The Efficacy of Electron Beam Irradiated Bacterial Cellulose Membranes as Compared with Collagen Membranes on Guided Bone Regeneration in Peri-Implant Bone Defects
Abstract
:1. Introduction
2. Results
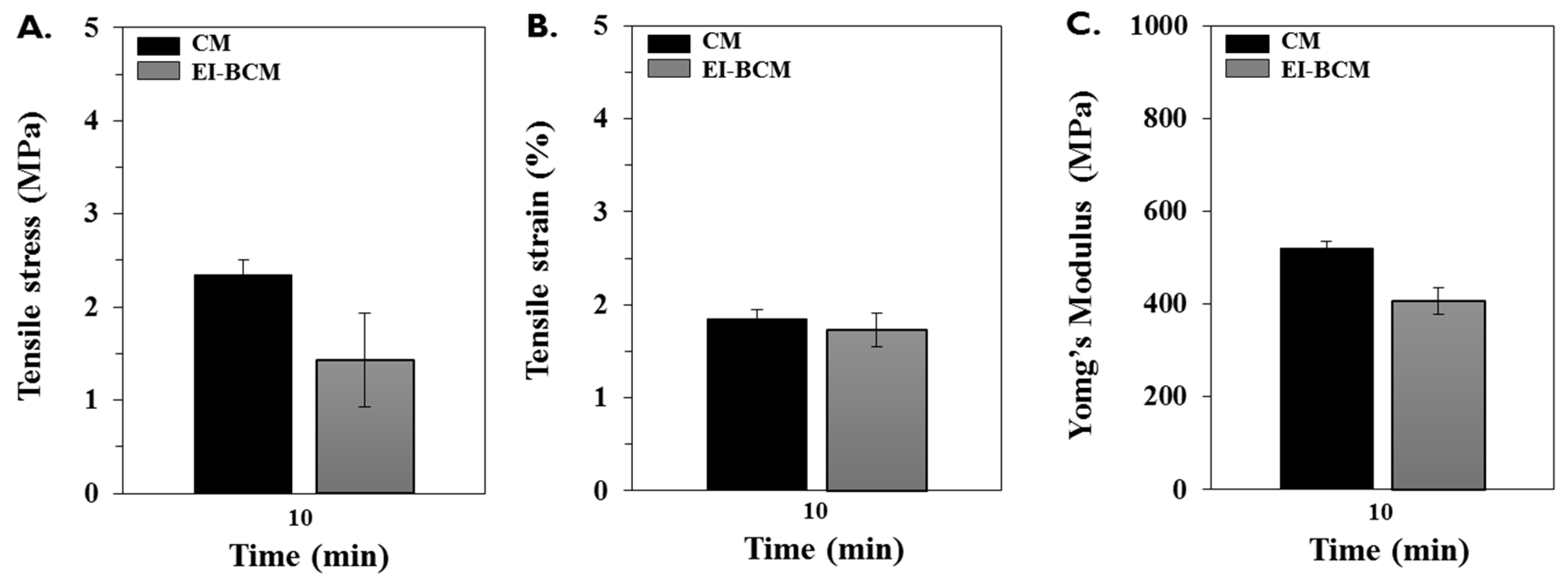
2.1. The Results of the Mechanical Studies
2.1.1. Scanning Electron Microscope (SEM) Morphologic Analysis
2.1.2. Measurement of Mechanical Strength
2.1.3. Porosity Analysis
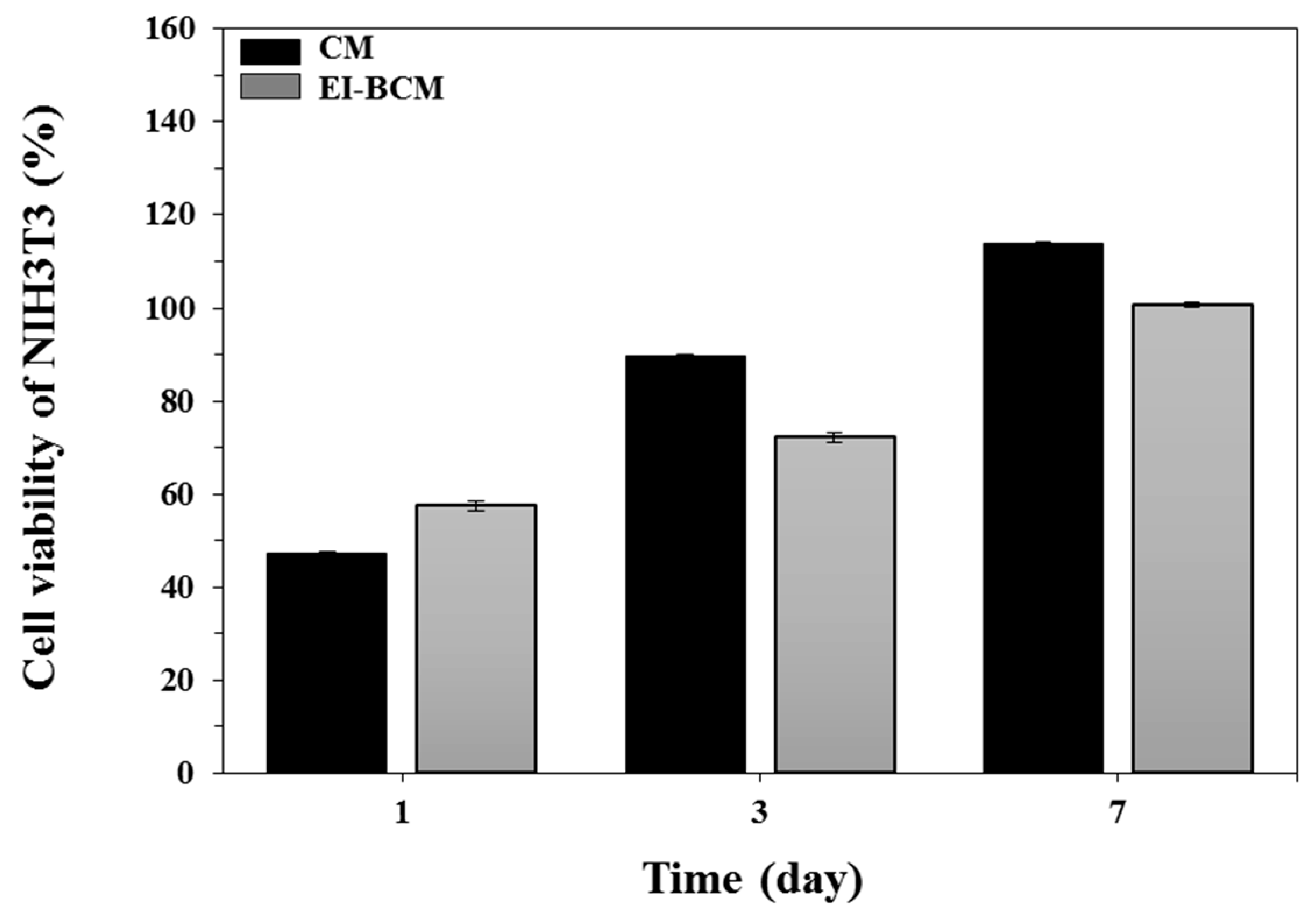
2.2. Results of In Vitro
2.2.1. Cell Viability
2.2.2. Immunofluorescent Staining Analysis of Cell Adhesion and Proliferation on the Membranes
2.3. The Results of In Vivo Studies
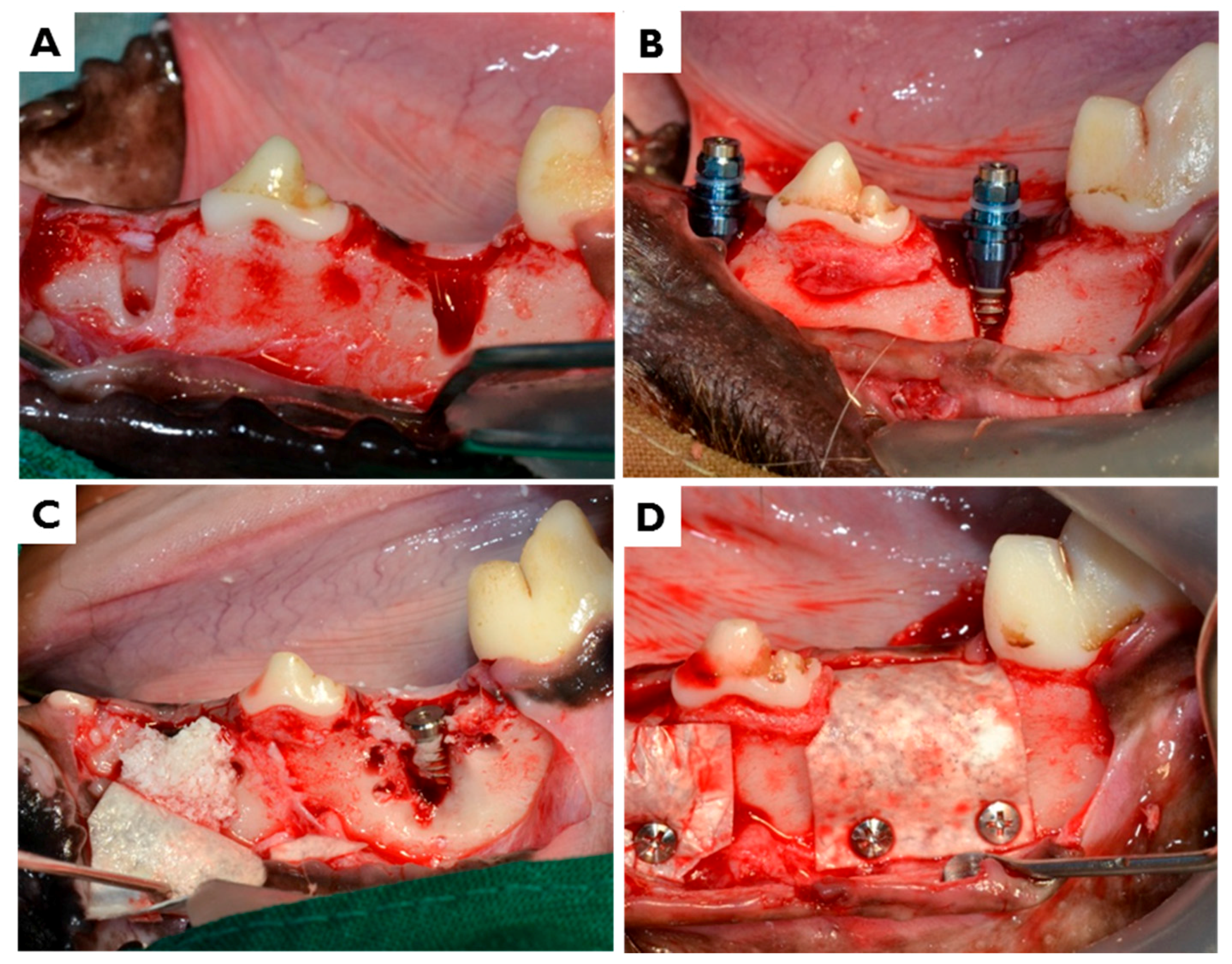
2.3.1. Clinical Findings
2.3.2. Volumetric Analysis Using Micro-Computed Tomography (μCT)
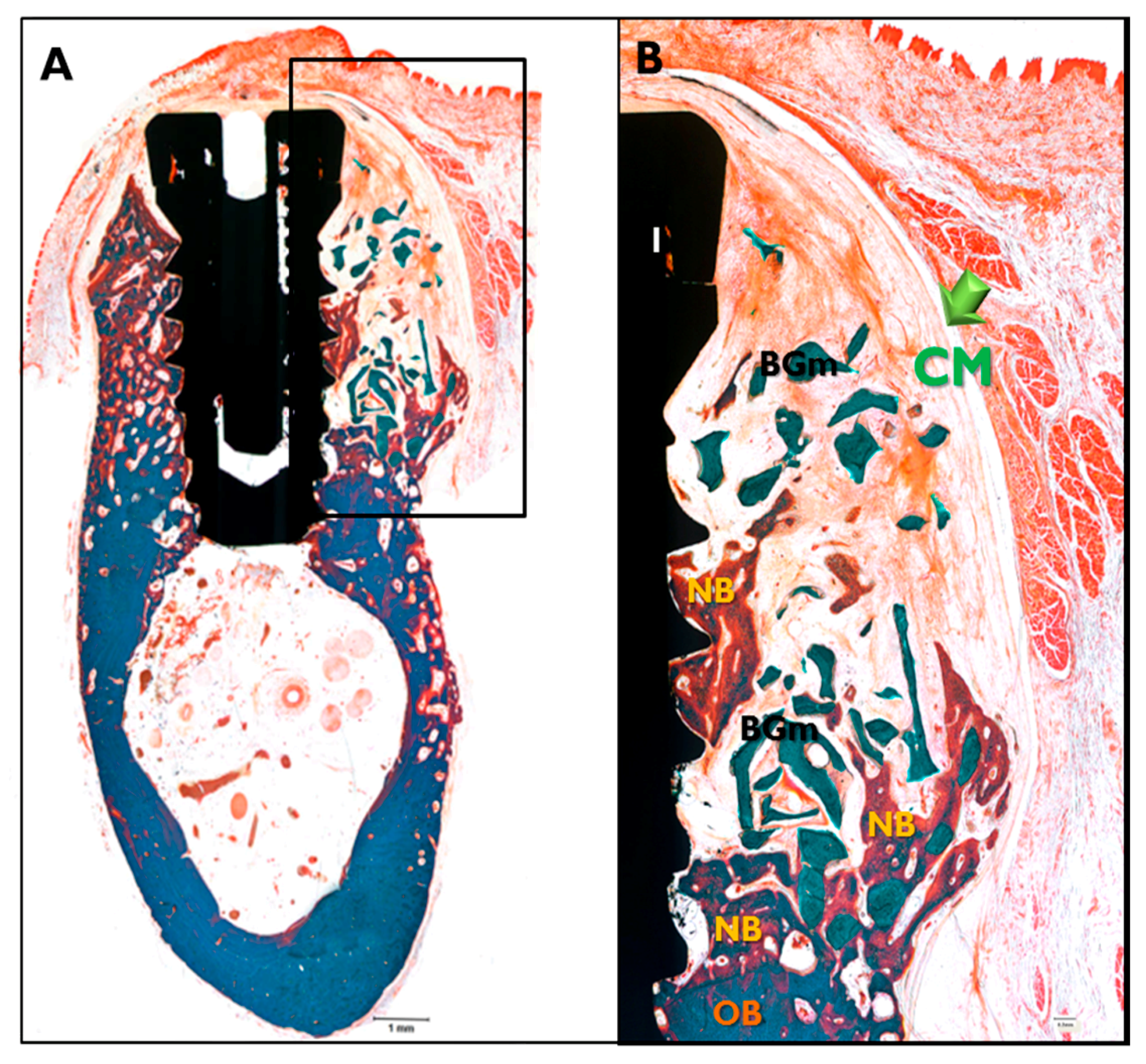
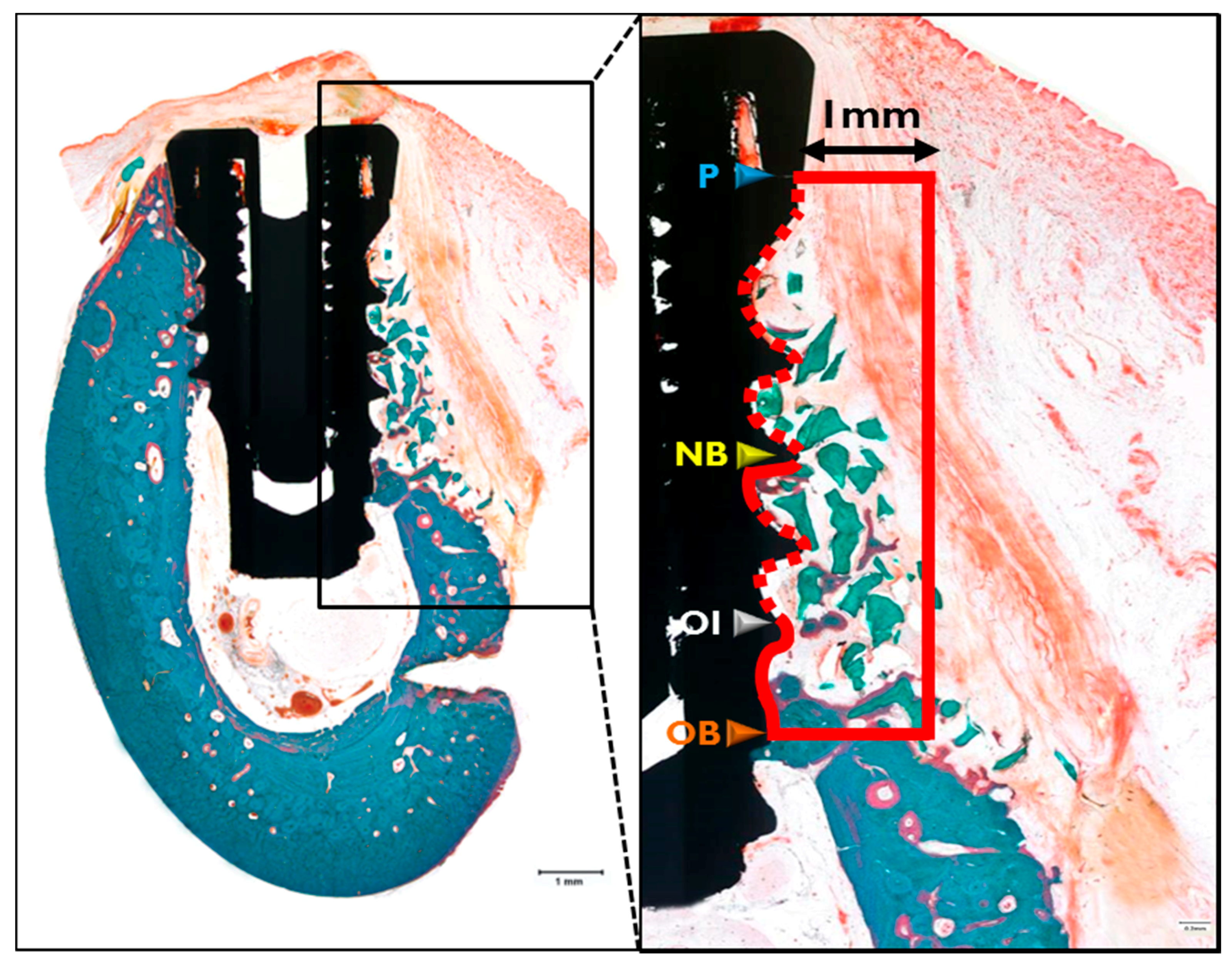
2.3.3. Histologic Findings
2.3.4. Histometric Analysis
3. Discussion
4. Materials and Methods
4.1. Preparation of Barrier Membranes
4.2. Fabrication of Electron Beam Irradiated Bacterial Cellulose Membranes
4.3. In Vitro Mechanical Studies
4.3.1. Scanning Electron Microscope (SEM) Analysis
4.3.2. Mechanical Strength Measurements
4.3.3. Porosity Analysis
4.4. In Vitro Cell Studies
4.4.1. Cell Culture
4.4.2. Cell Proliferation Assay
4.4.3. Immunofluorescent Staining
4.5. In Vivo Animal Studies
4.5.1. Experimental Animals
4.5.2. Surgical Procedures for Tooth Extraction
4.5.3. Surgical Procedures for Implant Placement and Guided Bone Regeneration (GBR)
4.5.4. Post-Operative Care and Sacrifice
4.5.5. Micro-Computed Tomography (μCT) Analysis
- Total augmented volume (TAV; mm3): volume occupied by total augmented bone volume within the ROI
- Non-mineralized tissue volume (NMV; mm3): volume occupied by non-mineralized tissue volume within the ROI
- Remaining bone substitute volume (RBV; mm3): volume occupied by remaining bone substitute volume within the ROI
- New bone volume (NBV; mm3): volume occupied by the new bone volume within the ROI
4.5.6. Histomorphometric Analysis
- New bone area (NBA; %): Area occupied by the new bone/AOI × 100
- Remaining bone substitute area (RBA; %): Area occupied by the remaining bone substitute/AOI × 100
- Bone-to-implant contact (BIC; %): Length of contact with new bone/total length of exposed threads × 100
- New bone-old bone (NB-OB; %): Distance from the most upper point of new bone to the most upper point of old bone /P-OB × 100
- New bone-old bone (NB-OB; %): Distance from the most upper point of new bone to the most upper point of old bone /P-OB × 100
- Osseointegration-old bone (OI-OB; %): Distance from the most upper point of osseointegration to the most upper point of old bone /P-OB × 100
4.6. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Rajwade, J.M.; Paknikar, K.M.; Kumbhar, J.V. Applications of bacterial cellulose and its composites in biomedicine. Appl. Microbiol. Biotechnol. 2015, 99, 2491–2511. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira Barud, H.G.; da Silva, R.R.; da Silva Barud, H.; Tercjak, A.; Gutierrez, J.; Lustri, W.R.; de Oliveira, O.B.J.; Ribeiro, S.J. A multipurpose natural and renewable polymer in medical applications: Bacterial cellulose. Carbohydr. Polym. 2016, 153, 406–420. [Google Scholar] [CrossRef] [PubMed]
- Ross, P.; Mayer, R.; Benziman, M. Cellulose biosynthesis and function in bacteria. Microbiol. Rev. 1991, 55, 35–58. [Google Scholar] [PubMed]
- Czaja, W.; Romanovicz, D.; Brown, R.M. Structural investigations of microbial cellulose produced in stationary and agitated culture. Cellulose 2004, 11, 403–411. [Google Scholar] [CrossRef]
- Brown, A.J. Xliii.—On an acetic ferment which forms cellulose. J. Chem. Soc. Trans. 1886, 49, 432–439. [Google Scholar] [CrossRef]
- Shoda, M.; Sugano, Y. Recent advances in bacterial cellulose production. Biotechnol. Bioprocess Eng. 2005, 10, 1–8. [Google Scholar] [CrossRef]
- Kurosumi, A.; Sasaki, C.; Yamashita, Y.; Nakamura, Y. Utilization of various fruit juices as carbon source for production of bacterial cellulose by Acetobacter xylinum NBRC 13693. Carbohydr. Polym. 2009, 76, 333–335. [Google Scholar] [CrossRef]
- Lee, K.Y.; Buldum, G.; Mantalaris, A.; Bismarck, A. More than meets the eye in bacterial cellulose: Biosynthesis, bioprocessing, and applications in advanced fiber composites. Macromol. Biosci. 2014, 14, 10–32. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.-Y.; Liang, H.-W.; Chen, L.-F.; Hu, B.-C.; Yu, S.-H. Bacterial cellulose: A robust platform for design of three dimensional carbon-based functional nanomaterials. Acc. Chem. Res. 2015, 49, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Nishiyama, Y.; Sugiyama, J.; Chanzy, H.; Langan, P. Crystal structure and hydrogen bonding system in cellulose Iα from synchrotron X-ray and neutron fiber diffraction. J. Am. Chem. Soc. 2003, 125, 14300–14306. [Google Scholar] [CrossRef] [PubMed]
- Gayathry, G.; Gopalaswamy, G. Production and characterisation of microbial cellulosic fibre from acetobacter xylinum. NISCAIR-CSIR 2014, 39, 93–96. [Google Scholar]
- Wan, Y.; Huang, Y.; Yuan, C.; Raman, S.; Zhu, Y.; Jiang, H.; He, F.; Gao, C. Biomimetic synthesis of hydroxyapatite/bacterial cellulose nanocomposites for biomedical applications. Mater. Sci. Eng. C 2007, 27, 855–864. [Google Scholar] [CrossRef]
- Helenius, G.; Bäckdahl, H.; Bodin, A.; Nannmark, U.; Gatenholm, P.; Risberg, B. In vivo biocompatibility of bacterial cellulose. J. Biomed. Mater. Res. Part A 2006, 76, 431–438. [Google Scholar] [CrossRef] [PubMed]
- Svensson, A.; Nicklasson, E.; Harrah, T.; Panilaitis, B.; Kaplan, D.; Brittberg, M.; Gatenholm, P. Bacterial cellulose as a potential scaffold for tissue engineering of cartilage. Biomaterials 2005, 26, 419–431. [Google Scholar] [CrossRef] [PubMed]
- Tuzlakoglu, K.; Bolgen, N.; Salgado, A.; Gomes, M.E.; Piskin, E.; Reis, R. Nano-and micro-fiber combined scaffolds: A new architecture for bone tissue engineering. J. Mater. Sci. Mater. Med. 2005, 16, 1099–1104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Novaes, A.B., Jr.; Novaes, A.B. Imz implants placed into extraction sockets in association with membrane therapy (gengiflex) and porous hydroxyapatite: A case report. Int. J. Oral Maxillofac. Implants 1992, 7, 536–540. [Google Scholar] [PubMed]
- Novaes, A., Jr.; Novaes, A. Bone formation over a tial6v4 (imz) implant placed into an extraction socket in association with membrane therapy (gengiflex). Clin. Oral Implants Res. 1993, 4, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, K.A.; LeBlanc, J.M.; Sheets, K.T.; Fox, R.W.; Gatenholm, P. Biomimetic design of a bacterial cellulose/hydroxyapatite nanocomposite for bone healing applications. Mater. Sci. Eng. C 2011, 31, 43–49. [Google Scholar] [CrossRef]
- Shi, Q.; Li, Y.; Sun, J.; Zhang, H.; Chen, L.; Chen, B.; Yang, H.; Wang, Z. The osteogenesis of bacterial cellulose scaffold loaded with bone morphogenetic protein-2. Biomaterials 2012, 33, 6644–6649. [Google Scholar] [CrossRef] [PubMed]
- Saska, S.; Scarel-Caminaga, R.M.; Teixeira, L.N.; Franchi, L.P.; Dos Santos, R.A.; Gaspar, A.M.M.; de Oliveira, P.T.; Rosa, A.L.; Takahashi, C.S.; Messaddeq, Y. Characterization and in vitro evaluation of bacterial cellulose membranes functionalized with osteogenic growth peptide for bone tissue engineering. J. Mater. Sci. Mater. Med. 2012, 23, 2253–2266. [Google Scholar] [CrossRef] [PubMed]
- Brånemark, P.; Adell, R.; Albrektsson, T.; Lekholm, U.; Lundkvist, S.; Rockler, B. Osseointegrated titanium fixtures in the treatment of edentulousness. Biomaterials 1983, 4, 25–28. [Google Scholar] [CrossRef]
- Gottlow, J.; Nyman, S.; Karring, T.; Lindhe, J. New attachment formation as the result of controlled tissue regeneration. J. Clin. Periodontol. 1984, 11, 494–503. [Google Scholar] [CrossRef] [PubMed]
- Rispoli, L.; Fontana, F.; Beretta, M.; Poggio, C.E.; Maiorana, C. Surgery Guidelines for Barrier Membranes in Guided Bone Regeneration (GBR). J. Otolaryngol. Rhinol. 2015, 1, 1–8. [Google Scholar]
- Zitzmann, N.U.; Naef, R.; Schärer, P. Resorbable versus nonresorbable membranes in combination with bio-oss for guided bone regeneration. Int. J. Oral Maxillofac. Implants 1997, 12, 844–852. [Google Scholar] [PubMed]
- Kasaj, A.; Reichert, C.; Götz, H.; Röhrig, B.; Smeets, R.; Willershausen, B. In vitro evaluation of various bioabsorbable and nonresorbable barrier membranes for guided tissue regeneration. Head Face Med. 2008, 4, 22. [Google Scholar] [CrossRef] [PubMed]
- Her, S.; Kang, T.; Fien, M.J. Titanium mesh as an alternative to a membrane for ridge augmentation. J. Oral Maxillofac. Surg. 2012, 70, 803–810. [Google Scholar] [CrossRef] [PubMed]
- Piattelli, A.; Scarano, A.; Corigliano, M.; Piattelli, M. Comparison of bone regeneration with the use of mineralized and demineralized freeze-dried bone allografts: A histological and histochemical study in man. Biomaterials 1996, 17, 1127–1131. [Google Scholar] [CrossRef]
- Kellomäki, M.; Niiranen, H.; Puumanen, K.; Ashammakhi, N.; Waris, T.; Törmälä, P. Bioabsorbable scaffolds for guided bone regeneration and generation. Biomaterials 2000, 21, 2495–2505. [Google Scholar] [CrossRef]
- Petersen, N.; Gatenholm, P. Bacterial cellulose-based materials and medical devices: Current state and perspectives. Appl. Microbial. Biotechnol. 2011, 91, 1277–1286. [Google Scholar] [CrossRef] [PubMed]
- Dugan, J.M.; Gough, J.E.; Eichhorn, S.J. Bacterial cellulose scaffolds and cellulose nanowhiskers for tissue engineering. Nanomedicine 2013, 8, 287–298. [Google Scholar] [CrossRef] [PubMed]
- Nwe, N.; Furuike, T.; Tamura, H. Selection of a biopolymer based on attachment, morphology and proliferation of fibroblast NIH/3T3 cells for the development of a biodegradable tissue regeneration template: Alginate, bacterial cellulose and gelatin. Process Biochem. 2010, 45, 457–466. [Google Scholar] [CrossRef]
- Zaborowska, M.; Bodin, A.; Bäckdahl, H.; Popp, J.; Goldstein, A.; Gatenholm, P. Microporous bacterial cellulose as a potential scaffold for bone regeneration. Acta Biomater. 2010, 6, 2540–2547. [Google Scholar] [CrossRef] [PubMed]
- Mendes, P.N.; Rahal, S.C.; Pereira-Junior, O.C.M.; Fabris, V.E.; Lenharo, S.L.R.; de Lima-Neto, J.F.; da Cruz Landim-Alvarenga, F. In vivo and in vitro evaluation of an acetobacter xylinum synthesized microbial cellulose membrane intended for guided tissue repair. Acta Vet. Scand. 2009, 51, 12. [Google Scholar] [CrossRef] [PubMed]
- Mello, L.R.; Feltrin, L.T.; Fontes Neto, P.T.; Ferraz, F.A. Duraplasty with biosynthetic cellulose: An experimental study. J. Neurosurg. 1997, 86, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Amorim, W.L.; Costa, H.O.; de Souza, F.C.; de Castro, M.G.; da Silva, L. Experimental study of the tissue reaction caused by the presence of cellulose produced. Braz. J. Otorhinolaryngol. 2009, 75, 200–207. [Google Scholar] [CrossRef]
- Rojas, J.; Bedoya, M.; Ciro, Y. Current trends in the production of cellulose nanoparticles and nanocomposites for biomedical applications. In Cellulose-Fundamental Aspects and Current Trends; Intech: Vigo, Spain, 2015; Volume 8, pp. 193–228. [Google Scholar]
- Chen, Y.M. In vitro cytotoxicity of bacterial cellulose scaffolds used for tissue-engineered bone. J. Bioact. Compat. Polym. 2009, 24, 137–145. [Google Scholar] [CrossRef]
- Chen, Y.M.; Xi, T.F.; Zheng, Y.F.; Zhou, L.; Wan, Y.Z. In vitro structural changes of nano-bacterial cellulose immersed in phosphate buffer solution. J. Biomim. Biomater. Tissue Eng. 2011, 10, 55–66. [Google Scholar] [CrossRef]
- Saska, S.; Barud, H.S.; Gaspar, A.M.M.; Marchetto, R.; Ribeiro, S.J.L.; Messaddeq, Y. Bacterial cellulose-hydroxyapatite nanocomposites for bone regeneration. Int. J. Biomater. 2011, 2011. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Wan, Y.; Li, L.; Liang, H.; Wang, J. Preparation and characterization of 2, 3-dialdehyde bacterial cellulose for potential biodegradable tissue engineering scaffolds. Mater. Sci. Eng. C 2009, 29, 1635–1642. [Google Scholar] [CrossRef]
- Czaja, W.K.; Young, D.J.; Kawecki, M.; Brown, R.M. The future prospects of microbial cellulose in biomedical applications. Biomacromolecules 2007, 8, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Czaja, W.K.; Kyryliouk, D.; DePaula, C.A.; Buechter, D.D. Oxidation of γ-irradiated microbial cellulose results in bioresorbable, highly conformable biomaterial. J. Appl. Polym. Sci. 2014, 131, 1–12. [Google Scholar] [CrossRef]
- Hu, Y.; Catchmark, J.M. In vitro biodegradability and mechanical properties of bioabsorbable bacterial cellulose incorporating cellulases. Acta Biomater. 2011, 7, 2835–2845. [Google Scholar] [CrossRef] [PubMed]
- Darwis, D.; Khusniya, T.; Hardiningsih, L.; Nurlidar, F.; Winarno, H. In Vitro degradation behaviour of irradiated bacterial cellulose membrane. Atom Indones. 2012, 38, 78–82. [Google Scholar] [CrossRef]
- Lee, S.-H.; Lim, Y.-M.; Jeong, S.I.; An, S.-J.; Kang, S.-S.; Jeong, C.-M.; Huh, J.-B. The effect of bacterial cellulose membrane compared with CM on guided bone regeneration. J. Adv. Prosthodont. 2015, 7, 484–495. [Google Scholar] [CrossRef] [PubMed]
- Chmielewski, A.G. Worldwide developments in the field of radiation processing of materials in the down of 21st century. Nukleonika 2006, 51, 3–9. [Google Scholar]
- Kim, S.M.; Fan, H.; Cho, Y.J.; Eo, M.Y.; Park, J.H.; Kim, B.N.; Lee, B.C.; Lee, S.K. Electron beam effect on biomaterials I: Focusing on bone graft materials. Biomater. Res. 2015, 19, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Laurell, B.; Föll, E.; AB, E.C. Electron-beam accelerators for new applications. In RadTech Europe 2011 Exhibition & Conference for Radiation Curing; Electron Crosslinking AB: Basel, Switzerland, 2011. [Google Scholar]
- Földváry, C.M.; Takács, E.; Wojnarovits, L. Effect of high-energy radiation and alkali treatment on the properties of cellulose. Radiat. Phys. Chem. 2003, 67, 505–508. [Google Scholar] [CrossRef]
- Petryayev, Y.P.; Boltromeyuk, V.V.; Kovalenko, N.I.; Shadyro, O.I. Mechanism of radiation-initiated degradation of cellulose and derivatives. Polym. Sci. USSR 1988, 30, 2208–2214. [Google Scholar] [CrossRef]
- Kim, S.M.; Eo, M.Y.; Kang, J.Y.; Myoung, H.; Choi, E.K.; Lee, S.K.; Lee, B.C.; Lee, J.H. Bony regeneration effect of electron-beam irradiated hydroxyapatite and tricalcium phosphate mixtures with 7 to 3 ratio in the calvarial defect model of rat. Tissue Eng. Regen. Med. 2012, 9, 24–32. [Google Scholar]
- Davis, J.R. Overview of biomaterials and their use in medical devices. In Handbook of Materials for Medical Devices; Illustrated Edition; ASM International: Materials Park, OH, USA, 2003; pp. 1–11. [Google Scholar]
- Khan, R.A.; Beck, S.; Dussault, D.; Salmieri, S.; Bouchard, J.; Lacroix, M. Mechanical and barrier properties of nanocrystalline cellulose reinforced poly (caprolactone) composites: Effect of gamma radiation. J. Appl. Polym. Sci. 2013, 129, 3038–3046. [Google Scholar] [CrossRef]
- Eo, M.Y.; Fan, H.; Cho, Y.J.; Kim, S.M.; Lee, S.K. Cellulose membrane as a biomaterial: From hydrolysis to depolymerization with electron beam. Biomater. Res. 2016, 20, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.; Jia, S.; Jia, Y.; Yang, H. The influence of fermentation conditions and post-treatment methods on porosity of bacterial cellulose membrane. World J. Microbiol. Biotechnol. 2010, 26, 125–131. [Google Scholar] [CrossRef]
- Gao, C.; Wan, Y.; Lei, X.; Qu, J.; Yan, T.; Dai, K. Polylysine coated bacterial cellulose nanofibers as novel templates for bone-like apatite deposition. Cellulose 2011, 18, 1555–1561. [Google Scholar] [CrossRef]
- Zellin, G.; Linde, A. Effects of different osteopromotive membrane porosities on experimental bone neogenesis in rats. Biomaterials 1996, 17, 695–702. [Google Scholar] [CrossRef]
- Lundgren, A.; Sennerby, L.; Lundgren, D. Guided jaw-bone regeneration using an experimental rabbit model. Int. J. Oral Maxillofac. Surg. 1998, 27, 135–140. [Google Scholar] [CrossRef]
- Bäckdahl, H.; Helenius, G.; Bodin, A.; Nannmark, U.; Johansson, B.R.; Risberg, B.; Gatenholm, P. Mechanical properties of bacterial cellulose and interactions with smooth muscle cells. Biomaterials 2006, 27, 2141–2149. [Google Scholar] [CrossRef] [PubMed]
- Fang, B.; Wan, Y.-Z.; Tang, T.-T.; Gao, C.; Dai, K.-R. Proliferation and osteoblastic differentiation of human bone marrow stromal cells on hydroxyapatite/bacterial cellulose nanocomposite scaffolds. Tissue Eng. Part A 2009, 15, 1091–1098. [Google Scholar] [CrossRef] [PubMed]
- Chu, P.K.; Chen, J.Y.; Wang, L.P.; Huang, N. Plasma-surface modification of biomaterials. Mater. Sci. Eng. R Rep. 2002, 36, 143–206. [Google Scholar] [CrossRef]
- Ahn, S.-J.; Shin, Y.M.; Kim, S.E.; Jeong, S.I.; Jeong, J.-O.; Park, J.-S.; Gwon, H.-J.; Seo, D.E.; Nho, Y.-C.; Kang, S.S. Characterization of hydroxyapatite-coated bacterial cellulose scaffold for bone tissue engineering. Biotechnol. Bioprocess Eng. 2015, 20, 948–955. [Google Scholar] [CrossRef]
- Pértile, R.; Moreira, S.; Andrade, F.; Domingues, L.; Gama, M. Bacterial cellulose modified using recombinant proteins to improve neuronal and mesenchymal cell adhesion. Biotechnol. Prog. 2012, 28, 526–532. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Andrade, F.K.; Moreira, S.M.; Domingues, L.; Gama, F.M. Improving the affinity of fibroblasts for bacterial cellulose using carbohydrate-binding modules fused to RGD. J. Biomed. Mater. Res. Part A 2010, 92, 9–17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miyamoto, T.; Takahashi, S.I.; Ito, H.; Inagaki, H.; Noishiki, Y. Tissue biocompatibility of cellulose and its derivatives. J. Biomed. Mater. Res. Part A 1989, 23, 125–133. [Google Scholar] [CrossRef] [PubMed]
- Buser, D.; Dahlin, C.; Schenk, R.K. Guided bone regeneration in implant dentistry. Chic. Quintessence 1994, 31–47. [Google Scholar]
- Fujihara, K.; Kotaki, M.; Ramakrishna, S. Guided bone regeneration membrane made of polycaprolactone/calcium carbonate composite nano-fibers. Biomaterials 2005, 26, 4139–4147. [Google Scholar] [CrossRef] [PubMed]
- Bartee, B.K.; Carr, J.A. Evaluation of a high-density polytetrafluoroethylene (n-PTFE) membrane as a barrier material to facilitate guided bone regeneration in the rat mandible. J. Oral Implantol. 1995, 21, 88–95. [Google Scholar] [PubMed]








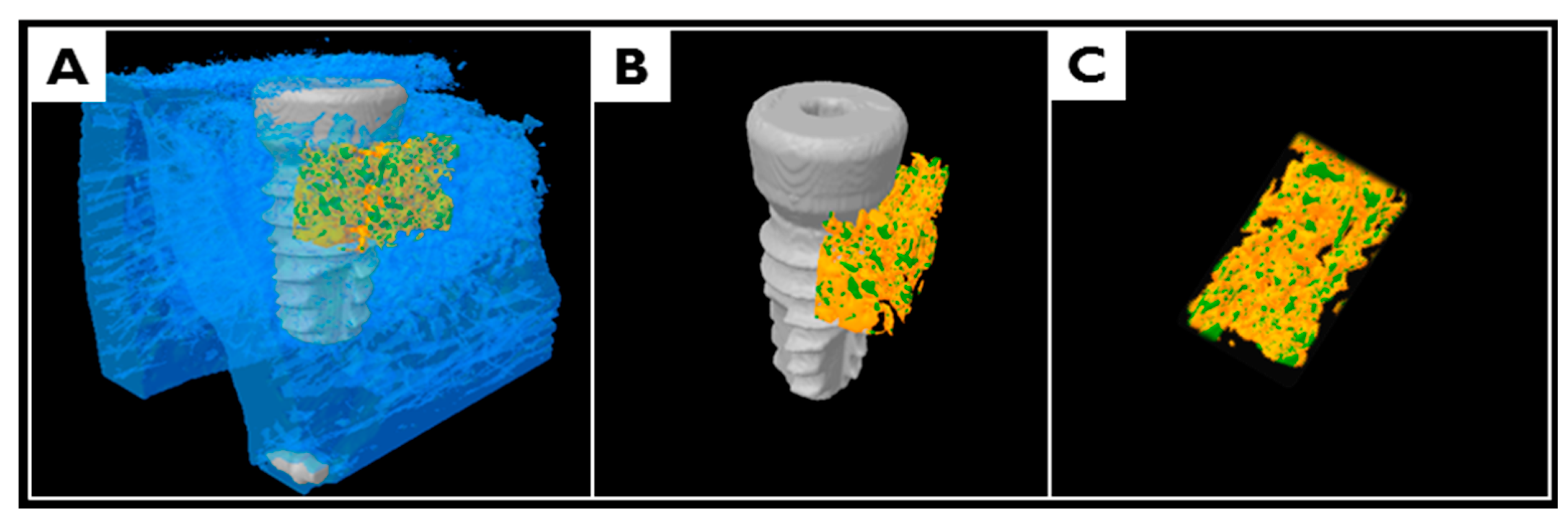
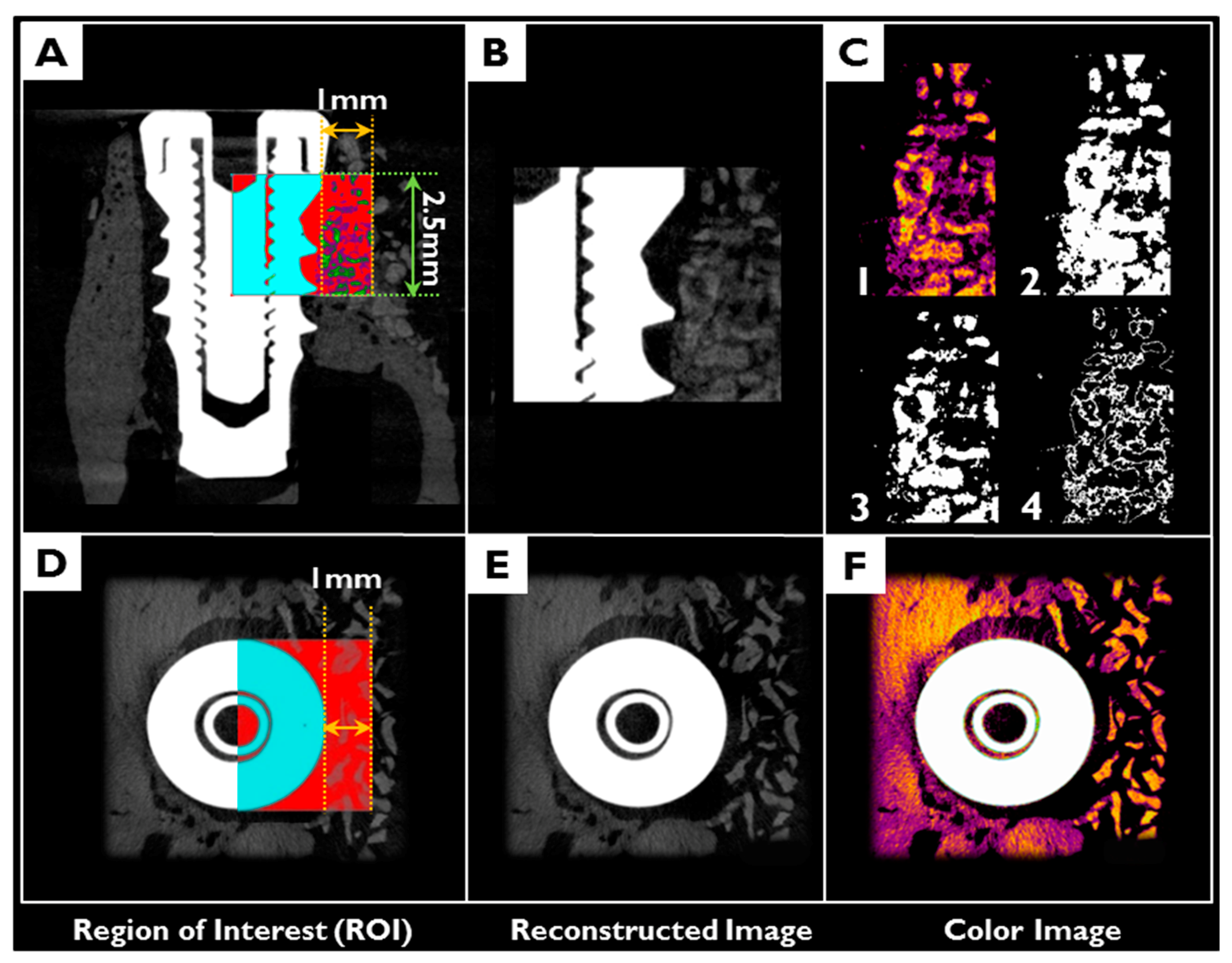
| Group (Membrane) | NBV (mm3) | TAV (mm3) | RBV (mm3) | NMV (mm3) |
|---|---|---|---|---|
| CM | 1.23 ± 0.86 | 14.28 ± 0.17 | 2.40 ± 2.36 | 10.65 ± 3.23 |
| EI-BCM | 1.14 ± 0.91 | 14.41 ± 0.16 | 2.31 ± 2.04 | 10.96 ± 2.85 |
| p | >0.05 | >0.05 | >0.05 | >0.05 |
| Group (Membrane) | NBA (%) | RBA (%) | BIC (%) | NB-OB (%) | OI-OB (%) |
|---|---|---|---|---|---|
| CM | 15.07 ± 9.95 | 12.49 ± 7.57 | 41.33 ± 13.52 | 54.67 ± 31.78 | 37.40 ± 16.63 |
| EI-BCM | 16.51 ± 13.00 | 9.05 ± 11.61 | 38.82 ± 25.74 | 44.35 ± 39.90 | 38.52 ± 37.60 |
| p | >0.05 | >0.05 | >0.05 | >0.05 | >0.05 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, S.-H.; An, S.-J.; Lim, Y.-M.; Huh, J.-B. The Efficacy of Electron Beam Irradiated Bacterial Cellulose Membranes as Compared with Collagen Membranes on Guided Bone Regeneration in Peri-Implant Bone Defects. Materials 2017, 10, 1018. https://doi.org/10.3390/ma10091018
Lee S-H, An S-J, Lim Y-M, Huh J-B. The Efficacy of Electron Beam Irradiated Bacterial Cellulose Membranes as Compared with Collagen Membranes on Guided Bone Regeneration in Peri-Implant Bone Defects. Materials. 2017; 10(9):1018. https://doi.org/10.3390/ma10091018
Chicago/Turabian StyleLee, So-Hyoun, Sung-Jun An, Youn-Mook Lim, and Jung-Bo Huh. 2017. "The Efficacy of Electron Beam Irradiated Bacterial Cellulose Membranes as Compared with Collagen Membranes on Guided Bone Regeneration in Peri-Implant Bone Defects" Materials 10, no. 9: 1018. https://doi.org/10.3390/ma10091018




