Isopropyl Myristate-Modified Polyether-Urethane Coatings as Protective Barriers for Implantable Medical Devices
Abstract
:1. Introduction
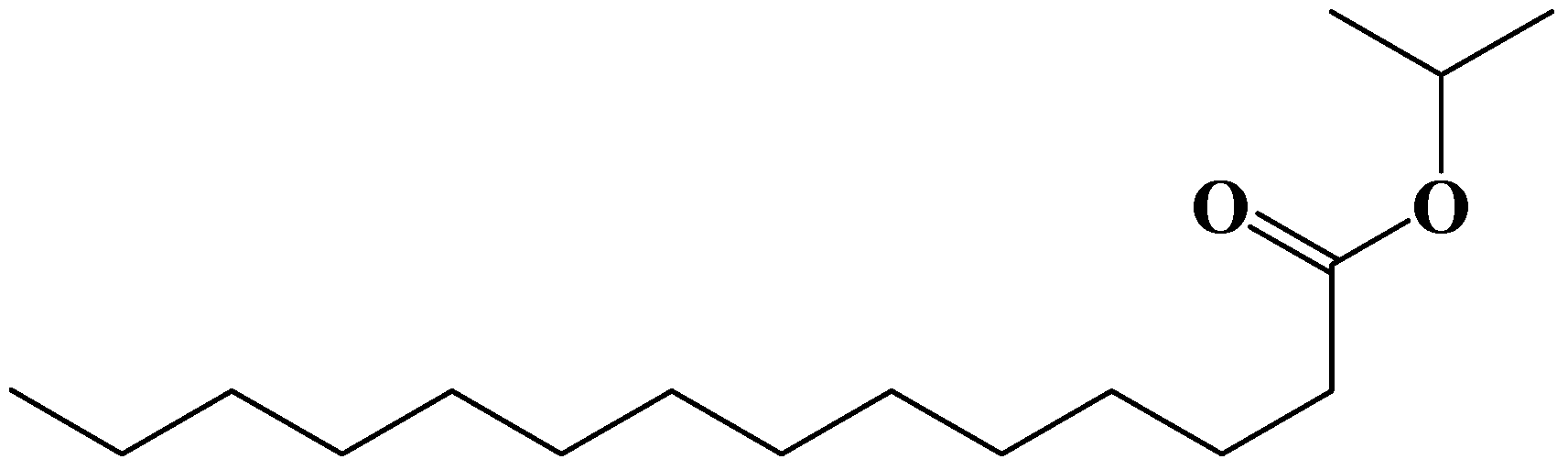
2. Experimental Section
2.1. Materials
2.2. Synthesis of polyurethane
2.3. Preparation of membranes
2.4. Characterisation
2.4.1. FTIR
2.4.2. Raman
2.4.3. Differential Scanning Calorimetry
2.4.4. Dynamic Mechanical Analysis
2.4.5. Mechanical Testing
2.4.6. Water contact angle
2.4.7. Water Uptake

3. Results and Discussion
| Sample ID | Hard segment in PEU (%) | PTMO (Mw) | IPM loading (Wt%) |
|---|---|---|---|
| PEU-1 | 35 | 1,000 | - |
| 0.5IPM-PEU-1 | 35 | 1,000 | 0.5 |
| 1IPM-PEU-1 | 35 | 1,000 | 1 |
| 2IPM-PEU-1 | 35 | 1,000 | 2 |
| 5IPM-PEU-1 | 35 | 1,000 | 5 |
| 10IPM-PEU-1 | 35 | 1,000 | 10 |
| PEU-2 | 25 | 2,000 | - |
| 0.5IPM-PEU-2 | 25 | 2,000 | 0.5 |
| 1IPM-PEU-2 | 25 | 2,000 | 1 |
| 2IPM-PEU-2 | 25 | 2,000 | 2 |
| 5IPM-PEU-2 | 25 | 2,000 | 5 |
| 10IPM-PEU-2 | 25 | 2,000 | 10 |
3.1. Spectroscopy analysis
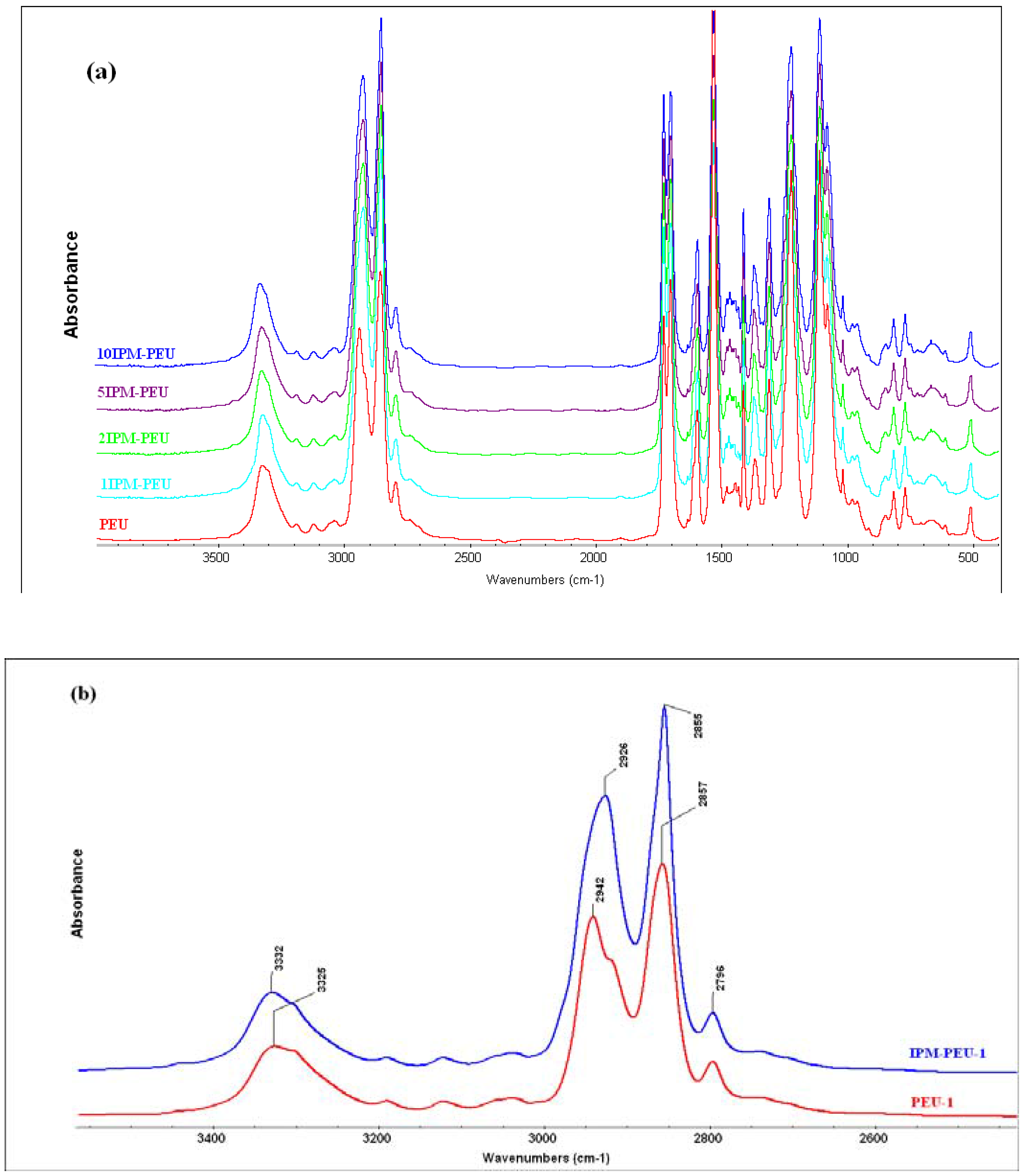
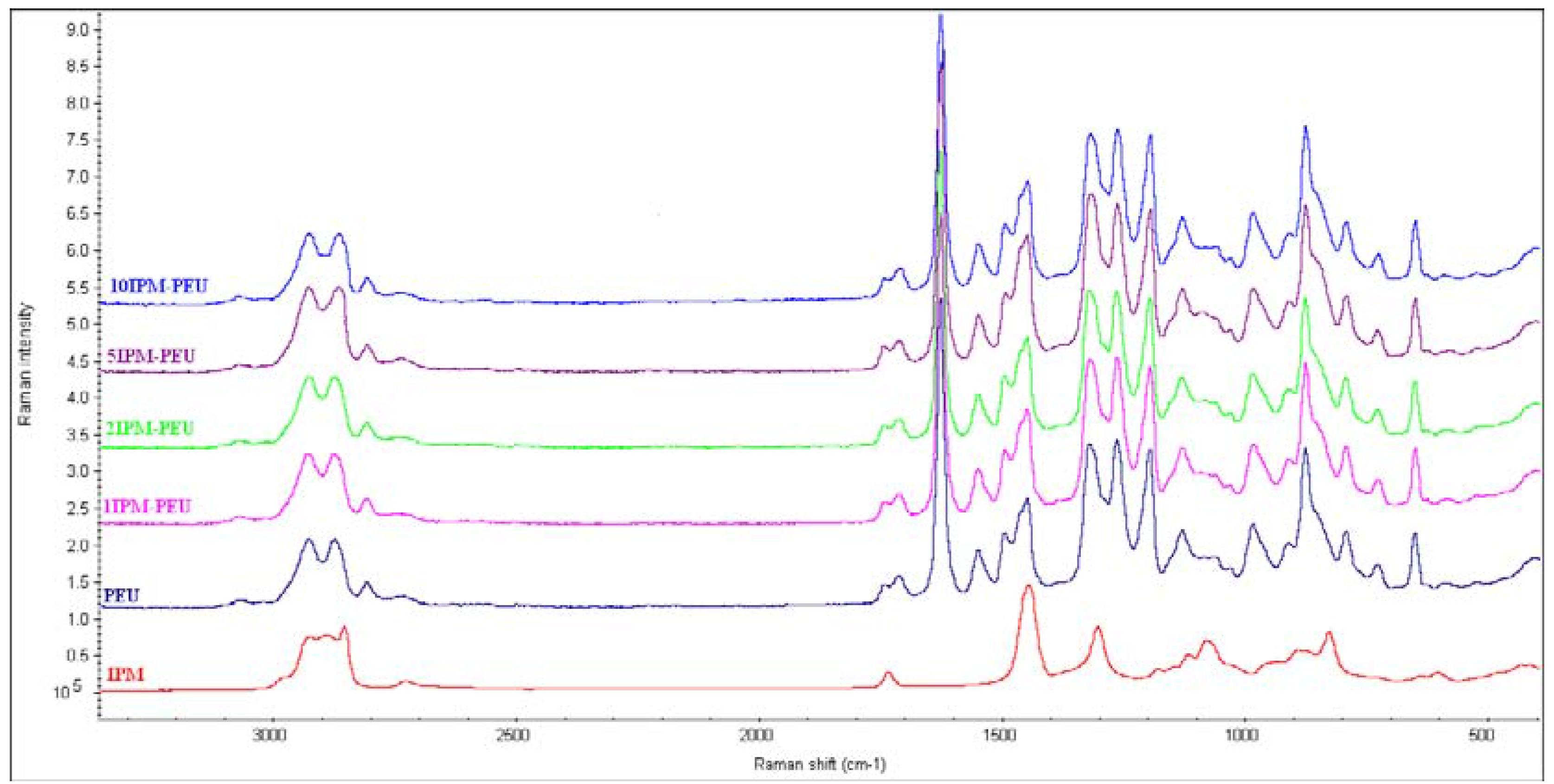
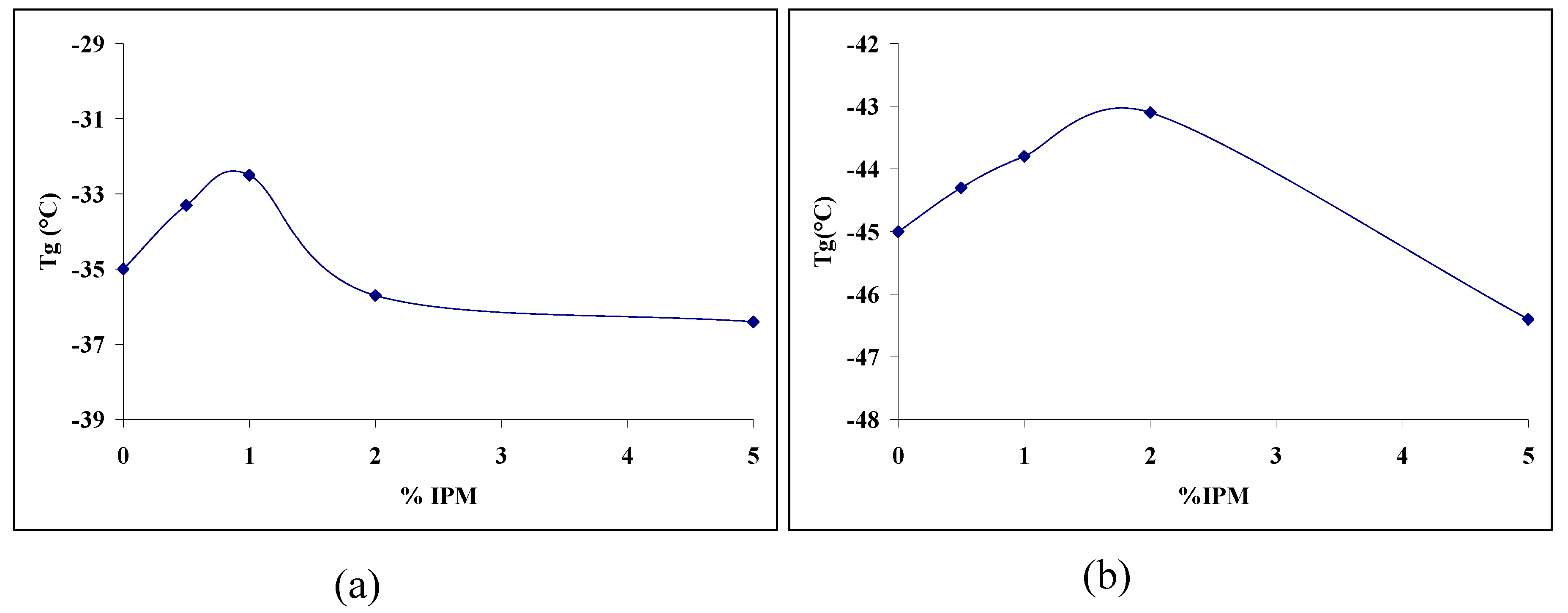
3.2. Thermal analysis
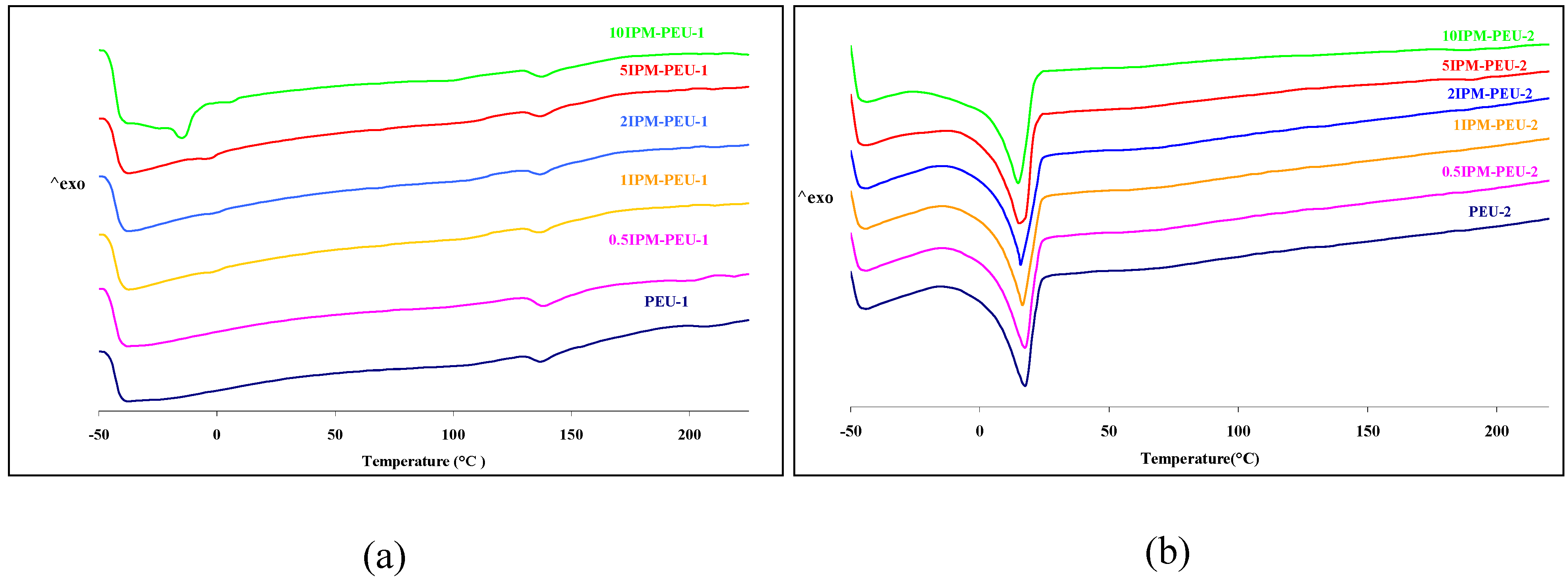
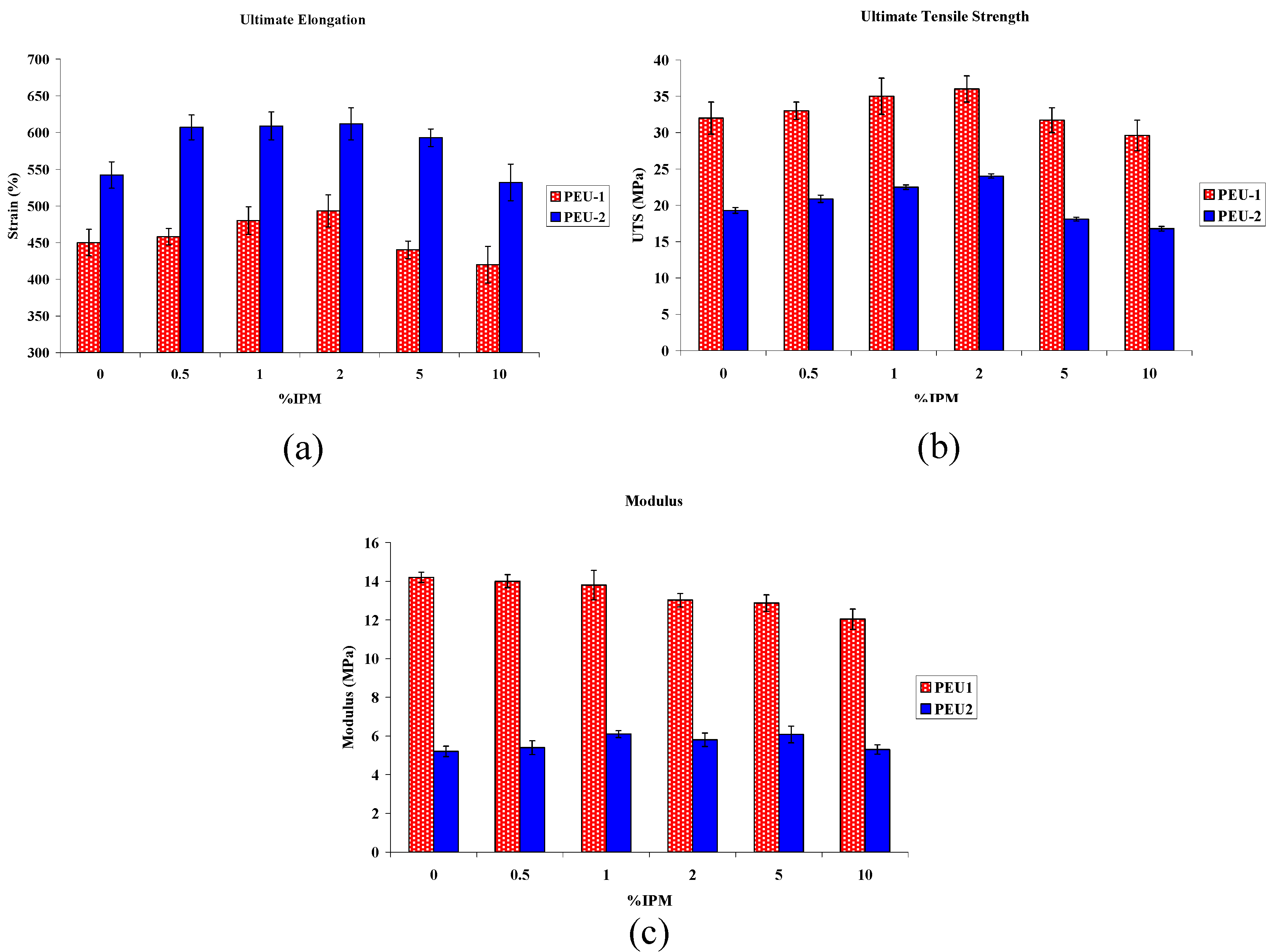
3.3. Mechanical Properties
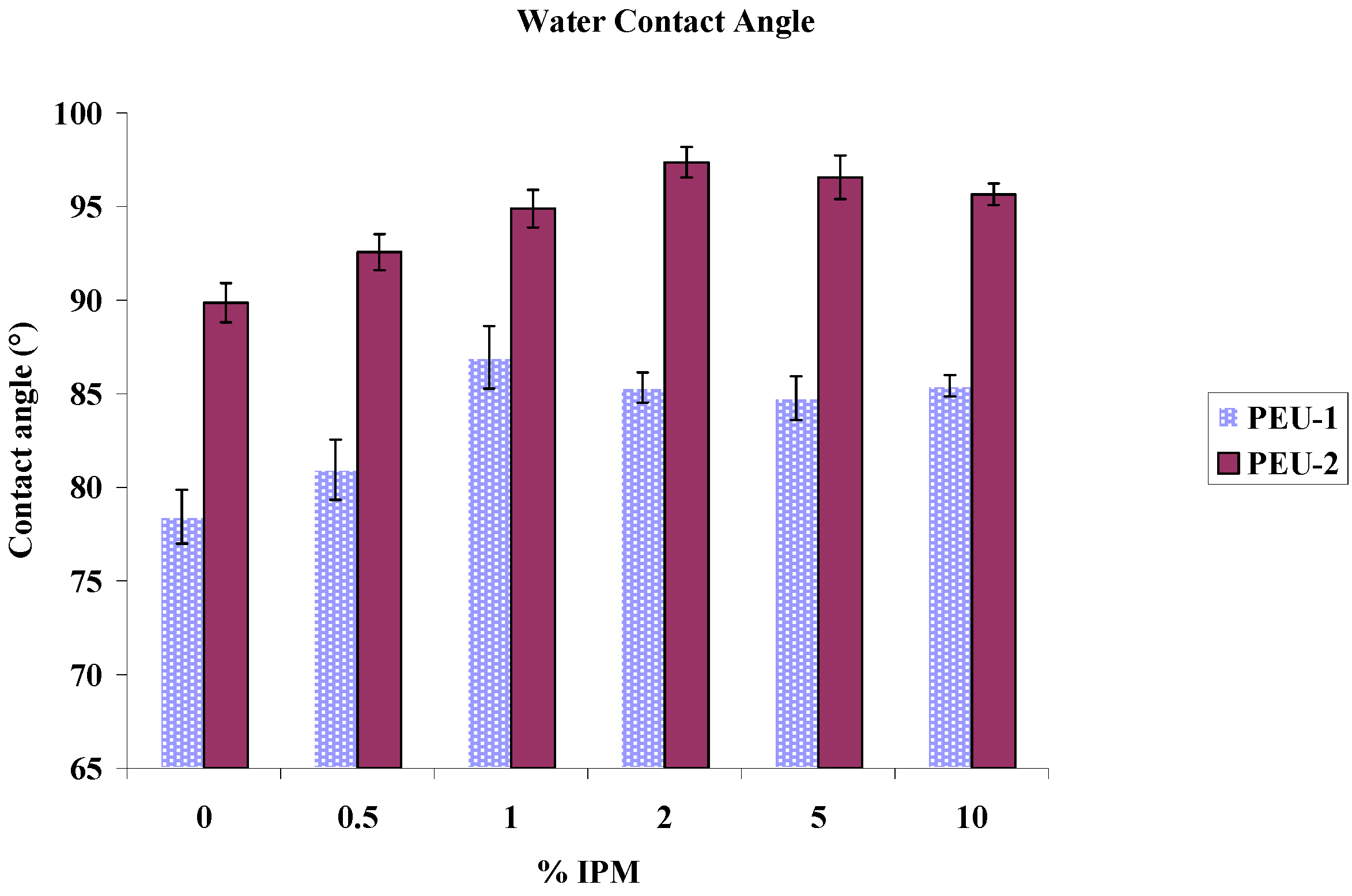
3.4. Contact Angle
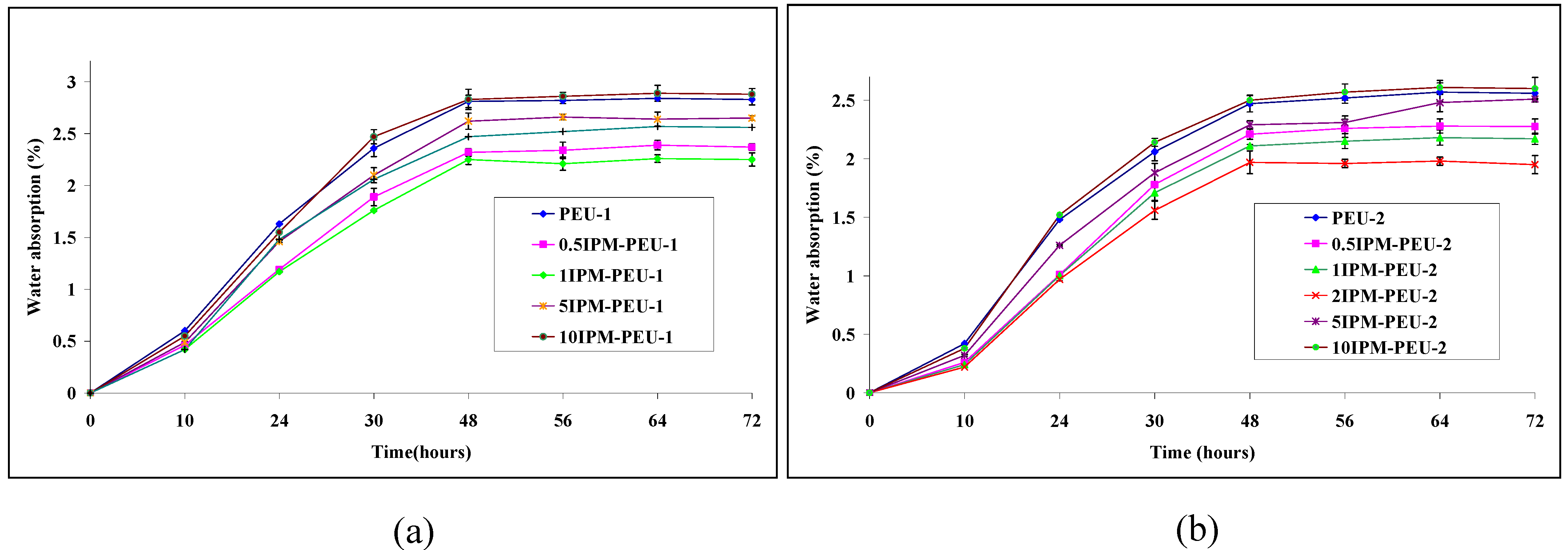
3.5. Water Uptake
4. Conclusions
Acknowledgements
References and Notes
- Hodgins, D.; Wasikiewicz, J.M.; Grahn, M.F.; Paul, D.; Roohpour, N.; Vadgama, P.; Silmon, A. M.; Cousins, B.; Verdon, B. Biocompatible materials development for new medical implants. Med.l Device Technol. 2007, 18, 32–35. [Google Scholar]
- Hierold, C.; Clasbrummel, B.; Behrend, D.; Scheiter, T.; Steger, M.; Oppermann, K.; Kapels, H.; Landgraf, E.; Wenzel, D.; Etzrodt, D. Low power integrated pressure sensor system for medical applications. Sensors Actuator. A-Phys. 1999, 73, 58–67. [Google Scholar] [CrossRef]
- Receveur, R.A.M.; Lindemans, F.W.; de Rooij, N.F. Microsystem technologies for implantable applications. J. Micromechanic. Microengineer. 2007, 17, R50–R80. [Google Scholar] [CrossRef]
- Dario, P.; Carrozza, M.C.; Benvenuto, A.; Menciassi, A. Micro-systems in biomedical applications. J. Micromechanic. Microengineer. 2000, 10, 235–244. [Google Scholar] [CrossRef]
- Donaldson, P.E.K. Encapsulating microelectronic implants in one-part silicone rubbers. Med. Biol. Eng. Comput. 1989, 27, 93–94. [Google Scholar] [CrossRef] [PubMed]
- Hodgins, D.; Bertsch, A.; Post, N.; Frischholz, M.; Volckaerts, B.; Spensley, J.; Wasikiewicz, J.M.; Higgins, H.; von Stetten, F.; Kenney, L. Healthy aims: Development new medical implants and diagnostic equipment. IEEE Pervasive Comput. 2008, 7, 14–21. [Google Scholar] [CrossRef]
- Wasikiewicz, J.M.; Roohpour, N.; Paul, D.; Grahn, M.; Ateh, D.; Rehman, I.; Vadgama, P. Polymeric barrier membranes for device packaging, diffusive control and biocompatibility. Appl. Surf. Sci. 2008, 255, 340–343. [Google Scholar] [CrossRef]
- Lamba, N.M.K.; Woodhouse, K.A.; Cooper, S. Polyurethane in Biomedical Application; CRC: Boca Raton, Florida, USA, 1997. [Google Scholar]
- Wright, J.I. Using Polyurethanes in Medical Applications. Med. Device Diagn. Ind. 2006, March, 98–109. [Google Scholar]
- Morimoto, N.; Iwasaki, Y.; Nakabayashi, N.; Ishihara, K. Physical properties and blood compatibility of surface-modified segmented polyurethane by semi-interpenetrating polymer networks with a phospholipid polymer. Biomaterials 2002, 23, 4881–4887. [Google Scholar] [CrossRef] [PubMed]
- Xu, R.J.; Manias, E.; Snyder, A.J.; Runt, J. Low penneability biomedical polyurethane nanocomposites. J. Biomed. Mater. Res. A 2003, 64A, 114–119. [Google Scholar] [CrossRef] [PubMed]
- Dube, N.; Al-Malaika, S.; Laroche, G.; Vermette, P. Additives in biomedical polyurethanes. In Biomedical Applications of Polyurethanes; Vermette, P., Griesser, H.J., Laroche, G., Guidoin, R., Eds.; Landes Bioscience: Georgetown, Texas, USA, 2001; Volume 6, pp. 55–76. [Google Scholar]
- Krishna, O.D.; Kim, K.; Byun, Y. Covalently grafted phospholipid monolayer on silicone catheter surface for reduction in platelet adhesion. Biomaterials 2005, 26, 7115–7123. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.D.; Luo, X.; Wang, X.F. Study on blends of thermoplastic polyurethane and aliphatic polyester: morphology, rheology, and properties as moisture vapor permeable films. Polym. Test. 2005, 24, 18–24. [Google Scholar] [CrossRef]
- Morimoto, N.; Watanabe, A.; Iwasaki, Y.; Akiyoshi, K.; Ishihara, K. Nano-scale surface modification of a segmented polyurethane with a phospholipid polymer. Biomaterials 2004, 25, 5353–5361. [Google Scholar] [CrossRef] [PubMed]
- Yoo, H.J.; Kim, H.D. Characteristics of crosslinked blends of Pellethene((R)) and multiblock polyurethanes containing phospholipid. Biomaterials 2005, 26, 2877–2886. [Google Scholar] [CrossRef] [PubMed]
- Reddy, S.M.; Vadgama, P.M. Surfactant-modified poly(vinyl chloride) membranes as biocompatible interfaces for amperometric enzyme electrodes. Anal. Chim. Acta 1997, 350, 77–89. [Google Scholar] [CrossRef]
- Pauliukaite, R.; Schoenleber, M.; Vadgama, P.; Brett, C.M.A. Development of electrochemical biosensors based on sol-gel enzyme encapsulation and protective polymer membranes. Anal. Bioanal. Chem. 2008, 390, 1121–1131. [Google Scholar] [CrossRef] [PubMed]
- Abraham, M.H.; Acree, W.E. Characterisation of the water-isopropyl myristate system. I. J. Pharm. 2005, 294, 121–128. [Google Scholar]
- Gupta, S.; Moulik, S.P. Biocompatible microemulsions and their prospective uses in drug delivery. J. Pharm. Sci. 2008, 97, 22–45. [Google Scholar] [CrossRef] [PubMed]
- Bremner, T.; Hill, D.J.T.; Killeen, M.I.; Odonnell, J.H.; Pomery, P.J.; StJohn, D.; Whittaker, A.K. Development of wear-resistant thermoplastic polyurethanes by blending with poly(dimethyl siloxane) .2. A packing model. J. Appl. Polym. Sci. 1997, 65, 939–950. [Google Scholar] [CrossRef]
- Stetzler, R.S.; Smullin, C.F. Determination of hydroxyl number of polyoxyalkylene ethers by acid-catalyzed acetylation. Anal. Chem. 1962, 34, 194–195. [Google Scholar] [CrossRef]
- McCarthy, S.J.; Meijs, G.F.; Mitchell, N.; Gunatillake, P.A.; Heath, G.; Brandwood, A.; Schindhelm, K. In-vivo degradation of polyurethanes: transmission-FTIR microscopic characterization of polyurethanes sectioned by cryomicrotomy. Biomaterials 1997, 18, 1387–1409. [Google Scholar] [CrossRef] [PubMed]
- Bai, C.Y.; Zhang, X.Y.; Dai, J.B.; Zhang, C.Y. Water resistance of the membranes for UV curable waterborne polyurethane dispersions. Prog. Org. Coating. 2007, 59, 331–336. [Google Scholar] [CrossRef]
- Garton, A. Inferared Spectroscopy of Polymer Blends, Composites and Surfcaes; Hanser Publisher: New York, NY, USA, 1992. [Google Scholar]
- Movasaghi, Z.; Rehman, S.; Rehman, I.U. Fourier transform infrared (FTIR) spectroscopy of biological tissues. Appl. Spectrosc. Rev. 2008, 43, 134–179. [Google Scholar] [CrossRef]
- Movasaghi, Z.; Rehman, S.; Rehman, I.U. Raman spectroscopy of biological tissues. Appl. Spectrosc. Rev. 2007, 42, 493–541. [Google Scholar] [CrossRef]
- Teo, L.S.; Chen, C.Y.; Kuo, J.F. Fourier transform infrared spectroscopy study on effects of temperature on hydrogen bonding in amine-containing polyurethanes and poly(urethane-urea)s. Macromolecules 1997, 30, 1793–1799. [Google Scholar] [CrossRef]
- Mattia, J.; Painter, P. A comparison of hydrogen bonding and order in a polyurethane and poly(urethane-urea) and their blends with poly(ethylene glycol). Macromolecules 2007, 40, 1546–1554. [Google Scholar] [CrossRef]
- Koberstein, J.T.; Galambos, A.F. Multiple melting in segmented polyurethane block copolymers. Macromolecules 1992, 25, 5618–5624. [Google Scholar] [CrossRef]
- Ambrose, R.J. Thermoplastic Polymer Additives - Theory and Practice; Wiley: New York, NY, USA, 1990; Vol. 28, p. 544. [Google Scholar]
- Eddy, S.; Warriner, K.; Christie, I.; Ashworth, D.; Purkiss, C.; Vadgama, P. The modification of enzyme electrode properties with nonconducting electropolymerized films. Biosens. Bioelectron. 1995, 10, 831–839. [Google Scholar] [CrossRef]
- Vadgama, P.; Ahmed, S. Modified polyurethane membrane sensors and analytical methods. US Patent 6200772B1.
- Lin, Y.-H.; Chou, N.-K.; Chen, K.-F.; Ho, G.-H.; Chang, C.-H.; Wang, S.-S.; Chu, S.-H.; Hsieh, K.-H. Effect of soft segment length on properties of hydrophilic/hydrophobic polyurethanes. Polym. Int. 2007, 56, 1415–1422. [Google Scholar] [CrossRef]
- Hill, D.J.T.; Killeen, M.I.; Odonnell, J.H.; Pomery, P.J.; StJohn, D.; Whittaker, A.K. Development of wear-resistant thermoplastic polyurethanes by blending with poly(dimethyl siloxane). 1. Physical properties. J. Appl. Polym. Sci. 1996, 61, 1757–1766. [Google Scholar] [CrossRef]
- Mascia, L. Thermoplastics:Materials Engineering; Elsevier Science: New York, NY, USA, 1989. [Google Scholar]
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Roohpour, N.; Wasikiewicz, J.M.; Moshaverinia, A.; Paul, D.; Rehman, I.U.; Vadgama, P. Isopropyl Myristate-Modified Polyether-Urethane Coatings as Protective Barriers for Implantable Medical Devices. Materials 2009, 2, 719-733. https://doi.org/10.3390/ma2030719
Roohpour N, Wasikiewicz JM, Moshaverinia A, Paul D, Rehman IU, Vadgama P. Isopropyl Myristate-Modified Polyether-Urethane Coatings as Protective Barriers for Implantable Medical Devices. Materials. 2009; 2(3):719-733. https://doi.org/10.3390/ma2030719
Chicago/Turabian StyleRoohpour, Nima, Jaroslaw M. Wasikiewicz, Alireza Moshaverinia, Deepen Paul, Ihtesham U. Rehman, and Pankaj Vadgama. 2009. "Isopropyl Myristate-Modified Polyether-Urethane Coatings as Protective Barriers for Implantable Medical Devices" Materials 2, no. 3: 719-733. https://doi.org/10.3390/ma2030719





