Analysis of Organic and Inorganic Contaminants in Dried Sewage Sludge and By-Products of Dried Sewage Sludge Gasification
Abstract
: Organic and inorganic contaminants in sewage sludge may cause their presence also in the by-products formed during gasification processes. Thus, this paper presents multidirectional chemical instrumental activation analyses of dried sewage sludge as well as both solid (ash, char coal) and liquid (tar) by-products formed during sewage gasification in a fixed bed reactor which was carried out to assess the extent of that phenomenon. Significant differences were observed in the type of contaminants present in the solid and liquid by-products from the dried sewage sludge gasification. Except for heavy metals, the characteristics of the contaminants in the by-products, irrespective of their form (solid and liquid), were different from those initially determined in the sewage sludge. It has been found that gasification promotes the migration of certain valuable inorganic compounds from sewage sludge into solid by-products which might be recovered. On the other hand, the liquid by-products resulting from sewage sludge gasification require a separate process for their treatment or disposal due to their considerable loading with toxic and hazardous organic compounds (phenols and their derivatives).1. Introduction
Gasification is regarded as a prospective and promising method for rendering sewage sludge (an example of unconventional biomass) harmless [1–4]. Unfortunately, apart from the valuable gas fuel produced, the process results in solid and liquid waste by-products [5–7]. The solidification of mineral substances during gasification produces solid products, mostly ash, but also char coal in some cases [5,6]. The formation of the latter depends on the composition of nonflammable inorganic substances in the sludge which considerably decreases the Debye characteristic temperatures of the ash [6]. Liquid products, i.e., tar, as a result of the condensation of the contaminants present in gas [7].
Sewage sludge, apart from energetically desirable compounds, is the source of toxic and hazardous organic and inorganic contaminants. Organic compounds identified in it include [8–11]: dioxins and furans, polychlorinated biphenyls (PCBs), organochlorine pesticides, adsorbed and extracted chloro derivatives, polycyclic aromatic hydrocarbons (PAHs), phenols and their derivatives, phthalates, sex hormones and others. The group of hazardous inorganic compounds assayed in sewage sludge contain primarily various heavy metals at a wide range of concentrations (mg/kg dry basis for raw sewage) [12–18]: arsenic 3–230, cadmium 1–3410, chromium 10–990,000, copper 80–2300, nickel 2–179, lead 13–465, and zinc 101–49,000.
As mentioned before, dried sewage sludge gasification produces waste by-products. This phenomenon accompanies all thermal treatment processes of both traditional and unconventional biomass (incineration, co-incineration and others) [1,3]. Thermal techniques usually transfer and accumulate contaminants in liquid and solid phases, which do not eliminate environmental hazards. It may be especially harmful while thermally processing sewage waste which is originally a source of various toxic and hazardous compounds. The preliminary research [19] found that the toxic effect of products produced during sewage sludge gasification depends on both the type of a sample tested (ash, char coal and tar) and sewage sludge used. That research was carried out using a Microtox® test with a Vibrio fischeri bacterial strain (luminescence method). Higher toxicity was found in the samples of ash that formed during gasification of sewage sludge, which appeared to be toxic, than for sludge of lower toxicity. As for tar samples, they were all toxic, regardless of the sludge gasified. Thus, from a cognitive perspective, the characteristics of such samples in terms of different contaminants which might be responsible for their toxicity are significant.
Taking into account the abovementioned aspects, this research covered multidirectional chemical instrumental activation analyses of dried sewage sludge and waste by-products formed during its gasification (ash, char coal and tar) in a fixed bed reactor. The tests were aimed at assessing transport and transformations of organic and inorganic contaminants during sewage sludge gasification in the sludge-gasification-solid and liquid waste by-products system.
2. Materials and Methods
2.1. Materials
Two different sewage sludges taken from wastewater treatment plants (WWTP) located in the West and North of Poland were selected for the research. Sewage Sludge 1 came from a WWTP operating in a mechanical and biological system while Sewage Sludge 2 was collected in a mechanical, biological and chemical WWTP with simultaneous phosphorus precipitation. The sludge produced at the plants was subject to fermentation and then, after dewatering, dried in a cylindrical drier on shelves heated up to 260 °C (Sewage Sludge 1) and using hot air at a temperature of 150 °C in a belt drier (Sewage Sludge 2). As a result, Sludge 1 took the form of granules while Sludge 2 was in the shape of irregular thin “pasta” (Figure 1).
The sludge was gasified in a fixed bed reactor using air as a gasifying agent fed to the reactor at a temperature of 25 °C and the amount of the agent corresponded to the ratio of excess air (λ) 0.18. The effect of the gasification parameters on producer gas yield, its composition and calorific value in particular, are discussed in detail in [6,20,21]. As far as the by-products are concerned, the gasification of Sewage Sludge 1 produced both ash (taken from the ash-pan) and char coal (taken from the inside of the reactor), while Sewage Sludge 2 produced ash only. The char coal formation in the case of the Sewage Sludge 1 was caused by a noncombustible inorganic compound. Those contaminants cause a significant reduction of the ash fusion temperatures. This influence is especially visible in the value of the temperature of the initial deformation [7].
2.2. Analysis of Selected Quality Indicators of Sewage Sludge and Gasification By-Products
The chemical qualitative analysis of the dried sewage sludge and waste by-products produced during gasification covered the concentrations of major elements (carbon, hydrogen, nitrogen, chlorine, fluorine, sulphur and oxygen), mineral elements (magnesium, calcium), phosphorus, alkaline metals (sodium and potassium), and selected heavy metals (zinc, selenium, lead, nickel, mercury, copper, chromium, cadmium and arsenic). The concentrations of the major elements were assayed, using automatic measurements with an infrared (IR) analyzer. Both the mineral elements and heavy metals were determined by plasma or absorption spectrometry. The assays carried out in the liquid products covered: total organic carbon (TOC) using an automatic analyzer, conductivity (conductometric analysis) and ammonia nitrogen concentration (spectrophotometry).
The authors of earlier works in this field [6,20,21] assayed moisture, volatile fractions, ash, heat of combustion and their calorific value as well. As for the ash, indicators characteristic of its tendency towards slagging and fouling of heated surfaces as well as agglomerate formation were determined [22]. The assays of moisture, volatile fractions and ash were conducted with the gravimetric methods given in the following standards PN-EN 14774-3:2010 [23], PN-EN 15402:2011 [24] and PN-EN 15403:2011 [25], respectively. The heat of combustion was determined by the calorimetric method while the calorific value was calculated, using the mass fractions of the major elements in a sample. The characteristics of two sewage sludges are given in Table 1.
2.3. Determination of Low-Molecular Organic Compounds with Gas Chromatography-Mass Spectrometry (GC-MS) Technique
The sewage sludge samples, as well as solid and liquid gasification waste by-products, were qualitatively and quantitatively analyzed using GC-MS to assess their contamination with low-molecular organic compounds. The assays were targeted at four main groups of contaminants commonly identified in sewage sludge [9,11], i.e., PAHs, pesticides, PCBs, phenols and their derivatives. The analysis employed the following Sigma-Aldrich (Poznan, Poland) standard solutions:
PAHs solution which contained 16 compounds [acenaphthene, acenaphthylene, anthracene, benzo(a)anthracene, benzo(a)pyrene, benzo(a)fluoranthene, benzo(b)fluoranthene, benzo(g,h,i)perylene, benzo(k)fluoranthene, chrysene, dibenzo(a,h)anthracene, phenanthrene, fluorene, indeno(1,2,3-cd)pyrene), naphthalene, pyrene] in a concentration of 100 ng/μL in toluene;
A solution of sixteen pesticides (aldrin, α-BHC, β-BHC, σ-BHC, dieldrin, α-endosulfan, β-endosulfan, endosulfan sulfate, endrin, endrin aldehyde, γ-BHC, heptachlor, heptachlor epoxide, 4,4′-DDE, 4,4′-DDE, 4,4′DDT) in a concentration of 200 ng/μL in n-hexane;
A solution of PCBs which contained six different derivatives [No. 28 (2,4,4′-PCB), 52 (2,2′,5,5′-PCB), No. 101 (2,2′,4,5,5′-PCB), No. 138 (2,2′,3,4,4′,5-PCB), No. 153 (2,2′,4,4′,5,5′-PCB), No. 180 (2,2′,3,4,4′,5,5′-PCB)] in a concentration of 10 ng/μL of the particular compounds prepared in isooctane;
A solution of ten phenols and their derivatives (pentachlorophenol, 2-methyl-4,6-dinitrophenol, 2-chlorophenol, 2-nitrophenol, 2,4-dichlorophenol, 2,4-dimethylphenol, 2,4-dinitrophenyl, 2,4,6-trichlorophenol, 4-chloro-3-methylphenol, 4-nitrophenol) in a concentration from 100 ng/μL to 250 ng/μL in methanol.
As a preliminary step in the chromatography assays, the solid samples were extracted with an organic solvent supported by ultrasounds. The analytical samples of sludge, ash or char coal (from 100 mg to 200 mg) were mixed with methylene chloride (1 mL) and placed in an ultrasonic bath (30 min). The extract produced was thickened and analyzed by GC-MS. The tar samples were thinned down to 10% in n-hexane and then analyzed chromatographically.
The chromatography analyses were performed using: a gas chromatograph coupled with a mass spectrometer (Saturn 2100 T GC-MS Varian, Warsaw, Poland) and equipped with a Supelco SLB™-5 ms column (30 m × 0.25 mm internal diameter, 0.25 μm film thickness), a split injector which maintained a constant temperature of 240 °C, helium (5 N) as a carrier gas with a flow rate of 1.1 mL/min. The temperature settings of the chromatography oven were as follows: 50 °C (4 min) → 8 °C /min → 260 °C → 4 °C/min → 300 °C (5 min). The temperature of the ion trap and ion source was 200 °C. The ions were within the m/z range of 40 to 400.
3. Results and Discussions
3.1. General Characteristics of Gasified Sewage Sludge
A comparison of the sewage sludge analyzed pointed to the conclusion that Sewage Sludge 1 exhibited a higher calorific value than Sewage Sludge 2 (Table 1). This was also confirmed by the analysis of gas composition and its calorific value shown in our papers [6,20,21]. However, the calorific value of the sewage sludge is lower than that of traditional biomass (wheat straw, rape straw, the sawdust of common osier, pine and oak) [6]. Our other paper [22] also compared the chemical composition of ash, sewage sludge and traditional biomass. It has been found that the sewage sludge exhibited much higher concentration of iron, titanium, phosphorus compounds and lower concentrations of potassium compounds. The above characteristics affect the typical indicators of ash depicting its tendency to slag and foul heated surfaces as well as form agglomerates. Compared with traditional biomass, sewage sludge tends to foul surfaces used for thermal treatment much less, but unfortunately, tends to slag and form agglomerates much more.
3.2. Characteristics of Sewage Sludge and Its Gasification Waste By-Products in Terms of Low-Molecular Organic Compounds
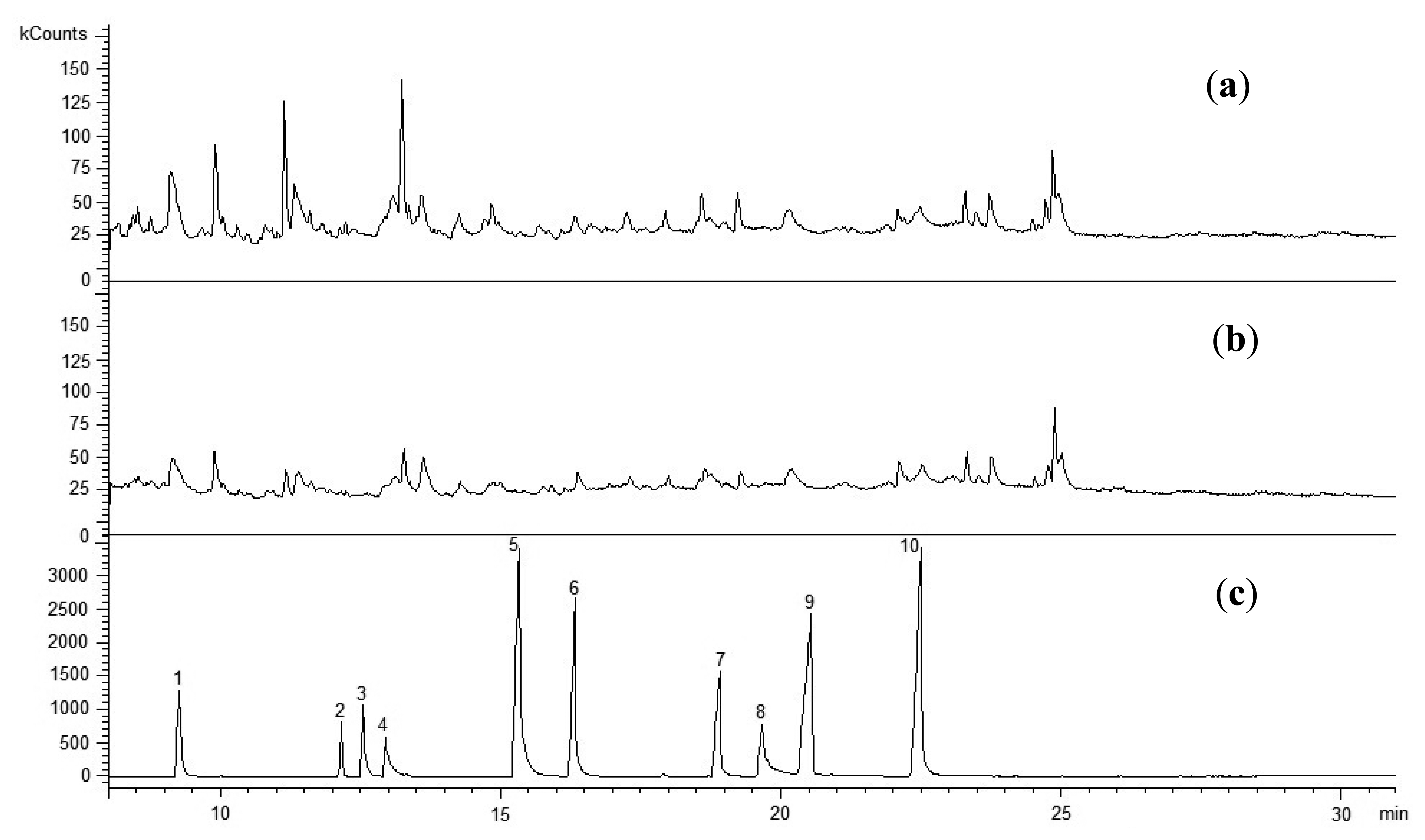
The chromatographic analyses of the dried sewage sludge extracts conducted herein confirmed their contamination with low-molecular organic compounds, PAHs in particular (Figure 2). Table 2 shows the concentrations of compounds assayed in the sewage sludge.
As mentioned above, the groups of organic contaminants assayed in the sewage sludge contained primarily PAHs. Sewage Sludge 1 revealed nine [phenanthrene, anthracene, benzo(a)fluoranthene, pyrene, chrysene, benzo(b)fluoranthene, dibenzo(a,h)anthracene, benzo(g,h,i)perylene, indeno(1,2,3-cd)pyrene], while Sewage Sludge 2 revealed eight [acenaphthene, benzo(a)fluoranthene, pyrene, benzo(a)anthracene, chrysene, benzo(b)fluoranthene, benzo(a)pyrene, indeno(1,2,3-cd)pyrene] compounds from that group of contaminants [naphthalene, acenaphthylene, acenaphthene, fluorene, phenanthrene, anthracene, benzo(a)fluoranthene, pyrene, benzo(a)anthracene, chrysene, benzo(b)fluoranthene, benzo(k)fluoranthene, benzo(a)pyrene, dibenzo(a,h)anthracene, benzo(g,h,i)perylene, indeno(1,2,3-cd)pyrene]. However, the total concentration of PAHs (Table 2) was four times higher in Sewage Sludge 1 than in Sewage Sludge 2. The papers [8,9] also found that PAHs constitute the basic group of contaminants in sewage sludge.
The group of organic contaminants identified in the sewage sludge also contained pesticides and PCBs. It is worth mentioning that Sewage Sludge 1 revealed the presence of three pesticides (heptachlor, aldrin and endrin) from the group of those commonly found in the natural environment in Poland (heptachlor, hexachlorocyclohexane, heptachlor epoxide, aldrin, endrin) [26]. Like the group of PAHs, the total concentrations of pesticides and PCBs were higher in Sewage Sludge 1 than in Sewage Sludge 2. The sewage sludge samples did not reveal any low-molecular phenols or their derivatives.
The profile of the organic contaminant concentrations obtained for the sewage sludge was typical of the sludge produced during treatment of domestic wastewater with an addition of industrial wastewater [11]. Obviously, considering the concentrations of particular contaminant groups, the contribution of industrial wastewater was higher for Sewage Sludge 1 than for Sewage Sludge 2. This is also supported by the heavy metal concentrations, which will be discussed in the following.
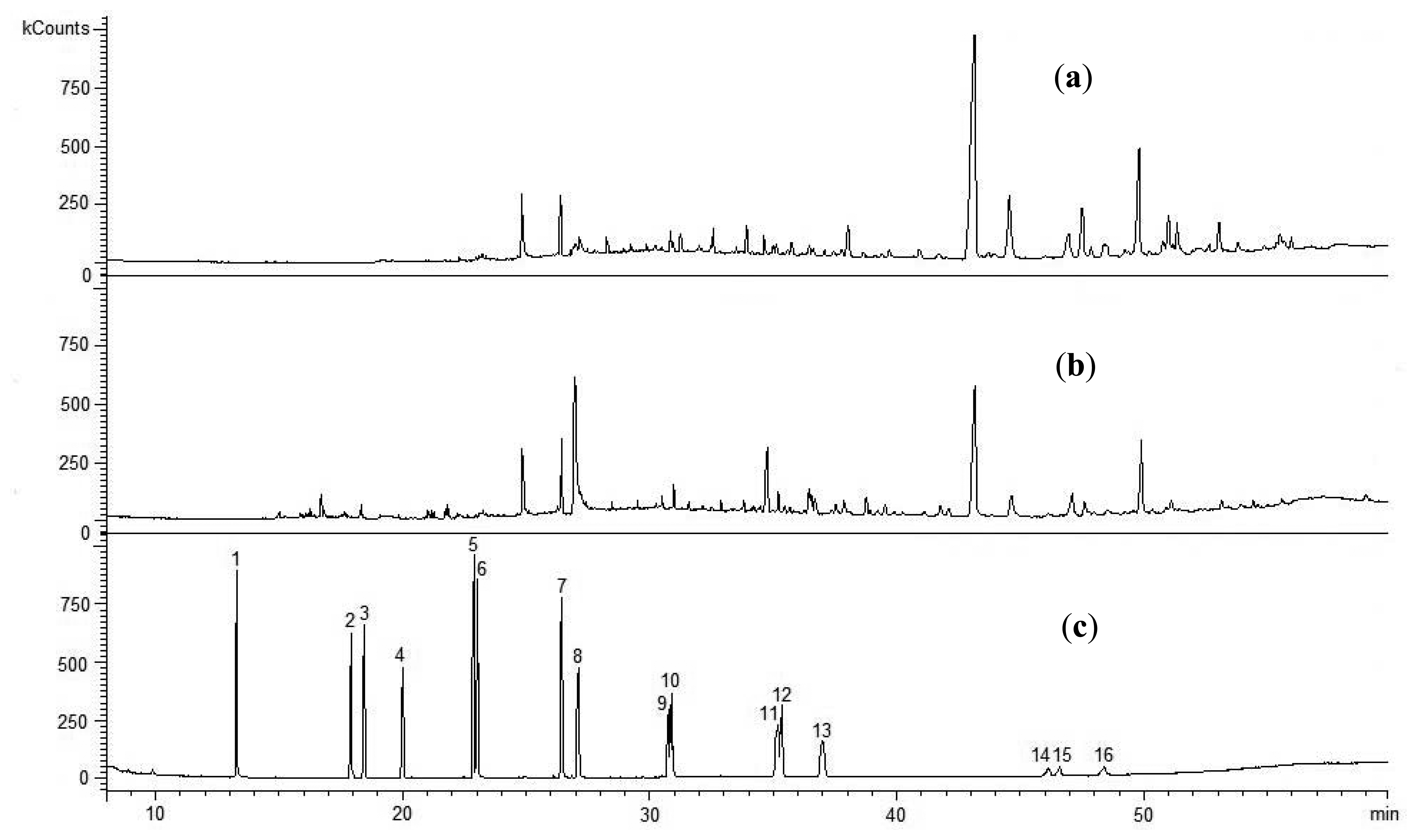
Figure 3 gives chromatograms prepared after analyzing the extracts of solid waste by-products, i.e., ash, char coal and gasified dried sewage sludge 1. The plots revealed that the waste by-products were not contaminated with organic contaminants initially identified in the sewage sludge. The chromatograms of ash and char coal showed just a couple of peaks deriving from unidentified organic compounds. However, it was possible to notice that there were more peaks in the chromatograms for ash than char coal. This resulted from the fact that the ash also contained an organic fraction which is difficult to decompose thermally [27]. A similar correlation was found while performing a comparative analysis of an extract from Sewage Sludge 2 and ash produced during its gasification. The ash extract did not reveal any compounds present in the sewage sludge before the gasification.
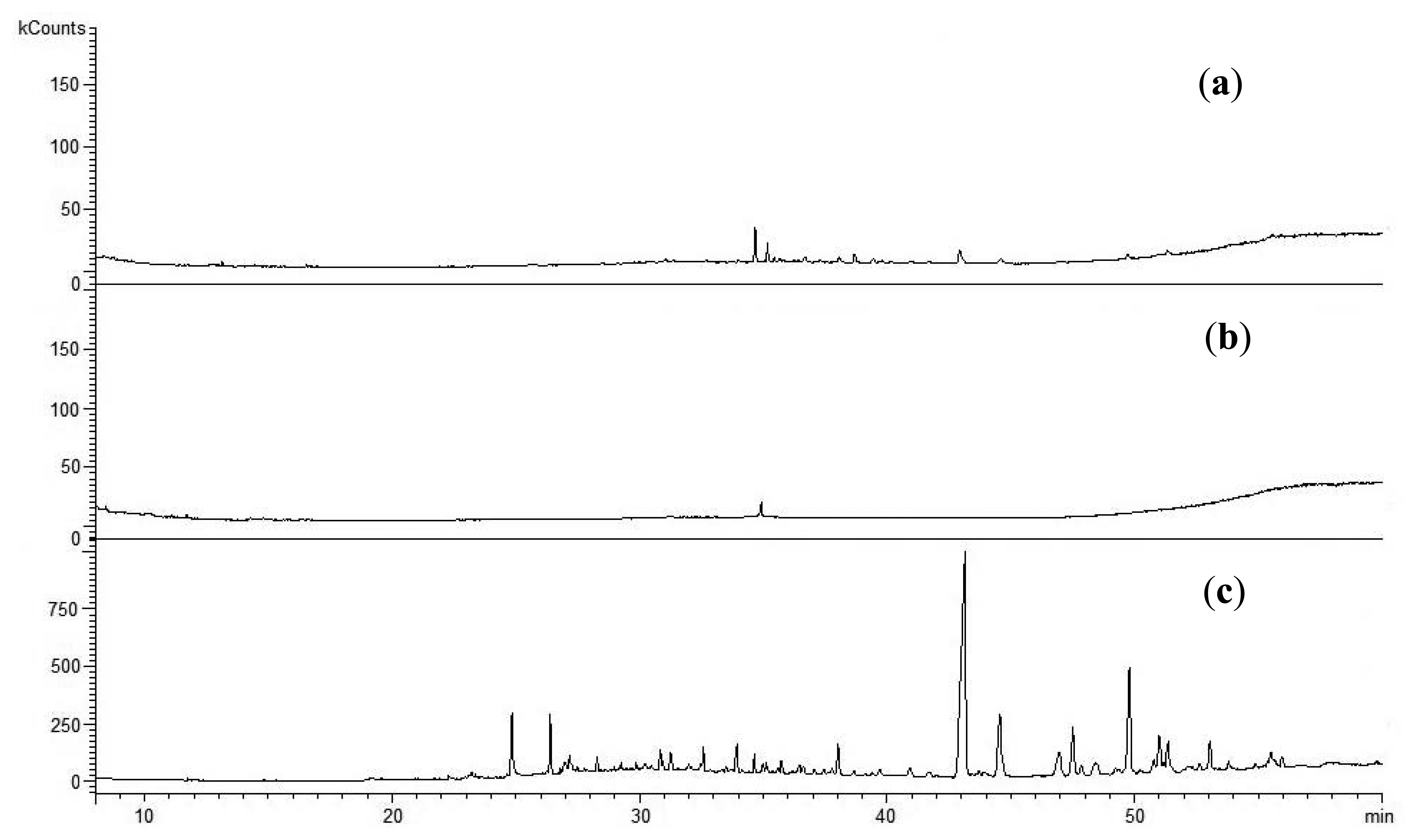
The tests of the liquid products (tar) formed during sewage sludge gasification determined TOC which directly measures the amount of different organic substances in the sample. The indicator was very high for both the tar produced during gasification of Sewage Sludges 1 and 2, being 20,950 mg TOC/L and 22,390 mg TOC/L, respectively. A chromatographic analysis of the tar samples showed their contamination mainly by phenols and their derivatives (Table 3).
The formation of those contaminants during the gasification of coal, biomass and waste is a common phenomenon, although they are usually accompanied by other aromatic and polyaromatic compounds [10]. It was also observed that the total concentration of phenols and their derivatives was almost eight times higher in the tar produced during the gasification of Sewage Sludge 1 than the tar from Sewage Sludge 2. Since Sewage Sludge 1 initially contained more organic contaminants (Table 2), a correlation between the properties of the tar and gasified sewage sludge might be found.
The drying temperature may also affect on the observed dependences. The drying temperature was much higher in the case of the Sewage Sludge 1 in comparison to Sewage Sludge 2. The higher drying temperature may affect on the tendency of the organic fraction from sewage sludge into the volatile organic compounds. As a result of the high drying temperature used sewage sludge has the properties similar to coal: firstly, hydrophobic properties by which storage of sludge is safer and without risk of the biological degradation and secondly, significantly improved regrind properties.
Phenols and their derivatives (Figure 4) are also identified in liquid waste by-products produced during biomass and waste gasification with water vapor [28–30]. It should also be mentioned that those components might act as a precursor of PAHs formation [31,32].
3.3. Assessment of Selected Inorganic Compounds Concentrations in Sewage Sludge and Gasification Waste By-Products
Thereafter, the paper assesses the concentrations of selected inorganic compounds in sewage sludge and gasification by-products.
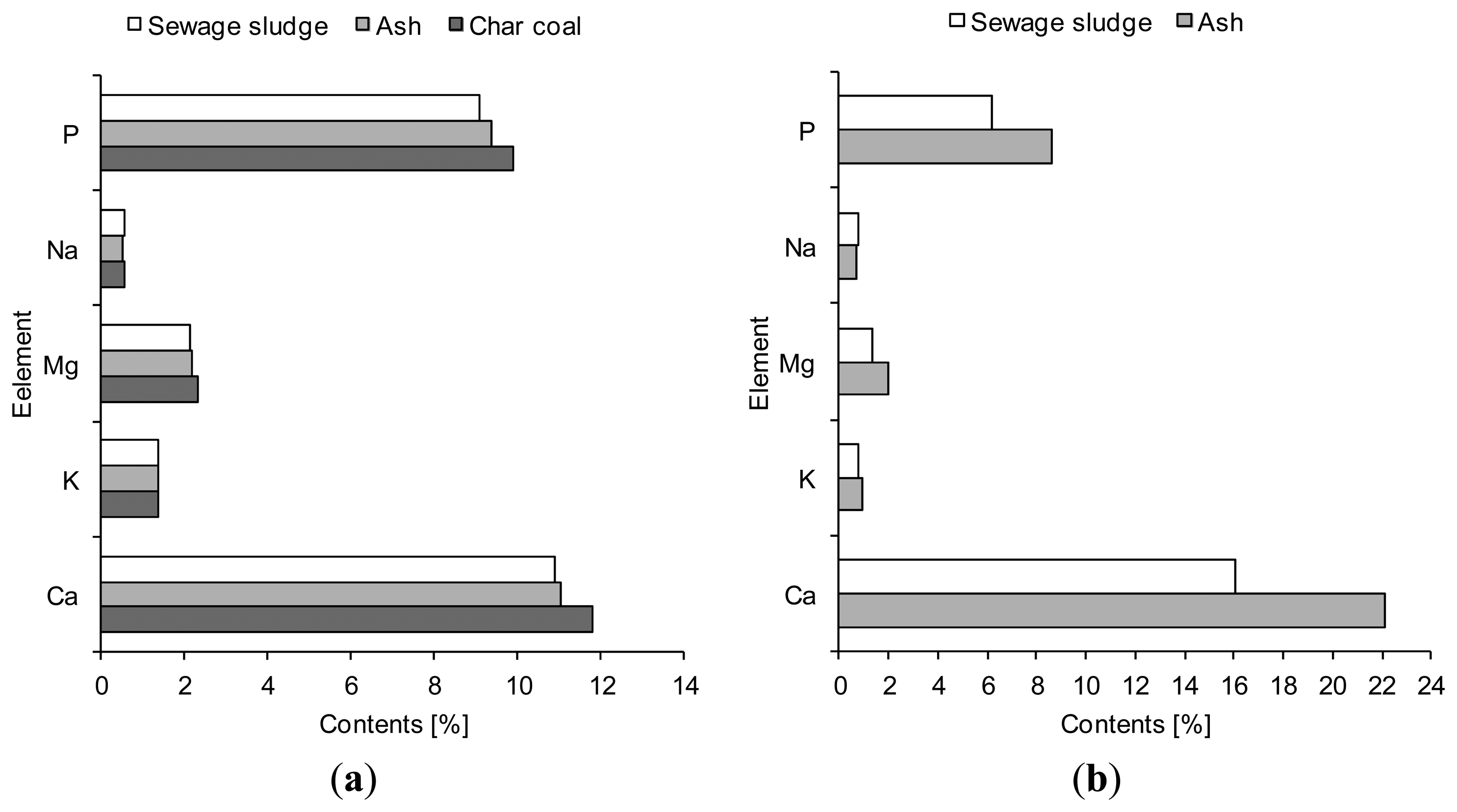
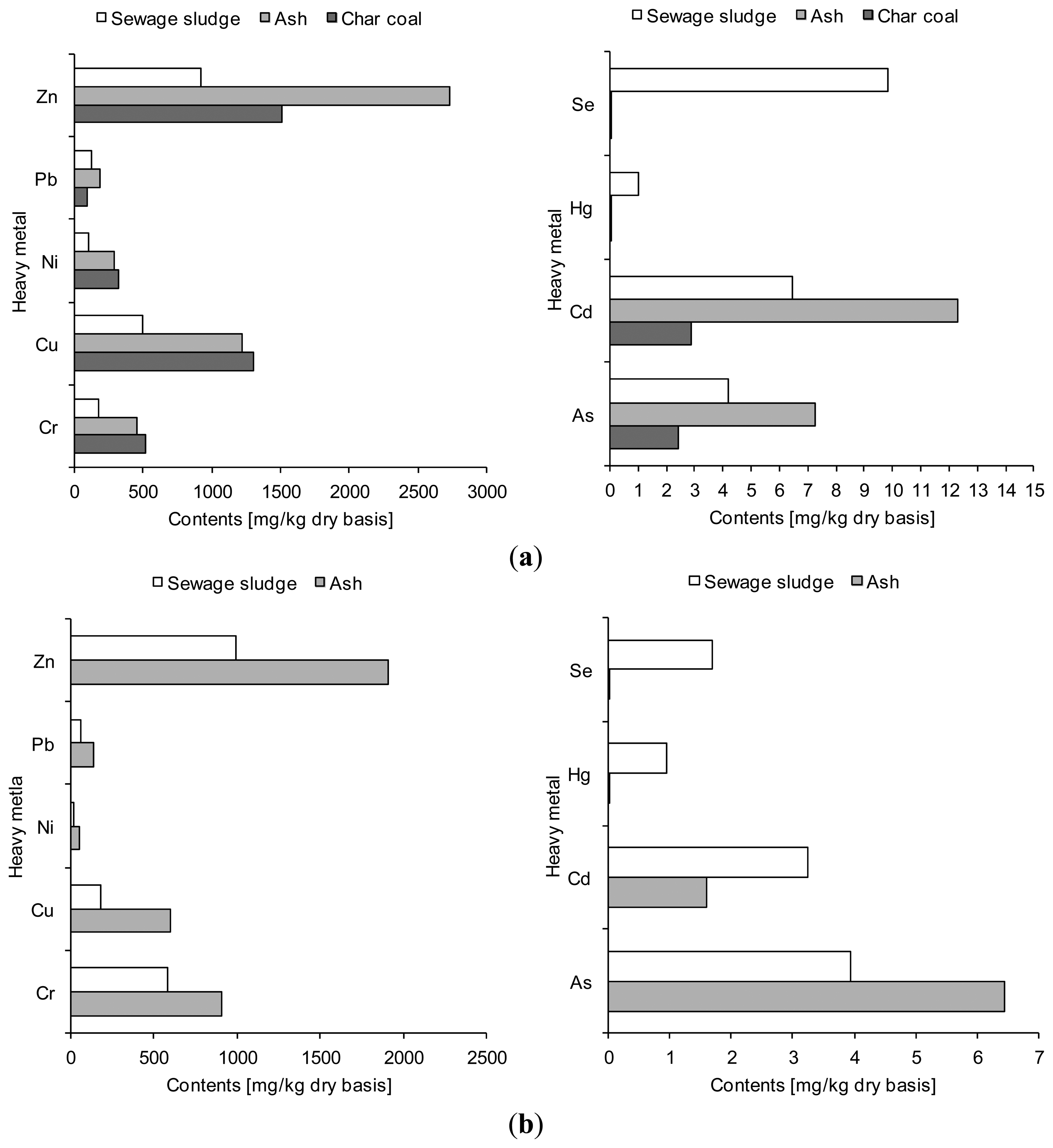
As for the original contamination of the sewage sludge, it was different in terms of mass contribution of particular elements, including heavy metals (Figures 5 and 6). On the other hand, the total concentration of, e.g., heavy metals were similar in both sewage sludges, being 1841 mg/kg dry wt. for Sewage Sludge 1 and 1848 mg/kg dry wt. for Sewage Sludge 2.
It has been found that the mass contribution of phosphorus and calcium in the solid products was higher than that assayed in the sewage sludge prior to thermal treatment, the highest accumulation being observed for Sewage Sludge 2 and the ash produced during its gasification (Figure 5b). Similar effects are given in paper [33] which points out that phosphorus concentration in the ash formed after sewage sludge gasification increased from 14.1 mg/kg dry basis to 20.6 mg/kg dry basis (an increase by 68%).
Similarly, the concentrations of heavy metals were higher in ash and char coal than sewage sludge (Figure 6). This applied to seven (zinc, lead, nickel, copper, chromium, cadmium, arsenic) out of nine heavy metals investigated (zinc, lead, nickel, copper, chromium, selenium, mercury, cadmium and arsenic), irrespective of the type of sewage sludge. Similar conclusions were drawn by the authors of the paper [13] who found an increase in the concentrations of cadmium (from 0.93 to 1.67 mg/kg dry basis), chromium (from 80.82 to 247.95 mg/kg dry basis), copper (from 580.36 to 922.14 mg/kg dry basis), lead (from 78.27 to 125.09 mg/kg dry basis) and zinc (from 402.09 to 637.50 mg/kg dry basis) in sewage sludge and ash samples after gasification. However, some differences in the accumulation of the solid waste by-products of particular heavy metals were spotted. For instance, zinc concentration in an ash sample produced during Sewage Sludge 2 gasification increased twofold, and even three-fold for Sewage Sludge 1.
The heavy metals presence in solid by-products generated during sewage sludge gasification has an impact on their potential treatment or utilization. As it is well known, one of the main obstacles to agricultural usage of the sewage sludge is the high content of heavy metals, which can penetrate from the sewage sludge to the soil causing their contamination [16]. From the results of this study, it can be concluded that concentration of the heavy metals in solid by products from sewage sludge gasification process is higher in comparison to sewage sludge. It should be noted that there is an ecological risk from those type of waste. This was confirmed by the toxicity analysis presented in the introduction part of this work. Additionally, some possibilities of the solid gasification by products treatment are indicated in the Subsection 3.4.
The liquid by-products (tar) produced during sewage sludge gasification demonstrated high conductivity, which proves their high contamination with various inorganic substances. The conductivity of the tar from Sewage Sludge 1 was 9800 μS/cm and 8170 μS/cm for Sewage Sludge 2. The hazardous inorganic contaminants occurring in the tar also included ammonia whose concentrations were 1090 mg NH4+/L (tar from Sewage Sludge 1) and 950 mg NH4+/L (tar from Sewage Sludge 2). Table 4 compares the physical and chemical characteristics of the tar produced during sewage sludge gasification with current Polish permissible standards for contaminants in the sewage resulting from the thermal treatment of waste in terms of, e.g., heavy metals [34]. As an effect of the condensation process in the end part of the gas pipe in the reactor, heavy metals from the gasification gas are transformed into tar.
It has been found that, out of eight heavy metals included in the government regulations (arsenic, cadmium, chromium, copper, mercury, nickel, lead and zinc), the concentrations of four metals (arsenic, cadmium, lead, zinc) in the tar from Sewage Sludge 1 and three metals (arsenic, cadmium and zinc) in the tar from Sewage Sludge 2 exceeded the standards. The same applies to the pH of the tar which fell outside the permissible range too (6.5–8.5).
Analyzing the sewage sludge and by-products after their gasification in terms of the heavy metals presence it is expected that gasification gas will be much less contaminated by these substances because the most of the inorganic substances originally present in the gasified sewage sludge undergoing accumulates in the solid by-products generated during the process. Such behaviors of heavy metals have been confirmed by other authors [13]. The organic compounds are transformed in the route of the complex thermochemical reactions accompanied by the completed decomposition of the compounds originally present in the sludge.
3.4. Suggestions for Further Research
The authors suggest that further research should concentrate on the recovery of phosphorus from solid products (ash and char coal) formed during the gasification of sewage sludge, e.g., through leaching phosphorus from ash or sinter with mineral acids. The application of ash resulting from gasification to the sorption of toxic and hazardous compounds (e.g., heavy metals) from different sewage is also worth considering. Similar research has already been carried out, using dried sewage sludge [35,36]. As for the tar produced during sewage sludge gasification, it requires an individual treatment system since it is contaminated by phenols, their derivatives and heavy metals (causing considerable toxicity of the samples, which was described in the preliminary research in paper [19]).
4. Conclusions
The waste by-products (solid and liquid) produced during sewage sludge gasification differ in the type of contaminants present in them. The concentrations of the contaminants depended on the characteristics of the sewage sludge treated thermally. The organic compounds present in the initial sewage sludge were not identified in the solid products (ash and char coal). They were contaminated mainly with inorganic compounds, including heavy metals. The liquid gasification products (tar) contained both toxic and hazardous organic (phenols and their derivatives) and inorganic (heavy metals) compounds. Higher concentrations of contaminants were found in the by-products formed after the thermal treatment of sewage sludge which was initially considerably contaminated with different groups of organic (PAHs, pesticides and PCBs) and inorganic (heavy metals among others) compounds, although paradoxically, the sludge displayed a higher calorific value.
Gasification promotes the migration of certain inorganic compounds, e.g., phosphorus or zinc from the sewage sludge to the solid phase formed after the sludge was treated thermally. This depended on both the type of the solid by-product (ash and char coal) and the sewage sludge used. Taking all that into account, the valorization of solid gasification products to recover valuable compounds or use them in the sorption of toxic and hazardous sewage might be worth considering in the future.
The high accumulation of heavy metals, including chromium, in the solid by-products (ash and char coal) and, e.g., phenols and their derivatives in the liquid products (tar) points to the ecological hazard they create. In the case of the gasification tar, it is necessary to develop a separate treatment system.
Acknowledgments
The paper has been prepared within the frame of the National Science Centre project based on decision no DEC-2011/03/D/ST8/04035.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Chun, Y.N.; Ji, D.W.; Yoshikawa, K. Pyrolysis and gasification characterization of sewage sludge for high quality gas and char production. J. Mech. Sci. Technol. 2013, 27, 263–272. [Google Scholar]
- Kang, S.W.; Dong, J.I.; Kim, J.M.; Lee, W.C.; Hwang, W.G. Gasification and its emission characteristics for dried sewage sludge utilizing a fluidized bed gasifier. J. Mater. Cycles Waste Manag. 2011, 13, 180–185. [Google Scholar]
- Werle, S.; Wilk, R.K. A review of methods for the thermal utilization of sewage sludge: The Polish perspective. Renew. Energy 2010, 35, 1914–1919. [Google Scholar]
- Werle, S. Sewage sludge gasification: Theoretical and experimental investigation. Environ. Prot. Eng. 2013, 39, 25–32. [Google Scholar]
- Nilsson, S.; Gómez-Barea, A.; Cano, D.F. Gasification reactivity of char from dried sewage sludge in a fluidized bed. Fuel 2012, 92, 346–353. [Google Scholar]
- Werle, S.; Dudziak, M. Gaseous Fuel Production from Granular Sewage Sludge via Air Gasification—Influence of the Waste Treatment Configuration on Sludge Properties and Producer Gas Parameters. Proceedings of the 8th Dubrovnik Conference on Sustainable Development of Energy, Water and Environment Systems, Dubrovnik, Croatia, 22–27 September 2013; pp. 1–10.
- Phuphuakrat, T.; Nipattummakul, N.; Namioka, T.; Kerdsuwan, S.; Yoshikawa, K. Characterization of tar content in the syngas produced in a downdraft type fixed bed gasification system from dried sewage sludge. Fuel 2010, 89, 2278–2284. [Google Scholar]
- Xu, Z.R.; Zhu, W.; Li, M. Influence of moisture content on the direct gasification of dewatered sludge via supercritical water. Int. J. Hydrog. Energy 2012, 37, 6527–3535. [Google Scholar]
- Xu, Z.R.; Zhu, W.; Li, M.; Zhang, H.W.; Gong, M. Quantitative analysis of polycyclic aromatic hydrocarbons in solid residues from supercritical water gasification of wet sewage sludge. Appl. Energy 2013, 102, 476–483. [Google Scholar]
- Aznar, M.; San Anselmo, M.; Manyà, J.J.; Murillo, M.B. Experimental study examining the evolution of nitrogen compounds during the gasification of dried sewage sludge. Energy Fuels 2009, 23, 3236–3245. [Google Scholar]
- Berset, J.D.; Holzer, R. Quantitative determination of polycyclic aromatic hydrocarbons, polychlorinated biphenyls and organochlorine pesticides in sewage sludges using supercritical fluid extraction and mass spectrometric detection. J. Chromatogr. A. 1999, 852, 545–558. [Google Scholar]
- Marrero, T.W.; McAuley, B.P.; Sutterlin, W.R.; Morris, J.S.; Manahan, S.E. Fate of heavy metals and radioactive metals in gasification of sewage sludge. Waste Manag. 2004, 24, 193–198. [Google Scholar]
- Li, L.; Xu, Z.R.; Zhang, C.; Bao, J.; Dai, X. Quantitative evaluation of heavy metals in solid residues from sub- and super-critical water gasification of sewage sludge. Bioresour. Technol. 2012, 121, 169–175. [Google Scholar]
- Abad, E.; Martínez, K.; Planas, C.; Palacios, O.; Caixach, J.; Rivera, J. Priority organic pollutant assessment of sludges for agricultural purposes. Chemosphere 2005, 61, 1358–1369. [Google Scholar]
- Fuentes, A.; Lloréns, M.; Sáez, J.; Isabel Aguilar, M.; Ortuño, J.F.; Meseguer, V.F. Comparative study of six different sludges by sequential speciation of heavy metals. Bioresour. Technol. 2008, 99, 517–525. [Google Scholar]
- Szymański, K.; Janowska, B.; Jastrzębski, P. Heavy metal compounds in wastewater and sewage sludge. Annu. Set Environ. Prot. 2011, 13, 83–100. [Google Scholar]
- Cai, Q.-Y.; Mo, C.-H.; Wu, Q.-T.; Zeng, Q.-Y.; Katsoyiannis, A. Concentration and speciation of heavy metals in six different sewage sludge-composts. J. Hazard. Mater. 2007, 147, 1063–1072. [Google Scholar]
- Karvelas, M.; Katsoyiannis, A.; Samara, C. Occurrence and fate of heavy metals in the wastewater treatment process. Chemosphere 2003, 53, 1201–1210. [Google Scholar]
- Werle, S.; Dudziak, M. Evaluation of toxicity of sewage sludge and gasification waste-products. Przem. Chem. 2013, 92, 1350–1353, in Polish. [Google Scholar]
- Werle, S.; Dudziak, M. Impact of Wastewater Treatment Processes and the Method of Sludge Treatment on Combustible Properties of Sludge Derived Fuel and Gas from Gasification. Proceedings of the 40th International Conference of Slovak Society of Chemical Engineering (SSCHE), Tatranské Matliare, Slovakia, 27–31 May 2013.
- Werle, S.; Dudziak, M. Influence of Wastewater Treatment Processes and the Method of Sludge Treatment on Sludge-Derived Fuel Properties and Gasification Gas Parameters. Proceedings of the 26th International Conference on Efficiency, Cost, Optimization, Simulation and Environmental Impact of Energy Systems, Guilin, China, 16–19 July 2013.
- Werle, S. Influence of sewage sludge properties on the thermal management processes. Zesz. Naukowe Uniw. Zielonogórs 2013, 151, 106–112, in Polish. [Google Scholar]
- Solid Biofuels—Methods for Moisture Determining Using Drier Method. Part 3—Moisture Analysis in General Sample; Standards PN-EN 14774-3:2010; Polish Committee for Standardization: Warsaw, Poland, 2010.
- Solid Recovered Fuels—Determination of Volatile Content; Standards PN-EN 15402:2011; Polish Committee for Standardization: Warsaw, Poland, 2011.
- Solid Recovered Fuels—Determination of Ash Content; Standards PN-EN 15403:2011; Polish Committee for Standardization: Warsaw, Poland, 2011.
- Tomza-Marciniak, A.; Witczak, A. Distribution of endocrine-disrupting pesticides in water and fish from the Oder river, Poland. Acta Ichthyol. Piscat. 2010, 40, 1–9. [Google Scholar]
- San Miguel, G.; Dominguez, M.P.; Hernandez, M.; Sanz-Perez, F. Characterization and potential applications of solid particle produced at a biomass gasification plant. Biomass Bioenergy 2012, 47, 134–144. [Google Scholar]
- Sinag, A.; Kruse, A.; Schwarzkopf, V. Key compounds of the hydropyrolysis of glucose in supercritical water in the presence of K2CO3. Ind. Eng. Chem. Res. 2003, 42, 3516–3521. [Google Scholar]
- Kruse, A.; Krupka, A.; Schwarzkopf, V.; Gamard, C.; Hanningsen, T. Influence of proteins on the hydrothermal gasification and liquefaction of biomass. 1. Comparison of different feedstocks. Ind. Eng. Chem. Res. 2005, 44, 3013–3020. [Google Scholar]
- Zhang, L.H.; Xu, C.B.; Champagne, P. Energy recovery from secondary pulp/paper mill sludge and sewage sludge with supercritical water treatment. Bioresour. Technol. 2010, 101, 2713–2721. [Google Scholar]
- Morf, P.; Hasler, P.; Nussbaumer, T. Mechanisms and kinetics of homogeneous secondary reactions of tar from continuous pyrolysis of wood chips. Fuel 2002, 81, 843–853. [Google Scholar]
- Zhang, B.P.; Xiong, S.J.; Xiao, B.; Yu, D.K.; Jia, X.Y. Mechanism of wet sludge pyrolysis in a tubular furnace. Int. J. Hydrog. Energy 2011, 36, 355–363. [Google Scholar]
- Zhu, W.; Xu, Z.R.; Li, L.; He, C. The behavior of phosphorus in sub- and super-critical water gasification of sewage sludge. Chem. Eng. J. 2011, 171, 190–196. [Google Scholar]
- The Polish Ordinance of Ministry for the Environment from 24 July 2006 about on the Conditions Which Must Be Fulfilled While Discharging Waste Water to Water or Ground and on Substances Particularly Dangerous for The Water Environment; Journal of Laws No 137 Item 984; Ministry of the Environment: Warsaw, Poland, 2006.
- Yang, C.; Wang, J.; Lei, M.; Xie, G.; Zeng, G.; Luo, S. Biosorption of zinc(II) from aqueous solution by dried activated sludge. J. Environ. Sci. 2010, 22, 675–680. [Google Scholar]
- Thawornchaisit, U.; Pakulanon, K. Application of dried sewage sludge as phenol biosorbent. Bioresour. Technol. 2007, 98, 140–144. [Google Scholar]

| Element/Parameter | Sewage Sludge 1 | Sewage Sludge 2 | |
|---|---|---|---|
| Proximate analysis *, % (as received) | Moisture | 5.30 | 5.30 |
| Volatile matter | 51.00 | 49.00 | |
| Ash | 36.50 | 44.20 | |
| Ultimate analysis, % (dry basis) | C | 31.79 | 27.72 |
| H | 4.36 | 3.81 | |
| N | 4.88 | 3.59 | |
| O (by difference) | 20.57 | 18.84 | |
| S | 1.67 | 1.81 | |
| F | 0.013 | 0.003 | |
| Cl | 0.22 | 0.03 | |
| Calorific value * | Higher heating value (HHV), MJ/kg dry basis | 14.05 | 11.71 |
| Lower heating value (LHV), MJ/kg dry basis | 12.96 | 10.75 | |
| Compound | Retention time | Concentration, μg/kg dry basis | ||
|---|---|---|---|---|
| Sewage Sludge 1 | Sewage Sludge 2 | |||
| PAHs | acenaphthene | 18.45 | n.d. | 80.84 |
| phenanthrene | 22.89 | 511.12 | n.d. | |
| anthracene | 23.03 | 200.03 | n.d. | |
| benzo(a)fluoranthene | 26.46 | 44.78 | 126.48 | |
| pyrene | 27.08 | 187.22 | 123.86 | |
| benzo(a)anthracene | 27.13 | n.d. | 35.15 | |
| chrysene | 30.76 | 108.14 | 23.79 | |
| benzo(b)fluoranthene | 30.90 | 700.51 | 53.62 | |
| benzo(a)pyrene | 37.05 | n.d. | 46.11 | |
| dibenzo(a,h)anthracene | 46.13 | 101.54 | n.d. | |
| benzo(g,h,i)perylene | 46.61 | 209.44 | n.d. | |
| indeno(1,2,3-cd)pyrene | 48.39 | 370.62 | 131.48 | |
| Sum | - | 2,433.40 | 621.33 | |
| Pesticides | heptachlor | 24.16 | 4.14 | n.d. |
| aldrin | 25.04 | 3.13 | 1.28 | |
| endrin | 28.09 | 11.58 | n.d. | |
| Sum | - | 18.85 | 1.28 | |
| PCBs | 2,2′,5,5′-PCB | 24.79 | 9.75 | 7.90 |
| 2,2′,4,5,5′-PCB | 28.82 | 33.33 | n.d. | |
| 2,2′,4,4′,5,5′-PCB | 29.47 | 23.78 | 4.57 | |
| Sum | - | 66.86 | 12.47 | |
n.d.: Not detected.
| Compound | Retention time | Concentration, μg/L | |
|---|---|---|---|
| Sewage Sludge 1 | Sewage Sludge 2 | ||
| 2-Chlorophenol | 9.28 | 211.84 | 57.20 |
| 2-Nitrophenol | 12.55 | 89.52 | n.d. |
| 2,4-Dichlorophenol | 12.95 | 361.56 | n.d. |
| 4-Chloro-3-Methylphenol | 15.35 | 9.27 | 1.98 |
| 2,4,6-Trichlorophenol | 16.33 | 62.32 | 46.33 |
| Pentachlorophenol | 22.50 | 57.97 | 53.90 |
| Sum | - | 792.48 | 100.23 |
n.d.: Not detected.
| Indicator/heavy metal | Unit | Tar from sewage sludge gasification | The maximum concentration indicator for sewage sludge from the waste thermal treatment process [34] | |
|---|---|---|---|---|
| 1 | 2 | |||
| pH | - | 4.39 | 4.24 | 6.5–8.5 |
| As | mg/L | 0.29 | 0.16 | 0.15 |
| Cd | mg/L | 0.13 | 0.06 | 0.05 |
| Cr | mg/L | 0.02 | 0.01 | 0.50 |
| Cu | mg/L | 0.02 | <0.01 | 0.50 |
| Hg | mg/L | <0.01 | <0.01 | 0.03 |
| Ni | mg/L | 0.01 | <0.01 | 0.50 |
| Pb | mg/L | 0.38 | 0.17 | 0.20 |
| Zn | mg/L | 5.60 | 5.73 | 1.50 |
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Werle, S.; Dudziak, M. Analysis of Organic and Inorganic Contaminants in Dried Sewage Sludge and By-Products of Dried Sewage Sludge Gasification. Energies 2014, 7, 462-476. https://doi.org/10.3390/en7010462
Werle S, Dudziak M. Analysis of Organic and Inorganic Contaminants in Dried Sewage Sludge and By-Products of Dried Sewage Sludge Gasification. Energies. 2014; 7(1):462-476. https://doi.org/10.3390/en7010462
Chicago/Turabian StyleWerle, Sebastian, and Mariusz Dudziak. 2014. "Analysis of Organic and Inorganic Contaminants in Dried Sewage Sludge and By-Products of Dried Sewage Sludge Gasification" Energies 7, no. 1: 462-476. https://doi.org/10.3390/en7010462





