Effects of Biodiesel Blend on Marine Fuel Characteristics for Marine Vessels
Abstract
:1. Introduction
2. Calculation Methods for Determining Blended-Fuel Properties
| Fuel parameter | Unit | ISO8217 RMA | ISO8217 DMA | EN 14214 biodiesel |
|---|---|---|---|---|
| Cetane number, min | - | 20 | 40 | 51 |
| Sulfur, max | ppmw | 45,000 | 15,000 | 10 |
| Density (at 15 °C) | kg/m3 | 920 max | 890 max | 860 to 900 |
| Flash point, min | °C | 60.0 | 60.0 | 120.0 |
| Carbon residue, max | wt.% | 2.5 | 0.3 | 0.05 * |
| Kinematic viscosity (at 40 °C) | mm2/s | 10.0 max | 2.0 to 6.0 | 3.5 to 5.0 |
| Heating value | MJ/kg | 40 | 42 | 38 |
| Water content, max | ppm | 3,000 | - | 500 |
| Acid number, max | mg KOH/g | 2.5 | 0.5 | 0.5 |
3. Results and Discussion
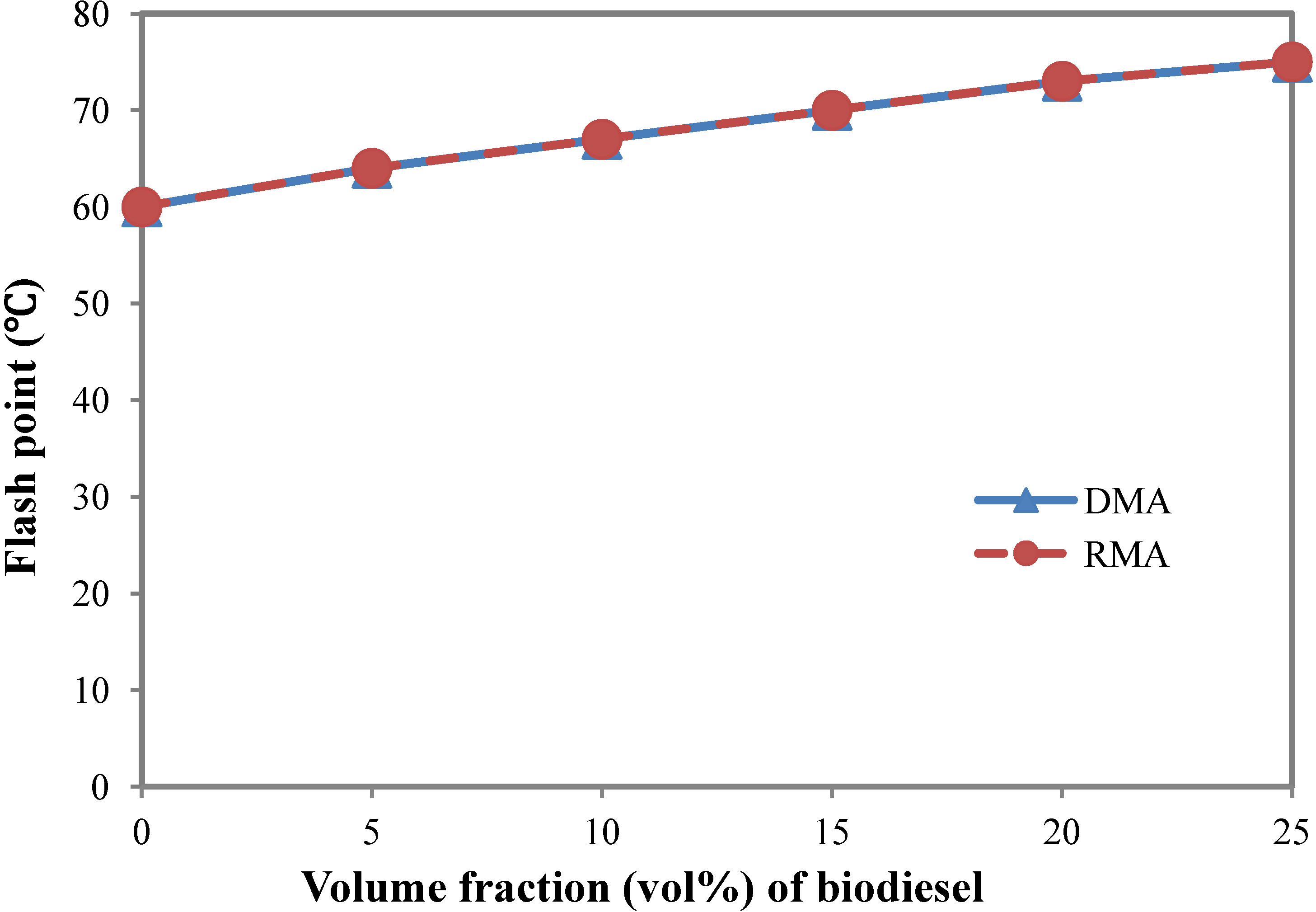
3.1. Flash Point
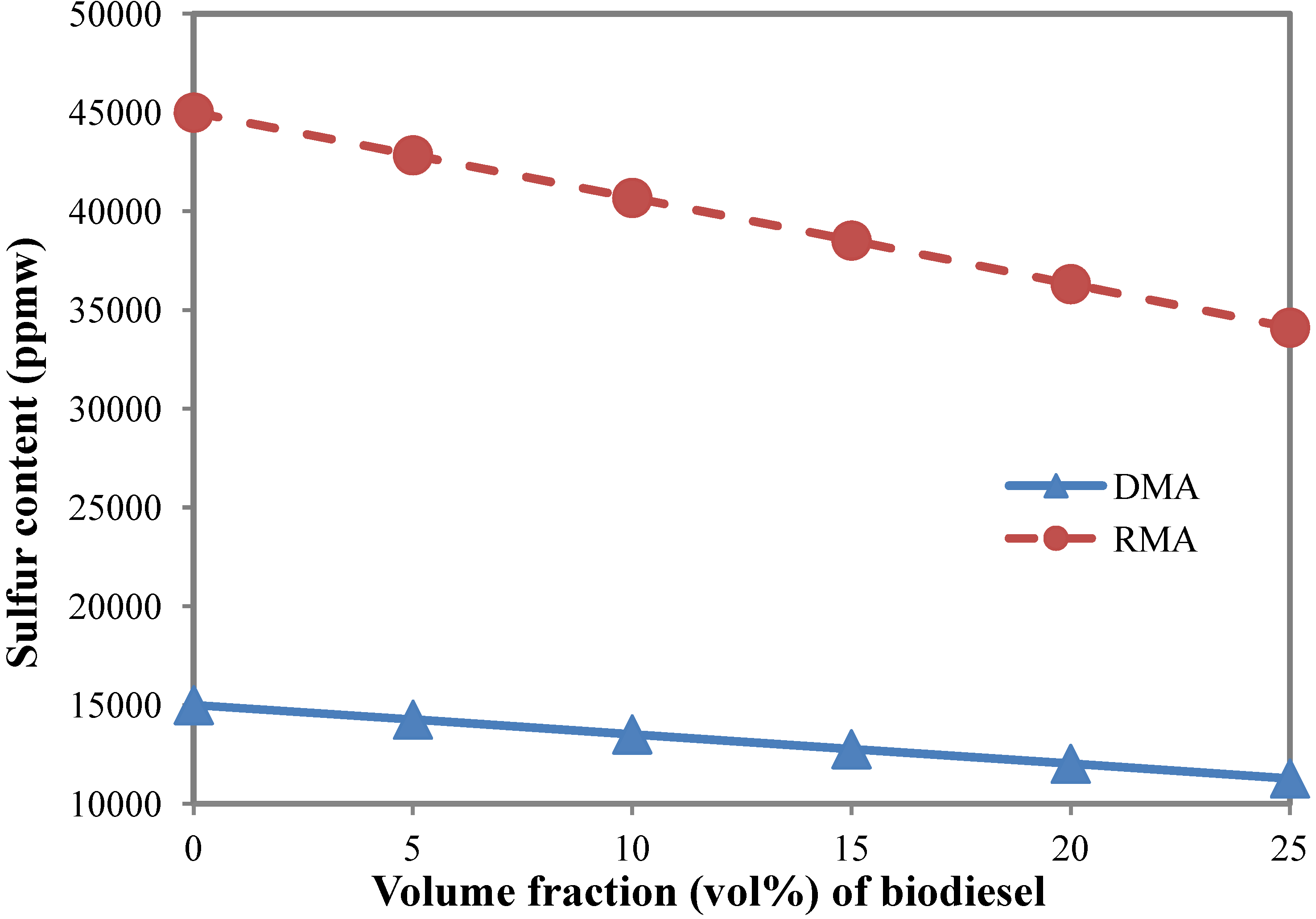
3.2. Sulfur Content
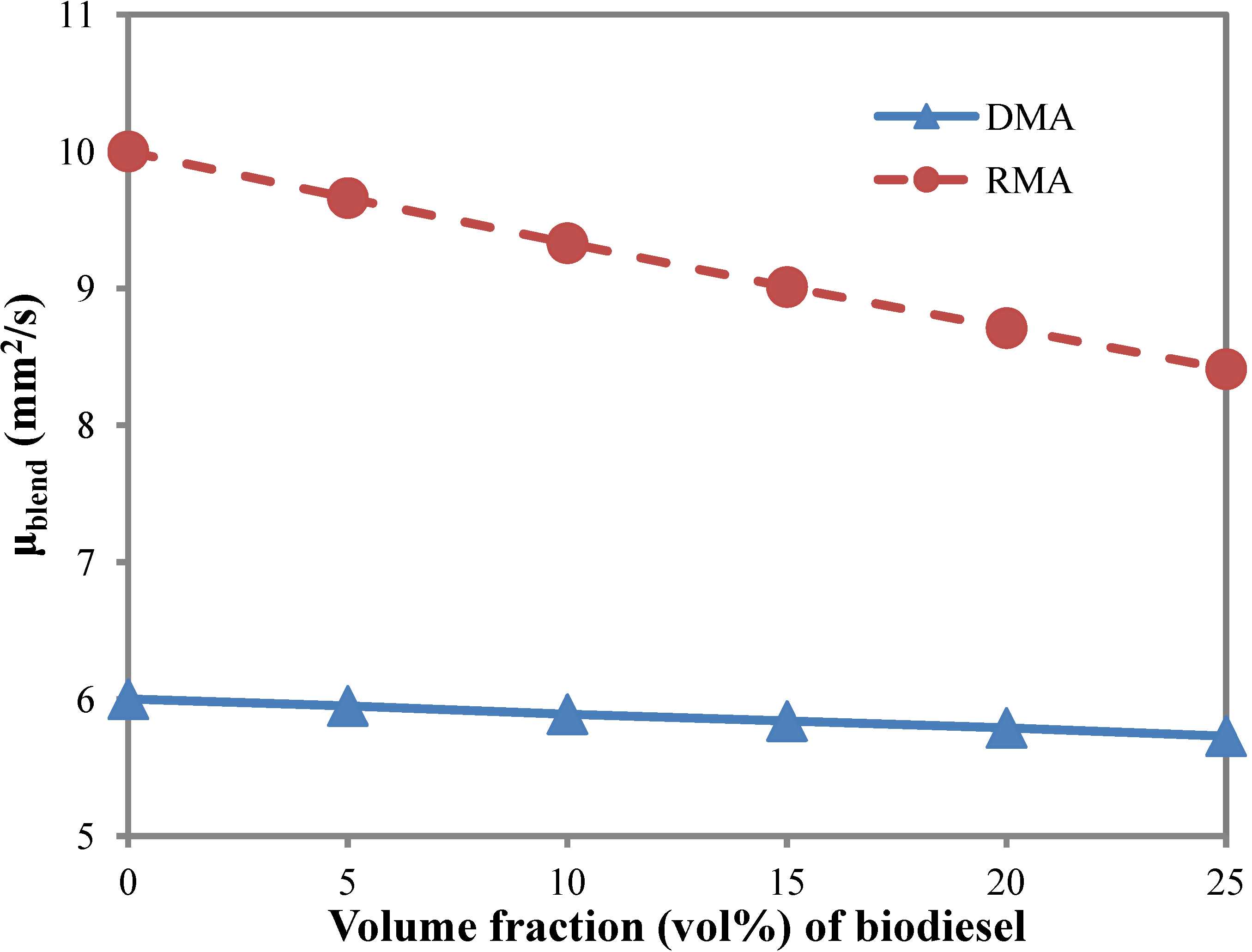
3.3. Kinematic Viscosity
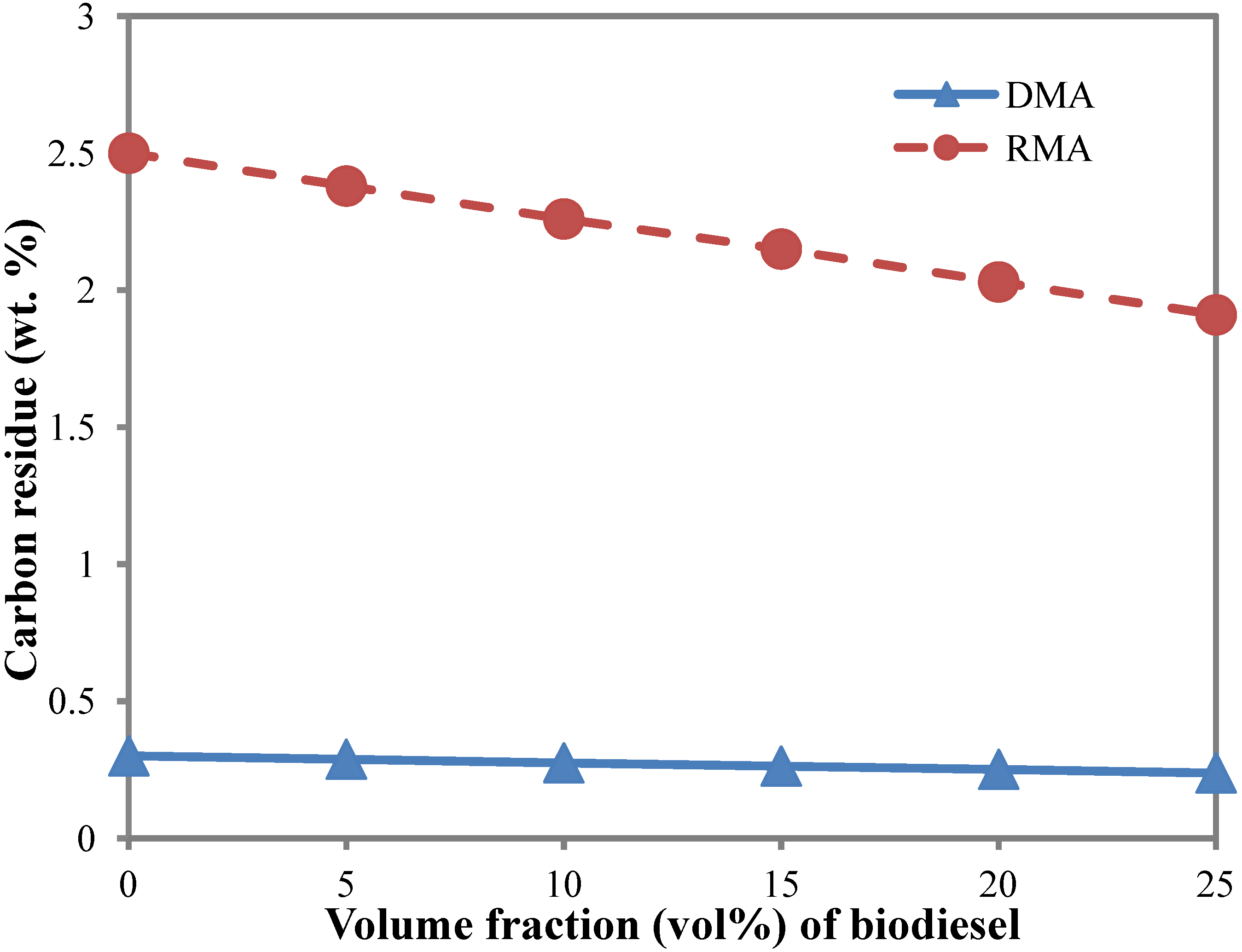
3.4. Carbon Residue
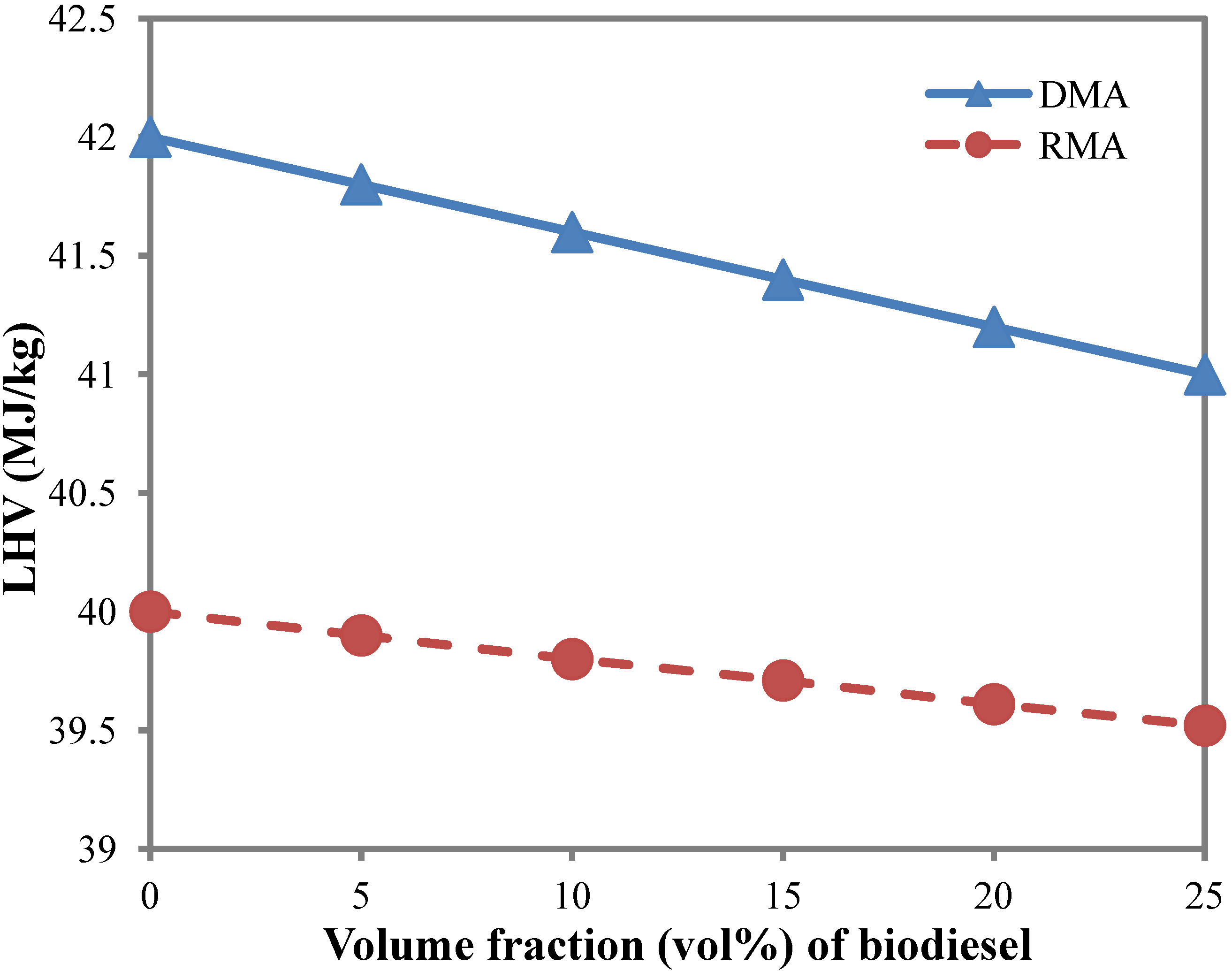
3.5. Lower Heating Value (LHV)
4. Conclusions
- (1)
- Residual marine fuel RMA has the worst fuel characteristics compared with distillate marine fuel DMA and biodiesel. Biodiesel blending could improve the fuel properties of residual marine fuel RMA more significantly compared with distillate marine fuel DMA.
- (2)
- Biodiesel has a significantly higher flash point than marine fuels DMA and RMA. A 20 vol% biodiesel blending in marine fuel RMA could increase its flash point by 20%, thus increasing fuel safety during operation and storage periods.
- (3)
- The fuel sulfur limit of marine fuel oil reaches as high as 4.5 wt% based on the ISO 8217 specification of residual marine fuel. Biodiesel contains almost no sulfur. Only a 23.0 vol% biodiesel blending in marine fuel RMA would reduce the sulfur content to below 3.5 wt%, which would meet the requirement of the 2008 MARPOL Annex VI Amendment [21].
- (4)
- Biodiesel blending decreases the kinematic viscosity of marine fuel. A 20 vol% biodiesel blending in marine fuel RMA could reduce its kinematic viscosity by 12.9%.
- (5)
- Much less carbon residue is released after the burning of biodiesel compared with marine fuel RMA. The carbon residue could be decreased by 23.6% if 25 vol% biodiesel were blended into residual marine fuel RMA. The combustion characteristics of the blended marine fuel would also be greatly improved.
- (6)
- The lower heating value of the blended marine fuel DMA and RMA with 20 vol% biodiesel is found to decrease by 1.9% and 0.1%, respectively.
Acknowledgments
Conflicts of Interest
References
- Lin, C.Y.; Huang, T.H. Cost-benefit evaluation of using biodiesel as an alternative fuel for fishing boats in Taiwan. Mar. Policy 2012, 36, 103–107. [Google Scholar] [CrossRef]
- Nayyar, P. The Use of Biodiesel Fuels in the U.S. Marine; Report for the U.S. Maritime Administration (MARAD); Prime Inc.: Washington, DC, USA, 2010; pp. 65–79. [Google Scholar]
- Lin, C.Y.; Chen, W.C. Effects of potassium sulfide content in marine diesel fuel oil on emission characteristics of marine furnaces under varying humidity of inlet air. Ocean Eng. 2006, 33, 1260–1270. [Google Scholar] [CrossRef]
- Deniz, C.; Kilic, A.; Civkaroglu, G. Estimation of shipping emissions in Candarli Gulf, Turkey. Environ. Monit. Assess. 2010, 171, 219–228. [Google Scholar] [CrossRef] [PubMed]
- Eyring, V.; Köhler, H.W.; van Aardenne, J.; Lauer, A. Emissions from international shipping: The last 50 years. J. Geophys. Res. 2005, 110, D17305. [Google Scholar] [CrossRef]
- Prpic, J.; Faltinsen, O.D. Estimation of ship speed loss and associated CO2 emission in a seaway. Ocean Eng. 2012, 44, 1–10. [Google Scholar] [CrossRef]
- Jaichandarl, S.; Annamalai, K. The status of biodiesel as an alternative fuel for diesel engine—An overview. J. Sust. Energy Environ. 2011, 2, 71–75. [Google Scholar]
- Usta, N.; Aydogan, B.; Con, A.H.; Uguzdogam, E.; Ozkal, S.G. Properties and quality verification of biodiesel produced from tobacco seed oil. Energy Convers. Manag. 2011, 52, 2031–2039. [Google Scholar]
- Mejia, J.D.; Salgado, N.; Orrego, C.E. Effect of blends of diesel and palm-castor biodiesels on viscosity, cloud point and flash point. Ind. Crop. Prod. 2012, 43, 791–797. [Google Scholar] [CrossRef]
- Krisnangkura, K.; Yimsuwan, T.; Pairintra, R. An empirical approach in predicting biodiesel viscosity at various temperatures. Fuel 2006, 85, 107–113. [Google Scholar] [CrossRef]
- Benjumea, P.; Agudelo, J.; Agudelo, A. Basic properties of palm oil biodiesel-diesel blends. Fuel 2008, 87, 2069–2075. [Google Scholar] [CrossRef]
- Saxena, P.; Jawale, S.; Joshipura, M.H. A review on prediction of properties of biodiesel and blends of biodiesel. Proc. Eng. 2013, 51, 395–402. [Google Scholar] [CrossRef]
- Tesfa, B.; Mishra, R.; Gu, F.; Powles, N. Prediction models for density and viscosity of biodiesel and their effects on fuel supply system in CI engines. Renew. Energy 2010, 35, 2752–2760. [Google Scholar] [CrossRef]
- García, M.; Gonzalo, A.; Sánchez, J.L.; Arauzo, J.; Peña, J.A. Prediction of normalized biodiesel properties by simulation of multiple feedstock blends. Bioresour. Technol. 2010, 101, 4431–4439. [Google Scholar] [CrossRef] [PubMed]
- Clements, L.D. Blending Rules for Formulating Biodiesel Fuel. In Liquid Fuel and Industrial Products from Renewable Resources, Proceedings of the 3rd Liquid Fuels Conference, Nashville, TN, USA, 15–16 September 1996; American Society of Agricultural and Engineers: St. Joseph, MI, USA, 1996; p. 44. [Google Scholar]
- Tesfa, B.; Gu, F.; Mishra, R.; Ball, A.D. LHV predication models and LHV effect on the performance of CI engine running with biodiesel blends. Energy Convers. Manag. 2013, 71, 217–226. [Google Scholar] [CrossRef]
- Marine Environment Protection Committee (MEPC) and International Maritime Organization (IMO). ISO 8217: 2010 Revised Specification of Marine Fuels; Document Number MEPC61/4/1; MEPC and IMO: London, UK, 2010. [Google Scholar]
- European Committee for Standardization (CEN). Automotive Fuels—Fatty Acid Methyl Esters (FAME) for Diesel Engines—Requirement Methods; EN 14214:2008; CEN: Brussels, Belgium, 2009. [Google Scholar]
- Knudsen, O.F.; Hassler, B. IMO legislation and its implementation: Accident risk, vessel deficiencies and national administrative practices. Mar. Policy 2011, 35, 201–207. [Google Scholar] [CrossRef]
- Mahajan, A.; Ahluwalia, A.S.; Mahajan, P. Properties of biodiesel produced from various oilseeds. Int. J. Res. Environ. Sci. Technol. 2011, 1, 26–29. [Google Scholar]
- International Maritime Organization (IMO). MARPOL Annex VI: Regulations for the Prevention of Air Pollution from Ships, Consolidated ed.; IMO: London, UK, 2006. [Google Scholar]
- Muñoz, M.; Moreno, F.; Monné, C.; Morea, J.; Terradillos, J. Biodiesel improves lubricity of new low sulphur diesel fuels. Renew. Energy 2011, 36, 2918–2924. [Google Scholar] [CrossRef]
- National Renewable Energy Laboratory (NREL) and U.S. Department of Energy. Biodiesel Handling and Use Guide, 4th ed.; Report Number NREL/TP-540-43672; Office of Scientific and Technical Information: Oak Ridge, TN, USA, 2009. [Google Scholar]
- Lin, C.Y.; Li, R.J. Fuel properties of biodiesel produced from the crude fish oil from the soapstock of marine fish. Fuel Proces. Technol. 2009, 90, 130–136. [Google Scholar] [CrossRef]
- Nabi, M.N.; Husted, J.E. Investigation of engine performance and emissions of a diesel engine with a blend of marine gas oil and synthetic diesel fuel. Environ. Technol. 2012, 33, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Pinzi, S.; Mata-Granados, J.M.; Lopez-Gimenez, F.J.; Luque de Castro, M.D.; Dorado, M.P. Influence of vegetable oils fatty-acid composition on biodiesel optimization. Bioresour. Technol. 2011, 102, 1059–1065. [Google Scholar] [CrossRef] [PubMed]
- Mehta, P.S.; Anand, K. Estimation of a lower heating value of vegetable oil and biodiesel fuel. Energy Fuels 2009, 23, 3893–3898. [Google Scholar] [CrossRef]
- Fregolente, P.B.L.; Fregolente, L.V.; Wolf Maciel, M.R. Water content in biodiesel, diesel, and biodiesel–diesel blends. J. Chem. Eng. Data 2012, 57, 1817–1821. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lin, C.-Y. Effects of Biodiesel Blend on Marine Fuel Characteristics for Marine Vessels. Energies 2013, 6, 4945-4955. https://doi.org/10.3390/en6094945
Lin C-Y. Effects of Biodiesel Blend on Marine Fuel Characteristics for Marine Vessels. Energies. 2013; 6(9):4945-4955. https://doi.org/10.3390/en6094945
Chicago/Turabian StyleLin, Cherng-Yuan. 2013. "Effects of Biodiesel Blend on Marine Fuel Characteristics for Marine Vessels" Energies 6, no. 9: 4945-4955. https://doi.org/10.3390/en6094945




