In Vitro Effectiveness of Microspheres Based on Silk Sericin and Chlorella vulgaris or Arthrospira platensis for Wound Healing Applications
Abstract
:1. Introduction
2. Methods
2.1. Materials
2.2. Preparation of Microalgae Extracts
2.3. Extraction of Silk Sericin
2.4. Microsphere Preparation by Spray Drying
2.5. Microsphere Characterization
2.5.1. Granulometric Analysis
2.5.2. Scanning Electron Microscopy (SEM)
2.5.3. Fourier Transform Infrared Spectroscopy (FTIR)
2.5.4. Simultaneous Thermogravimetric Analysis (TGA/DSC 1)
2.5.5. Determination of Protein Content
2.5.6. Determination of Carbohydrate Content
2.5.7. ROS-Scavenging Activity Assay
2.6. In Vitro Assays
2.6.1. Cytotoxicity Assay
2.6.2. Scratch Assay
2.6.3. Statistical Analysis
3. Results
3.1. Microsphere Characterization
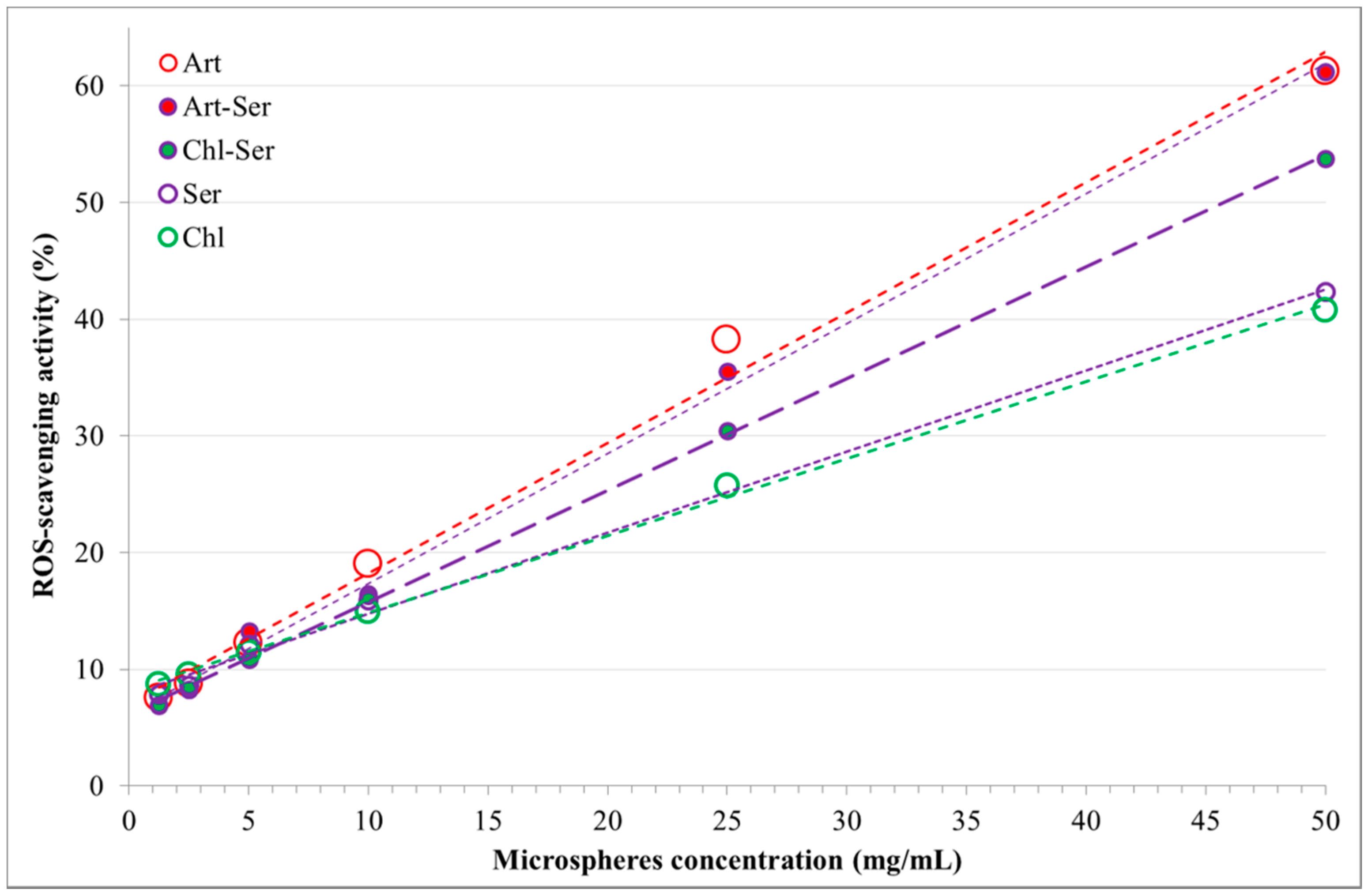
3.2. ROS-Scavenging Activity
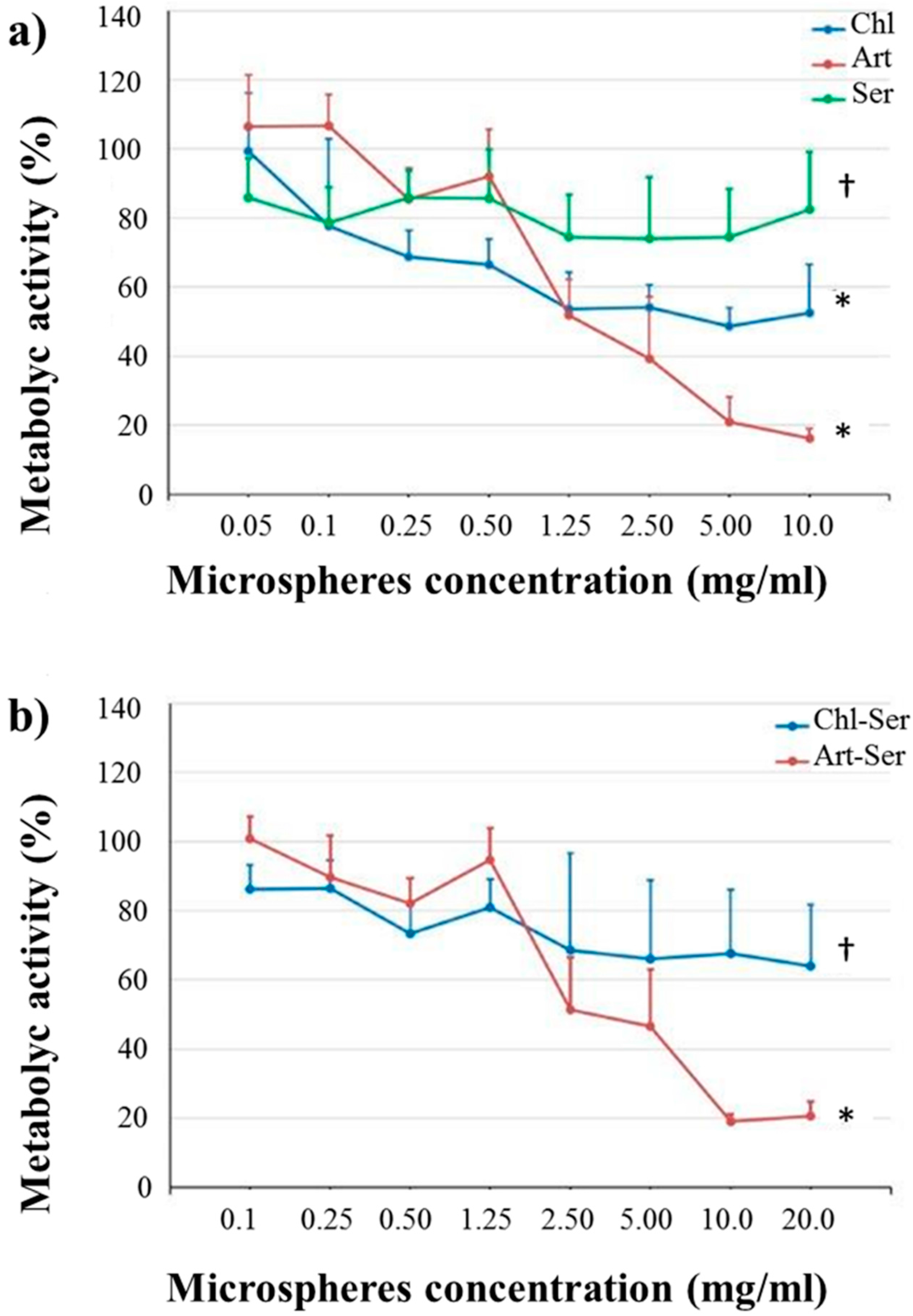
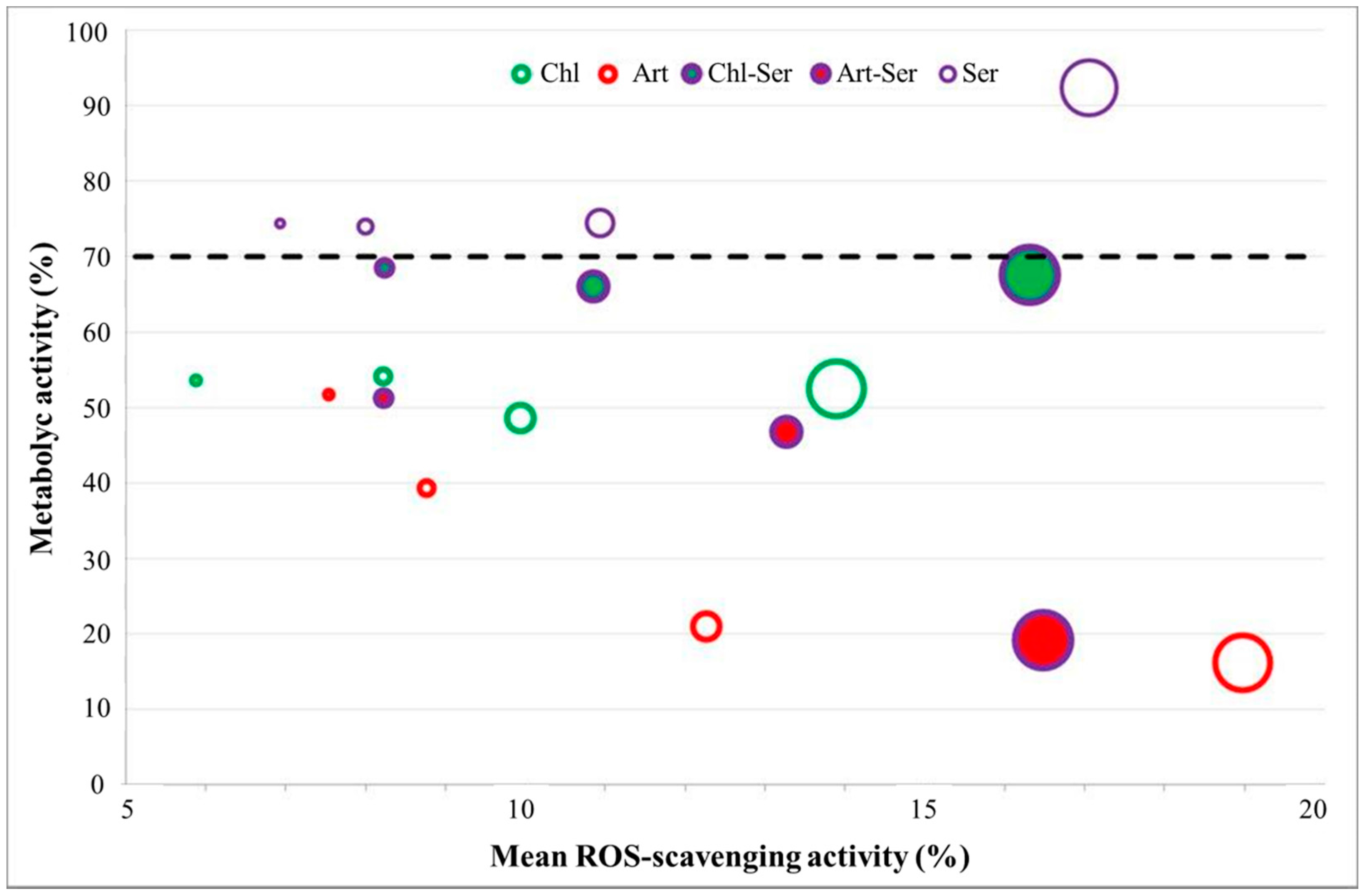
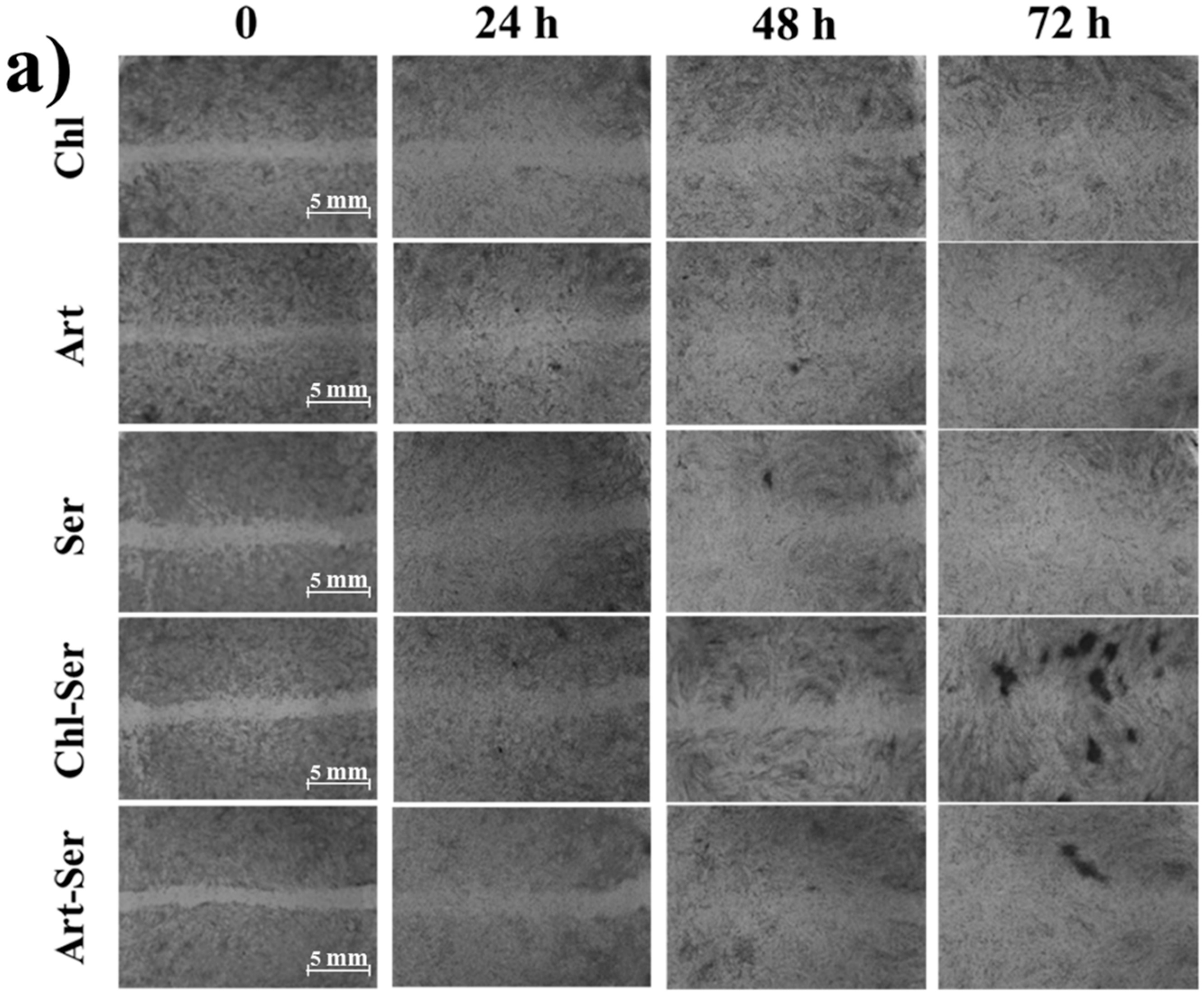
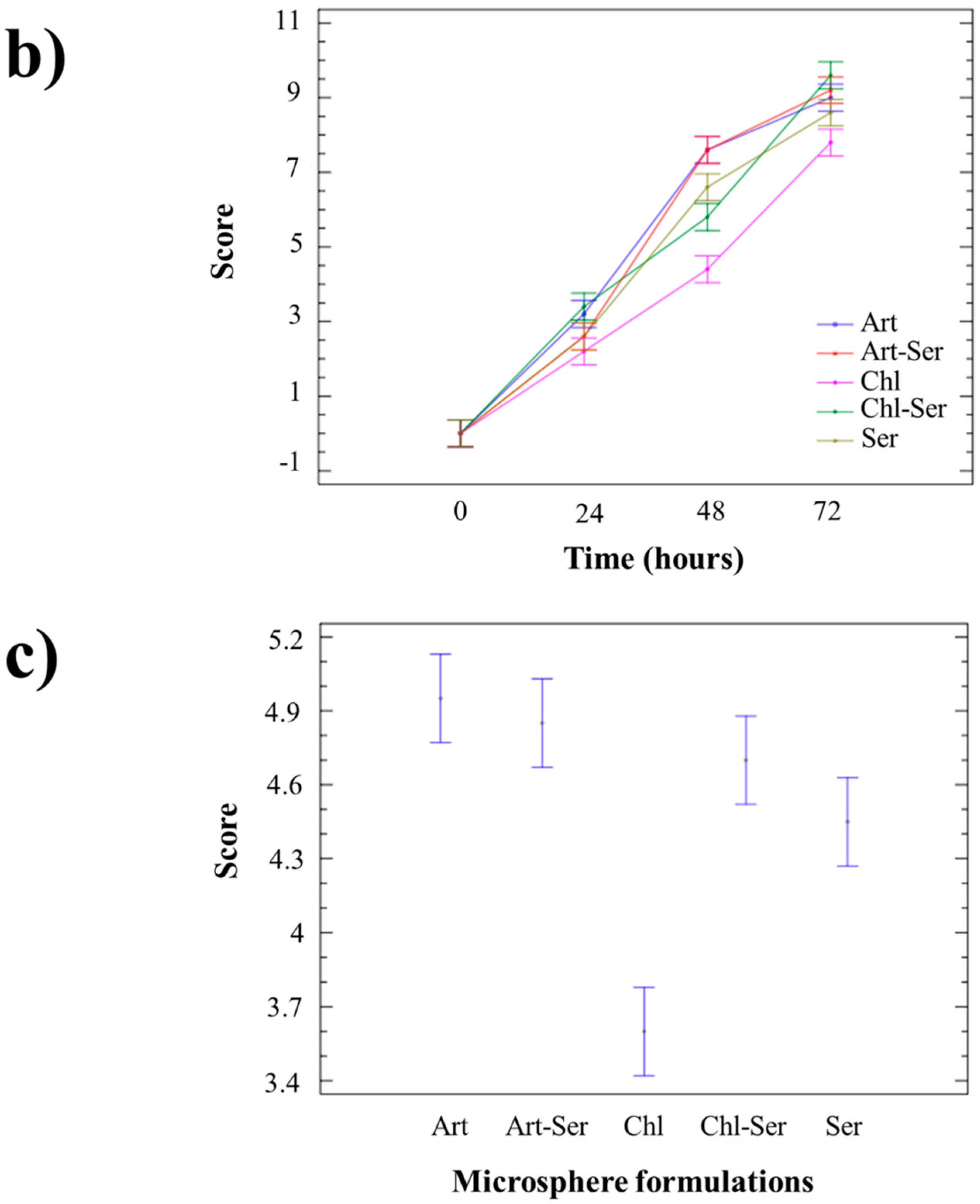
3.3. In Vitro Results
4. Discussion
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ANOVA | Analysis of variance |
| BCA | Bicinchoninic acid |
| BSA | Bovine serum albumin |
| DMSO | Dimethyl sulfoxide |
| DPPH | 2,2-Diphenyl-2-picrylhydrazyl hydrate |
| FTIR | Fourier transform infrared spectroscopy |
| GRAS | Generally recognized as safe |
| MTT | 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide |
| ROS | Reactive oxygen species |
| SEM | Scanning electron microscopy |
| TGA | Thermogravimetric Analysis |
References
- Gil-Chàvez, G.J.; Villa, J.A.; Ayala-Zavala, F.; Heredia, B.; Sepulveda, D.; Yahia, E.M.; González-Aguilar, G.A. Technologies for extraction and production of bioactive compounds to be used as nutraceuticals and food ingredients: An overview. Compr. Rev. Food Sci. Food. Saf. 2013, 12, 5–23. [Google Scholar] [CrossRef]
- Wang, H.M.; Chen, C.C.; Huynh, P.; Chang, J.S. Exploring the potential of using algae in cosmetics. Bioresour. Technol. 2015, 184, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Markou, G.; Nerantzis, E. Microalgae for high-value compounds and biofuels production: A review with focus on cultivation under stress conditions. Biotechnol. Adv. 2013, 31, 1532–1542. [Google Scholar] [CrossRef] [PubMed]
- De Morais, M.G.; Vaz Bda, S.; de Morais, E.G.; Costa, J.A. Biologically active metabolites synthesized by microalgae. BioMed Res. Int. 2015, 835761. [Google Scholar] [CrossRef] [PubMed]
- Plaza, M.; Santoyo, S.; Jaime, L.; Reina, G.G.B.; Herrero, M.; Señoráns, F.J.; Ibáñez, E. Screening for bioactive compounds from algae. J. Pharmac. Biomed. Anal. 2010, 51, 450–455. [Google Scholar] [CrossRef] [PubMed]
- Vàzquez, A.I.F.; Sànchez, C.M.D.; Delgado, N.G.; Alfonso, A.M.S.; Ortega, Y.S.; Sanchez, H.C. Anti-inflammatory and analgesic activities of red seaweed Dichotomaria obtusata. Braz. J. Pharm. Sci. 2011, 47, 111–118. [Google Scholar] [CrossRef]
- Al-Saif, S.S.A.; Abdel-Raouf, N.; El-Wazanani, H.A.; Aref, I.A. Antibacterial substances from marine algae isolated from Jeddah coast of Red sea, Saudi Arabia. Saudi J. Biol. Sci. 2014, 21, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Michalak, I.; Chojnacka, K. Algae as production systems of bioactive compounds. Eng. Life Sci. 2015, 15, 160–176. [Google Scholar] [CrossRef]
- Masojìdek, J.; Pràšil, O. The development of microalgal biotechnology in the Czech Republic. J. Ind. Microbiol. Biotechnol. 2010, 37, 1307–1317. [Google Scholar] [CrossRef] [PubMed]
- Demir, B.S.; Tükel, S.S. Purification and characterization of lipase from Spirulina platensis. J. Mol. Catal. B Enzym. 2010, 64, 123–128. [Google Scholar] [CrossRef]
- Cuellar-Bermudez, S.P.; Aguilar-Hernandez, I.; Cardenas-Chavez, D.L.; Ornelas-Soto, N.; Romero-Ogawa, M.A.; Parra-Saldivar, R. Extraction and purification of high-value metabolites from microalgae: Essential lipids, astaxanthin and phycobiliproteins. Microb. Biotechnol. 2015, 8, 190–209. [Google Scholar] [CrossRef] [PubMed]
- Kulshreshtha, A.; Zacharia, A.J.; Jarouliya, U.; Bhadauriya, P.; Prasad, G.B.; Bisen, P.S. Spirulina in health care management. Curr. Pharm. Biotechnol. 2008, 9, 400–405. [Google Scholar] [CrossRef] [PubMed]
- Small, E. Spirulina-food for the universe. Biodiversity 2011, 12, 255–265. [Google Scholar] [CrossRef]
- Deng, R.; Chow, T.J. Hypolipidemic, antioxidant, and anti-inflammatory activities of microalgae Spirulina. Cardiovasc. Ther. 2010, 28, 33–45. [Google Scholar] [CrossRef] [PubMed]
- Safi, C.; Zebib, B.; Merah, O.; Pontalier, P.; Vaca-Garcia, C. Morphology, composition, production, processing and applications of Chlorella vulgaris: A review. Renew. Sustain. Energy Rev. 2014, 35, 265–278. [Google Scholar] [CrossRef]
- Wang, H.; Pan, J.; Chen, C.; Chiu, C.; Yang, M.; Chang, H.; Chang, J. Identification of anti-lung cancer extract from Chlorella vulgaris C-C by antioxidant property using supercritical carbon dioxide extraction. Process Biochem. 2010, 45, 1865–1872. [Google Scholar] [CrossRef]
- Li, H.B.; Jiang, Y.; Chen, F. Isolation and purification of lutein from the microalga Chlorella vulgaris by extraction after saponification. J. Agric. Food Chem. 2002, 50, 1070–1072. [Google Scholar] [CrossRef] [PubMed]
- Cao, T.T.; Zhang, Y.Q. Processing and characterization of silk sericin from Bombyx mori and its application in biomaterials and biomedicine. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 61, 940–952. [Google Scholar] [CrossRef] [PubMed]
- Mori, M.; Rossi, S.; Ferrari, F.; Bonferoni, M.C.; Sandri, G.; Chlapanidas, T.; Torre, M.L.; Caramella, C. Sponge-like dressings based on the association of chitosan and sericin for the treatment of chronic skin ulcers. I. Design of experiments—Assisted development. J. Pharm. Sci. 2016, 105, 1180–1187. [Google Scholar] [CrossRef] [PubMed]
- Mori, M.; Rossi, S.; Ferrari, F.; Bonferoni, M.C.; Sandri, G.; Riva, F.; Tenci, M.; Del Fante, C.; Nicoletti, G.; Caramella, C. Sponge-like dressings based on the association of chitosan and sericin for the treatment of chronic skin ulcers. II. Loading of the hemoderivative platelet lysate. J. Pharm. Sci. 2016, 105, 1188–1195. [Google Scholar] [CrossRef] [PubMed]
- Fan, J.; Wu, L.; Chen, L.; Mao, X.; Ren, F. Antioxidant activities of silk sericin from silkworm Bombyx mori. J. Food Biochem. 2009, 33, 74–88. [Google Scholar] [CrossRef]
- Chlapanidas, T.; Faragò, S.; Lucconi, G.; Perteghella, S.; Galuzzi, M.; Mantelli, M.; Avanzini, M.A.; Tosca, M.C.; Marazzi, M.; Vigo, D.; et al. Sericins exhibit ROS-scavenging, anti-tyrosinase, anti-elastase, and in vitro immunomodulatory activities. Int. J. Biol. Macromol. 2013, 58, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Tsubouchi, K.; Igarashi, Y.; Takasu, Y.; Yamada, H. Sericin enhances attachment of cultured human skin fibroblasts. Biosci. Biotechnol. Biochem. 2005, 69, 403–405. [Google Scholar] [CrossRef] [PubMed]
- Chlapanidas, T.; Perteghella, S.; Leoni, F.; Faragò, S.; Marazzi, M.; Rossi, D.; Martino, E.; Gaggeri, R.; Collina, S. TNF-α blocker effect of naringenin-loaded sericin microparticles that are potentially useful in the treatment of psoriasis. Int. J. Mol. Sci. 2014, 15, 13624–13636. [Google Scholar] [CrossRef] [PubMed]
- Kuda, T.; Eda, M.; Kataoka, M.; Nemoto, M.; Kawahara, M.; Oshio, S.; Takahashi, H.; Kimura, B. Anti-glycation properties of the aqueous extract solutions of dried algae products and effect of lactic acid fermentation on the properties. Food Chem. 2016, 192, 1109–1115. [Google Scholar] [CrossRef] [PubMed]
- Faragò, S.; Lucconi, G.; Perteghella, S.; Vigani, B.; Tripodo, G.; Sorrenti, M.; Catenacci, L.; Boschi, A.; Faustini, M.; Vigo, D.; et al. A dry powder formulation from silk fibroin microspheres as a topical auto-gelling device. Pharm. Dev. Technol. 2016, 21, 453–462. [Google Scholar] [CrossRef] [PubMed]
- Lucconi, G.; Chlapanidas, T.; Martino, E.; Gaggeri, R.; Perteghella, S.; Rossi, D.; Faragò, S.; Vigo, D.; Faustini, M.; Collina, S.; et al. Formulation of microspheres containing Crataegus monogyna Jacq. extract with free radical scavenging activity. Pharm. Dev. Technol. 2013, 19, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Smith, P.K.; Krohn, R.I.; Hermanson, G.T.; Mallia, A.K.; Gartner, F.H.; Provenzano, M.D.; Fujimoto, E.K.; Goeke, N.M.; Olson, B.J.; Klenk, D.C. Measurement of protein using bicinchoninic acid. Anal. Biochem. 1985, 150, 76–85. [Google Scholar] [CrossRef]
- DuBois, M.; Gilles, K.; Hamilton, J.; Rebers, P.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Chaudhuri, D.; Ghate, N.B.; Deb, S.; Panja, S.; Sarkar, R.; Rout, J.; Mandal, N. Assessment of the phytochemical constituents and antioxidant activity of a bloom forming microalgae Euglena tuba. Biol. Res. 2014, 47, 24. [Google Scholar] [CrossRef] [PubMed]
- Liang, C.C.; Park, A.Y.; Guan, J.L. In vitro scratch assay: A convenient and inexpensive method for analysis of cell migration in vitro. Nat. Protoc. 2007, 2, 329–333. [Google Scholar] [CrossRef] [PubMed]
- Chlapanidas, T.; Perteghella, S.; Faragò, S.; Boschi, A.; Tripodo, G.; Vigani, B.; Crivelli, B.; Renzi, S.; Dotti, S.; Preda, S.; et al. Platelet lysate and adipose mesenchymal stromal cells on silk fibroin nonwoven mats for wound healing. J. Appl. Polym. Sci. 2016, 133. [Google Scholar] [CrossRef]
- De Morais, M.G.; Stillings, C.; Dersch, R.; Rudisile, M.; Pranke, P.; Costa, J.A.V.; Wendorff, J. Preparation of nanofibers containing the microalga Spirulina (Arthrospira). Bioresour. Technol. 2010, 101, 2872–2876. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Shin, C.; Min, S.K.; Jung, S-M.; Shim, H.S. In vitro evaluation of the effects of electrospun PCL nanofiber mats containing the microalgae Spirulina (Arthrospira) extract on primary astrocytes. Colloids Surf. B Biointerfaces 2012, 90, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Cha, B.-G.; Kwan, H.W.; Park, A.R.; Kim, S.H.; Park, S.-Y.; Kim, H.-J.; Kim, I.-S.; Lee, K.H.; Park, Y.H. Structural characteristics and biological performance of silk fibroin nanofiber containing microalgae Spirulina extract. Byopolymers 2014, 101, 307–318. [Google Scholar] [CrossRef] [PubMed]
- Oh, H.; Kim, M.K.; Lee, K.H. Preparation of sericin microparticles by electrohydrodynamic spraying and their application in drug delivery. Macromol. Res. 2011, 19, 266–272. [Google Scholar] [CrossRef]
- Aramwit, P.; Palapinyo, S.; Srichana, T.; Chottanapund, S.; Muangman, P. Silk sericin ameliorates wound healing and its clinical efficacy in burn wounds. Arch. Dermatol. Res. 2013, 305, 585–594. [Google Scholar] [CrossRef] [PubMed]
- Syarina, P.N.; Karthivashan, G.; Abas, F.; Arulselvan, P.; Fakurazi, S. Wound healing potential of Spirulina platensis extracts on human dermal fibroblast cells. EXCLI J. 2015, 14, 385–393. [Google Scholar] [CrossRef] [PubMed]
- Zailan, N.; Abdul Rashid, A.H.; Das, S.; Abdul Mokti, N.A.; Hassan Basri, J.; Teoh, S.L.; Wan Ngah, W.Z.; Mohd Yusof, Y.A. Comparison of Chlorella vulgaris dressing and sodium alginate dressing: An experimental study in rats. Clin. Ther. 2010, 161, 515–521. [Google Scholar]
- Napavichayanun, S.; Aramwit, P. Effect of animal products and extracts on wound healing promotion in topical applications: A review. J. Biomater. Sci. Polym. Ed. 2017, 28, 703–729. [Google Scholar] [CrossRef] [PubMed]
- Degim, Z. Use of microparticulate systems to accelerate skin wound healing. J. Drug Target. 2008, 16, 437–448. [Google Scholar] [CrossRef] [PubMed]
- Dehnad, D.; Jafari, S.M.; Afrasiabi, M. Influence of drying on functional properties of food biopolymers: From traditional to novel dehydration techniques. Trends Food Sci. Technol. 2016, 57, 116–131. [Google Scholar] [CrossRef]
- Leach, G.; Oliveira, G.; Morais, R. Spray-drying of Dunaliella salina to produce a β-carotene rich powder. J. Ind. Microbiol. Biotechnol. 1998, 20, 82–85. [Google Scholar] [CrossRef]
- Orset, S.; Leach, G.C.; Morais, R.; Young, A.J. Spray-drying of the microalga Dunaliella salina: Effects on β-carotene content and isomer composition. J. Agric. Food Chem. 1999, 47, 4782–4790. [Google Scholar] [CrossRef] [PubMed]
- Mishara, M.; Mishara, B. Formulation optimization and characterization of spray dried microparticles for inhalation delivery of doxycycline hyclate. Yakugaku Zasshi 2011, 113, 1813–1825. [Google Scholar] [CrossRef]
- Raula, J.; Eerikäinen, H.; Kauppinen, E.I. Influence of the solvent composition on the aerosol synthesis of pharmaceutical polymer nanoparticles. Int. J. Pharm. 2004, 284, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Genç, G.; Narin, G.; Bayrakcar, O. Spray drying as a method of producing silk sericin powders. J. Achiev. Mater. Manuf. Eng. 2009, 37, 78–86. [Google Scholar]
- Dunnill, C.; Patton, T.; Brennan, J.; Barrett, J.; Dryden, M.; Cooke, J.; Leaper, D.; Georgopoulos, N.T. Reactive oxygen species (ROS) and wound healing: The functional role of ROS and emerging ROS-modulating technologies for augmentation of the healing process. Int. J. Wound 2017, 14, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Safafar, H.; van Wagenen, J.; Møller, P.; Jacobsen, C. Carotenoids, phenolic compounds and tocopherols contribute to the antioxidative properties of some microalgae species grown on industrial wastewater. Mar. Drugs 2015, 13, 7339–7356. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, L.C.; Ho, J.A.; Shieh, M.C.; Lu, I.W. Antioxidant and antiproliferative activities of Spirulina and Chlorella water extracts. J. Agric. Food Chem. 2005, 53, 4207–4212. [Google Scholar] [CrossRef] [PubMed]


| Formulation | A. platensis Extract | C. vulgaris Extract | Silk Sericin |
|---|---|---|---|
| Chl | 0 | 100 | 0 |
| Art | 100 | 0 | 0 |
| Ser | 0 | 0 | 100 |
| Chl-Ser | 0 | 50 | 50 |
| Art-Ser | 50 | 0 | 50 |
| Formulation | Protein Content (μg Proteins/mg Microspheres) | Carbohydrate Content (μg Carbohydrates/mg Microspheres) |
|---|---|---|
| Chl | 162.74 ± 2.11 a | 97.45 ± 3.29 a |
| Art | 217.41 ± 21.26 b | 100.50 ± 6.67 a |
| caSer | 1104.89 ± 46.07 c | 17.14 ± 0.05 b |
| Chl-Ser | 640.94 ± 8.02 d | 46.48 ± 0.10 c |
| Art-Ser | 714.35 ± 21.95 e | 36.53 ± 3.45 d |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bari, E.; Arciola, C.R.; Vigani, B.; Crivelli, B.; Moro, P.; Marrubini, G.; Sorrenti, M.; Catenacci, L.; Bruni, G.; Chlapanidas, T.; et al. In Vitro Effectiveness of Microspheres Based on Silk Sericin and Chlorella vulgaris or Arthrospira platensis for Wound Healing Applications. Materials 2017, 10, 983. https://doi.org/10.3390/ma10090983
Bari E, Arciola CR, Vigani B, Crivelli B, Moro P, Marrubini G, Sorrenti M, Catenacci L, Bruni G, Chlapanidas T, et al. In Vitro Effectiveness of Microspheres Based on Silk Sericin and Chlorella vulgaris or Arthrospira platensis for Wound Healing Applications. Materials. 2017; 10(9):983. https://doi.org/10.3390/ma10090983
Chicago/Turabian StyleBari, Elia, Carla Renata Arciola, Barbara Vigani, Barbara Crivelli, Paola Moro, Giorgio Marrubini, Milena Sorrenti, Laura Catenacci, Giovanna Bruni, Theodora Chlapanidas, and et al. 2017. "In Vitro Effectiveness of Microspheres Based on Silk Sericin and Chlorella vulgaris or Arthrospira platensis for Wound Healing Applications" Materials 10, no. 9: 983. https://doi.org/10.3390/ma10090983













