Continuous Fermentation of Clostridium tyrobutyricum with Partial Cell Recycle as a Long-Term Strategy for Butyric Acid Production
Abstract
:1. Introduction
1.1. Background
| Parameters | Free cells (pH 6.0) | Immobilized cells (pH 6.0) | Immobilized cells (pH 5.5) | Extractive fermentation (pH 5.5) | Complete cell recycling (pH 5.8) | Partial cell recycling (pH 6.8) |
|---|---|---|---|---|---|---|
| ODmax | 5.8 | 11.5 | 8.2 | 8.1 | 35 | 15.1 |
| Butyrate concentration (g/L) | 16.3 | 43.4 | 20.4 | 301 | 33.0 | 7.58 |
| Butyrate yield (g/g) | 0.34 | 0.42 | 0.38 | 0.45 | 0.41 | 0.45 |
| Acetate yield (g/g) | 0.120 | 0.095 | 0.115 | 0.111 | 0.012 | 0.02 |
| Butyrate productivity (g/Lh) | 0.193 | 6.77 | 5.11 | 7.37 | 5.3 | 1.13 |
| Product selectivity | 0.74 | 0.81 | 0.77 | 0.80 | 0.97 | 0.95 |
| Operation days | 2 | 2 | 2 | 14 | 24 | 120 |
| Reference | [1] | [11] | [11] | [11] | [17] | This study |
1.2. Theory
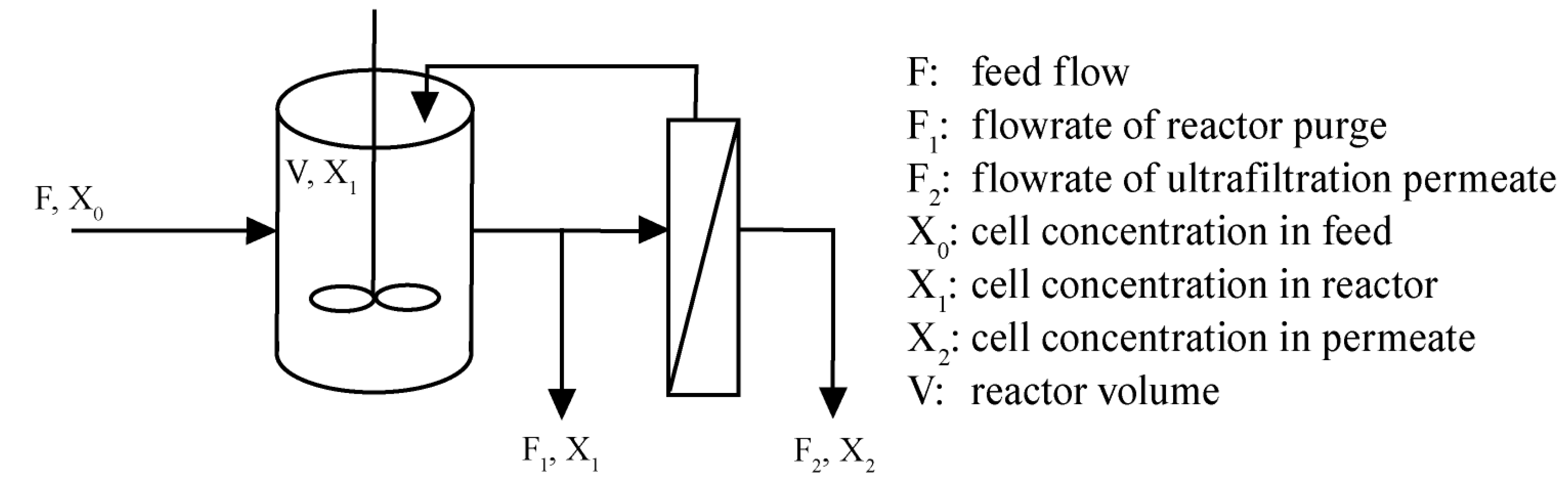
2. Results and Discussion
2.1. Batch Fermentation Kinetics with Clostridium Tyrobutyricum
| Run | Time (h) | Butyric acid(g/L) | Acetic acid (g/L) | Glucose (g/L) | Cell mass (g/L ) | Δx/Δt (g/Lh) | Instantaneous productivity of butyric acid (g/Lh) | Instantaneous productivity of acetic acid (g/Lh) | Instantaneous selectivity of butyric acid |
|---|---|---|---|---|---|---|---|---|---|
| Run #1 | 0 | 1.29 | 0.52 | 17 | 0.8 | - | - | - | - |
| 1 | 1.48 | 0.63 | 16.5 | 0.9 | 0.1 | 0.271 | 0.157 | 0.63 | |
| 4.7 | 2.22 | 1.16 | 14.2 | 1.42 | 0.135 | 0.301 | 0.193 | 0.609 | |
| 8.3 | 3.31 | 1.68 | 11.1 | 2.0 | 0.167 | 0.433 | 0.206 | 0.677 | |
| 12.9 | 5.23 | 2.36 | 6.2 | 2.68 | 0.152 | 0.596 | 0.211 | 0.739 | |
| 16.6 | 7.13 | 2.72 | 1.7 | 2.77 | 0.081 | 0.734 | 0.139 | 0.841 | |
| 18.0 | 7.95 | 2.75 | 0.03 | 3.1 | 0.071 | 0.806 | 0.031 | 0.963 | |
| Run #2 | 0 | 0.4 | 0.21 | 12 | 0.6 | - | - | - | - |
| 10 | 1.71 | 0.97 | 7.4 | 1 | 0.09 | 0.131 | 0.076 | 0.633 | |
| 23.3 | 4.38 | 2.41 | 3.06 | 2.4 | 0.241 | 0.20 | 0.108 | 0.649 | |
| 24.4 | 4.65 | 2.455 | 2.4 | 2.65 | 0.636 | 0.245 | 0.041 | 0.857 | |
| 26.7 | 4.85 | 2.462 | 1.86 | 2.75 | 0.1 | 0.087 | 0.003 | 0.967 | |
| 27.5 | 5.13 | 2.5 | 1.26 | 2.77 | 0.0625 | 0.35 | 0.048 | 0.879 | |
| 28.5 | 5.77 | 2.71 | 0.96 | 2.75 | −0.04 | 0.64 | 0.21 | 0.751 |
2.2. Long-Term Fermentation Performance with C. Tyrobutyricum
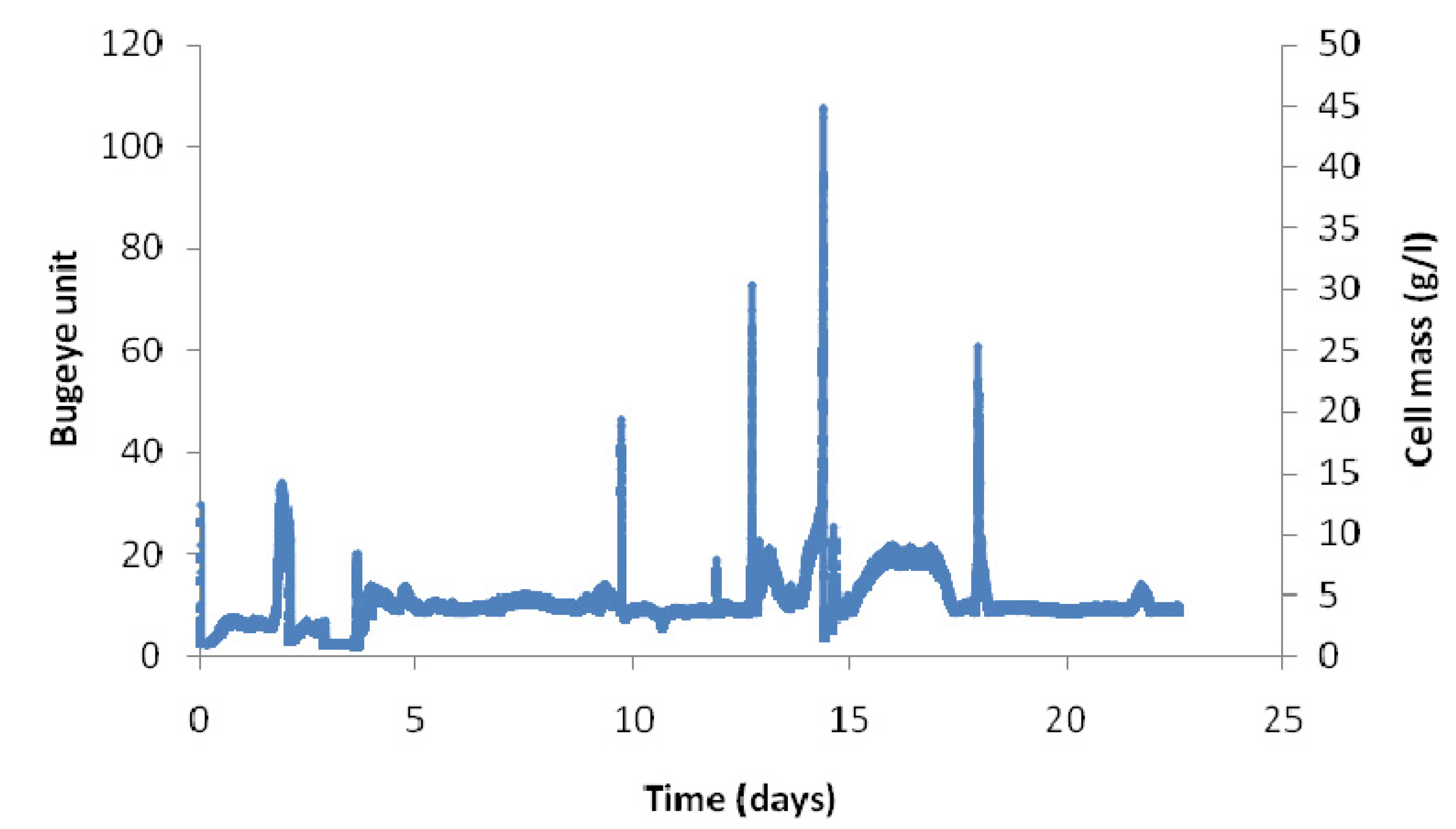
2.3. Effect of Dilution Rate on Continuous Fermentation Performance with No Cell Recycle
| Dilution rate (h−1) | R | wt% butyric | wt% acetic | wt% lactic | Cell mass (g/L) | Yield of butyric (g/g sugar) | Productivity of butyric (g/Lh) | YP/S |
|---|---|---|---|---|---|---|---|---|
| 0.077 | 0 | 33.3 | 9.1 | 57.6 | 4.34 | 0.22 | 0.28 | 0.66 |
| 0.077 | 95 | 95.8 | 3.2 | 0 | 15.1 | 0.446 | 0.567 | 0.47 |
| 0.15 | 0 | 19.3 | 14.7 | 66 | 4 | 0.143 | 0.36 | 0.74 |
| 0.15 | 80 | 88.9 | 7.2 | 3.8 | 14.3 | 0.435 | 1.06 | 0.49 |
| 0.15 | 95 | 94.3 | 4 | 1.7 | 14.8 | 0.442 | 1.11 | 0.47 |
| 0.21 | 0 | 35.3 | 40.4 | 24.2 | 4.01 | 0.221 | 0.79 | 0.63 |
| 0.21 | 40 | 23.4 | 17.3 | 59.3 | 5.3 | 0.184 | 0.65 | 0.79 |
| 0.21 | 80 | 70.9 | 16.3 | 12.7 | 14.1 | 0.332 | 1.17 | 0.47 |
| 0.21 | 95 | 53 | 14.6 | 32.4 | 10.8 | 0.318 | 1.12 | 0.6 |
| 0.38 | 0 | 30.2 | 48.8 | 21 | 2.4 | 0.074 | 0.48 | 0.25 |
| 0.38 | 26 | 9.5 | 14.4 | 76.1 | 4.2 | 0.066 | 0.43 | 0.69 |
| 0.38 | 43 | 5.8 | 11.1 | 83.2 | 4.5 | 0.051 | 0.33 | 0.88 |
| 0.38 | 80 | 37.2 | 10.3 | 52.5 | 13.9 | 0.149 | 0.96 | 0.40 |
| 0.38 | 95 | 42.5 | 12.8 | 44.7 | 15.5 | 0.159 | 1.26 | 0.37 |
2.4. Effects of Recycle Rate on Continuous Fermentation
2.5. Correlation of Productivity with Cell Growth Rate
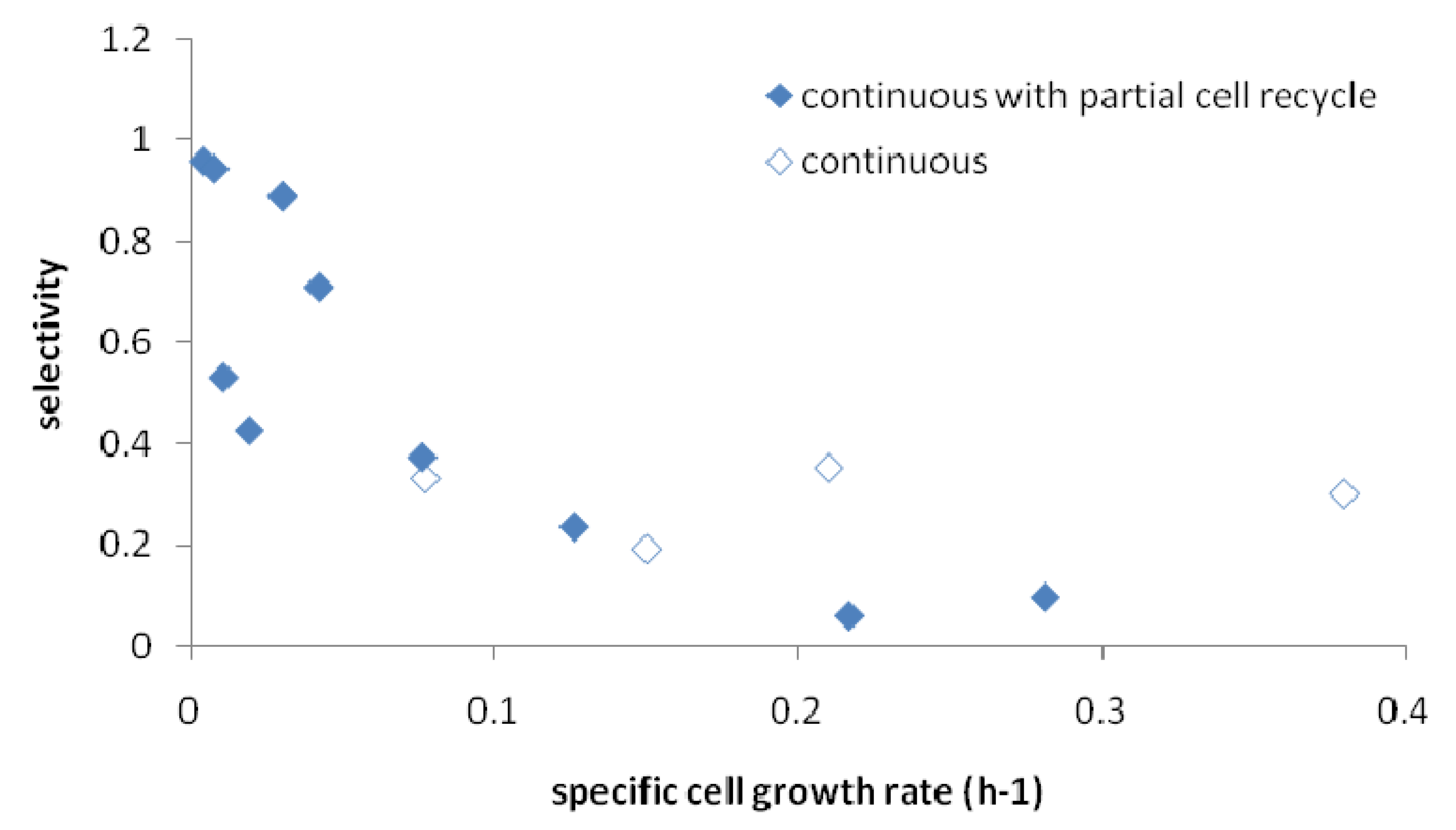
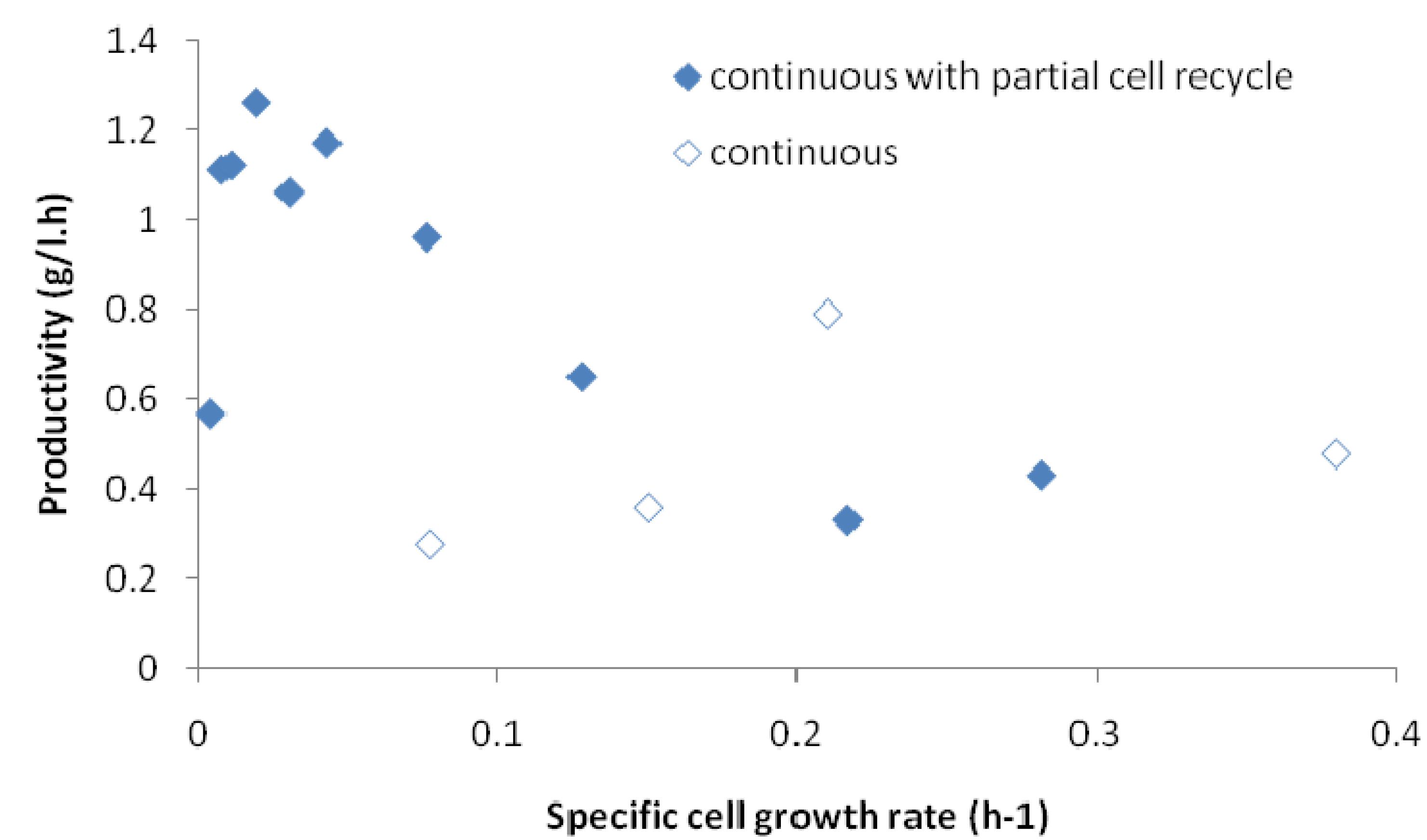
3. Experimental Section
3.1. Cultures and Medium
3.2. Fermentation Modes
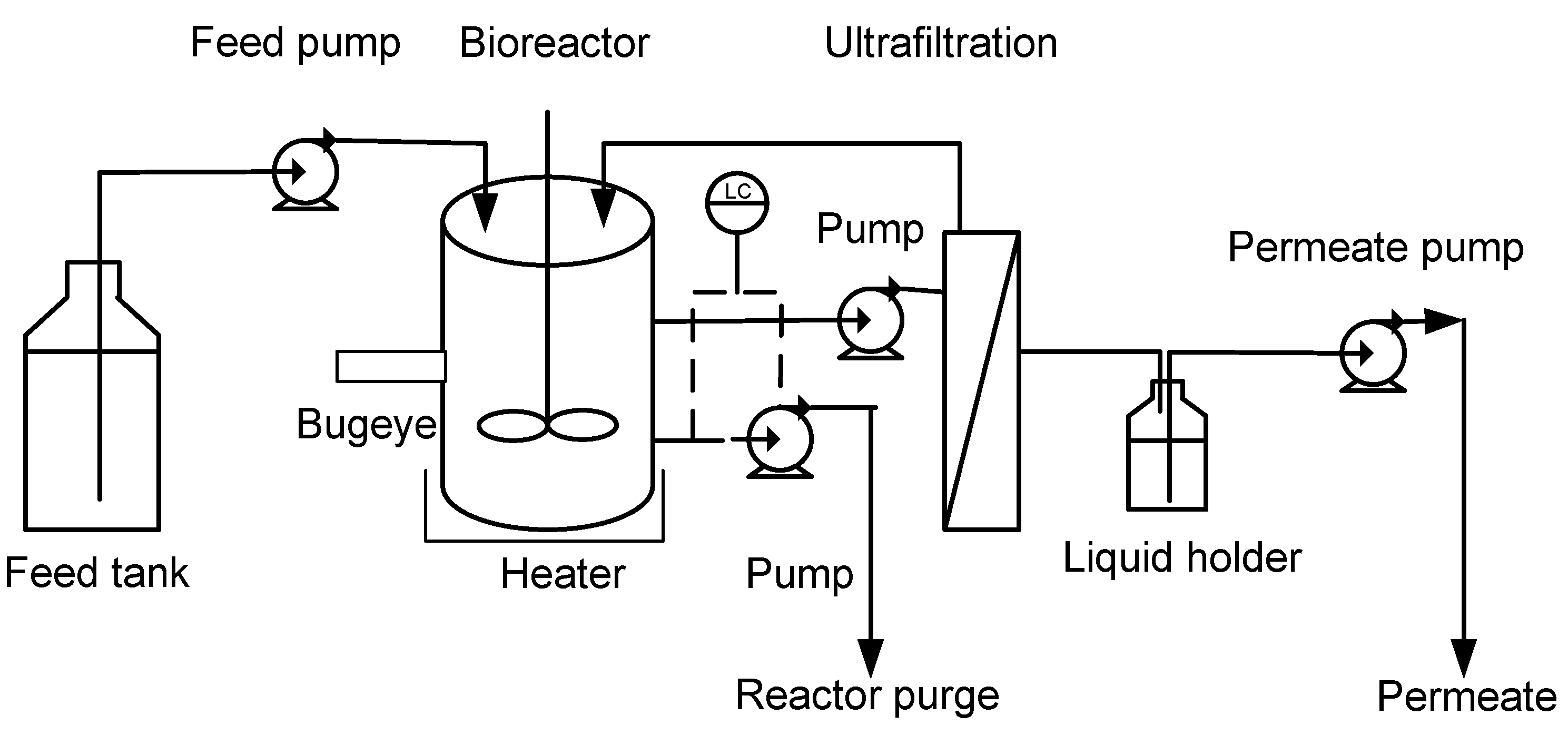
3.3. Analytical Methods
4. Conclusions
Acknowledgments
References
- Zigova, J.; Sturdik, E.; Vandak, D.; Schlosser, S. Butyric acid production by Clostridium butyricum with integrated extraction and pertraction. Process. Biochem. 1999, 34, 835–843. [Google Scholar] [CrossRef]
- Ramey, D.E. Continuous Two Stage, Dual Path Anaerobic Fermentation of Butanol and Other Organic Solvents Using Two Different Strains of Bacteria. U.S. Patent 5,753,474, 19 May 1998. [Google Scholar]
- Binder, T.; Canavera, D.; Cavalieri, R.; Chen, S.; Conway, R.; Downing, M.; Drumm, L.; Eidman, V.; Foster, H.; Green, K. Vision for bioenergy and biobased products in the United States: Bioeconomy for a sustainable future. In Proceedings of the Biomass Research and Development Initiative Vision Workshop, Sacramento, CA, USA, 2–3 March 2006.
- Zhang, C.; Yang, H.; Yang, F.; Ma, Y. Current progress on butyric acid production by fermentation. Curr. Microbiol. 2009, 59, 656–663. [Google Scholar] [CrossRef] [PubMed]
- Qureshi, N.; Ezeji, T.C.; Ebener, J.; Dien, B.S.; Cotta, M.A.; Blaschek, H.P. Butanol production by Clostridium beijerinckii. Part I: Use of acid and enzyme hydrolyzed corn fiber. Biosource Technol. 2008, 99, 5915–5922. [Google Scholar] [CrossRef]
- Ramey, D.E. Production of Butyric Acid and Butanol; DE-F-G02–00ER86106; U.S. Department of Energy: Morgantown, WV, USA, 2006.
- Potts, T.M.; Du, J.; Paul, M.; May, P.; Beitle, R.; Hestekin, J. The production of butanol from Jamaica Bay macroalgae. Environ. Prog. Sustain. Energy 2012, 31, 29–36. [Google Scholar] [CrossRef]
- Tashiro, Y.; Takeda, K.; Kobayashi, G.; Sonomoto, K.; Ishizaki, A.; Yoshino, S. High butanol production by Clostridium saccharoberbutylacetonicum N1-4 in fed-batch culture with pH-stat continuous butyric acid and glucose feeding method. J. Biosci. Bioeng. 2004, 98, 263–268. [Google Scholar] [CrossRef]
- Potts, T.M.; Ackerson, M.; Hestekin, J.A. The production of fuel grade butanol from algal carbohydrates utilizing a hybrid biological and chemical process. In Presented at AIChE Annual Meeting, Minneapolis, MN, USA, 16–21 October 2011.
- Weigel, J.; Seung-Uk, K.U.K.; Kohring, G.W. Clostridium thermobutyricum: A moderate thermophile isolated from a cellulolytic culture that produces butyrate as the major product. Int. J. Syst. Bacteriol. 1989, 39, 199–204. [Google Scholar] [CrossRef]
- Wu, Z.; Yang, S.T. Extractive fermentation for butyric acid production from glucose by clostridium tyrobutyricum. Biotechnol. Bioeng. 2003, 82, 93–102. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Yang, S.T. Kinetics of butyric acid fermentation of glucose and xylose by clostridium tyrobutyricum wild type and mutant. Process Biochem. 2006, 41, 801–808. [Google Scholar] [CrossRef]
- Liu, X.; Zhu, Y.; Yang, S.T. Butyric acid and hydrogen production by clostridium tyrobutyricum ATCC 25755 and mutants. Enzyme Microb. Technol. 2006, 38, 521–528. [Google Scholar] [CrossRef]
- Zhu, Y.; Yang, S.T. Effect of pH on metabolic pathway shift in fermentation of xylose by clostridium tyrobutyricum. J. Biotechnol. 2004, 110, 143–157. [Google Scholar] [CrossRef] [PubMed]
- Van Andel, J.G.; Zoutberg, G.R.; Crabbendam, P.M.; Breure, A.M. Glucose fermentation by clostridium butyricum grown under a self generated gas atmosphere in chemostat culture. Appl. Microbiol. Biotechnol. 1985, 23, 21–26. [Google Scholar] [CrossRef]
- Michel-Savin, D.; Marchal, R.; Vandecasteele, J.P. Butyrate production in continuous culture of clostridium tyrobutyricum: Effect of end-product inhibition. Appl. Microbiol. Biotechnol. 1990, 33, 127–131. [Google Scholar]
- Michel-Savin, D.; Marchal, R.; Vandecasteele, J.P. Control of the selectivity of butyric acid production and improvement of fermentation performance with clostridium tyrobutyricum. Appl. Microbiol. Biotechnol. 1990, 32, 387–392. [Google Scholar] [CrossRef]
- Zhu, Y. Enhanced Butyric Acid Fermentation by Clostridium Tyrobutyricum Immobilized in a Fibrous-Bed Reactor. Ph.D. Dissertation, Ohio State University, Columbus, OH, USA, 2003. [Google Scholar]
- Arora, M.B.; Hestekin, J.A.; Snyder, S.W.; St. Martin, E.J.; Lin, Y.J.; Donnelly, M.I.; Millard, C.S. The separative bioreactor: A continuous separation process for the simultaneous production and direct capture of organic acids. Sep. Sci. Technol. 2007, 42, 2519–2538. [Google Scholar] [CrossRef] [PubMed]
- Du, J.; Lorenz, N.; Beitle, R.R.; Hestekin, J.A. Application of wafer-enhanced electrodeionization in continuous fermentation process to produce butyric acid with Clostridium tyrobutyricum. Sep. Sci. Technol. 2012, 47, 43–51. [Google Scholar] [CrossRef]
- Hu, W.; Dodge, T.C. Cultivation of mammalian cells in bioreactors. Biotechnol. Prog. 1985, 1, 4–10. [Google Scholar] [CrossRef]
- Cheetham, P.S.J.; Blunt, K.W.; Bucke, C. Physical studies on cell immobilization using calcium alginate gels. Biotechnol. Bioeng. 1979, 21, 2155–2168. [Google Scholar] [CrossRef]
- Karel, S.F.; Libicki, S.B.; Robertson, C.R. The immobilization of whole cells: Engineering principles. Chem. Eng. Sci. 1985, 40, 1321–1354. [Google Scholar] [CrossRef]
- Shuler, M.L.; Kargi, F. Bioprocess Engineering: Basic Concepts, 2nd ed.; Prentice Hall: Upper Saddle River, NY, USA, 2002. [Google Scholar]
- Mason, C.A.; Hamer, G.; Bryers, J.D. The death and lysis of microorganisms in environmental processes. FEMS Microbiol. Lett. 1986, 39, 373–401. [Google Scholar] [CrossRef]
- Ferras, E.; Minier, M.; Goma, G. Acetonobutylic fermentation: Improvement of performances by coupling continuous fermentation and ultrafiltration. Biotechnol. Bioeng. 1986, 28, 523–533. [Google Scholar] [CrossRef] [PubMed]
- Young, J.; Lee, P.J.; di Gioia, A.D. Fast HPLC Analysis for Fermentation Ethanol Process. Waters 2007, 720001896EN. Available online: http://www.waters.com/waters/library.htm?cid=511436&lid=1512671 (accessed on 26 July 2012). [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Du, J.; McGraw, A.; Lorenz, N.; Beitle, R.R.; Clausen, E.C.; Hestekin, J.A. Continuous Fermentation of Clostridium tyrobutyricum with Partial Cell Recycle as a Long-Term Strategy for Butyric Acid Production. Energies 2012, 5, 2835-2848. https://doi.org/10.3390/en5082835
Du J, McGraw A, Lorenz N, Beitle RR, Clausen EC, Hestekin JA. Continuous Fermentation of Clostridium tyrobutyricum with Partial Cell Recycle as a Long-Term Strategy for Butyric Acid Production. Energies. 2012; 5(8):2835-2848. https://doi.org/10.3390/en5082835
Chicago/Turabian StyleDu, Jianjun, Amy McGraw, Nicole Lorenz, Robert R. Beitle, Edgar C. Clausen, and Jamie A. Hestekin. 2012. "Continuous Fermentation of Clostridium tyrobutyricum with Partial Cell Recycle as a Long-Term Strategy for Butyric Acid Production" Energies 5, no. 8: 2835-2848. https://doi.org/10.3390/en5082835
APA StyleDu, J., McGraw, A., Lorenz, N., Beitle, R. R., Clausen, E. C., & Hestekin, J. A. (2012). Continuous Fermentation of Clostridium tyrobutyricum with Partial Cell Recycle as a Long-Term Strategy for Butyric Acid Production. Energies, 5(8), 2835-2848. https://doi.org/10.3390/en5082835





