Fractionation for Biodiesel Purification Using Supercritical Carbon Dioxide
Abstract
: In recent years, biodegradable and alternative biodiesel has attracted increased attention worldwide. Producing biodiesel from biomass involves critical separation and purification technology. Conventional technologies such as gravitational settling, decantation, filtration, water washing, acid washing, organic solvent washing and absorbent applications are inefficient, less cost effective and environmentally less friendly. In this study supercritical carbon dioxide (SC-CO2) with few steps and a low environmental impact, was used for biodiesel fractionation from impure fatty acid methyl ester (FAME) solution mixes. The method is suitable for application in a variety of biodiesel production processes requiring subsequent stages of purification. The fractionation and purification was carried out using continuous SC-CO2 fractionation equipment, consisting of three columns filled with stainless steel fragments. A 41.85% FAME content solution mix was used as the raw material in this study. Variables were a temperature range of 40–70 °C, pressure range of 10–30 MPa, SC-CO2 flow rate range of 7–21 mL/min and a retention time range of 30–90 min. The Taguchi method was used to identify optimal operating conditions. The results show that a separated FAME content of 99.94% was verified by GC-FID under optimal fractionation conditions, which are a temperature of 40 °C of, a pressure level of 30MPa and a flow rate of 7 mL/min of SC-CO2 for a retention time of 90 min.1. Introduction
As a result of population growth, industrial development and transportation expansion, energy demands are increasing. Due to problems associated with the widespread use of fossil fuels, future supply constraints, global climate change and other energy concerns, it is increasingly necessary to develop renewable energy sources such as biofuels (biodiesel or ethanol), biomass, and other alternative energies.
Biodiesel is an alternative fuel formulated specifically for diesel engines and has similar physicochemical properties to conventional diesel [1]. Generally, biodiesel is a mixture of fatty acid methyl esters (FAMEs), which can be produced primarily from oils or lipids of food crops, waste food by-products [2] and microalgae [3,4]. Biodiesel obtained from energy crops not only affects the environment more favorably, but also can help develop new industries, such as the agro-energy industry, thereby creating employment opportunities and boosting regional development. For these reasons, this renewable and environmentally friendly biofuel has the potential to provide alternative energy sources in the future by replacing exhaustible fossil fuels as the main energy supply. Despite the enormous benefits of biodiesel, food crops shortages, expensive feedstock, high processing costs and complicated purification steps have acted as barriers to its development [5].
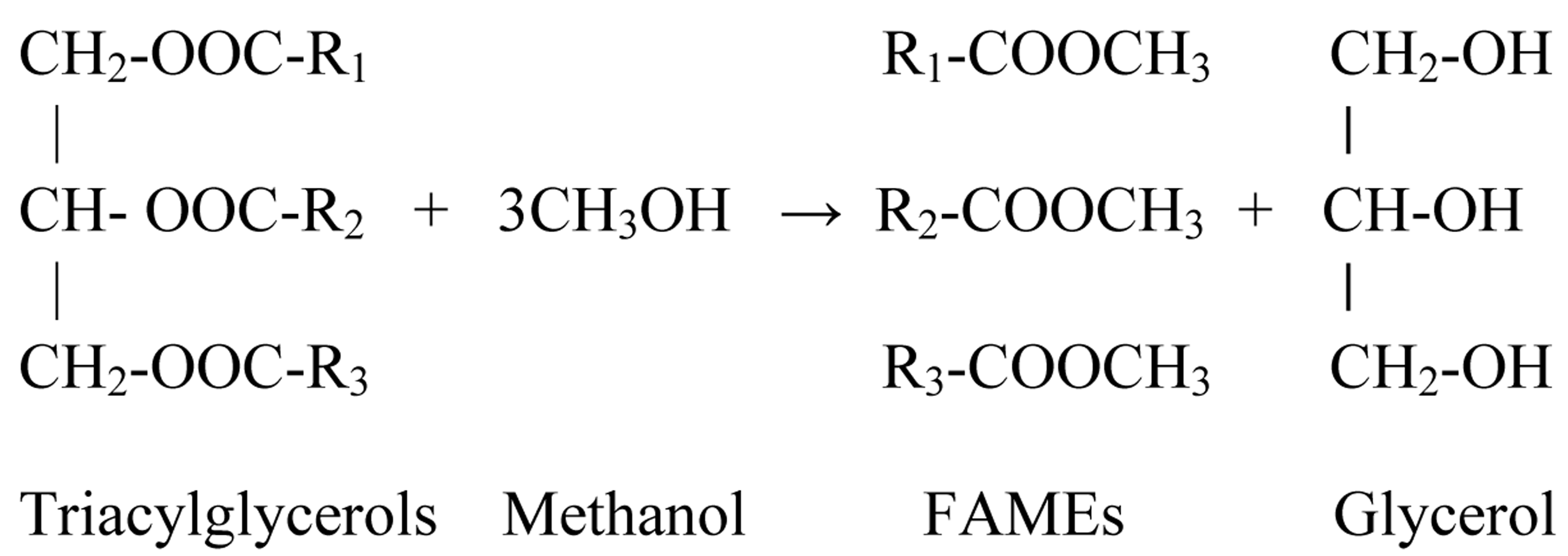
As presented in Scheme 1, biodiesel transesterification is a chemical reaction involving the transesterification of triacylglycerols into FAMEs, by replacing the glycerol of the triacylglycerols with a short-chain alcohol. This may be done using different processes [6]. Most commonly, biodiesel production can be catalyzed by utilizing alkaline, acidic, enzymatic, or other kinds of catalysts [7]. In addition, biodiesel can be processed from triacylglycerols via non-catalytic transesterification with supercritical alcohol [8,9].
In the production of biodiesel from biomass, separation and purification are critical technical requirements. Even though recent transesterification technology obtained high conversion yields in a short time, the production is energy intensive and the catalyst has to be removed from the product at the end. Because free fatty acids (FFA) and water will interfere with the process, recovery of glycerol is laborious and alkaline wastewater requires further treatment. The presence of water lowers the activity of the catalyst, while the FFA reacts with the catalyst to produce a saponified product. The formation of soap reduces the biodiesel yield and quality. Accordingly, it would cause significant difficulty in the following separation and purification processes. Thus, biodiesel and its byproduct, glycerol, have to be refined by being washed with hot deionized water several times, leading to increased time, energy and water consumption [10].
Ineffective biodiesel separation and purification not only creates the risk of failing to meet international standards, but also may cause severe problems for diesel engines. After going through various biodiesel manufacturing processes, crude product may consist of the main product, FAME, and by-product water, glycerol, raw materials impurities, soap, raw oil, alcohol, acids, bases, catalyst and other minor components. As a result, most of the time further purification processes are needed. Many technologies have been used for biodiesel separation and purification such as gravitational settling, decantation, membrane separation, filtration, water washing, acid washing, and solvent washing. Other methods involve passing the crude biodiesel product over activated carbon or applying relatively strong oxidants, typical liquid-liquid multistage extractions, or by using distillation for separating the products from impurities. Yet most of these methods have been proven to be inefficient, time and energy consuming, and less cost effective [11].
There are several supercritical fluid related applications of biodiesel production, such as one in which SC-CO2 was used as a medium for enzymatic catalytic biodiesel production [12]. Supercritical alcohol can be used as a medium and raw material in non-catalytic biodiesel production. On the topic of reducing by products, such as glycerol, during biodiesel production process, supercritical methyl acetate [13,14] and supercritical dimethyl carbonate [15] were used to replace the supercritical alcohol. However, methyl acetate and dimethyl carbonate are toxic materials and lower ignition properties. Wei et al. developed an alternative methodology by using supercritical methanol plus SC-CO2 with acetic acid, in which higher FAME content was increased and glycerol byproduct was reduced by 30% [16].
SC-CO2 is a popular, green and environmentally friendly solvent, as is safe, cheap, and readily available, which is advantageous for industrial scale production. SC-CO2 operations can be diversified based on selectivity of the solvents and their characteristics. Reverchon and De Marco have completed a review study including mathematical modeling and various applications for natural product separation using supercritical fluids and fractionation [17]. Fractionation systems can be mounted to SC-CO2 equipment for simultaneously conducting two stages synthesis and separation of natural product material. SC-CO2 fractionation allows the fractionation of the mixture, while operating with some separators in a series at different pressures and temperatures. With the alteration of pressure or temperature, the density of SC-CO2 changes, and as a result there is a change in solvent properties. Because the scope of such operations changes the solvent properties, this induces the selective precipitation of different compound families as a function of their different saturation conditions in the SC-CO2. Compared with SC-CO2 extraction from solids, the packed fractionation column is capable of continuous steady state operations that allow for the processing of large quantities of liquid mixtures in a short time. Therefore, the lipophilic characteristics of the SC-CO2 are advantageous when polar compounds like FAMEs can be fractionated from a mixed biodiesel. Rezayat and Ghaziaskar used a continuous-supercritical-fluid-fractionation setup to extract glycerol acetates from a mixture of glycerol acetates, in which the single fractionation column consists of a sample container and an extraction column [18]. A selective separation of the oil components based on their polarity and molecular weight can be attained. This involves the continuous contact between SC-CO2 and the liquid mixture in a pressurized precipitation vessel. If the process is performed at optimized conditions, the liquid solution can be fractionated by SC-CO2 and part of the elements precipitate in a non-soluble liquid phase at the bottom of the high pressure vessel.
Enormous efforts have been directed toward replacing the conventional methods of purification of natural materials with new SC-CO2 fractionation techniques. In this work, impure, crude biodiesel with low FAME content was used to investigate the feasibility of purification with SC-CO2 in a continuous fractionation column. The goal is to recover FAME at or above the 96.5% purity international standard for FAME (ASTM D6751 for USA and Canada and EN14214 for Europe) [19].
2. Experimental Section
2.1. Experimental Planning
The experimental plan for this research was carried out using the Taguchi design method [20] and an analysis of variance (ANOVA). The Taguchi method systematically applies design and analysis principles to experiments, allowing the use of smaller amounts of experimental data to effectively identify improvements in the production process. This method can be employed to design processes and improve product quality by using economically optimized manufacturing processes which may involve multiple factors at different levels. With the Taguchi method, orthogonal arrays are used to reduce the parameter numbers of the experimental trials to a practical yet effective level. Where a great number of independent variables are involved in the design of an experiment, the Taguchi method allows for more-sophisticated analyses of such variables with a smaller number of experiments.
The signal-to-noise ratio (S/N ratio, η) is a ratio of the average and standard deviation calculated from the experimental data. The S/N ratio is the way the Taguchi method statistically measures the quality of fractionation. The S/N ratio equation depends on the criterion of the three quality characteristics to be optimized: higher-the-better, normal-the-better, and smaller-the-better. In this work on the fractionation of biodiesel with higher FAME contents, the S/N ratio of FAMEs in biodiesel (the higher-the-better function) was used as follows [Equation (1)]:
A method utilizing SC-CO2 to fractionation biodiesel resulted in higher FAME contents. This method may assist in meeting international standards for biodiesel production. A number of independent variables are involved in the fractionation of biodiesel, including the fractionation temperature, fractionation pressure, SC-CO2 flow rate and retention time. Taguchi L9 34 table was used, including standard Taguchi L9 orthogonal array, requiring nine experimental trials (Table 1). In this study, the influences of four variables (fractionation temperature, fractionation pressure, SC-CO2 flow rate and retention time) on FAME contents were investigated. The experimental error was determined by conducting two replicate experiments with detailed statistical analyses of the results. The levels and values of all variables applied in the experiments are presented in Table 2.
2.2. Fractionation Procedure
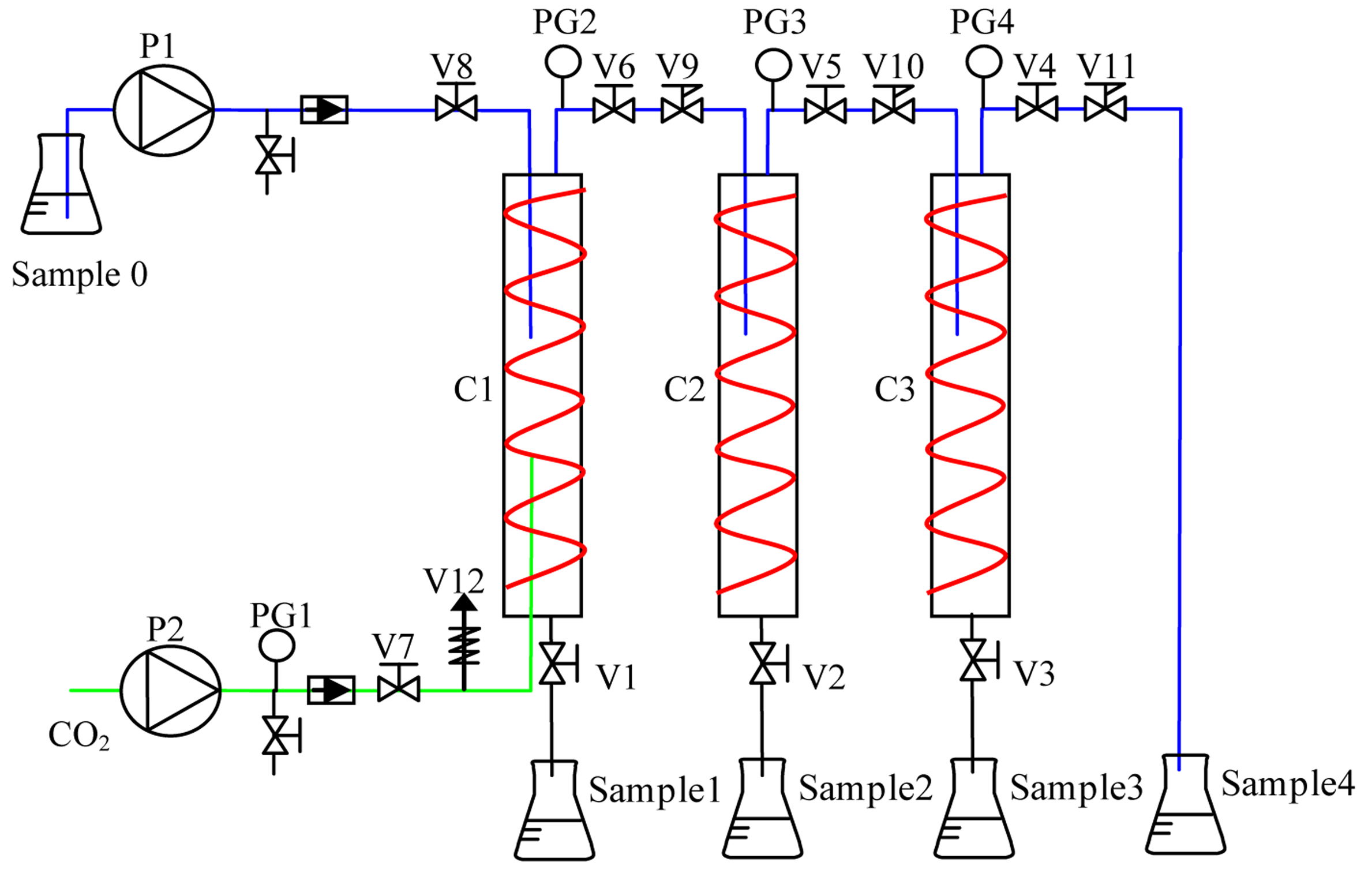
In this study, an impure FAME mixing solution was used in a SC-CO2 fractionation system (Lian-Sheng, Taichung, Taiwan) which was obtained from the waste biodiesel sample. All runs of the experiment were performed in three 500 mL columns (high pressure vessels with a heater jacked inside) made of stainless steel (SS 316). They were packed with stainless steel fragments. The schematic diagram of SC-CO2 fractionation is shown in Figure 1. In a typical run, the system was heated with an electrical heater jacket and the power was adjusted to create a system temperature of (40–70 °C). If the first column of system was pressurized at 30 MPa, the second and the third column were reduced by increments of 5 MPa, respectively. In other words, the second column was at 25 MPa and the third one at 20 Mpa. The system was pressurized with bottom feeding CO2 (Jing-Shang Gas, Kaohsiung, Taiwan) using a high pressure meter pump (Teledyne Isco, Lincoln, NE, USA). After the setup of fractionation conditions, a sample (70 mL) was loaded and pumped into the first column from the top using a high-pressure liquid pump with a 6 mL/min flow rate and maintained at a SC-CO2 flow rate of 7–21 mL/min for a retention time of 30–90 min. The sample was pumped out at the middle of the column and encountered SC-CO2. A soluble sample with pressured SC-CO2 was guided to the next column for separation under controlled pressure and temperature. After completion of each fractionation experiment run, the high-pressure valves of each outlet were opened to depressurize and collect the samples. If the first column was loaded with a SC-CO2 of 30 MPa, four samples were all collected. If the pressure started at 10 MPa, two or less samples were collected due to the low amount of SC-CO2 and low solubility.
2.3. Analysis of FAME Content
FAME concentrations of each sample after vacuum evaporation were analyzed (using a modified version of ISO5508) using a gas chromatographic (GC) system (Hewlett-Packard 5890 Packard Series II, Ramsey, MN, USA), equipped with a DB-wax capillary vessel column (30 m × 0.25 mm ID × 0.25 μm film thickness of polyethylene glycol). The detector and injector temperatures were both set to 250 °C. The oven temperature was maintained at a constant 200 °C. Methyl-heptadecanoate (heptadecanoic acid methyl ester) was obtained from Sigma-Aldrich (St. Louis, MO, USA) as an internal standard. All of the analytical assays were performed in duplicate, and mean values are presented. According to the GC integral data, the concentration of FAMEs was calculated by Equation (2):
3. Results and Discussion
3.1. FAME Content
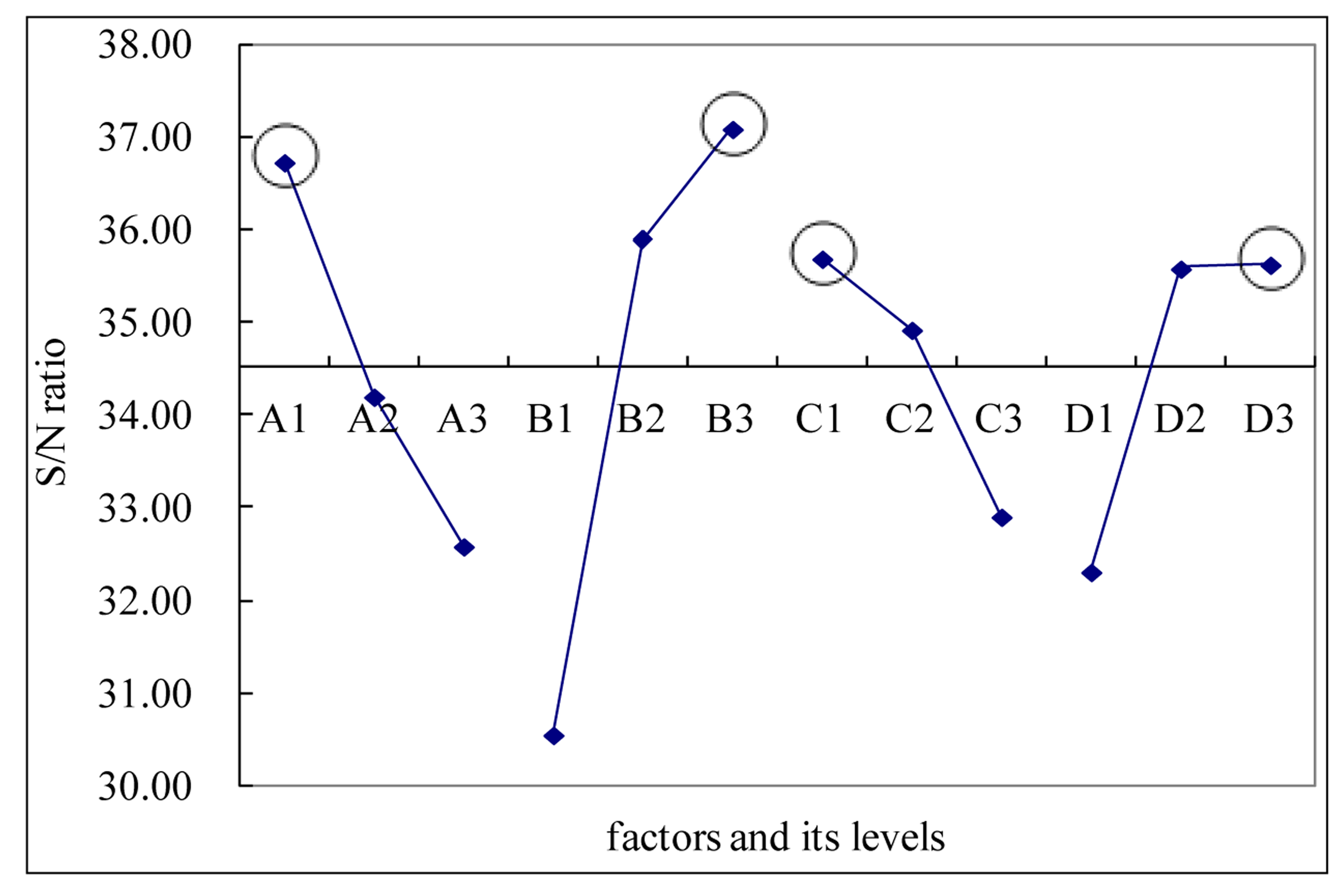
A range of SC-CO2 fractionation conditions was investigated for biodiesel production based on FAME content. Before the fractionation process, the original mixed solution sample has 11.59% evaporable solution and 88.41% oil, with FAME content of 41.85%. After completing the fractionation and collecting samples, the weight and FAME contents were calculated, respectively. In Table 3, the FAME content can be obtained based on a known concentration of HAME (10 mg/mL). The S/N equation depends on the criterion for the quality characteristic to be optimized. The objective in this study was to find the optimum conditions for fractionation which recovers a high amount of the FAME content. Therefore, the higher-the-better loss function [Equation (1)] was used to calculate S/N ratio. The response graph is shown in Figure 2. Figure 2 showed that the optimal conditions for biodiesel purification are A1 (40 °C fractionation temperature), B3 (30 MPa pressure), C1 (7 mL/min SC-CO2 flow rate) and D3 (90 min retention time). A confirmation run was conducted based on these optimum conditions in which the corresponding production of FAME content was 99.94%.
ANOVA is a method to demonstrate how control factors and parameters affect results. Table 4 shows the ANOVA values for biodiesel purification, demonstrating that each fractionation factor significantly affected FAME concentrations in the biodiesel fractionation system. The biodiesel sample obtained was also tested by using them in a diesel generator, which ran smoothly and showed no differences in performance from regular diesel.
3.2. Effect of Fractionation Temperature
In the SC-CO2 process, the temperature and pressure are the main factors to control the behavior of SC-CO2. The effects of the fractionation temperature on the purification of FAME were studied. Results showed that with higher fractionation temperatures, lower FAME content was obtained. Lower temperatures would lead to a higher density and higher viscosity, and more other compounds in the samples could be removed to the next column from the previous column. In this work, more FAME was collected from the first column due to its poor solubility. Accordingly, FAME content in the samples obtained from the first column outlet (Sample 1) in each experiment was higher than the others. At a temperature of 40 °C and a pressure of 30MPa, SC-CO2 demonstrates 0.91g/cm3 high density characteristics in which a good purification can be accomplished.
3.3. Effect of Fractionation Pressure
Pressure is also one of the main factors in controlling the characteristics of a SC-CO2 system. With increased system pressure, the density and solubility of the SC-CO2 is enhanced to carry more impure substances to the next column to achieve the desired separation and purification effects. The results show that higher fractionation pressure, results in better FAME content and better biodiesel purification. The lower fractionation pressure leads to lower solubility and density. At a pressure 10 MPa and a temperature 70 °C, SC-CO2 has a density of 0.25g/cm3. Therefore, an optimized purification effect and higher FAME content was obtained with a fractionation pressure of 30 MPa. Though the high pressure is beneficial to purification, the safety requirements of the container device and the capacity needs of pressurizing pumps are both high, which will increase the cost of production.
3.4. Effect of SC-CO2 Flow Rate
If the SC-CO2 flow rate from the bottom of the column is too fast, insoluble substances like FAMEs will be carried away to the next column. In theory, a slower flow rate for carrying the SC-CO2 will lead to better separation. On the other hand, if the flow rate of SC-CO2 is too slow, the impure soluble substances may remain in the column and not be carried to the next column. In this study, the optimal flow rate in the experiment is 7 mL/min, as it showed the best purification effect.
3.5. Effect of Retention Time
Under the proper flow rate, increased retention times (90 min) for all columns in the process resulted in better purification. Even though all columns were packed to gain the compounds solubility in SC-CO2 in the limited retention time and column space, if retention time was insufficient, the SC-CO2 couldn't carry all the impure substances in the samples away. This would lead to poor purification and a poor conversion rate of biodiesel at shorter retention times.
4. Conclusions
In this research, optimization of a purification process was successfully carried out and it was demonstrated that 99.94% of FAME content can be produced by using SC-CO2 starting with impure biodiesel. It was also demonstrated that the quality of biodiesel from this process meets the international standards ASTM D6751 and EN14214 (96.5% FAMEs). Most SC-CO2 processes are replacing organic solvents so there is also a green chemistry gain for the process. In this biodiesel purification SC-CO2 is not only replacing water solvent, it is replacing the water solution of salts, other additives and their recovery. Without the saponification water in the separated FAME there is no need to evaporate water after separation. The fractionation developed in this work has shown its potential to serve as an alternative to the traditional purification methods, such as chemical or enzymatic methods. The flexibility and adaptability of this process may make it viable for commercial production processes; however, research on this topic is still in progress. The process has been demonstrated to be able to produce high conversion rates for biodiesel. However analyses still need to be carried out with respect to the scaling up of fractionation and purification systems and cost-effectiveness analyses still need to be carried out.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Salvi, B.L.; Panwar, N.L. Biodiesel resources and production technologies—A review. Renew. Sustain. Energy Rev. 2012, 16, 3680–3689. [Google Scholar]
- Borugadda, V.B.; Goud, V.V. Biodiesel production from renewable feedstocks: Status and opportunities. Renew. Sustain. Energy Rev. 2012, 16, 4763–4784. [Google Scholar]
- Halim, R.; Danquah, M.K.; Webley, P.A. Extraction of oil from microalgae for biodiesel production: A review. Biotechnol. Adv. 2012, 30, 709–732. [Google Scholar]
- Rawat, I.; Kumar, R.R.; Mutanda, T.; Bux, F. Biodiesel from microalgae: A critical evaluation from laboratory to large scale production. Appl. Energy 2013, 103, 444–467. [Google Scholar]
- Hasheminejad, M.; Tabatabaei, M.; Mansourpanah, Y.; Khatami Far, M.; Javani, A. Upstream and downstream strategies to economize biodiesel production. Bioresour. Technol. 2011, 102, 461–468. [Google Scholar]
- Marchetti, J.M.; Miguel, V.U.; Errazu, A.F. Possible methods for biodiesel production. Renew. Sustain. Energy Rev. 2007, 11, 1300–1311. [Google Scholar]
- Tariq, M.; Ali, S.; Khalid, N. Activity of homogeneous and heterogeneous catalysts, spectroscopic and chromatographic characterization of biodiesel: A review. Renew. Sustain. Energy Rev. 2012, 16, 6303–6316. [Google Scholar]
- Sawangkeaw, R.; Bunyakiat, K.; Ngamprasertsith, S. A review of laboratory-scale research on lipid conversion to biodiesel with supercritical methanol (2001–2009). J. Supercrit. Fluids 2010, 55, 1–13. [Google Scholar]
- Tan, K.T.; Gui, M.M.; Lee, K.T.; Mohamed, A.R. An optimized study of methanol and ethanol in supercritical alcohol technology for biodiesel production. J. Supercrit. Fluids 2010, 53, 82–87. [Google Scholar]
- Deshpande, A.; Anitescu, G.; Rice, P.A.; Tavlarides, L.L. Supercritical biodiesel production and power cogeneration: Technical and economic feasibilities. Bioresour. Technol. 2010, 101, 1834–1843. [Google Scholar]
- Atadashi, I.M.; Aroua, M.K.; Aziz, A.A. Biodiesel separation and purification: A review. Renew. Energy 2011, 36, 437–443. [Google Scholar]
- Rodrigues, A.R.; Paiva, A.; Silva, M.G.; Simões, P.; Barreiros, S. Continuous enzymatic production of biodiesel from virgin and waste sunflower oil in supercritical carbon dioxide. J. Supercrit. Fluids 2011, 56, 259–264. [Google Scholar]
- Saka, S.; Isayama, Y. A new process for catalyst-free production of biodiesel using supercritical methyl acetate. Fuel 2009, 88, 1307–1313. [Google Scholar]
- Tan, K.T.; Lee, K.T.; Mohamed, A.R. A glycerol-free process to produce biodiesel by supercritical methyl acetate technology: An optimization study via Response Surface Methodology. Bioresour. Technol. 2010, 101, 965–969. [Google Scholar]
- Ilham, Z.; Saka, S. Dimethyl carbonate as potential reactant in non-catalytic biodiesel production by supercritical method. Bioresour. Technol. 2009, 100, 1793–1796. [Google Scholar]
- Wei, C.Y.; Huang, T.C.; Chen, H.H. Biodiesel production using supercritical methanol with carbon dioxide and acetic acid. J. Chem. 2013, 2013. Article ID 789594. Available online: http://dx.doi.org/10.1155/2013/789594 (accessed on 13 February 2013). [Google Scholar]
- Reverchon, E.; De Marco, I. Supercritical fluid extraction and fractionation of natural matter. J. Supercrit. Fluids 2006, 38, 146–166. [Google Scholar]
- Rezayat, M.; Ghaziaskar, H.S. Continuous extraction of glycerol acetates from their mixture using supercritical carbon dioxide. J. Supercrit. Fluids 2011, 55, 937–943. [Google Scholar]
- EN 14214. Available online: http://en.wikipedia.org/wiki/EN_14214 (accessed on 14 February 2014).
- Taguchi, G. Introduction to Quality Engineering; Asian Productivity Organization: Tokyo, Japan, 1990. [Google Scholar]
| L9 | Factors | |||
|---|---|---|---|---|
| A | B | C | D | |
| Run | Temperature | Pressure | Flow rate | Retention time |
| 1 | −1 | −1 | −1 | −1 |
| 2 | −1 | 0 | 0 | 0 |
| 3 | −1 | +1 | +1 | +1 |
| 4 | 0 | −1 | 0 | +1 |
| 5 | 0 | 0 | +1 | −1 |
| 6 | 0 | +1 | −1 | 0 |
| 7 | +1 | −1 | +1 | 0 |
| 8 | +1 | 0 | −1 | +1 |
| 9 | +1 | +1 | 0 | −1 |
| Factors | Parameter | Levels and values | ||
|---|---|---|---|---|
| Low (−1) | Medium (0) | High (1) | ||
| A | Temperature (°C) | 40 | 55 | 70 |
| B | Pressure (MPa) | 10 | 20 | 30 |
| C | Flow rate (mL/min) at 4 °C | 7 | 14 | 21 |
| D | Retention time (mins) | 30 | 60 | 90 |
| Run | FAME content of sample 1 | AVG of Sample weight (g) at each outlet | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Test1 (%) | Test2 (%) | AVG | SD | S/N ratio (db) | 1 | 2 | 3 | 4 | |
| 1 | 37.56 | 40.13 | 38.84 | 1.82 | 31.77 | 52.64 | 0.00 | 0.00 | 0.00 |
| 2 | 99.42 | 91.89 | 95.65 | 5.32 | 39.59 | 28.33 | 4.83 | 7.95 | 2.96 |
| 3 | 85.62 | 89.39 | 87.51 | 2.66 | 38.83 | 1.21 | 0.76 | 1.97 | 41.07 |
| 4 | 36.36 | 41.94 | 39.15 | 3.94 | 31.79 | 54.90 | 0.07 | 0.00 | 0.00 |
| 5 | 35.88 | 42.62 | 39.25 | 4.76 | 31.78 | 26.95 | 4.66 | 10.11 | 0.00 |
| 6 | 93.11 | 86.47 | 89.79 | 4.69 | 39.05 | 47.70 | 1.29 | 4.11 | 0.97 |
| 7 | 23.18 | 28.59 | 25.88 | 3.83 | 28.12 | 54.01 | 0.06 | 0.00 | 0.00 |
| 8 | 69.00 | 61.67 | 65.34 | 5.19 | 36.26 | 40.73 | 8.34 | 5.49 | 1.44 |
| 9 | 44.38 | 49.56 | 46.97 | 3.67 | 33.40 | 33.37 | 3.87 | 5.90 | 3.89 |
| CT | 99.87 | 100 | 99.94 | 0.09 | 39.99 | 4.49 | 1.37 | 13.58 | 34.02 |
Notes: AVG, average; SD, standard deviation; S/N, signal-to-noise ratio; CT, Confirmation Test.
| Factors | SS | DF | MS | F | CF |
|---|---|---|---|---|---|
| A | 2,404.61 | 2 | 1,202.31 | 70.28 | 100.00% |
| B | 5,412.15 | 2 | 2,706.08 | 158.19 | 100.00% |
| C | 601.17 | 2 | 300.58 | 17.57 | 99.92% |
| D | 2,732.17 | 2 | 1,366.09 | 79.86 | 100.00% |
| Error | 153.96 | 9 | 17.11 | - | - |
| Total | 11,304.06 | 17 | - | - | - |
Notes: A: temperature; B: pressure; C: flow rate; D: retention time; SS: sum of squares; DF: degrees of freedom; MS: mean square; F: F ratio, CF: confidence.
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wei, C.-Y.; Huang, T.-C.; Yu, Z.-R.; Wang, B.-J.; Chen, H.-H. Fractionation for Biodiesel Purification Using Supercritical Carbon Dioxide. Energies 2014, 7, 824-833. https://doi.org/10.3390/en7020824
Wei C-Y, Huang T-C, Yu Z-R, Wang B-J, Chen H-H. Fractionation for Biodiesel Purification Using Supercritical Carbon Dioxide. Energies. 2014; 7(2):824-833. https://doi.org/10.3390/en7020824
Chicago/Turabian StyleWei, Chao-Yi, Tzou-Chi Huang, Zer-Ran Yu, Be-Jen Wang, and Ho-Hsien Chen. 2014. "Fractionation for Biodiesel Purification Using Supercritical Carbon Dioxide" Energies 7, no. 2: 824-833. https://doi.org/10.3390/en7020824
APA StyleWei, C.-Y., Huang, T.-C., Yu, Z.-R., Wang, B.-J., & Chen, H.-H. (2014). Fractionation for Biodiesel Purification Using Supercritical Carbon Dioxide. Energies, 7(2), 824-833. https://doi.org/10.3390/en7020824




