Effect of Mixing Driven by Siphon Flow: Parallel Experiments Using the Anaerobic Reactors with Different Mixing Modes
Abstract
:1. Introduction
2. Results and Discussion
2.1. Acceptable Organic Loading Rates and Hydraulic Retention Time
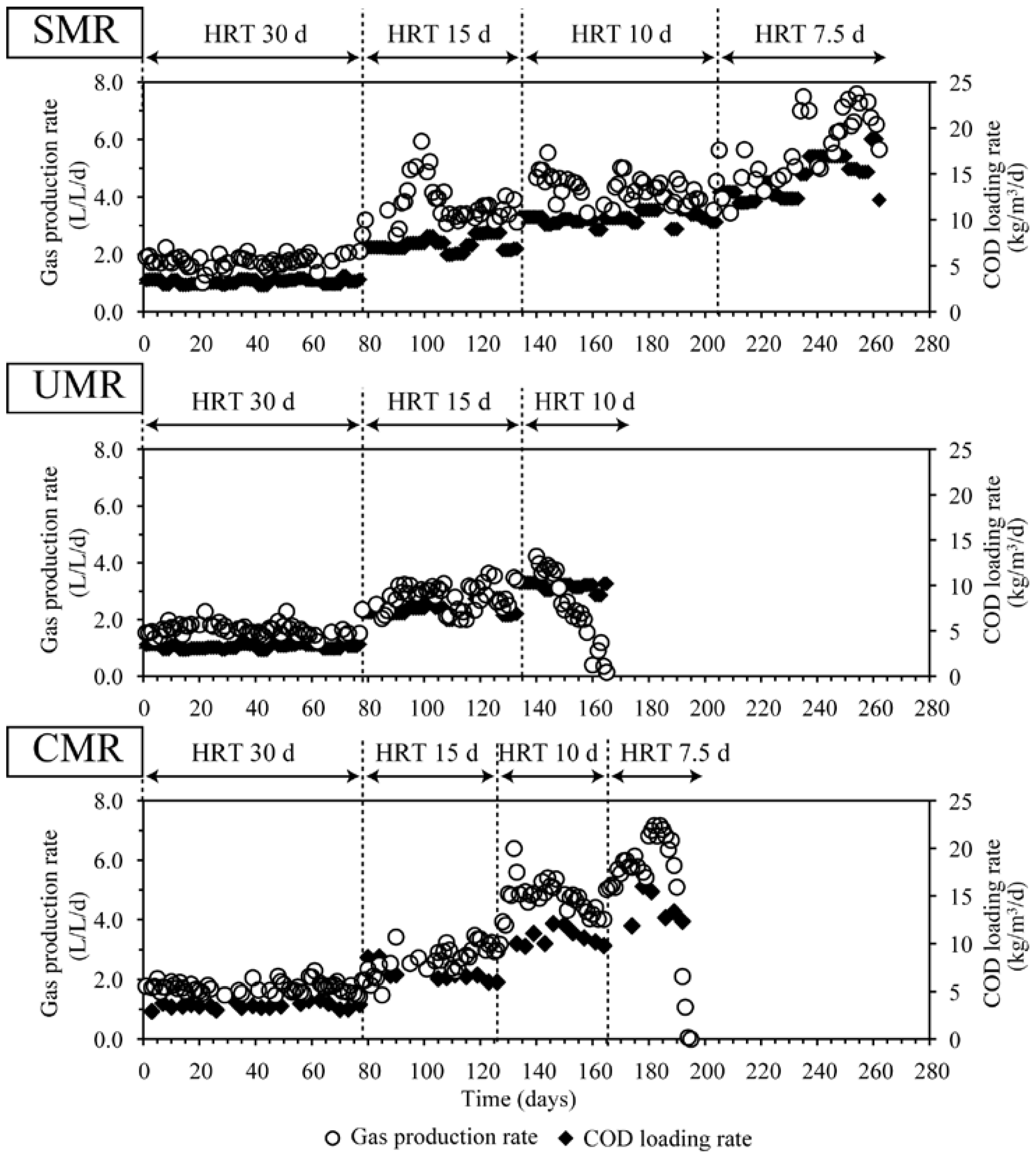
2.2. COD Reduction and Methane Conversion
| Reactor | SMR | UMR | CMR | ||||||
|---|---|---|---|---|---|---|---|---|---|
| HRT (d) | 30 | 15 | 10 | 7.5 | 30 | 15 | 30 | 15 | 10 |
| COD in the effluent sludge (g/L) | 7.1 ± 1.1 | 23.0 ± 7.4 | 18.1 ± 7.7 | 26.0 ± 4.2 | 10.9 ± 2.6 | 22.3 ± 7.9 | 20.1 ± 1.6 | 18.3 ± 1.6 | 33.5 ± 5.2 |
| COD reduction rate (RedCOD) (%) | 93.2 ± 1.4 | 82.6 ± 5.4 | 82.1 ± 6.2 | 73.9 ± 6.0 | 89.8 ± 2.4 | 81.3 ± 8.3 | 83.1 ± 2.7 | 80.2 ± 4.0 | 67.1 ± 3.4 |
| Methane production rate (L/L/day) | 1.1 ± 0.1 | 2.1 ± 0.2 | 2.5 ± 0.3 | 3.7 ± 0.5 | 0.9 ± 0.2 | 1.6 ± 0.3 | 1.1 ± 0.1 | 1.8 ± 0.2 | 2.7 ± 0.3 |
| Methane conversion rate (MeCOD) (%) | 92.1 ± 7.5 | 83.4 ± 2.1 | 71.0 ± 10.2 | 67.9 ± 7.7 | 75.1 ± 10.9 | 63.3 ± 14.3 | 83.6 ± 9.2 | 81.2 ± 11.5 | 71.5 ± 3.2 |
| Total COD recovery efficiency (RecCOD) (%) | 99.0 ± 8.4 | 102.0 ± 11.2 | 88.9 ± 9.9 | 89.2 ± 8.2 | 85.6 ± 11.9 | 84.0 ± 17.0 | 102.0 ± 10 | 100.3 ± 11.9 | 102.8 ± 8.0 |
2.3. VFA, Alkalinity and Solid Distribution inside The Reactors
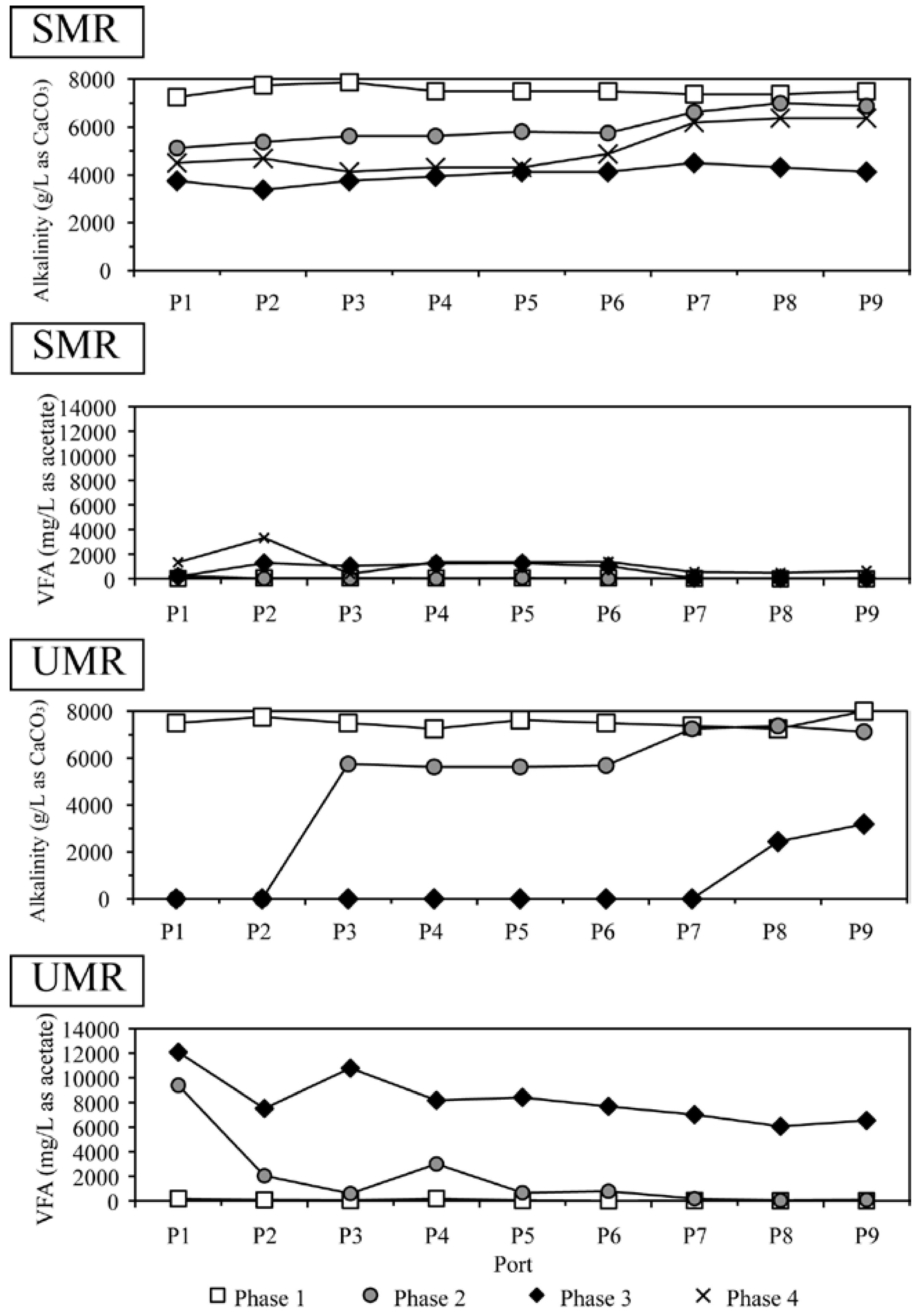
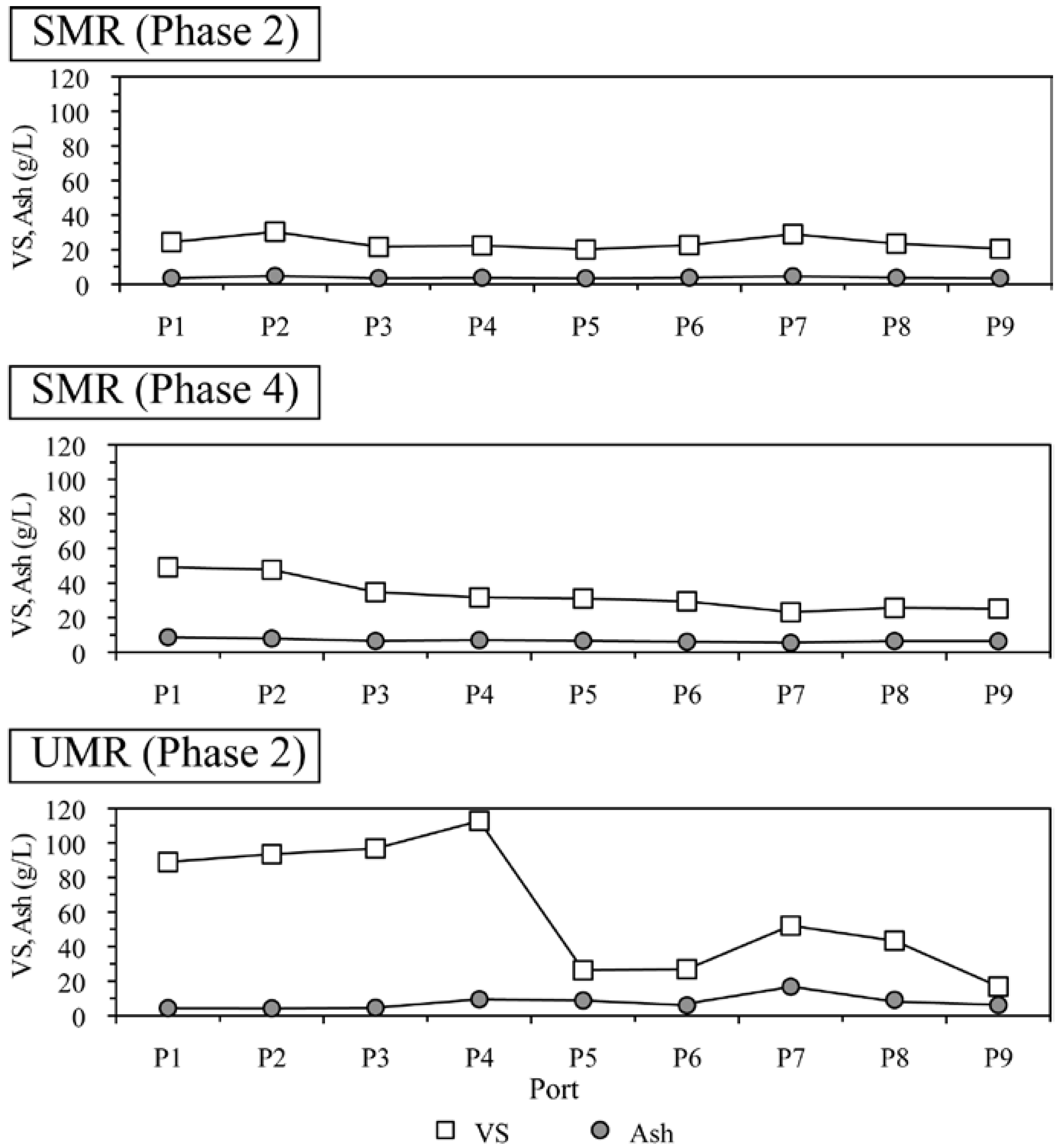
2.4. Microbial Community Structure Responsible for Methane Generation
| OTU | Closest strain (Accession number) | Identity | Clones obtained |
|---|---|---|---|
| SMROTU1 | Methanospirillum hungatei strain JF1 (NR_042789) | 97% | 18 |
| SMROTU2 | Methanosaeta concilii strain Opfikon (NR_028242) | 99% | 11 |
| SMROTU3 | Methanosarcina thermophila (NR_044725) | 98% | 1 |
| UMROTU1 | Methanosaeta concilii strain Opfikon (NR_028242) | 99% | 20 |
| UMROTU2 | Methanobacterium beijingense (NR_028202) | 99% | 4 |
| UMROTU3 | Methanospirillum hungatei strain JF1 (NR_042789) | 97% | 3 |
| UMROTU4 | Methanosarcina mazei strain DSM 2053 | 96% | 1 |
| UMROTU5 | Methanosarcina thermophila (NR_044725) | 98% | 1 |
| UMROTU6 | Methanosaeta concilii strain Opfikon (NR_028242) | 90% | 1 |
| CMROTU1 | Methanosarcina mazei strain DSM 2053 (NR_041956) | 98% | 18 |
| CMROTU2 | Methanospirillum hungatei strain JF1 (NR_042789) | 97% | 8 |
| CMROTU3 | Methanospirillum hungatei strain JF1 (NR_042789) | 92% | 1 |
| CMROTU4 | Methanospirillum hungatei strain JF1 (NR_042789) | 92% | 1 |
| CMROTU5 | Methanoculleus bourgensis strain MS2 (NR_042786) | 99% | 1 |
3. Experimental Section
3.1. Experimental Apparatus
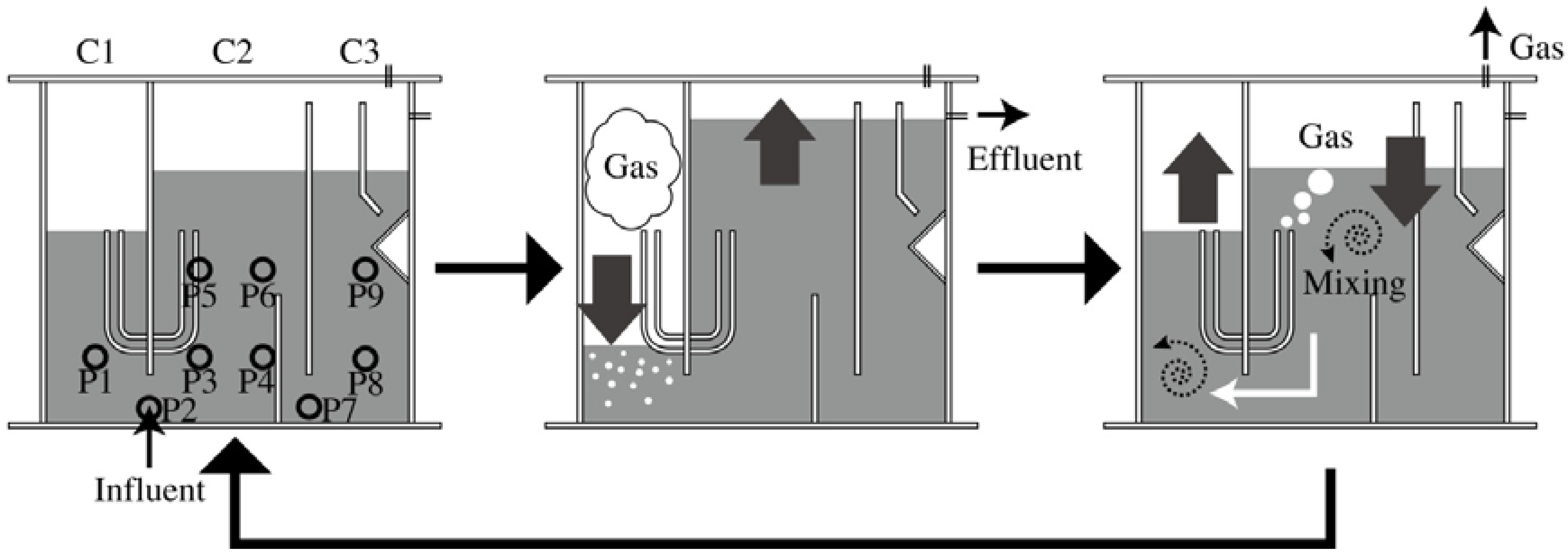
3.2. Feedstock and Seed Sludge
3.3. Chemical Analysis
3.4. Cloning Analysis of 16S rRNA Gene
4. Conclusions
Conflict of Interest
References
- Martí-Herrero, J. Reduced hydraulic retention times in low-cost tubular digesters. Biomass Bioenergy 2011, 35, 4481–4484. [Google Scholar] [CrossRef]
- Rajendran, K.; Aslanzadeh, S.; Taherzadeh, M.J. Household biogas digesters—A review. Energies 2012, 5, 2911–2942. [Google Scholar] [CrossRef]
- Pathkak, H.; Jain, N.; Bhatia, A.; Mohanty, S.; Gupta, N. Global warming mitigation potential of biogas plants in India. Environ. Moint. Assess. 2009, 157, 407–418. [Google Scholar] [CrossRef]
- Rajvanshi, A.K. R & D strategy for lighting and cooking energy for rural households. Curr. Sci. 2003, 85, 437–442. [Google Scholar]
- Van Groenendaal, W.; Gehua, W. Microanalysis of the benefits of China’s family-size bio-digesters. Energy 2010, 35, 4457–4466. [Google Scholar]
- Stroot, P.G.; McMahon, K.D.; Mackie, R.I.; Raskin, L. Anaerobic codigestion of municipal solid waste and biosolids under various mixing conditions-I Digester performance. Water Res. 2001, 35, 1804–1816. [Google Scholar] [CrossRef] [PubMed]
- Karim, K.; Hoffmann, R.; Klasson, T.; Al-Dahhan, M.H. Anaerobic digestion of animal waste: Effect of mode of mixing. Water Res. 2005, 39, 3597–3606. [Google Scholar] [CrossRef] [PubMed]
- Kaparaju, P.; Buendia, I.; Ellegaard, L.; Angelidaki, I. Effects of mixing on methane production during thermophilic anaerobic digestion of manure: lab-scale and pilot-scale studies. Bioresour. Technol. 2008, 99, 4919–4928. [Google Scholar] [CrossRef] [PubMed]
- Karim, K.; Hoffmann, R.; Klasson, T.; Al-Dahhan, M.H. Anaerobic digestion of animal waste: Waste strength versus impact of mixing. Bioresour. Technol. 2005, 96, 1771–1781. [Google Scholar] [CrossRef] [PubMed]
- Borole, A.P.; Klasson, K.T.; Ridenour, W.; Holland, J.; Karim, K.; Al-Dahhan, M.H. Methane production in a 100-L upflow bioreactor by anaerobic digestion of farm waste. Appl. Biochem. Biotechnol. 2006, 131, 887–896. [Google Scholar] [CrossRef] [PubMed]
- Monteith, H.D.; Stephenson, J.P. Mixing Efficiencies in full-scale anaerobic digesters by Tracer methods. J. Water Pollut. Control Fed. 1981, 53, 78–84. [Google Scholar]
- Hamad, M.A.; Abdel Dayem, A.M.; El Halwagi, M.M. Evaluation of the performance of two rural biogas units of Indian and Chinese design. Energy Agric. 1981, 1, 235–250. [Google Scholar] [CrossRef]
- Kobayashi, T.; Li, Y.Y. Performance and characterization of a newly developed self-agitated anaerobic reactor with biological desulfurization. Bioresour. Technol. 2011, 102, 5580–5588. [Google Scholar] [CrossRef] [PubMed]
- Qi, W.K.; Hojo, T.; Li, Y.Y. Hydraulic characteristics simulation of an innovative self-agitation anaerobic baffled reactor (SA-ABR). Bioresour. Technol. 2013, in press. [Google Scholar]
- McMahon, K.D.; Stroot, P.G.; Mackie, R.I.; Raskin, L. Anaerobic codigestion of municipal solid waste and biosolids under various mixing conditions-II: Microbial population dynamics. Water Res. 2001, 35, 1817–1827. [Google Scholar] [CrossRef] [PubMed]
- Barampouti, E.M.; Mai, S.T.; Vlyssides, A.G. Dynamic modeling of the ratio volatile fatty acids/bicarbonate alkalinity in a UASB reactor for potato processing wastewater treatment. Environ. Monit. Assess. 2005, 110, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Gopala Krishna, G.V.; Kumar, P.; Kumar, P. Treatment of low strength complex wastewater using an anaerobic baffled reactor (ABR). Bioresour. Technol. 2008, 99, 8193–8200. [Google Scholar] [CrossRef] [PubMed]
- Roy, C.S.; Talbot, G.; Topp, E.; Beaulieu, C.; Palin, M.F.; Massé, D.I. Bacterial community dynamics in an anaerobic plug-flow type bioreactor treating swine manure. Water Res. 2009, 43, 21–32. [Google Scholar] [CrossRef] [PubMed]
- Namsree, P.; Suvajittanont, W.; Puttanlek, C.; Uttapap, D.; Rungsardthong, V. Anaerobic digestion of pineapple pulp and peel in a plug-flow reactor. J. Environ. Manag. 2012, 110, 40–47. [Google Scholar] [CrossRef]
- Nagao, N.; Tajima, N.; Kawai, M.; Niwa, C.; Kurosawa, N.; Matsuyama, T.; Yusoff, F.M.; Toda, T. Maximum organic loading rate for the single-stage wet anaerobic digestion of food waste. Bioresour. Technol. 2012, 118, 210–218. [Google Scholar] [CrossRef] [PubMed]
- Halalsheh, M.M.; Koppes, J.; den Elzen, J.; Zeeman, G.; Fayyad, M.; Lettinga, G. Effect of SRT and temperature on biological conversions and the related scum-forming potential. Water Res. 2005, 39, 2475–2482. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Jahng, D. Long-term anaerobic digestion of food waste stabilized by trace elements. Waste Manag. 2005, 32, 1509–1515. [Google Scholar] [CrossRef]
- Langenhoff, A.A.M.; Intrachandra, N.; Stuckey, D.C. Treatment of dilute soluble and colloidal wastewater using an anaerobic baffled reactor: Influence of hydraulic retention time. Water Res. 2000, 34, 1307–1317. [Google Scholar] [CrossRef]
- Boopathy, P.; Tilche, A. Anaerobic digestion of high strength molasses wastewater using hybrid anaerobic baffled reactor. Water Res. 1991, 25, 785–790. [Google Scholar] [CrossRef]
- Fukuzaki, S.; Nishio, N.; Nagai, S. Kinetics of the methanogenic fermentation of acetate. Appl. Environ. Microbiol. 1990, 56, 3158–3163. [Google Scholar] [PubMed]
- Karakashev, D.; Batstone, D.J.; Trably, E.; Angelidaki, I. Acetate oxidation is the dominant methanogenic pathway from acetate in the absence of Methanosaetaceae. Appl. Environ. Microbiol. 2006, 72, 5138–5141. [Google Scholar] [CrossRef] [PubMed]
- Nettmann, E.; Bergmann, I.; Pramschu fer, S.; Mundt, K.; Plogsties, V.; Herrmann, C.; Klocke, M. Polyphasic analyses of methanogenic archaeal communities in agricultural biogas plants. Appl. Environ. Microbiol. 2010, 76, 2540–2548. [Google Scholar] [CrossRef] [PubMed]
- Karakashev, D.; Batstone, D.J.; Angelidaki, I. Influence of environmental conditions on methanogenic compositions in anaerobic biogas reactors. Appl. Environ. Microbiol. 2005, 71, 331–338. [Google Scholar] [CrossRef] [PubMed]
- Vavilin, V.A.; Angelidaki, I. Anaerobic degradation of solid material: Importance of initiation centers for methanogenesis, mixing intensity, and 2D distributed model. Biotechnol Bioeng. 2005, 89, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, R.A.; Garcia, M.L.; Veskivar, M.; Karim, K.; Al-Dahhan, M.H.; Angenent, L.T. Effect of shear on performance and microbial ecology of continuously stirred anaerobic digesters treating animal manure. Biotechnol. Bioeng. 2008, 100, 38–48. [Google Scholar] [CrossRef] [PubMed]
- Schnürer, A.; Nordberg, A. Ammonia, a selective agent for methane production by syntrophic acetate oxidation at mesophilic temperature. Water Sci. Technol. 2008, 57, 735–740. [Google Scholar] [CrossRef] [PubMed]
- Grosskopf, R.; Janssen, P.H.; Liesack, W. Diversity and structure of the methanogenic community in anoxic rice paddy soil microcosms as examined by cultivation and direct 16S rRNA gene sequence retrieval. Appl. Environ. Microbiol. 1998, 64, 960–969. [Google Scholar] [PubMed]
- Lane, D.J. 16S/23S rRNA Sequencing. In Nucleic Acid Techniques in Bacterial Systematics; Stackebrandt, E.R., Goodfellow, M., Eds.; John Wiley and Sons Ltd.: Chichester, UK, 1991; pp. 115–175. [Google Scholar]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kobayashi, T.; Wu, Y.-P.; Xu, K.-Q.; Li, Y.-Y. Effect of Mixing Driven by Siphon Flow: Parallel Experiments Using the Anaerobic Reactors with Different Mixing Modes. Energies 2013, 6, 4207-4222. https://doi.org/10.3390/en6084207
Kobayashi T, Wu Y-P, Xu K-Q, Li Y-Y. Effect of Mixing Driven by Siphon Flow: Parallel Experiments Using the Anaerobic Reactors with Different Mixing Modes. Energies. 2013; 6(8):4207-4222. https://doi.org/10.3390/en6084207
Chicago/Turabian StyleKobayashi, Takuro, Ya-Peng Wu, Kai-Qin Xu, and Yu-You Li. 2013. "Effect of Mixing Driven by Siphon Flow: Parallel Experiments Using the Anaerobic Reactors with Different Mixing Modes" Energies 6, no. 8: 4207-4222. https://doi.org/10.3390/en6084207
APA StyleKobayashi, T., Wu, Y.-P., Xu, K.-Q., & Li, Y.-Y. (2013). Effect of Mixing Driven by Siphon Flow: Parallel Experiments Using the Anaerobic Reactors with Different Mixing Modes. Energies, 6(8), 4207-4222. https://doi.org/10.3390/en6084207




