Abstract
Agricultural residues, such as corn stover, wheat straw, and nut shells show promise as feedstocks for lignocellulosic biorefinery due to their relatively high polysaccharide content and low or no nutritional value for human consumption. Apricot pit shells (APS) were studied in this work to assess their potential for use in a biorefinery. Hot water extraction (HWE; 160 °C, 2 h), proposed to remove easily accessible hemicelluloses, was performed to evaluate the susceptibility of APS to this mild pretreatment process. The chemical composition of APS before and after HWE (EAPS) was analyzed by standard methods and 1H-NMR. A low yield of the remaining HW-extracted APS (~59%) indicated that APS are highly susceptible to this pretreatment method. 1H-NMR analysis of EAPS revealed that ~77% of xylan present in raw APS was removed along with ~24% of lignin. The energy of combustion of APS was measured before and after HWE showing a slight increase due to HWE (1.61% increase). Near infrared radiation spectroscopy (NIRS), proposed as a quick non-invasive method of biomass analysis, was performed. NIRS corroborated results of traditional analysis and 1H-NMR. Determination of antioxidizing activity (AOA) of APS extracts was also undertaken. AOA of organic APS extracts were shown to be more than 20 times higher than that of a synthetic antioxidizing agent.
1. Introduction
Apricot (Prunus armeniaca L.) fruit has been recommended for human consumption due to its numerous health benefits, including fiber, minerals, and a variety of vitamins, such as Vitamin A, Vitamin C, Vitamin K, thiamine, riboflavin, niacin, and panthotenic acid. Apricot fruit also possesses a relatively high antioxidizing capacity attributed to phenolic and carotenoid compounds [1,2,3,4]. The fruits can be used fresh, dried, or processed as fruit juice, jam, and jelly. Apricots may also be consumed canned (16% of US production [4]).
The most important apricot producers are Turkey, Iran, Italy, and Pakistan, while the USA has traditionally been in the group of the ten largest apricot producers in the world [5]. The world production of apricot increased more than 60% between 1994 and 2013 (1994: 2.564 × 106 tons; 2013: 4.111 × 106 tons [6]). However, the US apricot production decreased in the same period (1994: 138,980 tons; 2014: 55,360 tons). The US average annual production of apricot between 2004 and 2013 (65,394 tons) decreased more than 27% from the average production between 1994 and 2003 (90,618 tons [6]). California accounts for ~80% of the US apricot production [3] as an unfavorable climate precludes large scale production in much of the remaining US. However, cold and frost hardy apricot varieties coupled with proper horticultural techniques may lead to increased success in apricot production in non-ideal climates [3,7].
The use of apricot pit shells (APS), which account for ~10% of the fruit mass [8] and are currently considered to be a waste product, may increase profitability of the apricot industry and promote apricot production in the USA. APS may be used for the production of activated carbon or converted to a gas mixture composed of mostly carbon monoxide, carbon dioxide, and methane [8,9,10,11,12], while kernels, which contain dietary proteins, oils, and fiber, exhibit antioxidant and antimicrobial activities and may be used in the production of oils, benzaldehyde, and cosmetics [1,13]. Valorization of APS is also consistent with efforts to develop materials, fuels, and processes that utilize lignocellulosic materials in place of fossil fuels [14].
However, unlike starch-rich biomass, utilization of lignocellulosic biomass requires pretreatment for efficient fractionation due to its inherent recalcitrance. Hot-water extraction is a mild autohydrolysis process (160 °C, 2 h) that can selectively remove large amounts of xylans from hardwoods while retaining the carbohydrate structure for further processing, such as hydrolysis and fermentation [15,16]. In addition to removing xylans, hot-water extraction (HWE) results in an increase in cellulose and lignin content, an increase in porosity, a decreased ash content, and cleavage of acetyl groups. These modifications provide for more efficient delignification, enzymatic hydrolysis, and pyrolysis of hot-water extracted biomass as confirmed in recent studies on different hardwoods, including sugar maple, Paulownia species, and eucalyptus [17,18,19,20]. HWE also results in an increase in energy of combustion [21].
This study was focused on the effects of HWE to estimate the utility of this pretreatment in valorization of apricot pit shells (APS). The effects were estimated by analysis of chemical composition and energy of combustion of APS before and after HWE. The chemical composition before and after HWE was analyzed by combination of traditional/standard methods used in wood chemistry and newly developed spectral methods (1H-NMR and NIRS). Antioxidizing potential of organic-solvent- and HW-extracts was also estimated.
2. Results and Discussion
2.1. Characterization of Apricot Pit Shells (APS)
2.1.1. Chemical Composition of APS
Results of analysis of the chemical composition of apricot pit shells (APS) following standard and traditional methods of wood analysis are presented in Table 1.

Table 1.
Chemical composition of APS; Results of standard and traditional methods.
| % Oven Dried (OD) APS | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Ash | Extractives | Uronic acids | Lignin | Cellulose | |||||
| Hexane | EtOH:Tol | Methanol | Cold water (Hot water) | Klason | Acid soluble | Total | KH | ||
| 0.55 ± 0.049 | 0.067 ± 0.00048 | 0.92 ± 0.082 | 0.81 ± 0.023 | 0.027 ± 0.0023 (0.10 ± 0.0036) | 10.98 ± 0.81 | 29.48 ± 0.72 | 2.43 ± 0.15 | 31.91 ± 0.86 | 34.31 ± 1.47 |
Relatively low ash content (0.55%) determined in this study was lower than found by Cañellas et al. (1.67% [12]) but between the values reported by other research groups (0.17% [22]; 0.2% [10,11]; 0.3% [23]; 0.78% [9]; 1.0% [24]). The ash content of apricot pit shells appears to be less, on average, than recorded for other fruit stones, seeds, and nut shells (nut shells: 1.2%–3.8%; almond shells: 1.54%–2.3%; cherry stones: 0.9%; grape seeds: 2.5% [10,11]), demonstrating benefits of using apricot pit shells for combustion, pyrolysis, or carbonization.
The sequential Soxhlet extraction with organic solvents of increasing polarity showed that the content of polar extractives (e.g., phenolic compounds) was higher than the content of non-polar extractives (aliphatic compounds). Overall, the total extractive content (1.82% based on OD APS) was higher than reported by Yeganeh et al. (0.41% [23]) but lower than other results reported earlier: in accordance with Wei et al. [22], APS contain 5.2% extractives while oil and polyphenolic compounds are present in the amount of 1.6%–2.2% and 0.74%–0.9% respectively in accordance with the results of Cañellas et al. [12]. The low solubility of APS in cold and hot water indicates a minute amount of water-soluble carbohydrates, such as free sugars, glycosides, and starch.
The total lignin content represented by the acid-insoluble (Klason) and acid-soluble lignin was slightly less than a third of the APS mass (31.91%). The lignin content reported for APS earlier varies considerably and ranges from 23.4% to 51.43% [10,12,22,23]. It should be noted, however, that on average, nut shells and fruit stones tend to contain more lignin than wood (walnut shell: 33.3%–52.3%; almond shell: 20.4%–27%; hazelnut shell: 27.2%–43.01%; apricot stone: 23.4%–51.4%; wood: 20%–33%; with compression wood 37%–40%; [10,12,25,26,27]).
The Kürschner-Hoffer (KH) method of cellulose determination is based on delignification and acid hydrolysis of hemicelluloses. It is expected that ~25% of pentosans remain undissolved, thus increasing the amount of measured insoluble residue [28]. The Kürschner-Hoffer cellulose accounted for 34.31% of APS OD mass, which is consistent with the results of Cañellas et al (33.6% [12]) and in between results of other authors (22.4%–39.8%; [10,22,23]). Overall, literature data indicates that the cellulose content of nut shells and fruit stones is less, on average, than that in wood (almond shell: 22.36%–50.7%; walnut shell: 25.6%–34.5%; hazelnut shell: 26.7%–40.5%; apricot stone: 33.6%–39.75%; wood: 37%–45%; tension wood: 50%–65%; [10,12,22,25,26,27]).
In addition to traditional and standard methods of chemical analysis of lignocellulosics, APS were analyzed for their carbohydrate composition by 1H-NMR analysis of acid hydrolysate [29,30,31]. The spectrum showing the distinct peaks of glucose, xylose, galactose, rhamnose, mannose, furfural, and acetate is presented in Figure 1. The other expected peaks that may be assigned to characteristic lignocellulosic monosaccharides and their degradation products, including arabinose and hydroxymethyl furfural, were below detection limit. Quantification of observed hydrolysate components was achieved by comparison of the peak integrals with that of a known internal standard (glucosamine) as reported in the literature [30,31] (Table 2). The results converted to the content of corresponding polymers showed that the amounts of glucan (cellulose) and xylan in the APS were approximately the same (20.9 and 19.5%, respectively). Carbohydrate content as determined by 1H-NMR is low compared to literature values, indicating that the acid hydrolysis method used may not be ideal for quantification of carbohydrates in lignocellulosic material. One important reason may be loss of xylan to furfural degradation products during acid hydrolysis. Shin and Cho reported a loss of up to 38% of xylans when using a similar method, even though furfural was taken into account [32]. It is recommended that other methods of hydrolysis such as acid methanolysis be considered for quantification of lignocellulosic carbohydrates [33,34]. Xylans (19.54%) in APS appear to be comparably acetylated (5.32%) to xylan in sugar maple and aspen (acetyl groups: 3.7 and 3.8%, respectively; xylans: 13.8 and 12.4%, respectively; [35]).
Uronic acids were quantified using a decarboxylation method [36]. Uronic acids were found to account for ~11% of the original dry mass of APS, which is almost twice as much as the amount of uronic acid found for various hardwood species [34]. The main sources of uronic acids in lignocellulosic materials are xylans (4-O-methyl glucuronic acid and glucuronic acid) and pectins (galacturonic acid).
The total mass balance for the compositional analysis of APS (sum of ~93% of OD APS using carbohydrate values from 1H-NMR analysis) indicates a need to consider other constituents not quantified in this study (for example methoxyl groups) and to avoid potential degradation of carbohydrates (use of alternative methods, such as acid methanolysis).
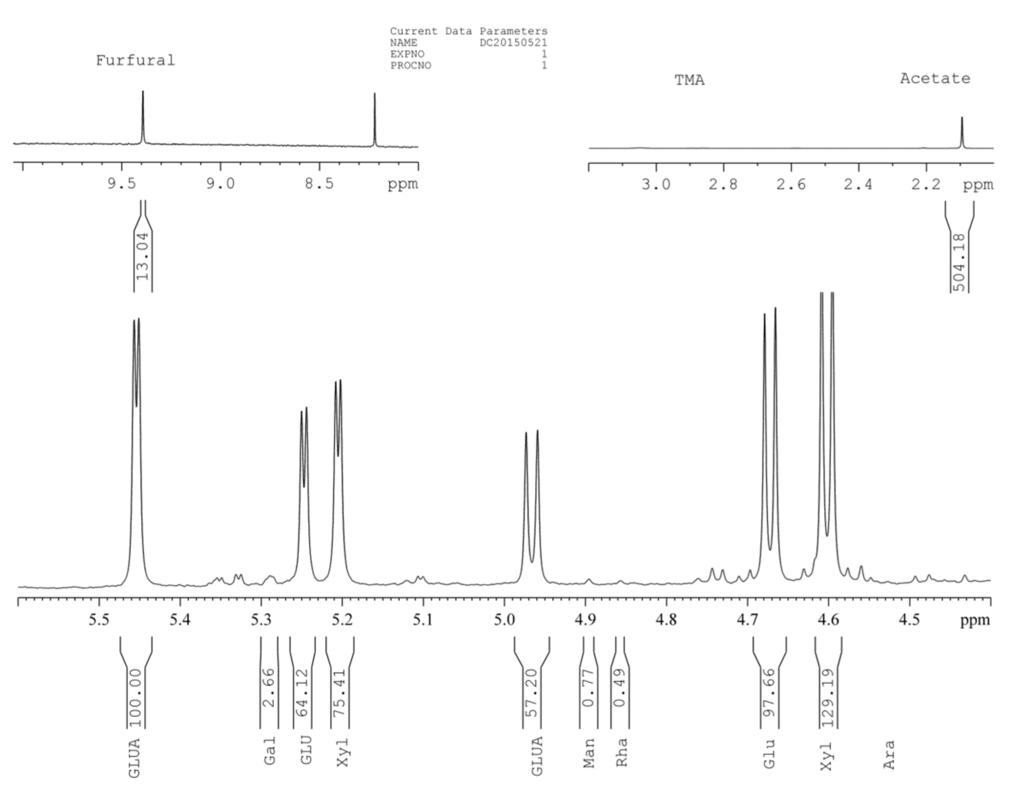
Figure 1.
1H-NMR spectrum of APS acid hydrolysate.

Table 2.
1H-NMR analysis of acid hydrolysate of APS, results based on OD mass APS.
| % OD APS | ||||||
|---|---|---|---|---|---|---|
| Glucose | Xylose | Galactose | Rhamnose | Mannose | Furfural | Acetate |
| 23.20 ± 1.42 | 22.20 ± 1.41 | 0.43 ± 0.089 | 0.030 ± 0.043 | 0.16 ± 0.082 | 1.42 ± 0.35 | 7.42 ± 0.10 |
2.1.2. Energy of Combustion of APS
Energy of combustion of APS was determined using dry APS (higher heating value; HHV). HHV of APS was 20.22 MJ/kg (standard deviation: 0.13) which is less than 22.08 MJ/kg reported by Kaynak et al. [24]. However, this value is higher than was found for fourteen other lignocellulosic species by Demirbaş, which had an average HHV of 19.14 MJ/kg (standard deviation: 1.14) [25].
2.1.3. Antioxidizing Activity (AOA) of APS Organic Solvent Extract
It has been reported that apricot fruit and apricot kernels have high antioxidizing capacities [1,2,3,4]. To assess whether the same was true for the pit shell, antioxidizing activity (AOA), analysis of APS toluene/ethanol (2/1 vol/vol) extract was performed. The IC50 (concentration of antioxidant at which 50% of present radicals are quenched) of the extract was found to be 0.029 mg/mL, which is ~23 times better than that of the synthetic antioxidizing agent butylated hydroxyanisole (0.653 mg/mL) but is still not as effective as the natural antioxidizing agent Vitamin E (0.001 mg/mL).
2.2. Effects of Hot Water Extraction on Apricot Pit Shells (APS)
2.2.1. Hot Water Extraction (HWE) of APS
HWE results demonstrated superior suitability of APS for this type of pretreatment. Solid extraction yield of hot water extracted APS (EAPS) was 59.23% (standard deviation 1.75), which indicates that slightly more than 40% of the APS total mass was extracted during HWE. HWE of wheat straw, sugar maple, and Paulownia species under the same conditions resulted in the dissolution of 29.0%–37.5% of total biomass [18,21].
2.2.2. Chemical Composition of EAPS
The results of analysis of the chemical composition of APS after HWE (EAPS) using traditional and standard methods are presented in Table 3, while the results of 1H-NMR analysis of acid hydrolysate of EAPS are shown in Figure 2 and Table 4. In accordance with the results of glucan analysis by 1H-NMR (APS: 20.9%; EAPS: 33.8%), the percent cellulose composition of EAPS as determined by the Kürschner-Hoffer method (52.78%) was higher than was found for APS (34.31%). The lignin content increased as well (31.91% to 40.93%), while the content of xylan, acetyl groups, uronic acids, and inorganics decreased substantially (19.53% to 7.47%, 5.32% to 1.71%, 10.98% to 0.41%, and 0.55% to 0.15%, respectively). The minute amounts of mannose, galactose, and rhamnose that were detected in the 1H-NMR spectrum of APS acid hydrolysate were not detectable in that of EAPS revealing that respective polysaccharides had been almost completely solubilized during HWE. It may be concluded that, even though it is considered as an underestimate of carbohydrate content (especially xylans [32]), 1H-NMR may be accepted as a comparative tool to assess the effects of HWE on APS.
The total mass balance for our analysis of EAPS (using 1H-NMR carbohydrate results) adds up to 90.3% of total EAPS.

Table 3.
Chemical composition of EAPS. Results of standard and traditional methods.
| % OD EAPS | ||||||
|---|---|---|---|---|---|---|
| Ash | Extractives | Uronic acids | Lignin | Cellulose | ||
| Dichloromethane | Klason | Acid soluble | Total | KH | ||
| 0.15 ± 0.038 | 1.49 N.D. | 0.41% ± 0.18 | 39.46 ± 0.47 | 1.47 ± 0.20 | 40.93 ± 0.27 | 52.78 ± 3.86 |
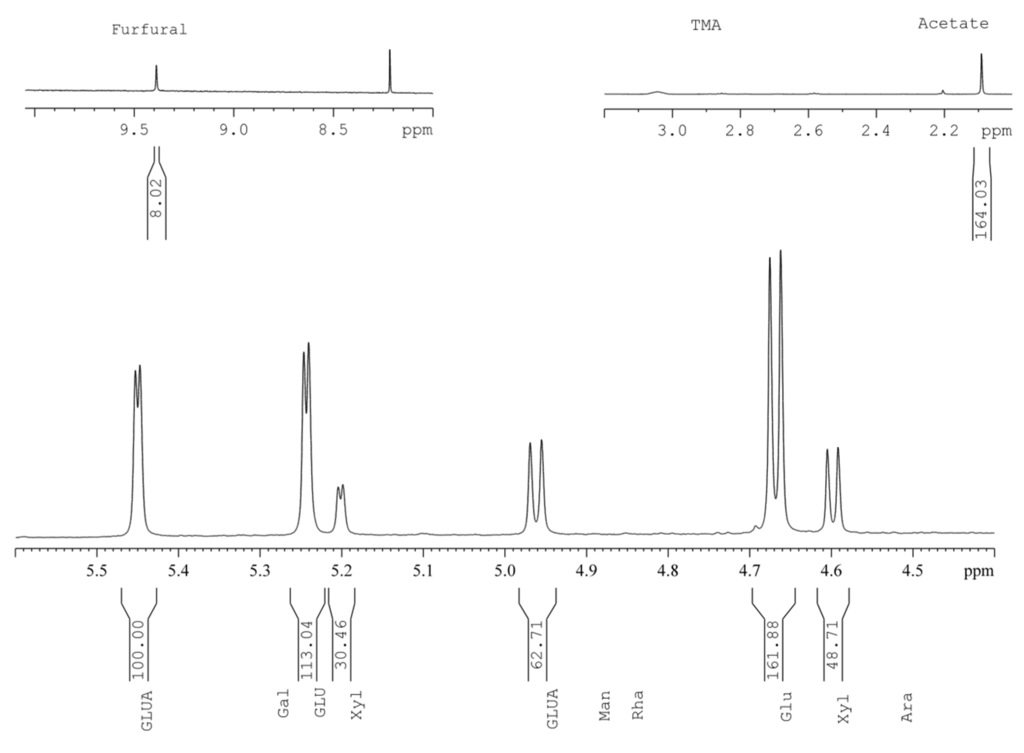
Figure 2.
1H-NMR spectrum of EAPS acid hydrolysate.

Table 4.
1H-NMR analysis of acid hydrolysate of EAPS, results based on OD mass of EAPS.
| % OD EAPS | |||
|---|---|---|---|
| Glucose | Xylose | Furfural | Acetate |
| 37.55 ± 0.69 | 8.49 ± 0.92 | 0.74 ± 0.23 | 2.38 ± 0.085 |
2.2.3. Effects of Hot Water Extraction (HWE) on APS
HWE as an autohydrolysis process is initiated by deacetylation of biomass, which in this case accounted to a total loss of 81% of originally present acetyl groups. The extracted acetic acid may be recovered from the hot-water extract by membrane separation and sold to increase profitability of biorefinery based on APS [15]. Even though other acidic compounds, such as uronic acids (which were almost completely solubilized during HWE of APS), contribute to a further decrease in pH, the process is triggered by deacetylation. The average final pH of HW-extract observed in experiments with APS was 3.42 (standard deviation: 0.079), which is consistent with a previously reported range of 3.2–3.8 [16].
HWE led to the dissolution of about 77% of originally present xylan. Dissolved xylans, which are present either as shorter chains, xylooligomers, or xylose, can be further processed in biorefinery operations to produce biofuels (ethanol, butanol) or bioplastics [15]. As indicated by the high removal of uronic acids as compared to the removal of xylans (98% vs. 77%), it can be concluded that highly acidified xylans were preferentially dissolved during HWE. At the same time, glucan (cellulose) was well retained (loss of ~4% glucan) verifying highly selective removal of xylan inherent to HWE [15,16]. It should be noted as well that with complete disappearance of mannose (as shown by 1H-NMR) a portion of the glucan removed might be due to the removal of glucomannan.
With decrease in pH during HWE easily accessible lignin fractions are hydrolyzed. Delignification occurs at a level ranging from 10%–15% on average for hardwoods, and reaching 28% for wheat straw (delignification degree based on originally present lignin in biomass; DD; [16,17,18,21]). The DD of 23.99% for APS was higher than reported for hardwoods. This finding may be indicative of a relatively high syringyl-to-guaiacyl ratio of the lignin present in APS [18].
Recovery of the HW-extracted lignin from the HWE liquor was not as efficient for APS as has been reported for hardwoods (~50% of extracted lignin; [16,18]). Only ~5% of the lignin extracted during HWE was recovered by the methods used (pH adjustment to 2.0 ± 0.01 with sulfuric acid, cold-settling, and centrifugation/decantation). Such a low lignin recovery yield indicates an extensive cleavage of lignin resulting in low molecular weight phenolic compounds that remain dissolved in the HW-extract due to high hydrophilicity even at low pH.
As it has been previously shown that HWE improves delignification efficiency [15,16,17,18], further delignification of EAPS was investigated by extraction with acetone:water (AW; 9/1 vol/vol), which has been shown to be highly selective for lignin extraction from HW-extracted hardwoods [17,18]. AW extraction of EAPS resulted in the removal of ~6.5% of the original EAPS dry mass, yielding a total mass removal of ~47% for both HWE and AW extractions together. The lignin content of the extracted solids was determined to be 97.4%, which represents a delignification degree of 15.3% due to AW extraction, and a total delignification degree of 35.3% for both HWE and AW extractions combined (based on original lignin in APS).
It is also expected that HWE results in the removal of inorganics that are soluble in water. More than 83% of the inorganics were dissolved from APS. This would be a beneficial feature of EAPS in the case of further processing, e.g., combustion, pyrolysis or the production of activated carbon. APS have been recently considered for these applications [8,9,10,11,22,23,24].
2.2.4. Energy of Combustion of EAPS
The higher heating value (HHV) of EAPS was determined to assess the effect of HWE on energy of combustion of APS. After HWE, energy of combustion increased ~4.1% (from 20.22 MJ/kg to 21.06 MJ/kg), which is a larger increase in energy of combustion than was found for sugar maple (2.9%) and wheat straw (3.0%) [21]. In his study, Demirbaş found a strong positive correlation between HHV and lignin content in biomass [25]. Results of this study corroborate the HHV-lignin correlation since EAPS, which is of higher lignin content than APS (40.93% as opposed to 31.91%), exhibited a higher HHV than APS. Along with the decrease in ash content, the increase in energy content after HWE is favorable for use of hot-water extracted lignocellulosics, including APS, for combustion purposes [24].
2.2.5. Antioxidizing Activity (AOA) of APS Hot-Water Extract
HW-extract of lignocellulosic biomass is expected to contain components with AOA [37]. The IC50 of evaporated supernatant was determined to be 0.116 mg/mL. This value indicates a lower AOA for HW-extract than for the organic extract of APS (0.029 mg/mL) but is still favorable as compared to the AOA of butylated hydroxyanisole (0.653 mg/mL).
2.2.6. Near Infrared Spectroscopy (NIRS) of APS and EAPS
Solid state NIRS analysis of APS and EAPS was performed. Quantitative analysis of lignocellulosic biomass using NIRS requires statistical models to analyze bond vibration overtones (OT) in the NIRS range. Existing models for lignocellulosic material are generally not applicable across species [38]. Due to the limited variety of APS and EAPS samples available a model specific to APS could not be constructed and, therefore, quantitative analysis was not feasible. Interpretation of spectra was conducted on raw spectra, as well as second derivative spectra. Key wavelengths are labeled in spectra according to numbers in Table 5.

Table 5.
Wavelengths of interest for NIRS analysis.
| Number | Wavelength (nm) | Bond vibration | Assignment | Reference |
|---|---|---|---|---|
| 1 | 1375 | 1st OT C-H stretch | Hemicelluloses | [39] |
| 2 | 1428 | 1st OT O-H stretch | Amorphous cellulose | [40] |
| 3 | 1725 | 1st OT C-H stretch | Hemicelluloses | [41] |
| 4 | 1907,1910 | 2nd OT C=O stretch | Hemicelluloses | [39] |
| 5 | 2267 | O-H/C-O stretch | Lignin | [39] |
Raw spectra showed near uniform reduction in absorption at associated wavelengths for all constituents. Thus, second derivative spectra proved more suitable for interpretation of chemical changes caused by HWE (Figure 3). Decrease of absorption band intensity at 1428 nm indicates a degradation of amorphous cellulose. Existing research has shown decreased absorption at this amorphous cellulose related band following hydrothermal treatment [42]. This is in agreement with wet chemical analysis indicating ~4% removal of cellulose, which is undoubtedly from amorphous regions of APS cellulose. Furthermore, degree of crystallinity of cellulose was calculated using NIR spectra and Equation (1) [42]:
in this equation, CI and CII represent the integral intensities of O-H bands corresponding to crystalline regions in cellulose (1550 nm ± 10 nm and 1590 nm ± 10 nm, respectively), and Am represents the integral intensity of O-H bands corresponding to amorphous regions in cellulose (<1428 nm). Results indicate that HWE leads to an increase in degree of crystallinity from 48.9% to 56.3%. This corroborates relatively low amount of glucans removed during HWE.
Decrease of absorption band at 1728 nm due to HWE confirms results of 1H-NMR, as this wavelength is hemicellulose-associated [43]. The peak found at 1890 nm (in raw spectra) is most likely a combination of bound water and hemicelluloses; this peak was greatly reduced in EAPS and APS following drying. HWE results in decreased hygroscopicity, which explains the decreased absorption band intensity for EAPS [44]. This reduction in hygroscopicity is related to the removal of hemicellulose sugars, which represent the most hydrophilic constituent of lignocellulosics. Decreased absorption intensities at 1910 nm, 1725 nm, and 1375 nm confirm results of 1H-NMR indicating ~77% reduction in total xylan sugars following HWE. Finally, absorption at 2267 nm indicates changes to lignin or phenolic compounds in APS following HWE. Absorption peaks at this wavelength have been attributed to O-H and C-H stretching vibrations as well as C-H combination vibrations associated with alkylbenzene structures [45]. This could indicate modification of lignin fraction during HWE resulting in altered residual lignin structures in EAPS. This could also be a result of an increase in phenolic hydroxyl groups due to HWE, as shown in the literature [18].
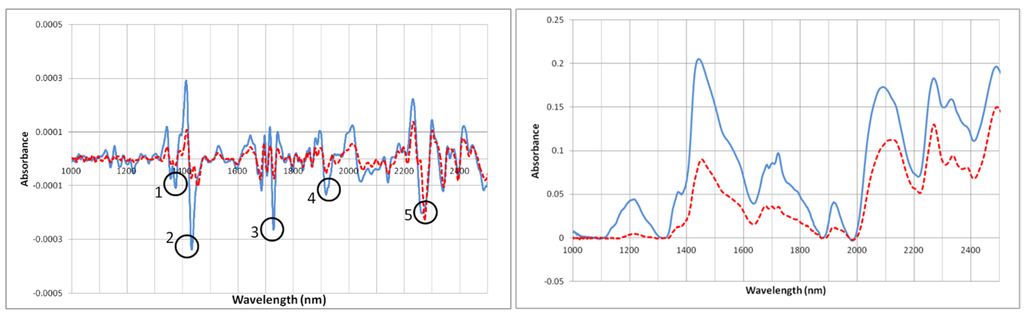
Figure 3.
Raw and 2nd derivative NIR spectra of APS (solid) and EAPS (dashed), labels correspond to Table 5.
3. Experimental Section
Apricot (Prunus armeniaca L.) pit shells (APS) were provided by Dr. Muhammad Zia-ul-Haq (Intellectual Property Organization: Chemical Sciences, Karachi, Pakistan). APS were air dried, milled using a Willey mill to 30 mesh, and stored in sealed plastic bags until use when the moisture content was determined using TAPPI Standard method T264 cm-07: Preparation of wood for chemical analysis.
Ash content was determined in accordance with TAPPI Standard T 211 om-02: Ash in wood, pulp, paper, and paperboard: combustion at 525 °C. Cellulose content was determined by the Kürschner-Hoffer (KH) method [28].
Lignin content, including acid soluble lignin, was determined in accordance with a modified Klason lignin method [46]. For lignin determination, APS were pre-extracted sequentially with hexane, toluene/ethanol (2/1 vol/vol), and methanol (Soxhlet apparatus), followed by cold water (hot-water solubility after the same sequential organic extraction was also determined). HW-extracted apricot pit shells (EAPS) were pre-extracted with dichloromethane (DCM) as it has been previously reported that common solvents used in pre-extraction for lignin determination dissolve lignin from HW-extracted biomass [17]. Soxhlet extractions were performed in accordance with TAPPI Standard T 204 cm-97: Solvent extractives of wood and pulp. Cold/hot-water solubility was determined in accordance with TAPPI Standard T 207 cm-99: Water solubility of wood and pulp. Pre-extraction with DCM was performed by sonication for two hours with fresh solvent added after one hour (Branson 3510 ultrasonic bath). All extraction results are an average of two experiments except dichloromethane.
Sugar content was determined by a modified acid hydrolysis procedure similar to that described by Alves et al. [30], followed by 1H-NMR analysis [30,31]. One hundred and fifty milligrams of APS (on oven-dry basis (OD); 30 mesh) was placed in a 15 mL screw-top pressure vessel along with 4.8 mL 72% sulfuric acid. This slurry was sonicated at room temperature for one hour after which 6.3 mL DI water were added to dilute the sulfuric acid to 40%. The mixture was then placed in a water bath at 80 °C for one hour with intermittent mixing. The lignin was allowed to precipitate in the cold room overnight. The next day the samples were filtered. All spectra were acquired at 30 °C with a Bruker AVANCE III 600 spectrometer (600 MHz 1H frequency) equipped with a 5 mm Cryo Prodigy BBO z-gradient probe. Data were acquired and processed in TOPSPIN v3.2 from Bruker BioSpin. Glucosamine was used as the internal standard [30,31]. The results are an average of four experiments.
Uronic acid content was quantified by decarboxylation using a modification of the method described by Barker et al involving digestion in 19% HCl at 145 °C for 2.5 h followed by titration of CO2 trapped in NaOH solution [36]. Due to the glassware available all reagent volumes were doubled. A blank value was determined in triplicate followed by a three-point standard curve in duplicate using 98+% glucuronic acid (Alfa Aesar). The curve fit had an R2 value of 0.913. Uronic acid content of APS and EAPS was determined based on the produced standard curve. Reported values are an average of two experiments.
Hot water extraction of the ground apricot seed shells (APS; 30 mesh) was performed in a 300 mL Parr reactor (4560 mini bench top reactor) at 160 °C; 20 min temperature ramp; two hours at maximal temperature; 5 grams of biomass (OD); 20:1 water-to-wood ratio. Extract was collected by filtration and the pH of the extract was recorded at room temperature. The extracted APS (EAPS) were washed thoroughly with cold water prior to being air-dried. The reported yield is an average of eight experiments. Following recovery of the HWE liquor, lignin precipitation and recovery was performed. The pH was dropped to 2.0 ± 0.01 by addition of 20% sulfuric acid and the pH-adjusted liquor was allowed to sit in the cold room for at least one week to allow for complete lignin precipitation. The liquor was then centrifuged and the residue was dried in the vacuum oven at 40 °C.
To assess the effect of HWE on delignification efficiency, acetone/water (AW; 9/1 vol/vol) extraction of EAPS was performed (120 °C, 45 min) in the Parr reactor described above. After rotary evaporation of the AW extract the lignin content of solid residue was determined by the Klason method, including acid soluble lignin [46].
Higher heating value (HHV) of oven-dried APS and EAPS samples were determined by calorimetry performed on a Parr Calorimeter 6200 instrument in accordance with ASTM method D5865-13: Standard test method for gross calorific value of coal and coke. Due to the difficulty in making a pellet out of the APS and EAPS material, benzoic acid was used to assist. The results are an average of three experiments. For comparison, HHV of sugar maple (Acer saccharum) and HW-extracted sugar maple was determined as well.
Antioxidizing activity (AOA) of two extracts of APS (toluene/ethanol 2/1 vol/vol, sonicator, and solids recovered from evaporation of HWE supernatant) were determined by a UV-VIS spectrometric method as described by Berger et al. [47]. Solid extract dissolved in methanol was used to quench the free radical 1,1-diphenyl-2-picrylhydrazyl (DPPH). Upon being quenched DPPH undergoes a color change that is detected by UV-VIS at 515 nm (Thermo Scientific, Genesys 10 UV-VIS). A serial dilution of the extracts was prepared to determine the IC50 of the extracts.
A Bruker FT-NIR Multi Purpose Analyzer spectrophotometer was used for collection of NIR spectra from ground APS and EAPS samples. Samples were all ground to 40 mesh as differences in particle size is known to influence scattering patterns, altering spectra shape [48]. Samples were scanned at 8 cm−1 resolution combining sixteen scans per spectra. Samples were vacuum-dried at 40 °C for 72 h prior to analysis.
4. Conclusions
It has been confirmed that apricot pit shells (APS) are a lignocellulosic biomass source with relatively high lignin and energy content. This makes APS an attractive material for various valorization methods, including pyrolysis and pelletization. Organic and aqueous extracts of APS were shown to have strong antioxidizing properties indicating that APS could potentially be a feedstock for the production of antioxidant supplements, preservatives, and stabilizers.
APS were shown to be rather uniquely susceptible to hot water extraction as a relatively mild pretreatment option for biorefinery type operations. Nearly 80% of total xylan was removed along with nearly 25% of lignin. Additional lignin was removed with subsequent acetone/water extraction resulting in a total delignification of ~35% without harsh conditions or chemicals. While most of the cellulose remained undissolved during HWE, NIRS results indicated that the crystallinity of the remaining cellulose increased. Removal of more than 80% of inorganics originally present in APS, along with the increase in energy of combustion, makes EAPS even more suitable for valorization.
NIR spectroscopy is remarkably simple to use, although its use as a quantitative tool is hindered by the need for multiple well-defined samples for production of a statistical model. In this study the results of NIR spectroscopy corroborated the results of traditional/standard methods combined with 1H-NMR.
Acknowledgments
This research was financially supported by USDA-McIntire-Stennis “Enhancing production of uniform high purity lignin” and National Science Foundation (NSF): “Technology Enhancement of Hot Water Extraction” research grants. We wish to extend our sincere gratitude to Muhammad Zia-ul-Haq (Intellectual Property Organization: Chemical Sciences, Karachi, Pakistan) for providing apricot pit shells. We also wish to thank David J. Kiemle (Analytical & Technical Services, SUNY-ESF) and Zachary Spangler for assistance in NMR and antioxidizing activity experiments, respectively.
Author Contributions
Derek Corbett: HWE of APS, solvent extractions, KH cellulose analysis, klason lignin analysis, ash analysis, acid hydrolysis and 1H-NMR sugars analysis, energy of combustion analysis, AOA analysis, uronic acids analysis, writing and editing. Neil Kohan: NIRS analysis, energy of combustion analysis, author of NIRS section, editing. Grazielle Machado: HWE of APS and acetone:water extractions of EAPS. Chengyan Jing: HWE of APS, klason lignin analysis, lignin recovery from HW-extract. Aditi Nagardeolekar: Valuable assistance with uronic acids determination. Dr. Biljana Bujanovic: Advising, writing, and editing.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Yiğit, D.; Yiğit, N.; Mavi, A. Antioxidant and antimicrobial activities of bitter and sweet apricot (Prunus armeniaca L.) kernels. Braz. J. Med. Biol. Res. 2009, 42, 346–352. [Google Scholar] [PubMed]
- Ali, S.; Masud, T.; Abbasi, K.S. Physico-chemical characteristics of apricot (Prunus armeniaca L.) grown in Northern Areas of Pakistan. Sci. Hortic. 2011, 130, 386–392. [Google Scholar] [CrossRef]
- Campbell, O.E.; Merwin, I.A.; Padilla-Zakour, O.I. Characterization and the effect of maturity at Harvest on the Phenolic and Carotenoid Content of Northeast USA Apricot (Prunus armeniaca) Varieties. J. Agric. Food Chem. 2013, 61, 12700–12710. [Google Scholar] [CrossRef] [PubMed]
- Campbell, O.E.; Padilla-Zakour, O.I. Phenolic and carotenoid composition of canned peaches (Prunus persica) and apricots (Prunus armeniaca) as affected by variety and peeling. Food Res. Int. 2013, 54, 448–455. [Google Scholar] [CrossRef]
- Ercisli, S. Apricot culture in Turkey. Sci. Res. Essays 2009, 4, 715–719. [Google Scholar]
- Food and Agriculture Organization of the United Nations. Available online: http://faostat3.fao.org/download/Q/QC/E (accessed on 1 May 2015).
- Lamb, R.; Stiles, W. Apricots for New York State. New York’s Food Life Sci. Bull. 1983, 100, 1–4. [Google Scholar]
- Soleimani, M.; Kaghazchi, T. Adsorption of gold ions from industrial wastewater using activated carbon derived from hard shell of apricot stones—An agricultural waste. Bioresour. Technol. 2008, 99, 5374–5383. [Google Scholar] [CrossRef] [PubMed]
- Aygün, A.; Yenisoy-Karakaş, S.; Duman, I. Production of granular activated carbon from fruit stones and nutshells and evaluation of their physical, chemical and adsorption properties. Microporous Mesoporous Mater. 2003, 66, 189–195. [Google Scholar] [CrossRef]
- Soleimani, M.; Kaghazchi, T. Agricultural waste conversion to activated carbon by chemical activation with phosphoric acid. Chem. Eng. Technol. 2007, 30, 649–654. [Google Scholar] [CrossRef]
- Savova, D.; Apak, E.; Ekinci, E.; Yardim, F.; Petrov, N.; Budinova, T.; Razvigorova, M.; Minkova, V. Biomass conversion to carbon adsorbents and gas. Biomass Bioenergy 2001, 21, 133–142. [Google Scholar] [CrossRef]
- Cañellas, J.; Femenia, A.; Rosselló, C.; Soler, L. Chemical composition of the shell of apricot seeds. J. Sci. Food Agric. 1992, 59, 269–271. [Google Scholar] [CrossRef]
- Gezer, İ.; Haciseferoğulları, H.; Demir, F. Some physical properties of Hacıhaliloğlu apricot pit and its kernel. J. Food Eng. 2003, 56, 49–57. [Google Scholar] [CrossRef]
- Lange, J.-P. Lignocellulose conversion: An introduction to chemistry, process and economics. Biofuels Bioprod. Biorefining 2007, 1, 39–48. [Google Scholar] [CrossRef]
- Amidon, T.E.; Bujanovic, B.; Liu, S.; Howard, J.R. Commercializing biorefinery technology: A case for the multi-product pathway to a viable biorefinery. Forests 2011, 2, 929–947. [Google Scholar] [CrossRef]
- Ye, P.; Cheng, L.; Ma, H.; Bujanovic, B.; Goundalkar, M.J.; Amidon, T.E. Biorefinery with Water. In The Role of Green Chemistry in Biomass Processing and Conversion; Xie, H., Gathergood, N., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2012; pp. 135–180. [Google Scholar]
- Gong, C.; Goundalkar, M.J.; Bujanovic, B.M.; Amidon, T.E. Evaluation of different sulfur-free delignification methods for hot-water extracted hardwood. J. Wood Chem. Technol. 2012, 32, 93–104. [Google Scholar] [CrossRef]
- Gong, C.; Bujanovic, B.M. Impact of hot-water extraction on acetone-water oxygen delignification of paulownia spp. and lignin recovery. Energies 2014, 7, 857–873. [Google Scholar] [CrossRef]
- Uygut, M.; Corbett, D.B.; Wood, C.; Zelie, M.; Bujanovic, B. Potential for enhancement of enzymatic hydrolysis of sugar maple (acer saccharum). In Proceedings of the 249th ACS National Meeting and Exposition, Denver, CO, USA, 22–26 March 2015.
- Mante, O.D.; Amidon, T.E.; Stipanovic, A.; Babu, S.P. Integration of biomass pretreatment with fast pyrolysis: An evaluation of electron beam (EB) irradiation and hot-water extraction (HWE). J. Anal. Appl. Pyrolysis 2014, 110, 44–54. [Google Scholar] [CrossRef]
- Murphy, A. Selected topics in the utilization of natural resources: Production of transportation fuel and potential biomass source for use in biorefinery. Bachelor’s Honors Thesis, Department of Paper and Bioprocess Engineering, College of Environmental Science and Forestry, State University of New York, Syracuse, NY, USA, 2014. [Google Scholar]
- Wei, L.; Xu, S.; Zhang, L.; Zhang, H.; Liu, C.; Zhu, H.; Liu, S. Characteristics of fast pyrolysis of biomass in a free fall reactor. Fuel Process. Technol. 2006, 87, 863–871. [Google Scholar] [CrossRef]
- Yeganeh, M.M.; Kaghazchi, T.; Soleimani, M. Effect of raw materials on properties of activated carbons. Chem. Eng. Technol. 2006, 29, 1247–1251. [Google Scholar] [CrossRef]
- Kaynak, B.; Topal, H.; Atimtay, A.T. Peach and apricot stone combustion in a bubbling fluidized bed. Fuel Process. Technol. 2005, 86, 1175–1193. [Google Scholar] [CrossRef]
- Demirbaş, A. Relationships between lignin contents and heating values of biomass. Energy Convers. Manag. 2001, 42, 183–188. [Google Scholar] [CrossRef]
- Demirbaş, A. Effect of temperature on pyrolysis products from four nut shells. J. Anal. Appl. Pyrolysis 2006, 76, 285–289. [Google Scholar] [CrossRef]
- Sjöström, E.; Westermark, U. Chemical composition of wood and pulps: Basic constituents and their distribution. In Analytical Methods in Wood Chemistry, Pulping, and Papermaking; Sjöström, P.D.E., Alén, P.D.R., Eds.; Springer: New York, NY, USA, 1999; pp. 1–19. [Google Scholar]
- Browning, B.L. Methods of Wood Chemistry; John Wiley & Sons, Inc.: New York, NY, USA, 1967; Volume 1. [Google Scholar]
- Bose, S.K.; Barber, V.A.; Alves, E.F.; Kiemle, D.J.; Stipanovic, A.J.; Francis, R.C. An improved method for the hydrolysis of hardwood carbohydrates to monomers. Carbohydr. Polym. 2009, 78, 396–401. [Google Scholar] [CrossRef]
- Alves, E.F.; Bose, S.K.; Francis, R.C.; Colodette, J.L.; Iakovlev, M.; van Heiningen, A. Carbohydrate composition of eucalyptus, bagasse and bamboo by a combination of methods. Carbohydr. Polym. 2010, 82, 1097–1101. [Google Scholar] [CrossRef]
- Kiemle, D.J.; Stipanovic, A.J.; Mayo, K.E. Proton NMR methods in the compositional characterization of polysaccharides. In Hemicelluloses: Science and Technology; Gatenholm, P., Tenkanen, M., Eds.; American Chemical Society: Washington, DC, USA, 2004; Volume 864, pp. 122–139. [Google Scholar]
- Shin, S.-J.; Cho, N.-S. Conversion factors for carbohydrate analysis by hydrolysis and 1H-NMR spectroscopy. Cellulose 2007, 15, 255–260. [Google Scholar] [CrossRef]
- Willför, S.; Pranovich, A.; Tamminen, T.; Puls, J.; Laine, C.; Suurnäkki, A.; Saake, B.; Uotila, K.; Simolin, H.; Hemming, J.; Holmbom, B. Carbohydrate analysis of plant materials with uronic acid-containing polysaccharides—A comparison between different hydrolysis and subsequent chromatographic analytical techniques. Ind. Crops Prod. 2009, 29, 571–580. [Google Scholar] [CrossRef]
- Willför, S.; Sundberg, A.; Pranovich, A.; Holmbom, B. Polysaccharides in some industrially important hardwood species. Wood Sci. Technol. 2005, 39, 601–617. [Google Scholar] [CrossRef]
- Mittal, A.; Chatterjee, S.G.; Scott, G.M.; Amidon, T.E. Modeling xylan solubilization during autohydrolysis of sugar maple and aspen wood chips: Reaction kinetics and mass transfer. Chem. Eng. Sci. 2009, 64, 3031–3041. [Google Scholar] [CrossRef]
- Barker, S.A.; Foster, A.B.; Siddiqui, I.R.; Stacey, M. Uronic acid determination. Talanta 1958, 1, 216–218. [Google Scholar] [CrossRef]
- Garrote, G.; Cruz, J.M.; Moure, A.; Domı́nguez, H.; Parajó, J.C. Antioxidant activity of byproducts from the hydrolytic processing of selected lignocellulosic materials. Trends Food Sci. Technol. 2004, 15, 191–200. [Google Scholar] [CrossRef]
- Via, B.K. Modeling Longleaf pine (Pinus palustris Mill) wood properties using near infrared spectroscopy. Ph.D. Thesis, Louisiana State University, Baton Rouge, LA, USA, 2004. [Google Scholar]
- Schwanninger, M.; Rodrigues, J.C.; Fackler, K. A review of band assignments in near infrared spectra of wood and wood components. J. Infrared Spectrosc. 2011, 19, 287. [Google Scholar] [CrossRef]
- Tsuchikawa, S.; Siesler, H.W. Near-infrared spectroscopic monitoring of the diffusion process of deuterium-labeled molecules in wood. Part I: Softwood. Appl. Spectrosc. 2003, 57, 667–674. [Google Scholar] [CrossRef] [PubMed]
- Sandak, A.; Sandak, J.; Negri, M. Relationship between near-infrared (NIR) spectra and the geographical provenance of timber. Wood Sci. Technol. 2011, 45, 35–48. [Google Scholar] [CrossRef]
- Inagaki, T.; Siesler, H.W.; Mitsui, K.; Tsuchikawa, S. Difference of the crystal structure of cellulose in wood after hydrothermal and aging degradation: A NIR spectroscopy and XRD study. Biomacromolecules 2010, 11, 2300–2305. [Google Scholar] [CrossRef] [PubMed]
- Mitsui, K.; Inagaki, T.; Tsuchikawa, S. Monitoring of hydroxyl groups in wood during heat treatment using NIR spectroscopy. Biomacromolecules 2007, 9, 286–288. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Hosseinaei, O.; Wang, S.; Zhou, Z. Influence of hemicellulose extraction on water uptake behavior of wood strands. Wood Fiber Sci. 2011, 43, 244–250. [Google Scholar]
- Chesson, A.; Ørskov, E.R. Physico-Chemical Characterisation of Plant Residues for Industrial and Feed Use; Elsevier Science Publishers Ltd: Essex, UK, 1989. [Google Scholar]
- Dence, C.W. The Determination of Lignin. In Methods in Lignin Chemistry; Lin, S.Y., Dence, C.W., Eds.; Springer: Berlin, Germany, 1992; pp. 33–61. [Google Scholar]
- Berger, J.M.; Rana, R.J.; Javeed, H.; Javeed, I.; Schulien, S.L. Radical quenching of 1, 1-diphenyl-2-picrylhydrazyl: A spectrometric determination of antioxidant behavior. J. Chem. Educ. 2008, 85, 408. [Google Scholar] [CrossRef]
- Shenk, J.S.; Workman, J.J.; Westerhaus, M.O. Application of NIR spectroscopy to agricultural products. Pract. Spectrosc. Ser. 2001, 27, 419–474. [Google Scholar]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).