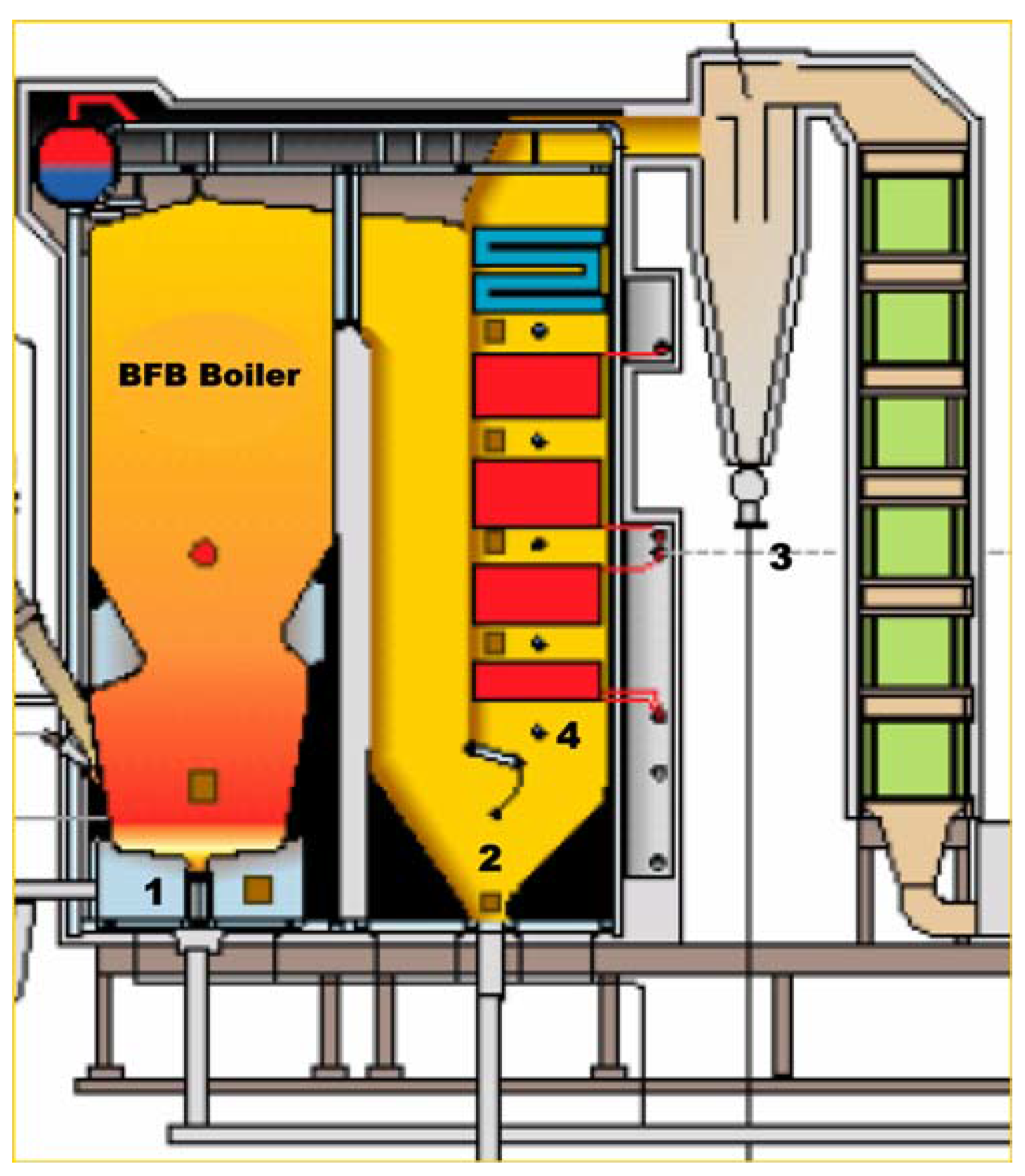
1. Introduction
Increasing demand for sustainable heat and power production, as well as problems landfilling wastes, has prompted heat and power plants to replace fossil fuels with alternatives such as combustible municipal solid wastes (MSW) or refuse-derived fuels (RDF) [
1,
2]. Combustion of MSW and RDF, however, can lead to serious fouling and corrosion problems, due to their physical and chemical properties. The major difficulties with thermally processing waste fuels are attributed to their heterogeneity, high concentrations of alkali and chlorine, and high content of inorganic incombustible materials [
3]. Fluidized-bed boilers, with their inherent fuel flexibility, have been developed to resolve problems concerning fuel heterogeneity and high ash content [
4]. High concentrations of alkali and chlorine compounds could cause severe corrosion of steel surfaces, particularly at temperatures above 450 °C. Potassium, sodium, and chlorine are known to be the main causes of severe high-temperature corrosion [
5]. In addition, interaction between alkali species and silica sand leads to the formation of low-temperature eutectics, which could cause the agglomeration/defluidization of fluidized-bed boilers [
6]. Formation of bed and fly ashes, which may contain large amounts of potentially toxic metal compounds, is also a problem when waste fuels are combusted. These ashes, which could harm the environment, are classified as hazardous materials and need special treatment and storage when landfilled [
7].
Regardless of these problems, the combustion of waste fuels is still an interesting option, considering their often high energy content, their abundance, and the economic aspects of combusting them. Improved boiler operation has been investigated by either modifying the boiler function or changing the fuel mix. For example, a previous full-scale project, the Reduced-Bed Temperature (RBT) project [
8], reduced the bed temperature by recirculating flue gas and spraying water on the fuel to modify the boiler function. The outcome was positive in terms of deposit formation rate, fly ash flow rate, reduced risk of bed agglomeration and some capture of chlorine in the bed [
8]. Co-combustion and additive use have also proved useful in alleviating the addressed problems. Co-combustion of secondary waste fuels, such as sewage sludge and animal waste, has previously shown benefits in some projects [
9,
10,
11]. Animal waste, a new fuel type, has proved beneficial when co-fired with wood chips, sorted MSW, and peat in commercial boilers [
11].
The present research investigated co-combustion of animal waste with a mix of sorted industrial and sorted household waste, in a full-scale bubbling fluidized bed (BFB) boiler. The paper investigates the impact of the new fuel composition on the boiler operation. Especially the effects on operational issues, such as deposition growth rate in the heat transfer area, bed agglomeration and flue gas emissions, were followed. The distribution of major ash forming elements such as Ca, S, Cl, Na, K and P in the bed and fly ashes were examined. It was particularly significant to find out how the high concentrations of calcium phosphate compounds in the animal waste and its high moisture content [
11] affected the ash transformation behavior in the bed area. Analysis of the formed deposit on the deposit rings was also performed to determine the amount of corrosive elements such as Cl and alkalis. A series of complementary lab-scale tests were also conducted to determine the effect of animal waste co-combustion on bed agglomeration temperature. The emissions of NO
x in the flue gas were measured, to identify whether high nitrogen content of animal waste [
11] may change NO
x emissions in positive or negative way.
3. Results and Discussion
3.1. Fuel Analysis
The results of the fuel analyses are presented in
Table 1. The ash content of the AW is almost half that of the ordinary MSW, making the ash content slightly lower in the resulting fuel mix. The calorific value of the fuel mix, however, decreased from 10.9 MJ kg
−1 to 9.4 MJ kg
−1 with the addition of wet AW.
Table 1.
Fuel analysis.
| Fuel | MSW (Ref) | AW | AW + MSW (AW) | Method |
|---|
| Moist, raw, wt-% | 37.5 | 60.9 | 46.4 | CEN/TS 14774-2 |
| Ash, dry, wt-% | 20.3 | 13.7 | 18.4 | CEN/TS 14775 |
| Dry sample, wt-% | C | 46.9 | 54.3 | 47.1 | CEN/TS 15104 |
| H | 6.20 | 7.90 | 6.30 | CEN/TS 15104 |
| N | 1.20 | 6.30 | 1.80 | CEN/TS 15104 |
| O | 24.3 | 17.3 | 25.3 | Calculated |
| S | 0.46 | 0.30 | 0.40 | CEN/TS 15289 |
| Cl | 0.70 | 0.40 | 0.70 | CEN/TS 15289 |
| LHV, raw, MJ kg−1 | 10.9 | 8.30 | 9.40 | CEN/TS 14918 |
| HHV, raw, MJ kg−1 | 18.8 | 25.0 | 19.7 | CEN/TS 14918 |
| Ash analysis, mg kg−1 ash | K | 3,898 | 2,050 | 2,926 | ASTM D 3682 |
| Na | 6,557 | 4,550 | 6,009 | ASTM D 3682 |
| Ca | 27,405 | 45,100 | 33,876 | ASTM D 3682 |
| Al | 11,611 | <100 | 9,447 | ASTM D 3682 |
| Mg | 2,740 | 1000 | 3,001 | ASTM D 3682 |
| Ti | 1,603 | <100 | 2,437 | ASTM D 3682 |
| Mn | 223 | <100 | 183 | ASTM D 3682 |
| P | 954 | 21,900 | 4,252 | ASTM D 3682 |
| Fe | 4,993 | 400 | 6,035 | ASTM D 3682 |
| Zn | 893 | 2,500 | 1,043 | ASTM D 3683 |
| Pb | 1,482 | <10 | 350 | ASTM D 3683 |
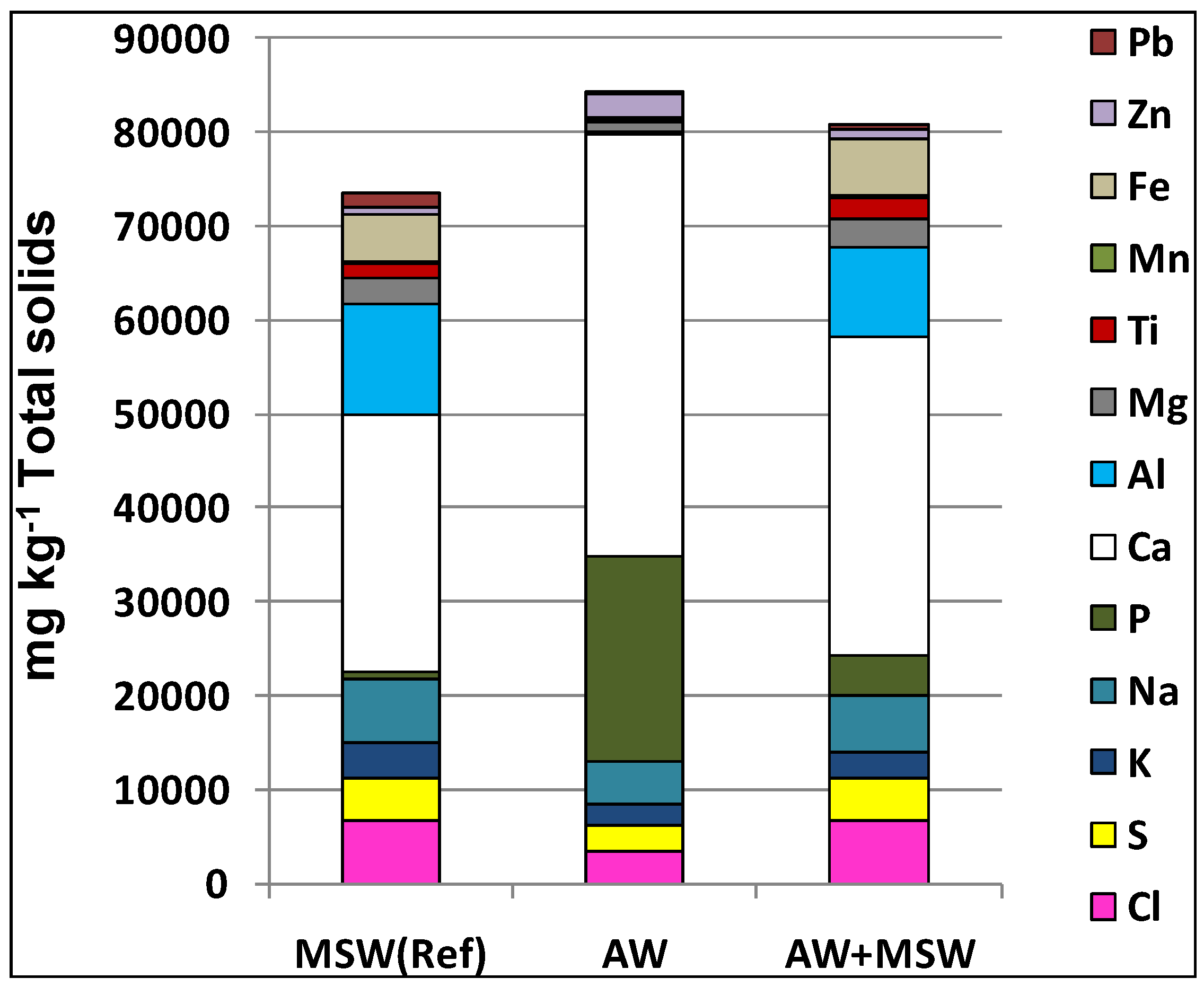
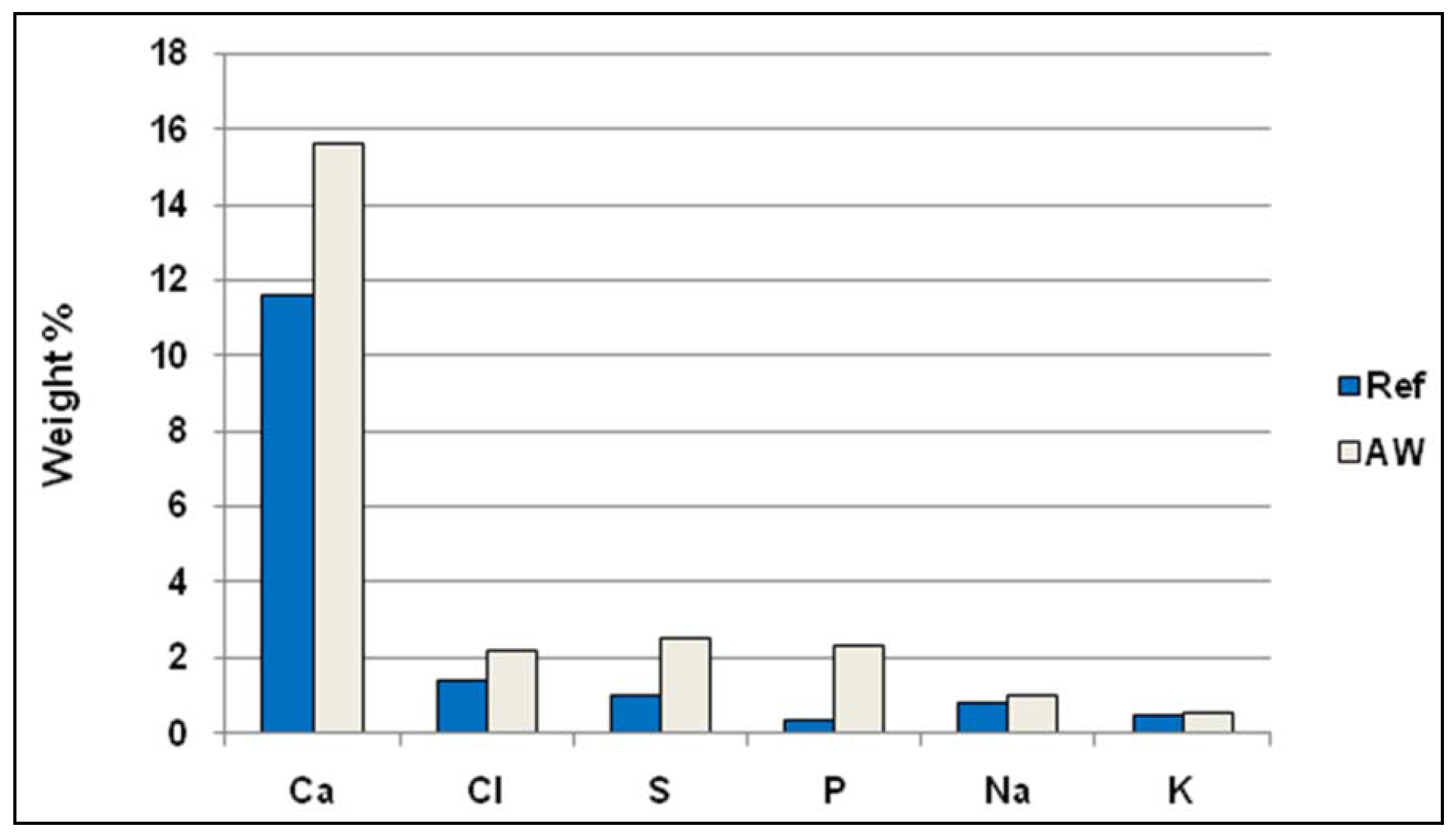
Table 1 also shows that the concentration of N is much higher in AW compared to the ordinary waste mix. The elemental analysis presented in
Figure 3 indicates significantly more P, and Ca and less S, Cl, Na, and K in AW than in the ordinary waste mix. The resulting fuel mix of ordinary waste fuel and AW contains a four times higher phosphorus concentration than does the ordinary waste mix. Note that, due to the characteristics of waste fuels, the composition of this fuel changes over time and was not exactly the same for all tests.
Figure 3.
Composition of fuels and fuel mix.
Figure 3.
Composition of fuels and fuel mix.
3.3. SEM-EDX Analysis on Ashes
SEM-EDX was employed to perform elemental analysis of the coating layers formed during combustion on the particles of sampled return sand and boiler ash. The analyses presented here focus mainly on the return sand, though the coating on the boiler ash was similar in nature. Two types of sand particles are present in the boiler, consisting of silicon oxides or feldspar. Therefore, it was important to determine whether their original composition could have affected the compounds bonding to their surface during combustion. The results, however, showed no indications of differences between the particles and, in contrast to previous findings, no signs could be found of alkali silicate melt on the inner surface of the silica sand particles [
6,
19,
20].
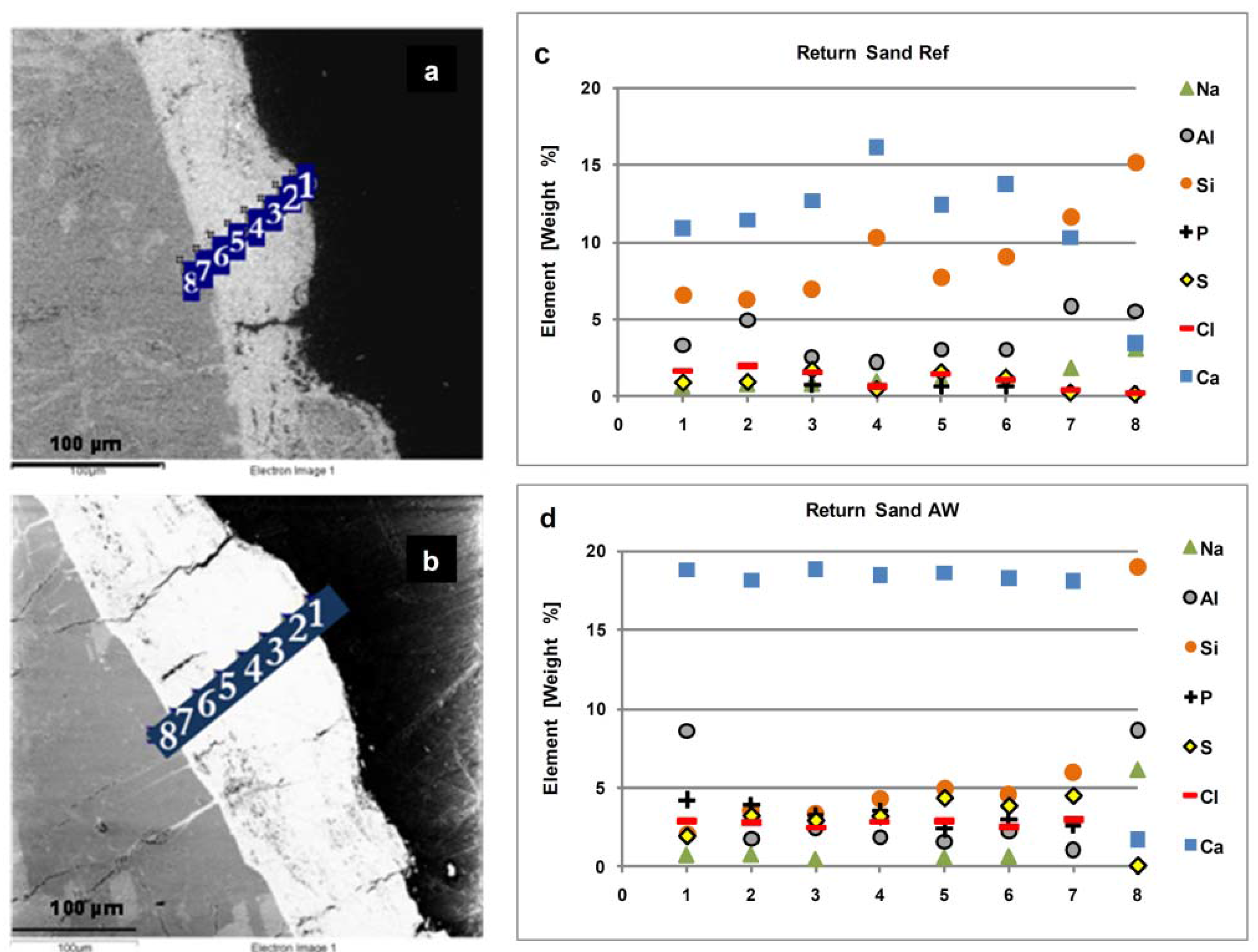
Figure 6a,b shows cross-sectional SEM-EDX spot analyses of the entire coating from its outer layer to the surface of the sand particles, for two return sands from the Ref and AW cases. The corresponding elemental distributions of Cl, S, Ca, Na, P, Al and Si are presented in
Figure 6c,d. Comparison of the two cases indicates that the concentrations of Ca, S, Cl and P are higher in the coating layer on return sand from the AW case. Si and Al, however, are found to be slightly lower in this case. The higher concentrations of Na, Si, and Al at point No. 8 in both cases reflect the original compositions of the feldspars in the sand particles.
Figure 6.
Cross-sectional SEM-EDX spot analysis of the coated return sands: (a) and (c) for the Ref case; (b) and (d) for the AW case.
Figure 6.
Cross-sectional SEM-EDX spot analysis of the coated return sands: (a) and (c) for the Ref case; (b) and (d) for the AW case.
To achieve an average coating composition, three of the analyzed sand particles were picked from each combustion test. The results presented in
Figure 7 confirm the higher concentrations of Ca, Cl, P, and S (which increased by 4.3, 1.3, 2.4 and 1.8 wt%, respectively) in the coating of the return sand particles in the AW case compared to the Ref case. The same coating composition was also found on the bed ash material and on the boiler ash particles. Higher capture of Ca, S, Cl and P compounds in the bed area could be connected to the lower rate of deposit formation in the super heater area. The results for alkali (Na, K), however, did not indicate any notable differences between the two cases, suggesting that the changed boiler conditions did not significantly affect the alkali capture in the bed.
Figure 7.
Average element concentrations in the coating surrounding the sand particles.
Figure 7.
Average element concentrations in the coating surrounding the sand particles.
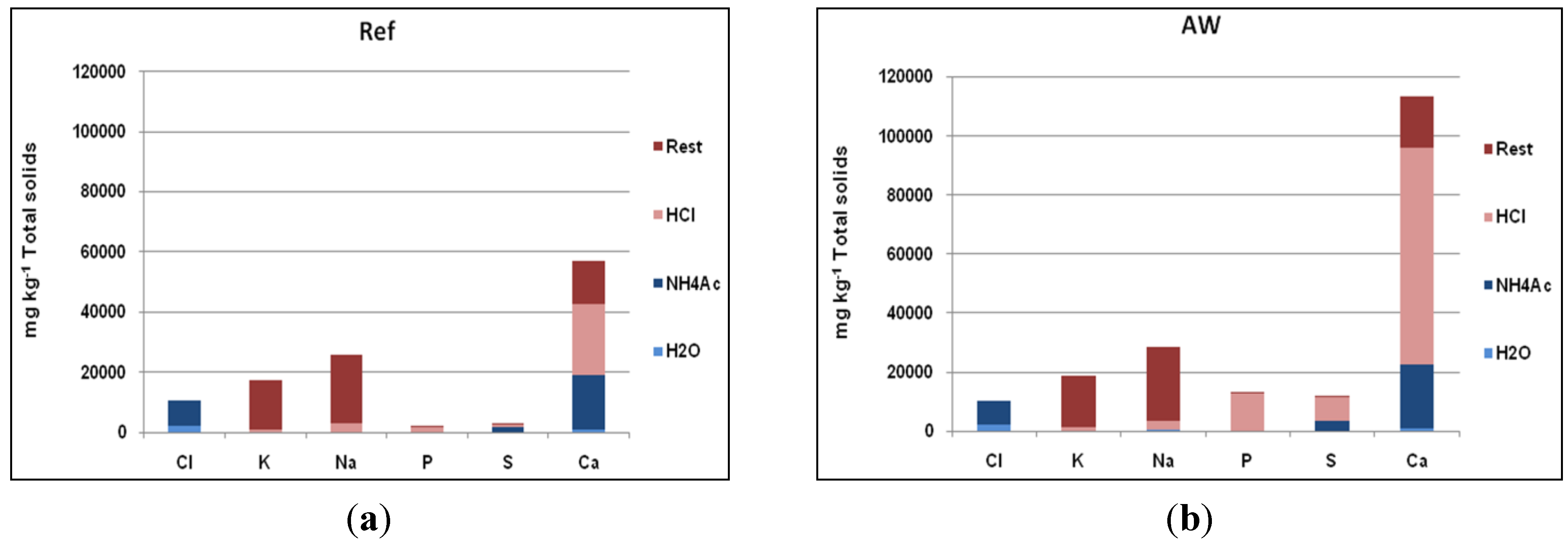
3.4. Chemical Fractionation Results for Return Sand and Cyclone Ash
The chemical fractionation results for the particles more or less support the results obtained using SEM-EDX. As
Figure 8 shows, higher concentrations of Ca, S, and P could be observed in the coating of the sand particles in the AW case, while the difference between the concentrations of Na and K are small. Note also that the largest part of the alkali found in the non-soluble fraction originates from the feldspar particles in the sand.
Figure 8.
Results of chemical fractionation analysis of return sands with coatings: (a) Ref; (b) AW.
Figure 8.
Results of chemical fractionation analysis of return sands with coatings: (a) Ref; (b) AW.
Comparing the two cases, the major differences in the concentrations of Ca, S, and P are found in the acid-soluble fractions. The water-and acetate-soluble fractions, in addition to the insoluble portions of Ca, P, and S are almost the same in both cases. All of the extra Ca, S, and P is found in acid-soluble forms in the AW case. This was partly explained by the fact that AW is rich in crushed bone containing Ca
10(PO
4)
6(OH)
2 and β-Ca
3(PO
4)
2 [
11,
12,
21], which is soluble in acid. But in addition, parts of the P introduced with the AW fuel derive from proteins in the soft tissue of the animals and are reactive during combustion [
22]. This reactive part of P dominates over Si in the competition for Ca
+2 to form calcium-phosphate, which is a stable compound [
23].
The results of chemical fractionation of cyclone ash are presented in
Figure 9. In addition, the concentration of acid-soluble Ca and P apparently increased in the AW case. The solubility of S changed drastically when adding AW. In the Ref case, the largest part of S occurred as acetate-soluble compounds, but in the AW case most S was found as acid-soluble compounds. The results do not indicate in which form this S is found in the ash. For the boiler ash, the results are almost similar to the cyclone ash.
Figure 9.
Results of chemical fractionation analysis of cyclone ash.
Figure 9.
Results of chemical fractionation analysis of cyclone ash.
3.5. XRD
The XRD results for boiler ash, cyclone ash, filter ash and fresh sand are presented in
Table 2. The differences between the three cases are very small, probably due to the original composition of the fresh sand. The ordinary composition of Swedish sands, which contain feldspars, make it difficult to distinguish, analytically, between the sands and ashes. Quartz (SiO
2), albite (NaAlSi
3O
8), microklin (KAlSi
3O
8) and klinoklor ((Mg,Al)
6(Si,Al)
4O
10(OH)
8) are compounds found in the fresh sand. Several of these compounds were also found in the ash, for example, quartz, albite and microklin in the boiler ash.
Despite the limitations of the analysis, one can conclude that no large changes occurred in the crystalline part of the ashes. The cyclone ash in all cases had a high NaCl content, but only the Ref case contained KCl. Regarding the fact that high concentrations of small NaCl crystals in the cyclone ash can impede good analytical results, the samples were washed with deionized water, dried, and then reanalyzed. However, the washing had only a small impact on the results in these cases. Calcium aluminum oxide, calcium aluminum silicate and quartz were the dominant compounds in all cases, and only one phosphate was detected in one case, that of AW.
3.6. Chemical Analysis of Ashes
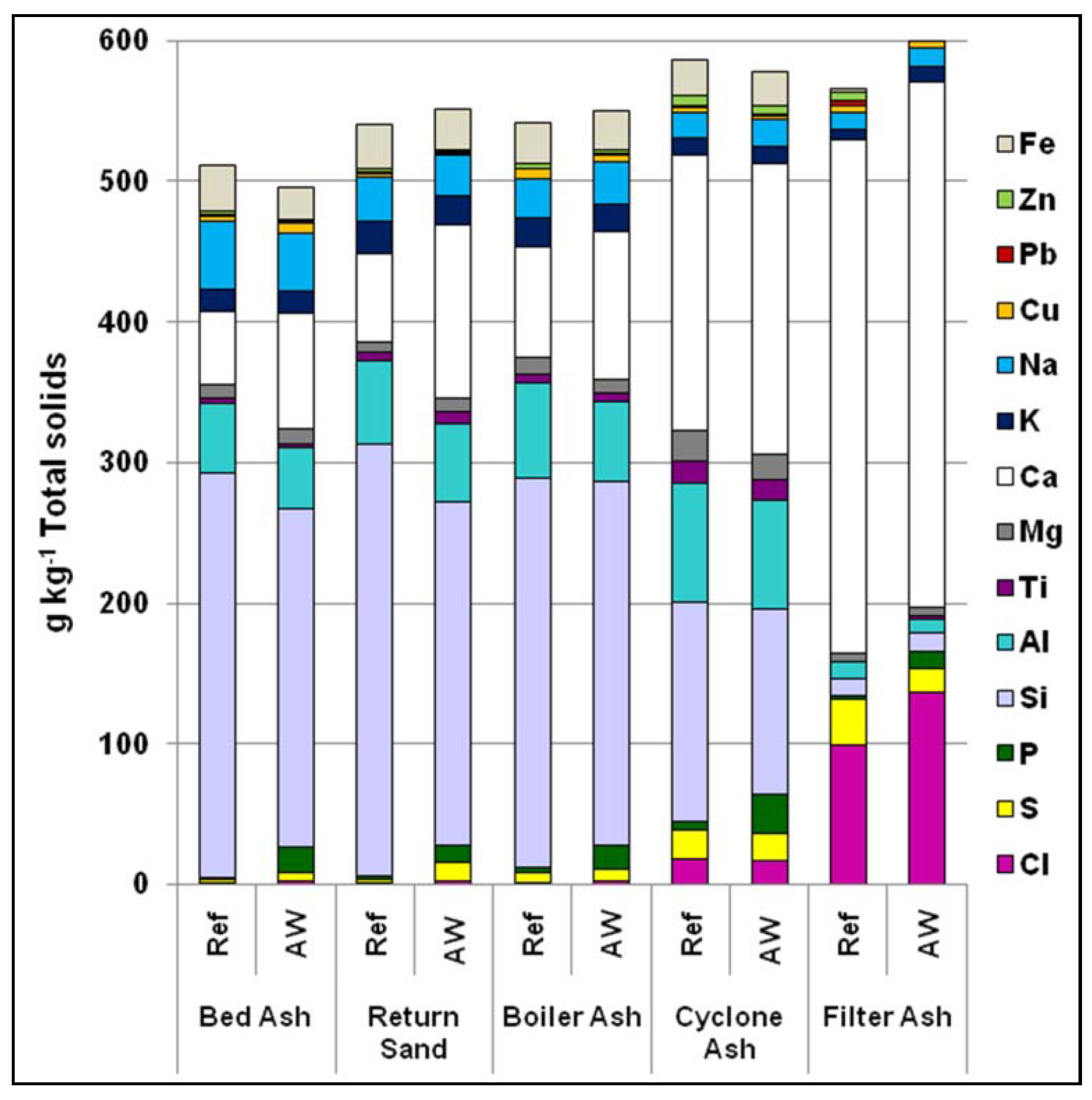
Five ash fractions were analyzed (see
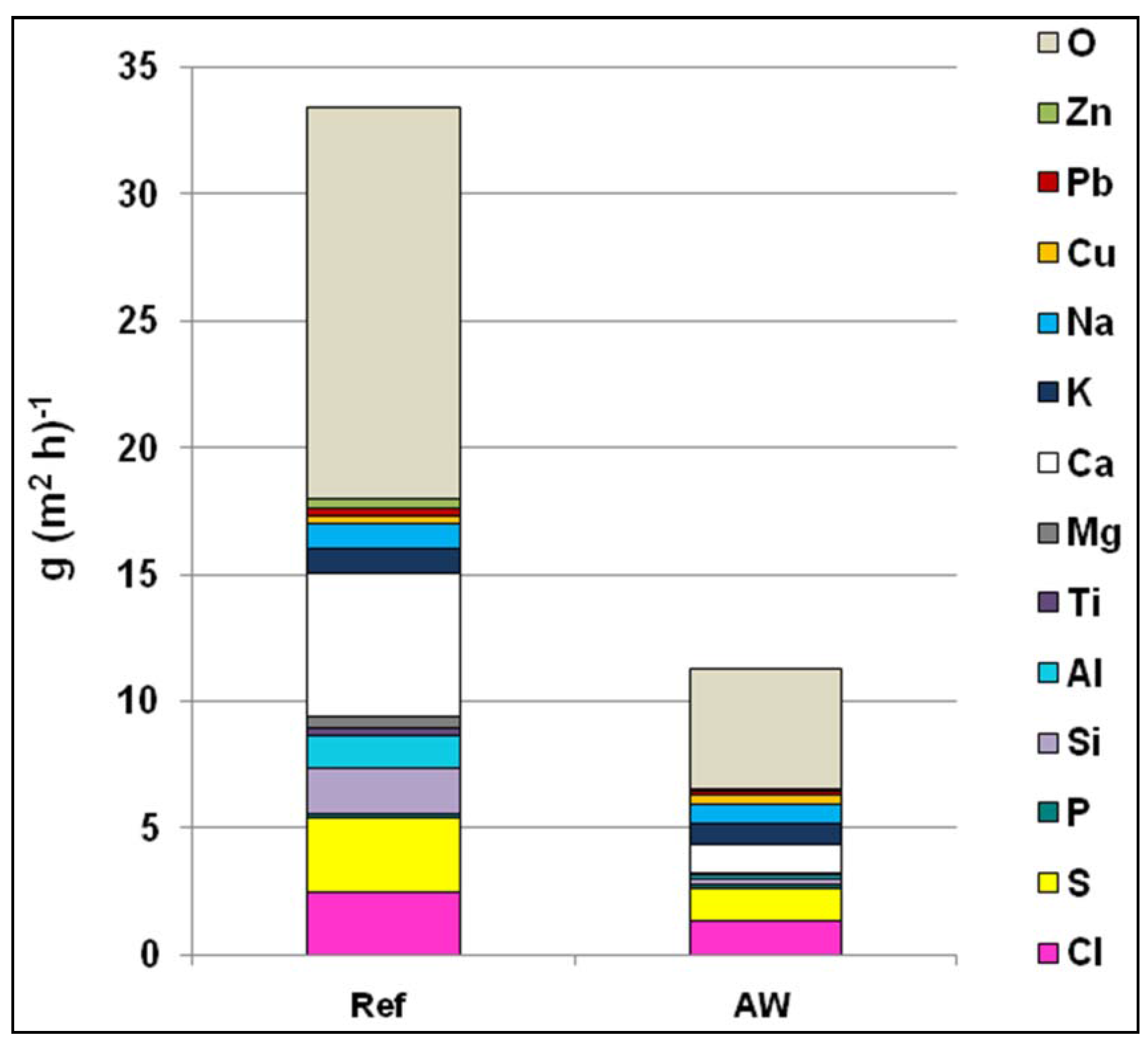
Figure 10). The comparison indicates increasing concentrations of S, Cl, and Ca when following the system from bed ash—the first fraction sampled—to filter ash. However, for Al, Si, and alkali the reverse is the case, with the lowest concentrations being found in the filter ash. This could be explained by the smaller amount of sand particles, consisting mostly of Si, Al, K, and Na, in the filter ash.
Once more, higher concentrations of Ca, P, S, and Cl could be observed in the ashes in the AW case. For S and Cl, this is more noteworthy given that S and Cl concentrations in the fuel in both the Ref and AW cases were fairly similar (
Figure 3). This may be linked to the lower bed temperature because the results are close to those from the RBT case, in which the lower bed temperature led to higher capture of S and Cl in the bed material coating [
8]. Ca and P, mainly introduced as bone tissue with the AW, seem to be enriched in the bed ashes. The results for K and Na in
Table 3 reconfirm that no major differences of the alkali concentration could be seen between the two cases.
Table 2.
XRD results from the Ref and AW cases.
Table 2.
XRD results from the Ref and AW cases.
| Compound | Boiler ash | Cyclone ash | Filter ash | Fresh Sand |
|---|
| Ref | AW | Ref | AW | Ref | AW |
|---|
| Ca(OH)2 | L | L | - | - | M | M | - |
| CaClOH | - | - | - | - | M | M | - |
| CaCO3 | - | - | L | L | L | L | - |
| CaO | L | L | T | T | - | - | - |
| CaSO4 | M | M | L | L | L | L | - |
| Ca5(PO4)3(OH,CI,F) | - | - | - | M | - | - | - |
| Na4Ca(SO4)3 | - | - | - | - | - | - | - |
| Ca3Al2O6 | M | M | M | M | - | - | - |
| Ca2Al2SiO7 | M | M | M | M | - | - | - |
| CaSiO3 | - | - | - | - | - | - | - |
| NaCl | - | - | L | L | M | M | - |
| KCl | - | - | L | | M | M | - |
| SiO2 | M | M | M | M | - | - | M |
| NaAlSi3O8 | M | M | - | - | - | - | M |
| KAlSi3O8 | L | M | - | - | - | - | M |
| Na2Si2O5 | - | - | - | - | - | - | - |
| Fe2O3 | T | - | - | - | - | - | - |
| Al metall | - | - | L | L | - | - | - |
| Fe metall | L | - | - | - | - | - | - |
| MgO | M | M | - | - | - | - | - |
| (Mg,Al)6(Si,Al)4O10(OH)8 Clinochlore | - | - | - | - | - | - | M |
Figure 10.
Elemental analysis of bed ash, return sand, boiler ash, cyclone ash, and filter ash, for the Ref and AW cases.
Figure 10.
Elemental analysis of bed ash, return sand, boiler ash, cyclone ash, and filter ash, for the Ref and AW cases.
Table 3.
Elemental analysis of ashes [g (kg ash)−1].
Table 3.
Elemental analysis of ashes [g (kg ash)−1].
| Element | Bed Ash | Return Sand | Boiler Ash | Cyclone Ash | Filter Ash |
|---|
| Ref | AW | Ref | AW | Ref | AW | Ref | AW | Ref | AW |
|---|
| Cl | 1.0 | 3.0 | 0.9 | 2.4 | 1.4 | 2.0 | 18 | 17 | 99 | 137 |
| S | 2.6 | 5.8 | 3.1 | 13 | 7.2 | 9.3 | 21 | 19 | 33 | 17 |
| P | 1.9 | 18 | 1.8 | 13 | 3.4 | 17 | 6.2 | 28 | 2.1 | 11 |
| Si | 287 | 241 | 307 | 244 | 277 | 258 | 156 | 132 | 13 | 14 |
| Al | 49 | 43 | 60 | 56 | 68 | 57 | 84 | 77 | 11 | 10 |
| Ti | 3.1 | 3.3 | 5.5 | 8.4 | 6.2 | 6.0 | 16 | 15 | 1.0 | 1.7 |
| Mg | 10 | 11 | 7.6 | 10 | 12 | 9.3 | 21 | 18 | 6.1 | 6.7 |
| Ca | 52 | 82 | 63 | 123 | 79 | 105 | 196 | 206 | 365 | 373 |
| K | 16 | 15 | 24 | 20 | 21 | 20 | 12 | 12 | 7.4 | 11 |
| Na | 48 | 42 | 31 | 30 | 28 | 30 | 19 | 19 | 12 | 14 |
| Cu | 4.6 | 7.0 | 3.0 | 1.4 | 6.6 | 5.3 | 4.1 | 3.5 | 5.3 | 4.8 |
| Pb | 0.7 | 0.4 | 0.7 | 0.3 | 0.6 | 0.4 | 0.9 | 0.7 | 3.5 | 2.4 |
| Zn | 2.5 | 1.9 | 3.0 | 2.1 | 3.8 | 2.5 | 7.0 | 5.8 | 5.2 | 3.3 |
| Fe | 33 | 23 | 31 | 29 | 29 | 28 | 26 | 24 | 3.4 | 2.3 |
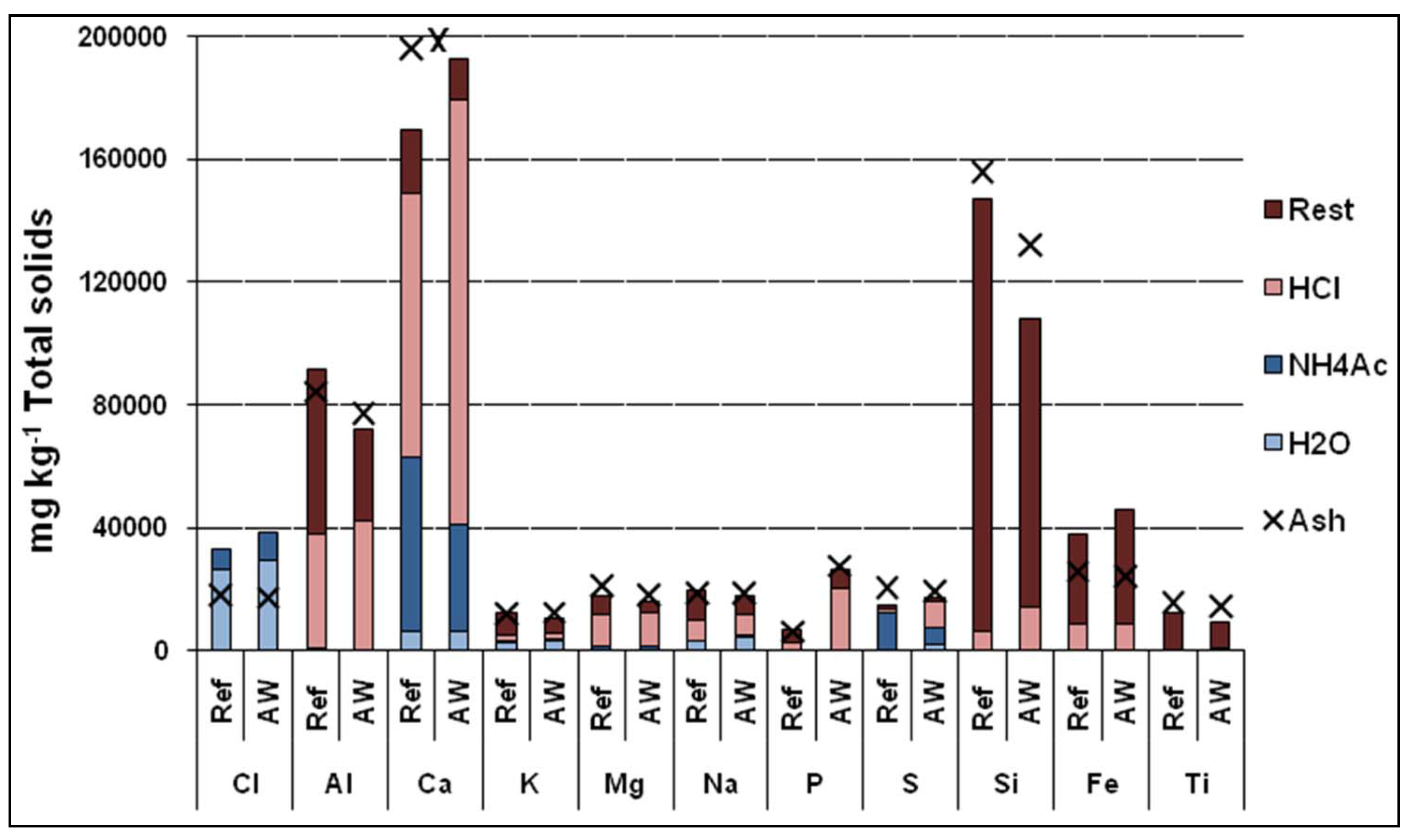
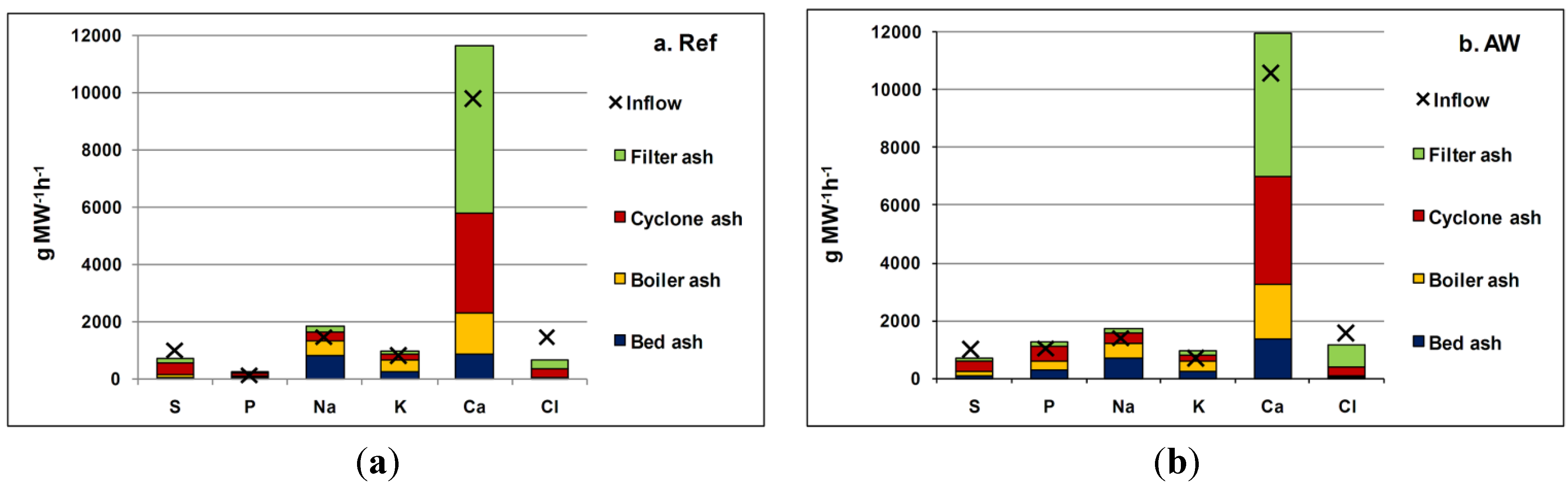
Figure 11 presents the elemental balances over the boiler for the various fractions. The results were calculated using the measured ash and fuel flows, in addition to the chemical compositions of both fuels and ashes. The fractions entering the system are fuel and lime and the fractions coming out are the ash fractions. The element balances agree very well, considering the size of the boiler.
Figure 11.
Element balances over the boiler: (a) Ref case; (b) AW case. X = The total inflow of the element to the boiler (fuel and lime) compared to the outflow (ash flows).
Figure 11.
Element balances over the boiler: (a) Ref case; (b) AW case. X = The total inflow of the element to the boiler (fuel and lime) compared to the outflow (ash flows).
Table 4 presents more detailed information about the proportion of elements in different ash fractions. As a result of AW co-combustion, the proportion of total P increased from 15% to 23% in the bed ash but decreased from 46% to 40% in the cyclone ash. The Ca concentration increased by 2.7 and 3.6 percentage points in the bed ash and boiler ash, respectively, but decreased by 6.6 percentage points in the filter ash. The S concentrations rose by approximately 8 percentage points in the bed ash and boiler ash, but declined by 1percentage points in the cyclone ash and 20 percentage points in the filter ash. The total alkali decreased by 13 percentage points in the bed ash, but increased in the boiler, cyclone and filter ashes. The greatest reduction in Cl concentration was observed in cyclone ash, in which it decreased by over 20 percentage points, while it increased by 1.5 percentage points in the bed ash.
Table 4.
Elemental analysis balance of the ashes (wt-% distribution).
Table 4.
Elemental analysis balance of the ashes (wt-% distribution).
| %Proportion in Ash | Bed Ash | Boiler Ash | Cyclone Ah | Filter Ash |
|---|
| Ref | AW | Ref | AW | Ref | AW | Ref | AW |
|---|
| P | 13.6 | 23.3 | 25.7 | 24.8 | 46.8 | 40.0 | 13.9 | 12.0 |
| Ca | 7.5 | 11.4 | 12.1 | 15.9 | 30.2 | 31.2 | 50.2 | 41.6 |
| S | 4.1 | 11.4 | 12.1 | 20.0 | 34.6 | 41.6 | 49.2 | 27.0 |
| K | 27.9 | 25.8 | 38.2 | 36.3 | 21.8 | 23.0 | 12.2 | 14.8 |
| Na | 44.3 | 39.3 | 27.2 | 31.0 | 18.3 | 19.3 | 10.2 | 10.3 |
| Cl | 0.9 | 2.2 | 1.3 | 1.6 | 16.7 | 14.1 | 81.1 | 82.1 |
3.7. Bed and Agglomeration Temperature
Adding AW to the fuel mix, reduced the bed temperature by 70 °C relative to that of the Ref case. The positive effects of AW co-combustion on the bed agglomeration are shown in
Figure 12a,b, in which visual inspection of the bed ashes for both cases shows considerably less amount of agglomerated particles in the AW case than do those from the Ref case. This difference could be explained by the change in temperature, the changed chemistry or both [
8].
Figure 12.
Bed ash samples: (a) Ref case, (b) AW case.
Figure 12.
Bed ash samples: (a) Ref case, (b) AW case.
The results of the lab-scale bed agglomeration test are presented in
Table 5. These results confirm that co-combustion of AW, with the normal waste fuel, increases the sintering temperature by 70–100 °C. However, the sintering temperatures in the tests are higher than the bed temperature in the full scale boiler.
Table 5.
Agglomeration temperatures for return sand.
Table 5.
Agglomeration temperatures for return sand.
| State | Unit | Ref | AW |
|---|
| Partial agglomeration | °C | 953 | 981 |
| Total agglomeration | °C | 993 | 1079 |
3.8. Emission
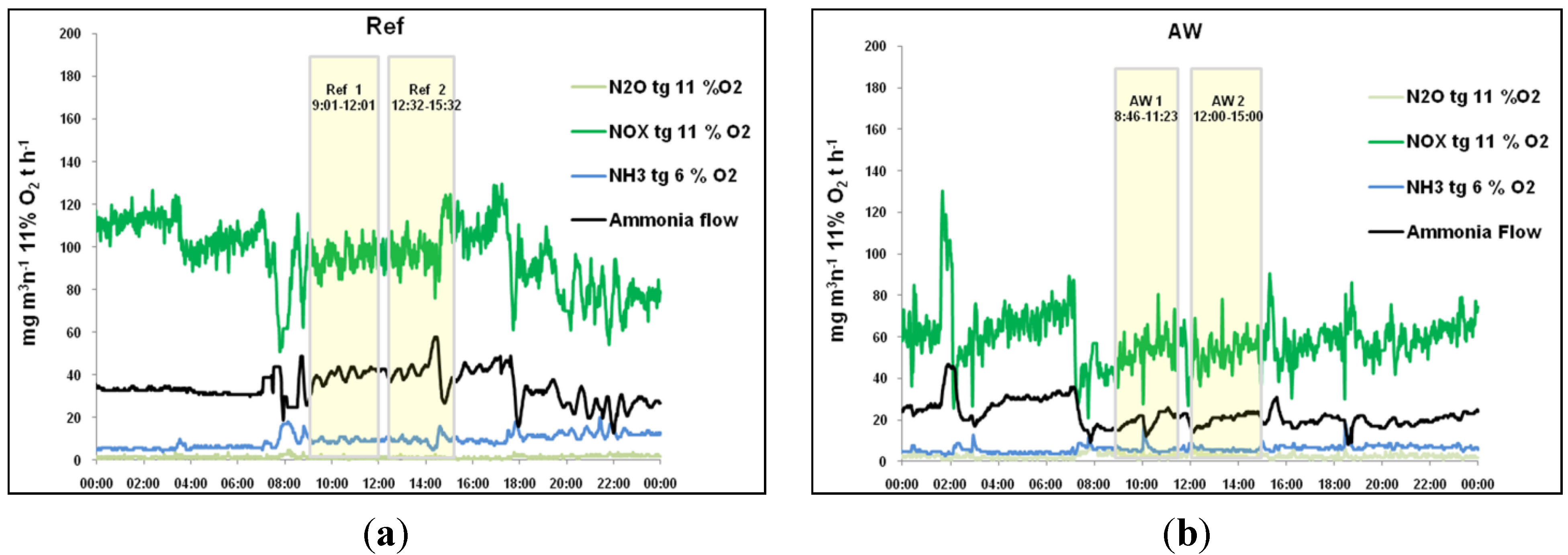
Co-combustion of AW with MSW raises concern about higher NO
x emissions, regarding high concentration of nitrogen in the fuel. However, flue gas analysis indicated a reduction in NO
x emissions of approximately 50% when AW was added to the fuel mix, as seen in
Figure 13. The NO
x-reduction effect of AW co-combustion has been reported previously [
11]. The decrease in NO
x concentration in the flue gas, despite the much higher nitrogen content of the AW, could be explained by the release of nitrogen as ammonia (NH
3) during combustion. In the AW, nitrogen is found as amino acids, which are decomposed during thermal treatment, releasing the amino-group. However, the lower bed temperature and lower primary air flow are other possible explanations. Adding AW also resulted in a 50% lower demand for ammonia injection into the boiler, to reduce NO
x emission. In addition, SO
2 emissions also declined from 11 to under 1 mg per normal cubic meter of flue gas in the case of AW addition (
Table 6). This might be due to a higher concentration of Ca and a lower concentration of S in the AW case fuel mix, leadings to the capture of S as CaSO
4 [
24].
Figure 13.
Emissions of N2O, NOx, and NH3 together with ammonia addition to boiler No. 1 in the Ref and AW cases: (a) Ref; (b) AW.
Figure 13.
Emissions of N2O, NOx, and NH3 together with ammonia addition to boiler No. 1 in the Ref and AW cases: (a) Ref; (b) AW.
Table 6.
Flue gas emissions.
Table 6.
Flue gas emissions.
| Compound | Unit | Ref | AW |
|---|
| NO
x | mg/Nm3 11% O2 | 95.5 | 53.0 |
| N2O | mg/Nm3 11% O2 | 1.4 | 3.0 |
| NH3 | mg/Nm3 11% O2 | 6.3 | 4.2 |
| SO2 | mg/Nm3 11% O2 | 11 | 0.5 |
| HCl | mg/Nm3 11% O2 | 8.1 | 8.0 |
4. Conclusions
Adding 20–30 wt% AW to the ordinary waste fuel mix reduced the bed temperature by 70–100 °C and effectively reduced the deposition growth rate in the super heater area. Despite higher concentrations of Cl, alkali and S for the deposits formed in the AW case, there were no indications of higher corrosion. Inspection of the boiler after almost one year of operation also revealed no signs of increased corrosion damage.
SEM-EDX analysis of the coatings formed on return sand and bed ash particles indicated higher concentrations of P, Ca, S, and Cl in the bed material in the AW case; this was confirmed by the chemical fractionation analysis. In addition, chemical fractionation indicated that more acid-soluble compounds of P, Ca, and S were formed in the AW case. The increased amount of Ca and P, in the form of bone tissue, and P from the soft tissue introduced into the boiler by the AW did not affect the combustion, but appeared as calcium phosphates in the ashes. Positive effects of AW co-combustion were also observed in the bed, where agglomerates were found in the Ref case but not in the AW case.
The emission data indicated lower NOx emissions in the AW case, despite a higher concentration of nitrogen in the AW fuel mix; this consequently reduced the rate of ammonia addition by 50%, relative to the Ref case. Later analyses indicated that the NOx emissions from the boiler decreased to minimum levels and that ammonia addition could be completely stopped when adding 15–20 wt% of AW. Furthermore, SO2 emissions declined during the co-combustion of AW, resulting in a decreased demand for added lime to capture sulfur.