A Two-Stage Continuous Fermentation System for Conversion of Syngas into Ethanol
Abstract
:1. Introduction
2. Results and Discussion
2.1. Cell Growth
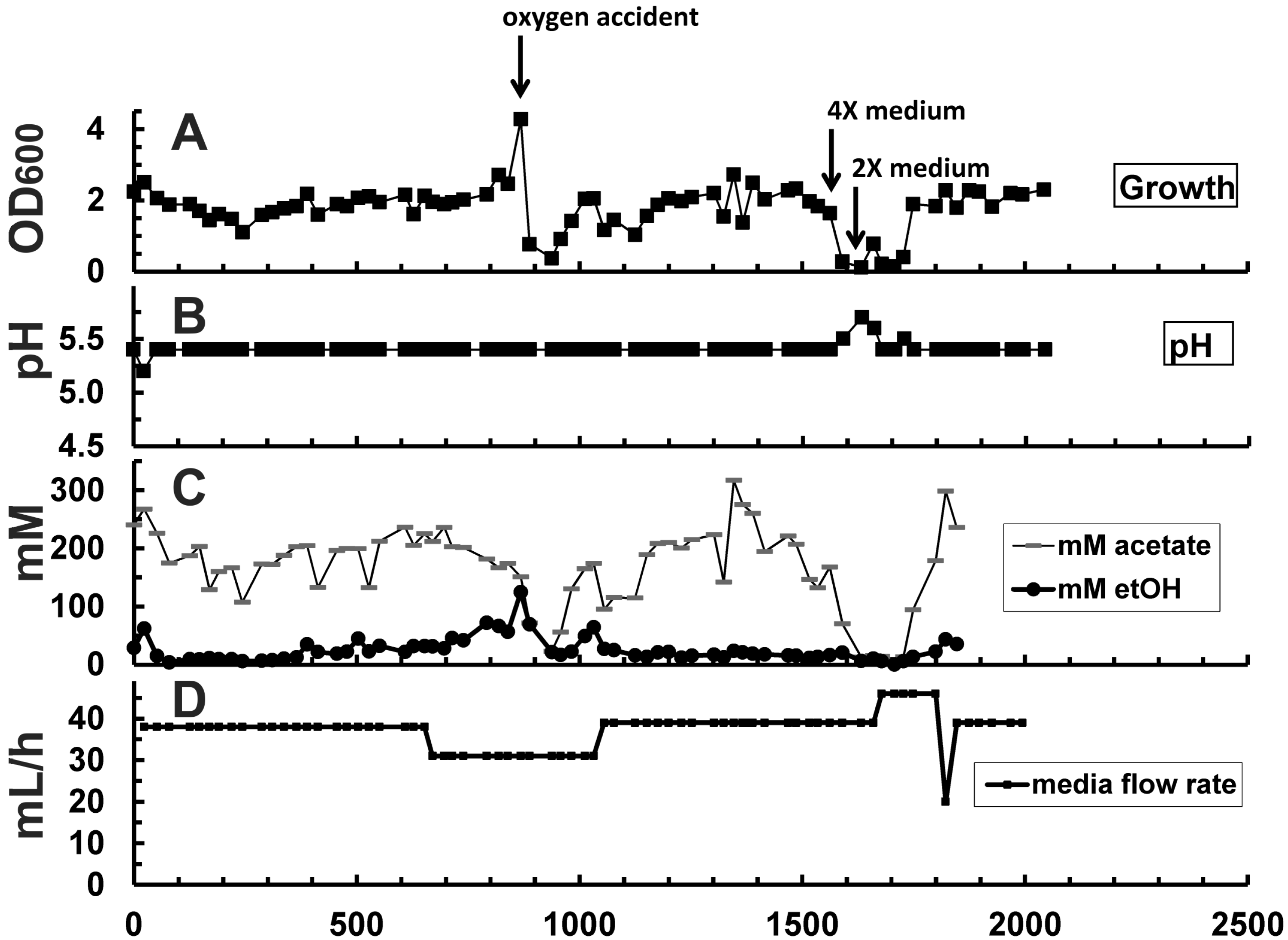
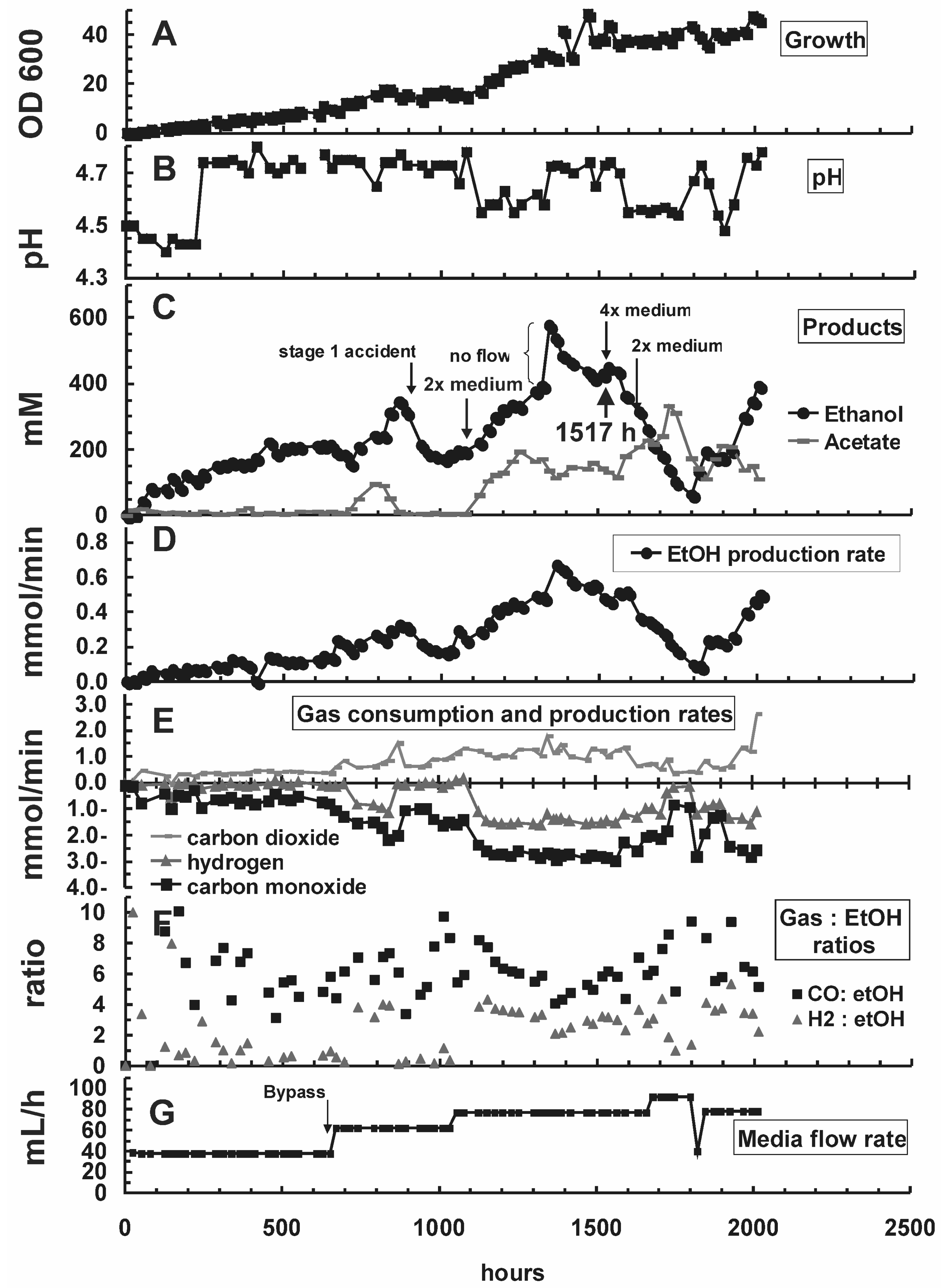
| Hour | Change in reactor condition (Figure 2B,G) | Effect on reactor performance (Figure 2A, C–E) | ||
|---|---|---|---|---|
| Stage one | Stage two | Stage one | Stage two | |
| 0–650 | D = 0.039 h−1 pH = 5.5 | D = 0.010 h−1 pH = 4.4–4.8 | Growth at equilibrium, OD600 at ~2.0 | Inoculation; OD600 increased at an initial rate of 0.37/day to ~9.9 |
| 650–888 | Activated media bypass channel (Figure 1), increased overall media flow rate by 50% to 62 mL/h; increased dilution rate in stage 2 relatively to stage 1 | OD600 increased to ~2.4 | OD600 increased to 17.8; higher reactor performance (Figure 2C–E) | |
| 888 | ACCIDENT: ambient air drawn into stage 1 | OD600 declined to 0.37, recovery completed at 1013 h | OD600 was not affected, but metabolic rates declined (Figure 2C–E) | |
| 1056 | D = 0.039 h−1 | D = 0.020 h−1 | OD600 decreased to ~2.0 | OD600 increased to ~30.0 |
| 1347 | ACCIDENT: Media pump stopped for several hours | Rapid increase of ethanol concentration from 393 to 576 mM; stabilized at 400–450 mM ethanol within 100 h after repairing the pump | ||
| 1366 | Raised gas recycle rate from 180 to 430 mL/min to increase syngas retention time and mass transfer | Stage 2: sudden rise of OD600 to 41.7 (Figure 2A) due to stir up of settled cells. OD600 stayed at higher level until end of experiment.Syngas consumption did not improve significantly (Figure 2E) | ||
| 1535–1632 | 2× medium was exchanged with 4× medium in the reservoir for 97 h to improve supply with nutrients | OD600 declined from 2.0 to 0.1. Recovery completed at 1749 h. | Salt shock did not affect OD600, but metabolic rates declined. Recovery started at 1800 h | |
| 1679 | D = 0.046 h−1 | D = 0.023 h−1 | New OD600 level settled at ~1.8 | New OD600 level settled at ~43.4 |
| 1800 | D = 0.039 h−1 | D = 0.020 h−1 | OD600 back at ~2.0 | Recovery from salt shock started |
| 2014 | End of experiment | Final OD600 of 46.2; ethanol concentration of 394 mM | ||
| Compound | Concentrations | ||
|---|---|---|---|
| Outlet stage 1 | Outlet stage 2 | Inlet stages 1 & 2 | |
| CO (G), (vol%) | 53 | 19 | 60 |
| H2 (G), (vol%) | 34 | 14 | 35 |
| CO2 (G), (vol%) | 13 | 63 | 5 |
| Ethanol (L), (mM) | 11.5 | 428.4 | NA |
| Acetic acid (L), (mM) | 146.5 | 142.5 | NA |
| Bacteria (g DW/L) | 0.476 | 9.34 | NA |
| Compounds | Rates [mmol/(L·min)] | ||
| Stage 1 | Stage 2 | Total | |
| CO in | 0.607 | 0.808 | 0.768 |
| CO out | 0.330 | 0.110 | 0.154 |
| CO consumption | 0.277 | 0.698 | 0.614 |
| H2 in | 0.354 | 0.471 | 0.448 |
| H2 out | 0.182 | 0.085 | 0.105 |
| H2 consumption | 0.172 | 0.386 | 0.343 |
| CO2 in | 0.051 | 0.067 | 0.064 |
| CO2 out | 0.085 | 0.371 | 0.314 |
| CO2 production | 0.034 | 0.303 | 0.250 |
| Ethanol production | 0.007 | 0.136 | 0.110 |
| Acetic acid production | 0.094 | 0.025 | 0.039 |
| Compounds | Efficiencies (%) | ||
| Stage 1 | Stage 2 | Total | |
| CO consumption | 46 | 86 | 80 |
| H2 consumption | 49 | 82 | 77 |
2.2. Substrate Consumption and Product Formation
3. Experimental Section
3.1. Biocatalyst and Growth Conditions
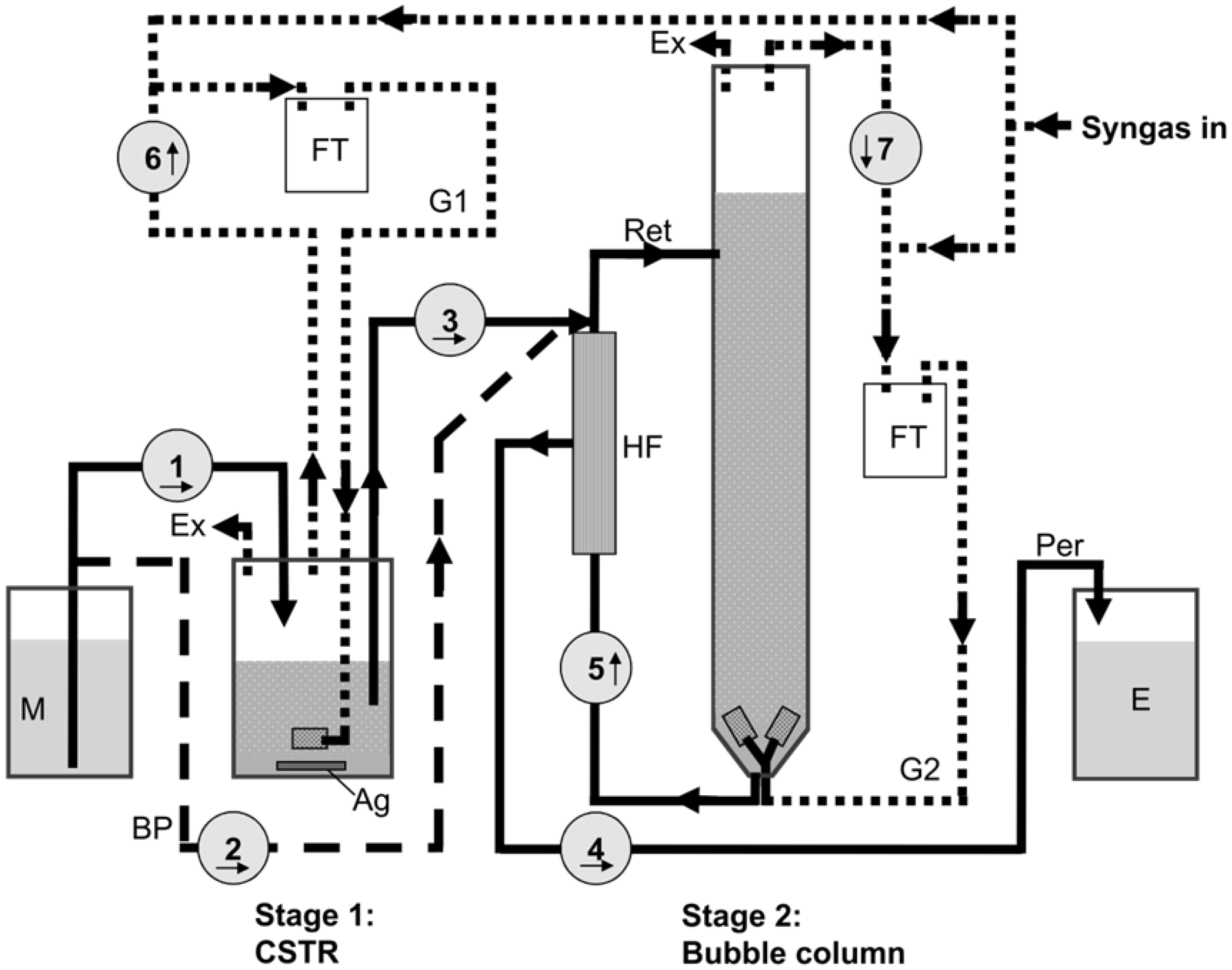
3.2. Reactor Setup
3.3. Analytical Procedures
4. Conclusions and Outlook
- -
- The relatively low ethanol concentration of 2% in the effluent requires advanced strategies for distillation to keep the energy balance of the entire process positive. A promising method has been described recently [24];
- -
- -
Acknowledgements
Conflict of Interest
References
- Köpke, M.; Held, C.; Hujer, S.; Liesegang, H.; Wiezer, A.; Wollherr, A.; Ehrenreich, A.; Liebl, W.; Gottschalk, G.; Dürre, P. Clostridium ljungdahlii represents a microbial production platform based on syngas. Proc. Natl. Acad. Sci. USA 2010, 107, 13087–13092. [Google Scholar] [CrossRef] [PubMed]
- Henstra, A.M.; Sipma, J.; Rinzema, A.; Stams, A.J. Microbiology of synthesis gas fermentation for biofuel production. Curr. Opin. Biotechnol. 2007, 18, 200–206. [Google Scholar] [CrossRef] [PubMed]
- Munasinghe, P.C.; Khanal, S.K. Biomass-derived syngas fermentation into biofuels: Opportunities and challenges. Bioresour. Technol. 2010, 101, 5013–5022. [Google Scholar] [CrossRef] [PubMed]
- Daniell, J.; Köpke, M.; Simpson, S.D. Commercial biomass syngas fermentation. Energies 2012, 5, 5372–5417. [Google Scholar] [CrossRef]
- Abubackar, H.N.; Veiga, M.C.; Kennes, C. Biological conversion of carbon monoxide: Rich syngas or waste gases to bioethanol. Biofuels Bioprod. Biorefin. 2011, 5, 93–114. [Google Scholar] [CrossRef]
- Gaddy, J.L.; Arora, D.K.; Ko, C.-W.; Phillips, J.R.; Basu, R.; Wikstrom, K.V.; Clausen, E.C. Methods for Increasing the Production of Ethanol from Microbial Fermentation. U.S. Patent 7,285,402 B2, 23 October 2003. [Google Scholar]
- Kundiyana, D.K.; Huhnke, R.L.; Wilkins, M.R. Syngas fermentation in a 100-L pilot scale fermentor: Design and process considerations. J. Biosci. Bioeng. 2010, 109, 492–198. [Google Scholar] [CrossRef] [PubMed]
- Klasson, K.T.; Ackerson, M.D.; Clausen, E.C.; Gaddy, J.L. Biological conversion of coal and coal-derived synthesis gas. Fuel 1993, 72, 1673–1678. [Google Scholar] [CrossRef]
- Klasson, K.; Elmore, B.; Vega, J.; Ackerson, M.; Clausen, E.; Gaddy, J. Biological production of liquid and gaseous fuels from synthesis gas. Appl. Biochem. Biotechnol. 1990, 24–25, 857–873. [Google Scholar] [CrossRef]
- Kundiyana, D.K.; Huhnke, R.L.; Wilkins, M.R. Effect of nutrient limitation and two-stage continuous fermentor design on productivities during “Clostridium ragsdalei” syngas fermentation. Bioresour. Technol. 2011, 102, 6058–6064. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.T.; Woods, D.R. Acetone-butanol fermentation revisited. Microbiol. Rev. 1986, 50, 484–524. [Google Scholar] [PubMed]
- Younesi, H.; Najafpour, G.; Mohamed, A.R. Ethanol and acetate production from synthesis gas via fermentation processes using anaerobic bacterium, Clostridium ljungdahlii. Biochem. Eng. J. 2005, 27, 110–119. [Google Scholar] [CrossRef]
- Worden, R.M.; Grethlein, A.J.; Jain, M.K.; Datta, R. Production of butanol and ethanol from synthesis gas via fermentation. Fuel 1991, 70, 615–619. [Google Scholar] [CrossRef]
- Abubackar, H.N.; Veiga, M.C.; Kennes, C. Biological conversion of carbon monoxide to ethanol: Effect of pH, gas pressure, reducing agent and yeast extract. Bioresour. Technol. 2012, 114, 518–522. [Google Scholar] [CrossRef] [PubMed]
- Richter, H.; Qureshi, N.; Heger, S.; Dien, B.; Cotta, M.A.; Angenent, L.T. Prolonged conversion of n-butyrate to n-butanol with Clostridium saccharoperbutylacetonicum in a two-stage continuous culture with in-situ product removal. Biotechnol. Bioeng. 2012, 109, 913–921. [Google Scholar] [CrossRef] [PubMed]
- Bredwell, M.D.; Worden, R.M. Mass-transfer properties of microbubbles. 1. Experimental studies. Biotechnol. Prog. 1998, 14, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Perez, J.M.; Richter, H.; Loftus, S.E.; Angenent, L.T. Biocatalytic reduction of short-chain carboxylic acids into their corresponding alcohols with syngas fermentation. Biotechnol. Bioeng. 2013, 110, 1066–1077. [Google Scholar] [CrossRef] [PubMed]
- Saxena, J.; Tanner, R.S. Effect of trace metals on ethanol production from synthesis gas by the ethanologenic acetogen, Clostridium ragsdalei. J. Ind. Microbiol. Biotechnol. 2011, 38, 513–521. [Google Scholar] [CrossRef] [PubMed]
- Buckel, W. Anaerobic Energy Metabolism. In Biology of the Prokaryotes; Lengeler, J.W., Drews, G., Schlegel, H.G., Eds.; Thieme: Stuttgart, Germany, 1999; pp. 278–326. [Google Scholar]
- Ramachandriya, K.D.; Wilkins, M.R.; Delorme, M.J.M.; Zhu, X.; Kundiyana, D.K.; Atiyeh, H.K.; Huhnke, R.L. Reduction of acetone to isopropanol using producer gas fermenting microbes. Biotechnol. Bioeng. 2011, 108, 2330–2338. [Google Scholar] [CrossRef] [PubMed]
- Köpke, M.; Mihalcea, C.; Liew, F.; Tizard, J.H.; Ali, M.S.; Conolly, J.J.; Al-Sinawi, B.; Simpson, S.D. 2,3-butanediol production by acetogenic bacteria, an alternative route to chemical synthesis, using industrial waste gas. Appl. Environ. Microbiol. 2011, 77, 5467–5475. [Google Scholar] [CrossRef] [PubMed]
- Datar, R.P.; Shenkman, R.M.; Cateni, B.G.; Huhnke, R.L.; Lewis, R.S. Fermentation of biomass-generated producer gas to ethanol. Biotechnol. Bioeng. 2004, 86, 587–594. [Google Scholar] [CrossRef] [PubMed]
- Website of Bio-Process Innovation, Inc. Available online: www.bio-process.com/process-technologies/fermentation (accessed on 30 July 2013).
- Vane, L.M.; Alvarez, F.R.; Huang, Y.; Baker, R.W. Experimental validation of hybrid distillation-vapor permeation process for energy efficient ethanol–water separation. J. Chem. Technol. Biotechnol. 2010, 85, 502–511. [Google Scholar]
- Xu, D.; Tree, D.R.; Lewis, R.S. The effects of syngas impurities on syngas fermentation to liquid fuels. Biomass Bioenergy 2011, 35, 2690–2696. [Google Scholar] [CrossRef]
- Phillips, J.R.; Remondet, N.M.; Atiyeh, H.K.; Wilkins, M.R.; Huhnke, R.L. Designing Syngas Fermentation Medium for Fuels and Bulk Chemicals Production. In Proceedings of 2011 ASABE Annual International Meeting, Louisville, KY, USA, 7–10 August 2011; paper # 1111052. pp. 1–12.
- Saxena, J.; Tanner, R. Optimization of a corn steep medium for production of ethanol from synthesis gas fermentation by Clostridium ragsdalei. World J. Microbiol. Biotechnol. 2012, 28, 1553–1561. [Google Scholar] [PubMed]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Richter, H.; Martin, M.E.; Angenent, L.T. A Two-Stage Continuous Fermentation System for Conversion of Syngas into Ethanol. Energies 2013, 6, 3987-4000. https://doi.org/10.3390/en6083987
Richter H, Martin ME, Angenent LT. A Two-Stage Continuous Fermentation System for Conversion of Syngas into Ethanol. Energies. 2013; 6(8):3987-4000. https://doi.org/10.3390/en6083987
Chicago/Turabian StyleRichter, Hanno, Michael E. Martin, and Largus T. Angenent. 2013. "A Two-Stage Continuous Fermentation System for Conversion of Syngas into Ethanol" Energies 6, no. 8: 3987-4000. https://doi.org/10.3390/en6083987




