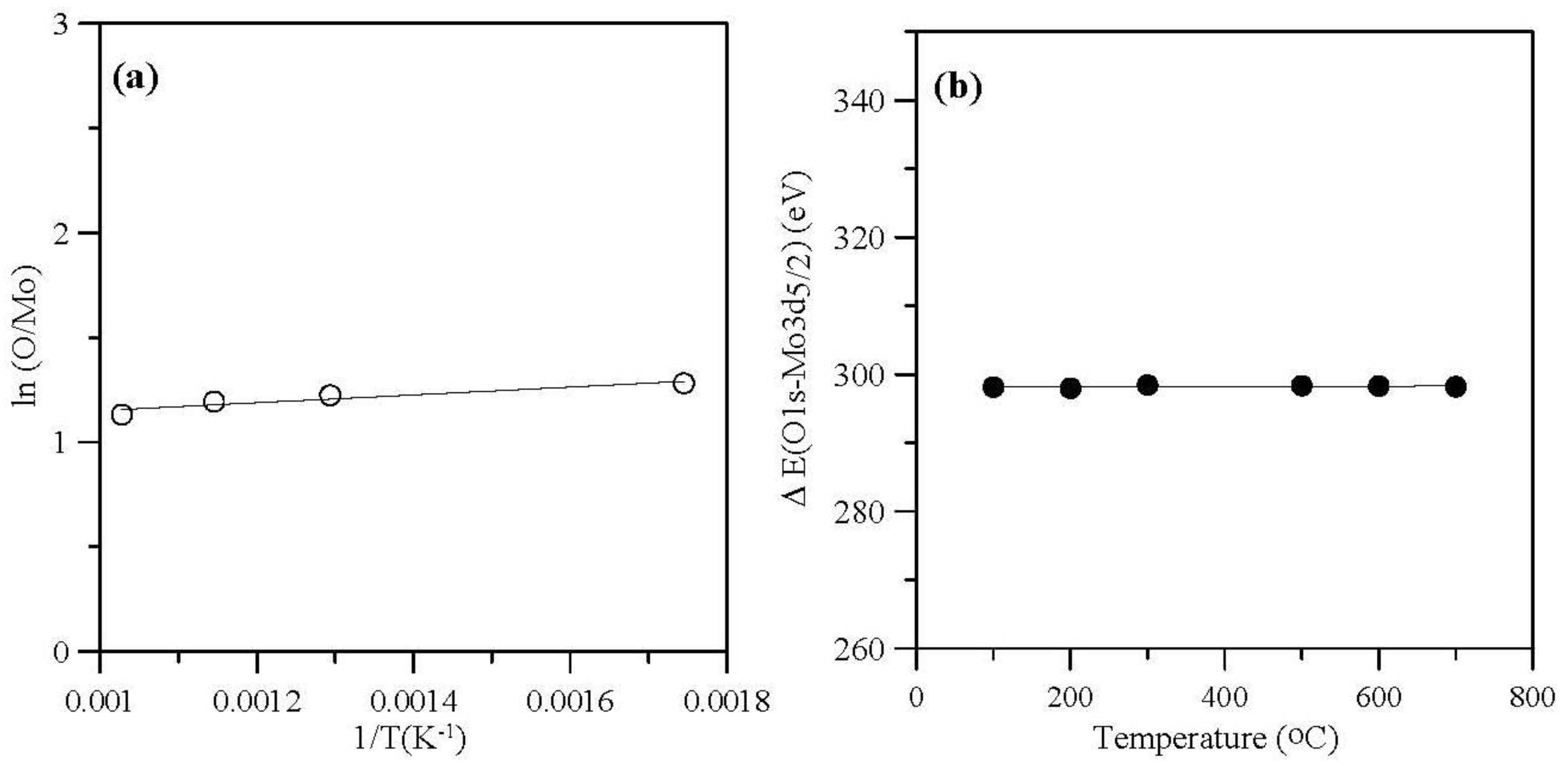
2.3. Effect of Sintering Times

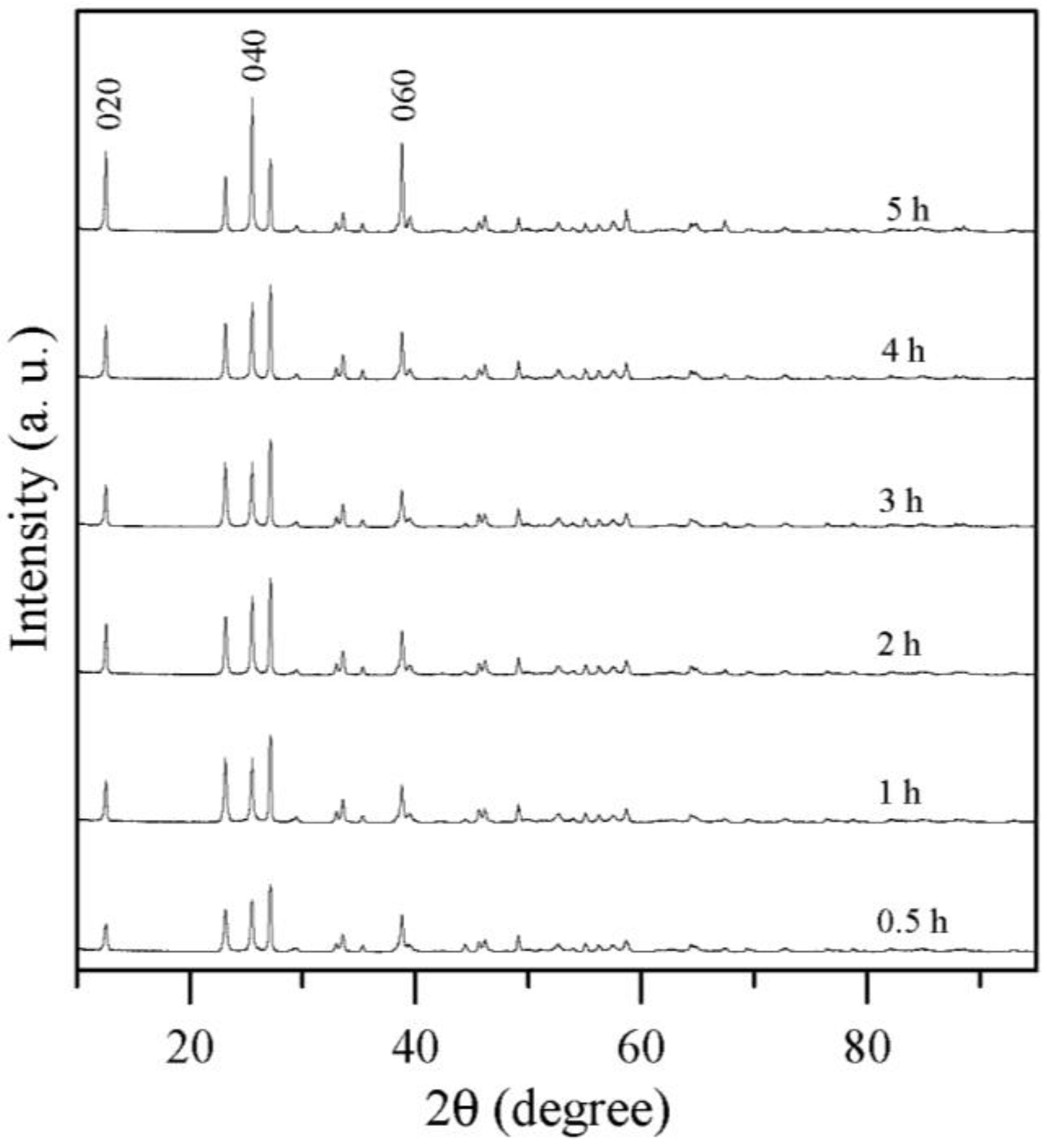
3. Experimental Section
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Cheng, L.; Shao, M.; Wang, X.; Hu, H. Single-crystalline molybdenum trioxide nanoribbons: Photocatalytic, photoconductive, and electrochemical properties. Chem. Eur. J. 2009, 15, 2310–2316. [Google Scholar] [PubMed]
- Yu, A.; Kumagai, N.; Liu, Z.; Lee, J.Y. Preparation of sodium molybdenum oxides by a solution technique and their electrochemical performance in lithium intercalation. Solid State Ion. 1998, 106, 11–18. [Google Scholar] [CrossRef]
- Queeney, K.T.; Friend, C.M. Site-selective surface reactions: Hydrocarbon oxidation processes on oxidized Mo (110). J. Phys. Chem. 2000, 104, 409–415. [Google Scholar] [CrossRef]
- Balendhran, S.; Deng, J.; Ou, J.Z.; Walia, S.; Scott, J.; Tang, J.; Wang, K.L.; Field, M.R.; Russo, S.; Zhuiykov, S.; Strano, M.S.; Medhekar, N.; Sriram, S.; Bhaskaran, M.; Kalantar-zadeh, K. Enhanced charge carrier mobility in two-dimensional high dielectric molybdenum oxide. Adv. Mater. 2013, 25, 109–114. [Google Scholar] [CrossRef] [PubMed]
- Rahmani, M.B.; Keshmiri, S.H.; Yu, J.; Sadek, A.Z.; Al-Mashat, L.; Moafi, A.; Latham, K.; Li, Y.X.; Wlodarski, W.; Kalantar-zadeh, K. Gas sensing properties of thermally evaporated lamellar MoO3. Sens. Actuator Chem. 2010, 145, 13–19. [Google Scholar] [CrossRef]
- Hussain, Z. Optical and electrochromic properties of heated and annealed MoO3 thin films. J. Mater. Res. 2001, 16, 2695–2708. [Google Scholar] [CrossRef]
- Balendhran, S.; Walia, S.; Nili, H.; Ou, J.Z.; Zhuiykov, S.; Kaner, R.B.; Sriram, S.; Bhaskaran, M.; Kalantar-zadeh, K. Two-Dimensional molybdenum trioxide and dichalcogenides. Adv. Funct. Mater. 2013, 23, 3952–3970. [Google Scholar] [CrossRef]
- Yao, D.D.; Ou, J.Z.; Latham, K.; Zhuiykov, S.; O’Mullane, A.P.; Kalantar-zadeh, K. Electrodeposited α- and β-Phase MoO3 films and investigation of their gasochromic properties. Cryst. Growth Des. 2012, 12, 1865–1870. [Google Scholar] [CrossRef]
- Kalantar-zadeh, K.; Tang, J.; Wang, M.; Wang, K.L.; Shailos, A.; Galatsis, K.; Kojima, R.; Strong, V.; Lech, A.; Wlodarski, W.; Kaner, R.B. Synthesis of nanometre-thick MoO3 sheets. Nanoscale 2010, 2, 429–433. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, C.V.; Muñoz-Espí, R.; Li, Q. Formation of molybdenum oxide nanostructures controlled by poly(ethylene oxide). Chin. J. Polym. Sci. 2009, 27, 11–22. [Google Scholar] [CrossRef]
- Ivanova, T.; Surtchev, M.; Gesheva, K. Investigation of CVD molybdenum oxide films. Mater. Lett. 2002, 53, 250–257. [Google Scholar] [CrossRef]
- Lee, Y.J.; Park, C.W.; Kim, D.G.; Nicholsa, W.T.; Oh, S.T.; Kim, Y.D. MoO3 Thin Film Synthesis by Chemical Vapor Transport of Volatile MoO3(OH)2. J. Ceram. Process. Res. 2010, 11, 52–55. [Google Scholar]
- Dong, W.; Dunn, B. Sol–gel Synthesis of Monolithic Molybdenum Oxide Aerogels and Xerogels. J. Mater. Chem. 1998, 8, 665–670. [Google Scholar] [CrossRef]
- Mizushima, T.; Fukushima, K.; Ohkita, H.; Kakuta, N. Synthesis of β-MoO3 through Evaporation of HNO3-added Molybdic Acid Solution and its Catalytic Performance in Partial Oxidation of Methanol. Appl. Catal. Gen. 2007, 326, 106–112. [Google Scholar] [CrossRef]
- Song, J.; Ni, X.; Gao, L.; Zheng, H. Synthesis of Metastable h-MoO3 by Simple Chemical Precipitation. Mater. Chem. Phys. 2007, 102, 245–248. [Google Scholar] [CrossRef]
- Zhao, Y.; Liu, J.; Zhou, Y.; Zhang, Z.; Xu, Y.; Naramoto, H.; Yamamoto, S. Preparation of MoO3 nanostructures and their optical properties. J. Phys. Condens. Matter 2003, 15, L547–L552. [Google Scholar] [CrossRef]
- Yang, X.; Tang, H.; Zhang, R.; Song, H.; Cao, K. NaCl-assisted hydrothermal synthesis of high-quality crystalline α-MoO3 Nanobelts. Cryst. Res. Technol. 2011, 4, 409–412. [Google Scholar] [CrossRef]
- Stoyanova, A.; Iordanova, R.; Mancheva, M.; Dimitriev, Y. Synthesis and structural characterization of MoO3 phases obtained from molybdic acid by addition of HNO3 and H2O2. J. Optoelectron. Adv. Mater. 2009, 11, 1127–1131. [Google Scholar]
- Lagashetty, A.; Havanoor, V.; Basavaraja, S.; Venkataraman, A. Synthesis of MoO3 and its polyvinyl alcohol nanostructured film. Bull. Mater. Sci. 2005, 28, 477–481. [Google Scholar] [CrossRef]
- Sowmiyanarayan, R.; Santhanalakshmi, J. Novel synthesis of MoO3 nanobelts and V2O5 nano flakes and the characterization. Nano Vis. 2012, 2, 49–60. [Google Scholar]
- Irmawati, R.; Shafizah, M. The Production of high purity hexagonal MoO3 through the acid washing of as-prepared solids. Int. J. Basic Appl. Sci. 2009, 9, 241–244. [Google Scholar]
- McEvoy, T.M.; Stevenson, K.J.; Hupp, J.T.; Dang, X. Electrochemical preparation of molybdenum trioxide thin films: effect of sintering on electrochromic and electroinsertion properties. Langmuir 2003, 19, 4316–4326. [Google Scholar] [CrossRef]
- Ramírez, I.J.; la Cruz, A.M. Synthesis of β-MoO3 by vacuum drying and its structural and electrochemical characterization. Mater. Lett. 2003, 57, 1034–1039. [Google Scholar] [CrossRef]
- Sasidharan, M.; Gunawardhana, N.; Noma, H.; Yoshio, M.; Nakashima, K. α-MoO3 hollow nanospheres as an anode material for li-ion batteries. Bull. Chem. Soc. Jpn. 2012, 8, 642–646. [Google Scholar] [CrossRef]
- Parviz, D.; Kazemeini, M.; Rashidi, A.M.; Jozani, K.J. Synthesis and characterization of MoO3 nanostructures by solution combustion method employing morphology and size control. J. Nanopart. Res. 2010, 12, 1509–1521. [Google Scholar] [CrossRef]
- Marquis, E.; Sanderson, T.J.; Keating, R.K.P. Epoxidation in the presence of molybdenum/alkali metal/ethylene glycol compolexes. U.S. Patent 4,845,251, 4 July 1989. [Google Scholar]
- Pak, J.J.; Jo, J.O.; Park, C.H.; Kang, J.G.; Shin, D.H. Recovery of molybdenum from spent acid by ammonia gas neutralization. Mater. Trans. 2008, 49, 202–207. [Google Scholar] [CrossRef]
- Weber, T.; Muijsers, J.C.; van Wolput, J.H.M.C.; Verhagen, C.P.J.; Niemantsverdriet, J.W. Basic Reaction steps in the sulfidation of crystalline MoO3 to MoS2, as studied by X-ray photoelectron and infrared emission spectroscopy. J. Phys. Chem. 1996, 100, 14144–14150. [Google Scholar] [CrossRef]
- Chen, D.; Liu, M.; Yin, L.; Li, T.; Yang, Z.; Li, X.; Fan, B.; Wang, H.; Zhang, R.; Li, Z.; et al. Single-crystalline MoO3 nanoplates: Topochemical synthesis and enhanced ethanol-sensing performance. J. Mater. Chem. 2011, 21, 9332–9342. [Google Scholar] [CrossRef]
- Pereira, L.G.; Soledade, L.E.B.; Ferreira, J.M.; Lima, S.J.G.; Fernandes Jr, V.J.; Araújo, A.S.; Paskocimas, C.A.; Longoe, E.; Santos, M.R.C.; Souza, A.G.; Santos, I.M.G. Influence of doping on the preferential growth of α-MoO3. J. Alloy. Compd. 2008, 459, 377–385. [Google Scholar] [CrossRef]
- Chen, M.; Waghmare, U.V.; Friend, C.M.; Kaxiras, E. A Density functional study of clean and hydrogen-covered α-MoO3 (010): electronic structure and surface relaxation. J. Chem. Phys. 1998, 109, 6854–6860. [Google Scholar] [CrossRef]
- Li, J.; Kwong, F.L.; Ng, D.H.L. Synthesis of a biomorphic molybdenum trioxide templated from paper. J. Am. Ceram. Soc. 2008, 91, 1350–1353. [Google Scholar] [CrossRef]
- Frost, R.L.; Čjka, J.; Dickfos, M.J. Raman and infrared spectroscopic study of the molybdate containing uranyl mineral calcurmolite. J. Raman Spectrosc. 2008, 39, 779–785. [Google Scholar] [CrossRef] [Green Version]
- Dhanasankar, M.; Purushothaman, K.K.; Muralidharan, G. Effect of temperature of annealing on optical, structural and electrochromic properties of sol-gel dip coated molybdenum oxide films. Appl. Surf. Sci. 2011, 257, 2074–2079. [Google Scholar] [CrossRef]
- Zakharova, G.S.; Täschner, C.; Volkov, V.L.; Hellmann, I.; Klingeler, R.; Leonhardt, A.; Buchner, B. Solid State Sci 2007, 9, 1028–1032.
- Morris, H.E. Reactions of ethyl alcohol. Chem. Rev. 1932, 10, 465–506. [Google Scholar] [CrossRef]
- Peng, Z. Nanofibers—Production, Properties and Functional Applications; InTech: Shanghai, China, 2011. [Google Scholar]
- Navgire, M.E.; Lande, M.K.; Gambhire, A.B.; Rathod, S.B.; Aware, D.V.; Bhitre, S.R. Effect of Poly(ethylene glycol) surfactant on carbon-doped MoO3 nanocomposite materials and its photocatalytic activity. Bull. Mater. Sci. 2011, 34, 535–541. [Google Scholar] [CrossRef]
- He, T.; Yao, J.J. Photochromism of molybdenum oxide. Photochem. Photobiol. Photochem. Rev. 2003, 4, 125–143. [Google Scholar] [CrossRef]
- Smith, R.L.; Rohrer, G.S. Scanning probe microscopy of cleaved molybdates: α-MoO3(010), Mo18O52(100), Mo8O23(010), and η-Mo4O11(100). J. Solid State Chem. 1996, 124, 104–115. [Google Scholar] [CrossRef]
- Song, L.X.; Xia, J.; Dang, Z.; Yang, J.; Wang, L.B.; Chen, J. Formation, structure and physical properties of a series of α-MoO3 nanocrystals: From 3D to 1D and 2D. CrystEngComm 2012, 14, 2675–2682. [Google Scholar] [CrossRef]
- Scherrer, P. Bestimmung der größe und der inneren struktur von kolloidteilchen mittels röntgenstrahlen (in German). Nachr. Ges. Wiss. Göttingen 1918, 2, 98–100. [Google Scholar]
- Scanlon, D.O.; Watson, G.W.; Payne, D.J.; Atkinson, G.R.; Egdell, R.G.; Law, D.S.L. Theoretical and experimental study of the electronic structures of MoO3 and MoO2. J. Phys. Chem. 2010, 114, 4636–4645. [Google Scholar]
- Li, Y.B.; Bando, Y.; Goldberg, D.; Kurashima, K. Low-temperature synthesis of large-scale single-crystal molybdenum trioxide (MoO3) nanobelts. Appl. Phys. Lett. 2002, 81, 5048–5050. [Google Scholar] [CrossRef]
- Kornelak, P.; Su, D.S.; Thomas, C.; Camra, J.; Weselucha-Briczyńska, A.; Toba, M.; Najbar, M. Field emission from MoO3 nanobelts. Catal. Today 2008, 137, 273–277. [Google Scholar] [CrossRef]
- Deng, X.; Quek, S.Y.; Biener, M.M.; Biener, J.; Kang, D.H.; Schalek, R.; Kaxiras, E.; Friend, C.M. Selective thermal reduction of single-layer MoO3 nanostructures on Au(111). Surf. Sci. 2008, 602, 1166–1174. [Google Scholar] [CrossRef]
- Ramana, C.V.; Atuchin, V.V.; Kesler, V.G.; Kochubey, V.A.; Pokrovsky, L.D.; Shutthanandan, V.; Becker, U.; Ewing, R.C. Growth and surface characterization of sputter-deposited molybdenum oxide thin films. Appl. Surf. Sci. 2007, 253, 5368–5374. [Google Scholar] [CrossRef]
- Dupin, J.C.; Gonbeau, D.; Vinatier, P.; Levasseur, A. Systematic XPS studies of metal oxides, hydroxides and peroxides. Phys. Chem. Chem. Phys. 2000, 2, 1319–1324. [Google Scholar] [CrossRef]
- Choi, W.S. XPS study of MoO3 interlayer between aluminum electrode and inkjet-printed zinc tin oxide for thin-film transistor. Trans. Electr. Electron. Mater. 2011, 12, 267–270. [Google Scholar] [CrossRef]
- Ramgir, N.S.; Late, D.J.; Bhise, A.B.; More, M.A.; Mulla, I.S.; Joag, D.S.; Vijayamohanan, K.J. ZnO multipods, submicron wires, and spherical structures and their unique field emission behavior. Phys. Chem. 2006, 110, 18236–18242. [Google Scholar] [CrossRef]
- Briggs, D.; Seah, M.P. Practical Surface Analsysis: Auger and X-ray Photoelectron Spectroscopy; John Wiley & Sons: Chichester, UK, 1990. [Google Scholar]
- Castañeda, S.I.; Montero, I.; Ripalda, J.M.; Díaz, N.; Galín, L.; Rueda, F. X-ray photoelectron spectroscopy study of low-temperature molybdenum oxidation process. J. Appl. Phys. 1999, 85, 8415–8418. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chiang, T.H.; Yeh, H.C. The Synthesis of α-MoO3 by Ethylene Glycol. Materials 2013, 6, 4609-4625. https://doi.org/10.3390/ma6104609
Chiang TH, Yeh HC. The Synthesis of α-MoO3 by Ethylene Glycol. Materials. 2013; 6(10):4609-4625. https://doi.org/10.3390/ma6104609
Chicago/Turabian StyleChiang, Tzu Hsuan, and Hung Che Yeh. 2013. "The Synthesis of α-MoO3 by Ethylene Glycol" Materials 6, no. 10: 4609-4625. https://doi.org/10.3390/ma6104609





